Abstract
Patients with psychiatric disorders such as bipolar disorder, depression and schizophrenia often take medication for prolonged periods; however, many of the psychotropic drugs in use today cause significant weight gain. The weight gained by psychiatric patients is often sufficient to adversely affect their health and many remain overweight, even after stopping treatment. Difficulties in quantifying weight gain and identifying those patients most likely to gain weight are compounded by the lack of a standard definition and the use of different means to report weight changes. For instance, many studies only report the raw change in weight without any reference to the patient's size, while others use perhaps more useful indices such as body mass index or percentage weight change. Furthermore, much of the information is derived from relatively shortterm studies usually lasting no more than 2 months. Clearly this does not compare to their clinical use. The situation is further complicated by variables of diagnosis, age, sex, duration of treatment, comorbidity and polypharmacy.
Definitions and risks of weight gain
With natural ageing, body weight inevitably and gradually increases [1]. The ideal body weight for a man or woman can be calculated in relation to their height, with any changes being expressed as a percentage of this. Using this method of measurement, overweight men are defined by being 10% more than their ideal body weight and women are so classified when they are 20% more than the ideal. Similarly, obesity is defined as an excess of 35% for men and 45% for women. Body mass index (BMI) is another means of measuring these changes. The BMI describes the relative weight of an individual in relation to their height and is expressed as kg/m2 [2]. Overweight is defined as having a BMI of 25–30, and any BMI above this indicates obesity [3]. In the general population, being obese or overweight confers a significant increased risk of developing many medical diseases [4] for instance, type II non-insulin dependent diabetes mellitus (NIDDM) [5], ischaemic heart disease [6], hypertension [7], stroke [8], gall bladder disease [9] and some cancers [10, 11]. This is particularly the case with abdominal obesity which is an independent predictor of morbidity. Weight-associated illnesses are also more likely in those that smoke, show insulin resistance and have high systolic blood pressure and blood glucose levels [12, 13].
Mechanisms of weight gain
