Abstract
Objectives:
Prescribing antipsychotic medications to children and adolescents with severe mental and developmental disorders is common; however, there is a lack of consensus on appropriate metabolic monitoring for this population. This review systematically evaluates studies examining metabolic monitoring of children and adolescents prescribed antipsychotic medication to understand the clinical practice of metabolic monitoring and identify opportunities to improve the safety of antipsychotic prescribing in this population.
Methods:
A systematic search for original research on metabolic monitoring in children and adolescents prescribed antipsychotics was conducted in six databases (PubMed, EMBASE, PsycINFO, The Cochrane Library [Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Trials, CENTRAL], Cochrane Methodology Register and Web of Science [Science and Social Science Citation Index]) from inception to February 2020 following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Studies were assessed for quality and findings summarised using narrative synthesis.
Results:
Fifteen papers were identified. Studies agreed on the need for metabolic monitoring; however, there was a gap between guideline-recommended practice and clinical practice. Variable rates of baseline and subsequent monitoring were reported for both physical and biochemical parameters, with particularly low rates for monitoring requiring venesection. Younger age was also associated with lower monitoring rates. Implementation of quality improvement activities (new guidelines, staff education and checklists) improved monitoring rates although the measurement of biochemical parameters still occurred in only a minority of children.
Conclusion:
Despite widespread awareness and concern regarding metabolic side-effects, monitoring occurred inconsistently and infrequently, particularly for biochemical parameters requiring venesection. Monitoring of anthropometric measures (weight, body mass index and waist circumference) with escalation to more laboratory testing where metabolic concerns are identified may improve monitoring. Minimising iatrogenic harm, through reduced antipsychotic prescription where possible, is a clinical priority in this population.
Introduction
The prescription of antipsychotic medication to children and adolescents has increased in recent years for both approved indications and off-label prescribing (Kaguelidou et al., 2020). Off-label prescribing commonly occurs for the management of mood disorders, disruptive behavioural disorders and autism spectrum disorders (Hollingworth et al., 2013; Morrison et al., 2013; Scott et al., 2018). While there has been a concerted effort to reduce the risk of cardiometabolic adverse effects arising from antipsychotic medication in adults (Melamed et al., 2019), metabolic monitoring in children has received relatively less attention. Cardiometabolic adverse effects start occurring soon after commencing antipsychotic medication (Chung et al., 2020; Nicol et al., 2018), making it essential that metabolic changes are detected early so adverse effects are managed to prevent morbidity and premature mortality in adult life (De Hert et al., 2011). In addition, these physical adverse effects can have a detrimental impact on a young person’s social and emotional wellbeing. For example, obesity places young people at greater risk of peer victimisation (Thomas et al., 2018), which in turn is a risk factor for mental illness (Moore et al., 2014) and adverse educational outcomes (Moore et al., 2015). Children prescribed antipsychotic medication may also experience stigma from peers and teachers (Hamrin et al., 2010). For these reasons, it is important that antipsychotic medication is used judiciously in this population and avoided whenever possible. For those children where the prescribing of antipsychotic medication is clinically necessary, appropriate monitoring for adverse effects is essential.
Adult guidelines for metabolic monitoring have been widely adopted, however, overall rates of monitoring remain sub-optimal (De Hert et al., 2011; Lambert et al., 2017). For children and adolescents prescribed antipsychotic medication, metabolic monitoring guidelines are available. For example, the Canadian Alliance for Monitoring Effectiveness and Safety of Antipsychotic Medications in Children (CAMESA) has developed recommendations for metabolic monitoring in children and adolescents (Pringsheim et al., 2012). These are based on a systematic review of adverse effects of antipsychotic medications reported in children and adolescent clinical trials. They provide comprehensive medication specific recommendations for anthropometric measures (height, weight, waist circumference [WC], blood pressure [BP] and body mass index [BMI]), laboratory investigations of biochemical parameters (e.g. fasting blood glucose [FBG], fasting blood lipids [FBL]) and the time intervals when these should be conducted. The CAMESA guideline recommendations closely resemble metabolic monitoring recommended for adults with schizophrenia prescribed antipsychotic medications (De Hert et al., 2011) because the side-effect profile for the medications is similar in both young and old. Where guidelines for metabolic monitoring of children and adolescents prescribed antipsychotic medications have been implemented, there have been low rates of adherence (Chen et al., 2018), for which the reasons are unclear.
This study systematically evaluated literature relating to metabolic monitoring of children and adolescents prescribed antipsychotic medication to inform the reasons for low rates of metabolic monitoring. The aim was to better understand systemic reasons as to why guideline adherence was low and to identify opportunities to improve the safety of prescribing of antipsychotic medication to children and adolescents.
Methods
A systematic review (outlined in Supplementary Figure 1) was conducted using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The review was prospectively registered with the PROSPERO International Prospective Register of Systematic Reviews (Registration ID: CRD42020145907).
A search strategy, documented in Supplementary Table 1, was developed with the assistance of a medical librarian and was used to search six databases for English-language, original research articles published in peer-reviewed journals from inception to 11 February 2020. These databases were PubMed, EMBASE, PsycINFO, The Cochrane Library (Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Trials [CENTRAL]), Cochrane Methodology Register and Web of Science (Science and Social Science Citation Index). Reference chaining of identified studies was also conducted.
The inclusion criteria for study selection were as follows: (a) articles published in English language; (b) study population of individuals aged < 18 years; (c) prescribed antipsychotic medication at a mental health service (either inpatient or outpatient); and (d) describe any intervention involving the screening or monitoring of metabolic parameters.
Study selection and data extraction
Papers were identified through screening of titles and abstracts independently by two authors (L.M. and A.A.). Full-text articles were reviewed in accordance with inclusion criteria. Eligibility assessment was then performed by two authors (L.M. and A.A.) independently. All articles were coded as either met or not met eligibility criteria. From those that met criteria, the following characteristics were extracted from each study where available: (a) study identifiers and description (including author, publication year, location, aims, participant details, study design and methodology, and sample sizes); (b) study findings and effect size, 95% confidence intervals (CIs) and any measured confounders; and (c) study recommendations and conclusions.
Quality assessment
The Cochrane Non-Randomised Studies Methods Working Group recommends the use of the Newcastle–Ottawa Scale (NOS) to evaluate study quality and bias (Wells et al., 2000). A modified version of the NOS (detailed in Supplementary Table 2) was applied to the identified cohort studies. A maximum of six points could be obtained. No points were allocated for the categories ‘demonstration that outcome of interest was not present at start of study’ and ‘adequate duration of follow up’ as these were not relevant to the identified studies.
Analysis of findings
Given the range of different monitoring procedures and outcomes, the outcome measures were not homogeneous enough to enable pooling. The findings of these studies were summarised using narrative synthesis. The findings were discussed between three child and adolescent psychiatrists (L.M., A.A. and J.S.), and clinical conclusions derived.
Results
Fifteen papers were identified for inclusion in this review comprising four studies of self-reported clinical practice (Curtis et al., 2012; McLaren et al., 2017; Rodday et al., 2015; Walter et al., 2008) (Table 1), five data linkage studies of metabolic monitoring in children and adolescents attending mental health services (Connolly et al., 2015; Hayden et al., 2019; Morrato et al., 2010; Okumura et al., 2018; Raebel et al., 2014) (Table 2) and six retrospective chart review studies to examine metabolic monitoring (Cotes et al., 2017; Coughlin et al., 2018; Featherston et al., 2018; Javaheri and McLennan, 2019; Kauffman et al., 2018; Wakefield et al., 2019) (Table 3). Of the 11 studies which reported data from child and adolescent patients, five provided information about the diagnoses of the children (Connolly et al., 2015; Coughlin et al., 2018; Featherston et al., 2018; Hayden et al., 2019; Morrato et al., 2010). The diagnoses provided were wide ranging and representative of children attending a specialist mental health service. When reported, second-generation antipsychotics were typically prescribed. Quality assessment scores for included studies are summarised in Supplementary Table 3. Self-reported clinical practice studies were not subjected to quality scoring. For the other studies, scores ranged from three to six, with most points being lost due to non-representative cohorts, the comparison cohort not being specified and cohorts not being comparable.
Psychiatrist self-reports on their opinions and practice of metabolic monitoring in children and adolescents.