It has been suggested that those who are obese have a reduced metabolic rate (RMR), but in fact their metabolic rate is consonant with their mass. However, it has been found that RMRs run in families and those with lower RMRs tend to gain weight [14]. It is thought that genetic factors may work through subtle changes in RMR, thermogenesis or nutrient partitioning (the metabolic fuels that are utilized) because only minor changes in any of these processes may produce weight changes over years. Though changes in hormones are often blamed for weight gain, in actual fact such hormonal changes are minor causes of major weight gain (> 10 kg) [14, 15]. Other causes of weight gain include fluid retention, with cardiac or renal failure for instance.
The mechanisms of weight gain that relate to the use of psychotropic medication can be conveniently considered as central and peripheral. See Figure 1.
Factors important in maintaining weight and the effects of psychotropic medications. MAOIs, monoamine oxidase inhibitors; SSRIs, selective serotonin reuptake inhibitors; TCAs, tricyclic antidepressants. ⊕, facilitation; ⊖, inhibition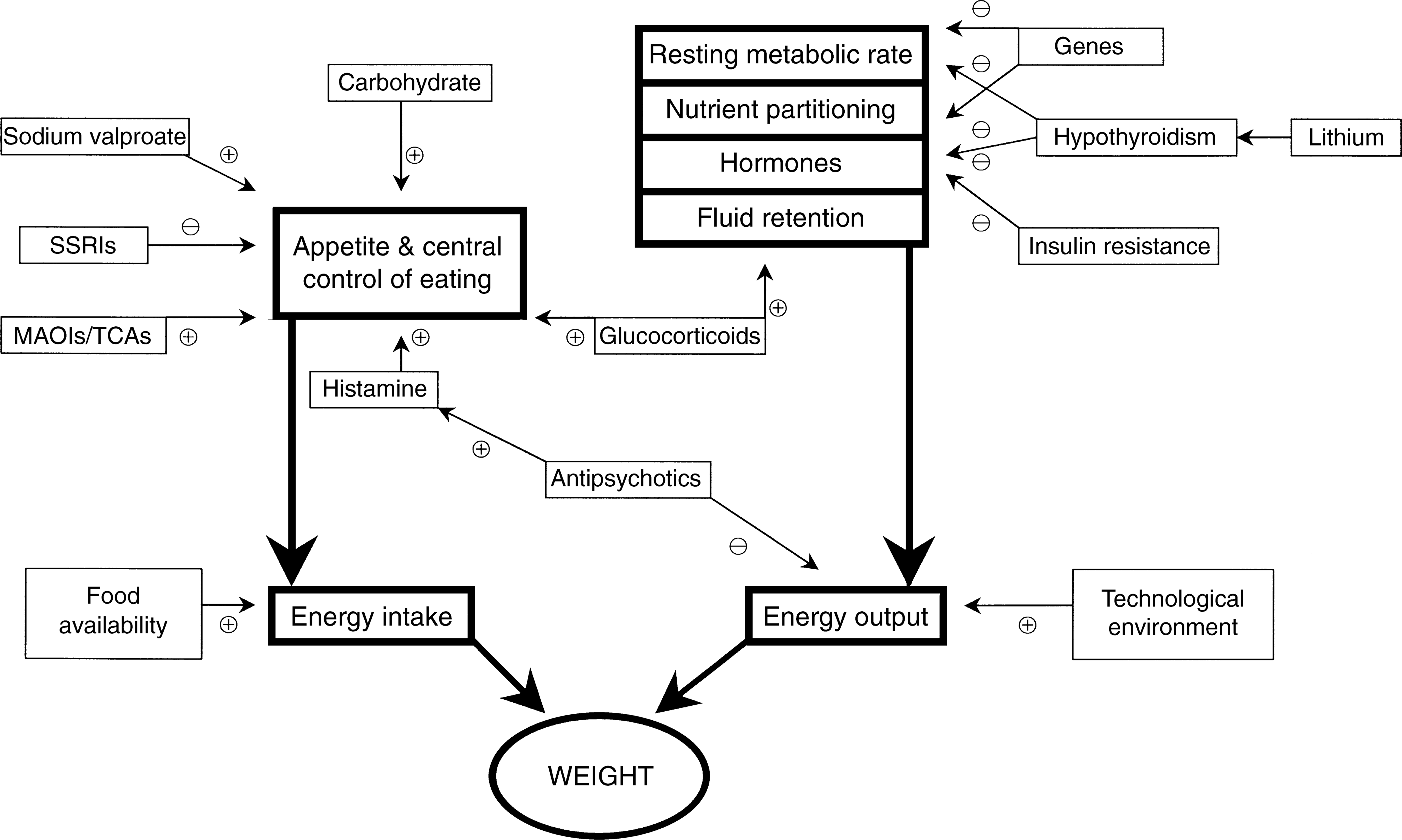
Central
Facilitation of serotonergic neurotransmission has been shown to reduce food intake and produce weight loss [16–18] whereas its blockade increases food consumption and causes weight gain [19–21]. It is unclear, however, which particular serotonin receptors are responsible for these changes though it is likely that both 5-HT2A and 5-HT2C receptors play a significant role [22]. Clinically, psychotropic medications which interact with these receptors influence appetite and produce weight change.
Histamine receptor H1 blockade is also associated with weight gain as is evidenced by the antihistamine astemizole [23]. Many psychotropic drugs have a strong affinity for the histamine H1 receptor, and this probably accounts for their effect on weight [24]. In practice, it is often observed that certain patients are more prone to weight gain and that this is perhaps familial or genetic. Almost 200 genes have been implicated in weight gain or regulation and obesity and these may influence weight by effects on basal metabolic rate, diet-induced thermogenesis, lipid oxidation or by altering habitual activity. For example, families with lower resting metabolic rates tend to gain weight more easily [14], while those with polymorphisms of uncoupling protein 2 (UPC2) find it more difficult to lose [25]. Genes may also alter the effect of regulatory peptides (for example those individuals who are unable to produce leptin [26, 27]).
Peripheral
Some familial syndromes such as insulin resistance and ‘syndrome X’ (characterized by insulin resistance, dyslipidaemia, glucose intolerance, central obesity, hypertension and atherosclerosis) increase the risk of gaining weight and developing associated illnesses [13, 28]. Similarly, insulin treatment and control of diabetes can produce weight gain. The exact mechanism of the gain is unknown.