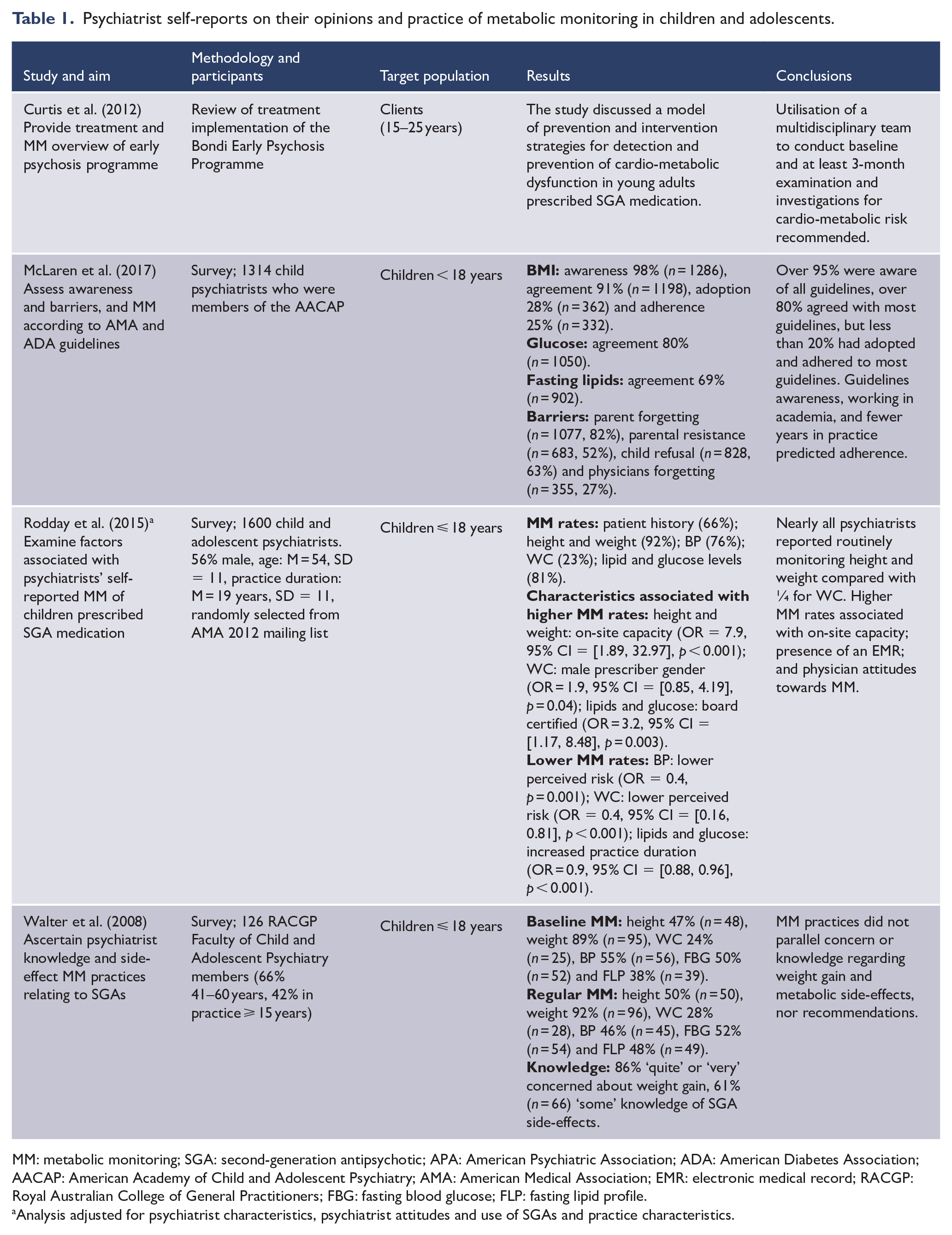
MM: metabolic monitoring; SGA: second-generation antipsychotic; APA: American Psychiatric Association; ADA: American Diabetes Association; AACAP: American Academy of Child and Adolescent Psychiatry; AMA: American Medical Association; EMR: electronic medical record; RACGP: Royal Australian College of General Practitioners; FBG: fasting blood glucose; FLP: fasting lipid profile.
Analysis adjusted for psychiatrist characteristics, psychiatrist attitudes and use of SGAs and practice characteristics.
Data linkage studies of metabolic monitoring of children and adolescents prescribed antipsychotic medication.
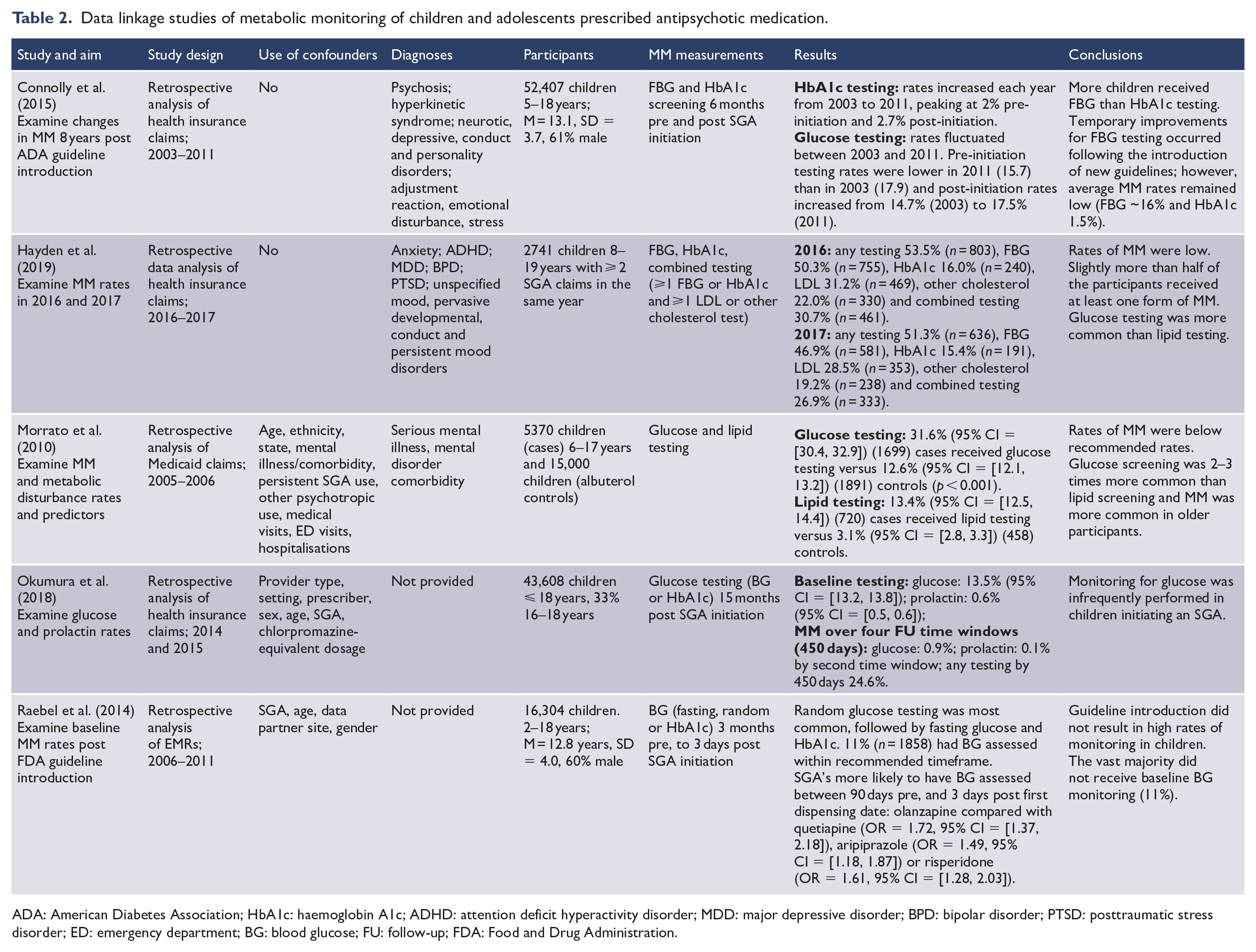
ADA: American Diabetes Association; HbA1c: haemoglobin A1c; ADHD: attention deficit hyperactivity disorder; MDD: major depressive disorder; BPD: bipolar disorder; PTSD: posttraumatic stress disorder; ED: emergency department; BG: blood glucose; FU: follow-up; FDA: Food and Drug Administration.
Retrospective medical record reviews of metabolic monitoring of children and adolescents prescribed antipsychotic medication.
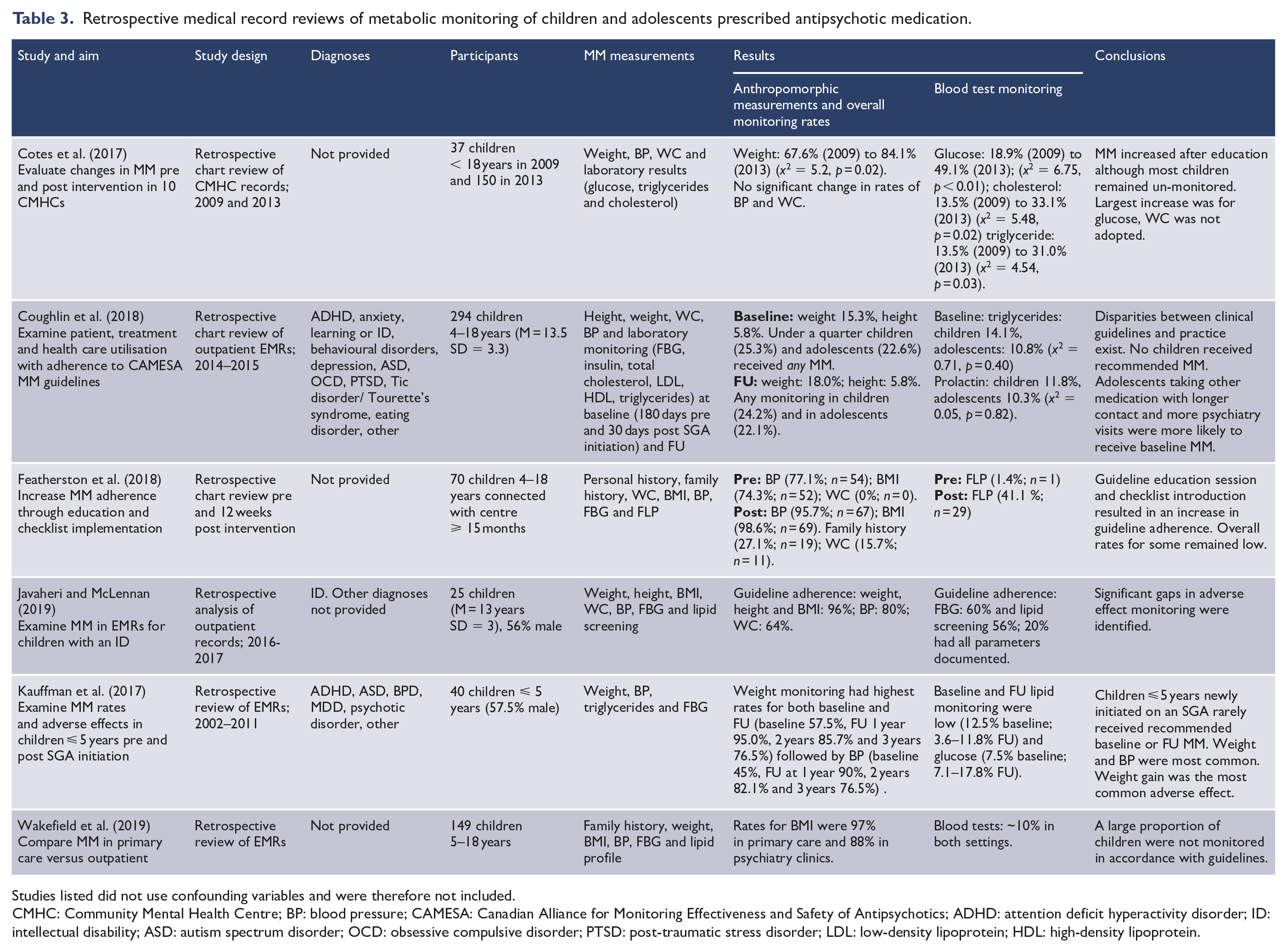
Studies listed did not use confounding variables and were therefore not included.
CMHC: Community Mental Health Centre; BP: blood pressure; CAMESA: Canadian Alliance for Monitoring Effectiveness and Safety of Antipsychotics; ADHD: attention deficit hyperactivity disorder; ID: intellectual disability; ASD: autism spectrum disorder; OCD: obsessive compulsive disorder; PTSD: post-traumatic stress disorder; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Studies describing self-reported metabolic monitoring
Four studies described clinician’s self-reports of metabolic monitoring in children and adolescents prescribed antipsychotic medication. Of these, two originated from Australia (Curtis et al., 2012; Walter et al., 2008) and two from the United States (McLaren et al., 2017; Rodday et al., 2015). Curtis et al. (2012) described a treatment and monitoring programme adopted by an Australian early psychosis service while the other three studies described the self-reported knowledge, opinions and monitoring practices of child and adolescent psychiatrists (McLaren et al., 2017; Rodday et al., 2015; Walter et al., 2008).
These articles concluded that the majority of psychiatrists had an awareness of, and agreement with, the need for metabolic monitoring; however, there were inconsistencies in the reported monitoring practices, with few basing these on current clinical guidelines. Walter et al. (2008) reported child psychiatrists (n = 126) varied in their baseline monitoring for both physical and biochemical parameters, with 24% measuring WC to 89% for weight, and 38% for FBL to 50% for FBG.
Weight and BMI were the most commonly monitored parameters reported by psychiatrists in all three survey-based studies (McLaren et al., 2017; Rodday et al., 2015; Walter et al., 2008). For two studies (McLaren et al., 2017; Walter et al., 2008), biochemical monitoring was less likely to be conducted than anthropometric measures of metabolic adverse effects. In contrast, Rodday et al. (2015) described high rates of self-reported biochemical investigations among surveyed psychiatrists (n = 1600) with 81% endorsing monitoring of FBG and lipids.
Studies examining the implementation of metabolic monitoring
Of the eight studies reporting on the implementation of metabolic monitoring, four were data linkage studies (Hayden et al., 2019; Morrato et al., 2010; Okumura et al., 2018; Raebel et al., 2014) and four were retrospective chart review studies (Coughlin et al., 2018; Javaheri and McLennan, 2019; Kauffman et al., 2018; Wakefield et al., 2019). Three data linkage studies originated from the United States (Hayden et al., 2019; Morrato et al., 2010; Raebel et al., 2014) and one from Japan (Okumura et al., 2018). Two retrospective chart reviews originated from the United States (Kauffman et al., 2018; Wakefield et al., 2019) and two from Canada (Coughlin et al., 2018; Javaheri and McLennan, 2019).
In all eight studies, there was an implementation gap between guideline-recommended practice and metabolic monitoring in clinical practice. Javaheri and McLennan (2019) (n = 25) found guideline adherence varied by parameter, with higher rates for anthropometric measurements (BMI 96% and BP 80%) compared with biochemical monitoring of children (FBG 60% and FBL 56%). Only 20% of all patients had all core recommended parameters documented (Javaheri and McLennan, 2019).
These findings were consistent with other studies which showed consistently low rates of adherence to guidelines for metabolic monitoring, particularly in relation to biochemical monitoring (Cotes et al., 2017; Coughlin et al., 2018; Featherston et al., 2018; Hayden et al., 2019; Javaheri and McLennan, 2019; Kauffman et al., 2018; McLaren et al., 2017; Morrato et al., 2010; Okumura et al., 2018; Raebel et al., 2014; Rodday et al., 2015; Wakefield et al., 2019; Walter et al., 2008). Low rates of baseline monitoring was also observed. Coughlin et al. (2018) (n = 294) reported approximately one-quarter of children (25.3%) and adolescents (22.6%) had any baseline monitoring recorded, and Okumura et al. (2018) reported only 13.5% participants (n = 43,608) had baseline FBG testing. Several studies showed higher rates of ongoing BMI monitoring; however, monitoring of WC, FBG and FBL was consistently low (Cotes et al., 2017; Hayden et al., 2019; Kauffman et al., 2018; Morrato et al., 2010; Wakefield et al., 2019). Rates of glucose monitoring were consistently higher than lipid monitoring (Connolly et al., 2015; Cotes et al., 2017; Hayden et al., 2019; Javaheri and McLennan, 2019; Morrato et al., 2010) with the exception of Kauffman et al. (2018) (n = 40) who found lipid monitoring was more commonly undertaken, although both biochemical investigations were performed in less than one in eight children prescribed antipsychotic medication. For example, baseline lipid monitoring was performed in 12.5% of the study population, with 3.6–11.8% having follow-up lipid monitoring compared with 7.5% having baseline glucose monitoring and 7.1–17.8% having follow-up glucose monitoring.
Younger age was associated with lower rates of monitoring. Raebel et al. (2014) found patients aged 16–18 years were twice as likely to receive glucose monitoring compared to children aged 2- to 4-year-olds. This was consistent with the findings of Kauffman et al. (2018) who reported that only approximately 1 in 10 children under the age of 5 years had metabolic parameters measured from blood. Okumura et al. (2018) and Raebel et al. (2014) were the only studies to distinguish between rates of monitoring for different antipsychotics. Those prescribed olanzapine were more likely to have biochemical metabolic monitoring than children prescribed risperidone (Raebel et al., 2014) or aripiprazole (Okumura et al., 2018).
Changes to metabolic monitoring practices following quality improvement interventions
Three studies (one data linkage [Connolly et al., 2015] and two retrospective chart reviews [Cotes et al., 2017; Featherston et al., 2018]) from the United States reported on changes to monitoring following quality improvement interventions. Connolly et al. (2015) (n = 52,407) found FBG testing increased from 14.7% to 16.6% in response to the introduction of new guidelines. Although this was statistically significant due to the large sample size, the absolute increase (1.9% of children) in monitoring was negligible (Connolly et al., 2015). Cotes et al. (2017) (n = 187) reported increased metabolic monitoring (glucose = 18.9–49.1%, cholesterol = 13.5–33.1%, triglycerides = 13.5–31.0% and weight = 67.6–84.1%) following education with audit and feedback, while Featherston et al. (2018) (n = 70) reported an increase in testing across all monitoring parameters (BP = 77.1–95.7%; BMI = 74.2–98.6%; WC = 0–15.7%; FBG = 8.6–38.6% and FBL = 1.4–41.4%) following the introduction of a checklist and staff education session.
Discussion
This systematic review showed that despite high rates of clinician self-reported awareness and monitoring of metabolic adverse effects arising from antipsychotic medication, adherence with guideline recommendations in clinical practice is very low. In particular, monitoring of biochemical parameters was undertaken in a small minority of children prescribed antipsychotic medications. There has been increased awareness in recent years of serious physical morbidity and premature mortality associated with all mental disorders (Plana-Ripoll et al., 2019a, 2019b), some of which is attributable to metabolic complications of antipsychotic medication (Barton et al., 2020). However, there is no evidence from the studies in this review of increased adherence to guidelines for metabolic monitoring during the time 2008–2020 in which they were conducted. Our study indicates that low rates of metabolic monitoring in children prescribed antipsychotics by clinicians is due to factors other than lack of awareness and education.
Interventions such as the use of audits and feedback to psychiatrists, monitoring reminder systems and education for patients and families about the importance of monitoring have been recommended to improve metabolic monitoring guideline adherence (McLaren et al., 2017). Studies demonstrating improvements in rates of metabolic monitoring (Connolly et al., 2015; Cotes et al., 2017; Featherston et al., 2018) used one or more of these interventions. The improvements in guideline adherence were only modest and it was unclear if increased monitoring was sustained due to the short duration of follow up in these studies.
This review highlights that knowledge and concern regarding metabolic side-effects are not sufficient to translate to adequate monitoring, suggesting that there are other barriers that need to be addressed. The awareness-to-adherence model used by McLaren et al. (2017) suggests that further education of practitioners is unlikely to result in significant monitoring improvements. Poor adherence and variable physician agreement with current guidelines internationally highlights the need for guidelines to be relevant and realistic, and tailored specifically to the child and adolescent population (Connolly et al., 2015).
Far from being confined to psychiatry, difficulties with guideline adoption and adherence by physicians is widespread. Mosca et al. (2005) demonstrated low rates of guideline adherence for cardiovascular monitoring by specialties including primary care, obstetrics and gynaecology, and cardiology in non-mental health populations. One of the barriers identified in the adoption of clinical guidelines are the perception by physicians that clinical guidelines lack flexibility and clinical relevance (Kenefick, 2008).
Given the harmful effects of antipsychotic medications to children, reducing the prescribing of these medications is a clinical priority. A recent review of interventions to reduce antipsychotic prescribing to children and adolescents found prior authorisation programmes (where the prescriber is required to obtain authority to prescribe antipsychotics to children) were the most effective (Mackie et al., 2021). There will, however, be a proportion of children and adolescents where there is a clear indication or clinical necessity for treatment with antipsychotic medication. For these children, monitoring of adverse metabolic effects is essential.
Current child and adolescent metabolic monitoring guidelines recommend monitoring of physical and biochemical parameters for metabolic health. Venesection in the child and adolescent populations is inherently difficult, with both parents and children often reluctant for blood tests to occur. An alternative to increase rates of metabolic monitoring of children prescribed antipsychotic medication is for prescribing clinicians to focus on the anthropomorphic measurements, which are easily implemented at the point of care, as a first step to screen for metabolic changes, with progression to testing of FBG and FBL if cardiometabolic adverse effects of weight gain and increased WC are identified. Given that some studies noted low rates of compliance with monitoring of WC, it would be important to explore reasons for this.
The increased adoption of electronic medical records also offers the opportunity to provide clinical decision-making support at the point of care (Kenefick, 2008). ‘Let’s Get Physical’ was an intervention implemented in an Australian mental health service which combined clinician education and feedback using data from electronic medical records to measure guideline adherence for metabolic monitoring in people prescribed clozapine. The intervention resulted in a fourfold improvement in the guideline adherence with over 75% of service user’s physical health being monitored (Wilson et al., 2014). Electronic medical records offer the opportunity for real time auditing of monitoring parameters such as weight and BMI to be efficiently conducted.
Limitations
This review has several limitations. Most significant is the paucity of studies that have been conducted on this topic to-date and lack of consistency in outcome measures, which prevents statistical analysis. The studies identified were all from high-income countries, and of participants attending specialist mental health services. Furthermore, studies were limited to English-language papers. Arguably, these factors limit the generalisability of the reported results. However, the findings present strong evidence of a need for practice improvement in child and adolescent mental health services. Data-linkage studies included in this review (Connolly et al., 2015; Hayden et al., 2019; Morrato et al., 2010; Okumura et al., 2018; Raebel et al., 2014) with large participant numbers only assessed measurement of biochemical parameters and did not have anthropometric measures. Although these studies showed very low rates of biochemical monitoring, this may not represent measurement of physical parameters (weight, BMI) which might have been undertaken by physicians.
Conclusion
In conclusion, despite high rates of awareness and concern regarding metabolic side-effects for children and adolescents prescribed antipsychotics, monitoring for these side-effects occurs at concerningly low rates, particularly for biochemical parameters where venesection is required. Where monitoring occurs, it is completed inconsistently, with frequent non-adherence to clinical guidelines.
Although guidelines have recommended laboratory investigations of all children on antipsychotics, the adoption of this in real-world clinical practice does not appear to be feasible. One potential solution may be to focus on monitoring of anthropometric measures such as weight, BMI and WC with clear guidance for escalation to more invasive testing should these indicate metabolic adverse effects. Prescriber education and auditing of clinical practice with electronic medical records could enhance monitoring.
Finally, there is very strong evidence of iatrogenic harm caused by antipsychotic medication prescribed to children and adolescents. Minimising the use of these agents, particularly in those children with mood and disruptive behavioural disorders where other psychosocial and pharmacological interventions may result in symptomatic and functional improvements without unnecessarily risking their metabolic health, is a clinical priority in the practice of child psychiatry.
Supplemental Material
sj-docx-1-anp-10.1177_00048674211009620 – Supplemental material for Monitoring of metabolic side-effects in children and adolescents prescribed antipsychotic medication: A systematic review
Supplemental material, sj-docx-1-anp-10.1177_00048674211009620 for Monitoring of metabolic side-effects in children and adolescents prescribed antipsychotic medication: A systematic review by Laura Mead, Alice Ayres, Julie A Blake and James G Scott in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