Lifestyle factors such as diet and activity level are obviously important. While these may be influenced by genotype, environment also exerts a significant effect. It is too easy to be sedentary and too difficult to choose the right amount and type of food from the abundance available.
There are a number of diseases, which can be associated with, or produce obesity. For instance thyroid hormones alter the body's basal metabolic rate and some psychotropic medications, particularly mood stabilizers, can produce thyroid dysfunction [29, 30]. The amount of weight gain with hypothyroidism tends to be of the order of 5–10 kg. Hypothyroidism is a common late side-effect of lithium therapy [31], however, much of the weight gain associated with lithium treatment has been shown to be independent of changes in thyroid function, implicating instead other mechanisms in the development of obesity [32].
Treatment with glucocorticoids increases weight. Both total energy intake and expenditure are increased [33]. There is also a redistribution of body fat. Beta-blockers, used for the treatment of hypertension or cardiac disease may reduce activity substantially as may the existence of osteoarthritis. In pregnancy, or states with elevated prolactin, there is an increase in body fatness and this may predispose to later weight or fat gain.
Antidepressants
For more than three decades, the tricyclic antidepressants (TCAs) have been associated with excessive weight gain [34]. Studies have reported a wide range of weight change with TCAs, from a loss of 0.4 kg a month to a gain of 4.1 kg, with the absolute amount being related to the duration of therapy and dose of antidepressant used. Significant weight gain is a problem associated in particular with the tertiary amine TCAs amitriptyline and imipramine. It is interesting to note that long-term studies report less weight gain than would be anticipated [35, 36]. The rate of weight gain also varies considerably, with some patients gaining up to 20 kg within 6 months of treatment [34], and others actually losing weight. Clinically, TCA-induced weight gain is an important issue for depressed patients with up to 50% of those discontinuing treatment because of it [34]. Furthermore, after stopping TCAs many patients experience significant weight loss vindicating their noncompliance [37].
Conventional monoamine oxidase inhibitors are also associated with weight gain, in particular phenelzine [38]. However, this seems to be less of a problem than with the TCAs. The selective reversible monoamine oxidase inhibitor, moclobemide, does not appear to have any significant effect on body weight [39, 40].
As mentioned earlier, drugs that enhance serotonin neurotransmission generally produce weight loss. In keeping with this, the selective serotonin reuptake inhibitors (SSRIs) have not been shown to increase body weight, and in some cases may in fact result in weight loss. This is thought to be due to a decreased appetite [34, 35]. However, the weight loss associated with the acute phase of SSRI treatment may not be seen with maintenance treatment, during which up to one-third of patients can instead gain a small amount of weight. This is most likely to be occur with the use of paroxetine, which is thought to enhance appetite, but has also been noted with fluoxetine and citalopram [41, 42].
Lithium and other mood stabilizers
More than half the patients on long-term lithium therapy gain significant weight, and a considerable number develop obesity [43]. The average weight gain over a decade is approximately 10 kg, but others gain much more. Overall, almost one-quarter of patients maintained on lithium become clinically obese [36, 44]. Lithium-associated weight gain is dose-dependent and unlikely to occur with plasma concentrations less than 0.8 mmol/L [39], and nowadays, most patients are maintained on doses that result in plasma concentrations of about 0.6 mmol/L. However, to achieve a therapeutic response it is often necessary to maintain higher plasma levels; despite clinical benefit, patients frequently discontinue lithium because of such weight gain [45]. This usually occurs early in the course of treatment, and it is of note that weight gain is more likely in women than in men and those who are already overweight [46].
Lithium is thought to promote weight gain by altering the metabolism of carbohydrate and fats, enhancing appetite, increasing fluid intake and producing endocrine changes including hypothyroidism. It seems likely that lithium also has an effect on body weight independent of these factors, however, the exact mechanism has not yet been elucidated [36]. Lithium, probably the most effective mood stabilizer in the treatment of bipolar disorder, is no longer the most widely prescribed and a number of other compounds are now in common usage when lithium fails or is withdrawn because of adverse effects or non-compliance [47]. Sodium valproate and carbamazepine are routinely used in the treatment of bipolar affective disorder, and some more recently marketed anticonvulsants, such as gabapentin, lamotrigine and topiramate, are currently being evaluated as antimanic agents and mood stabilizers. As with lithium, the most common reason patients discontinue valproate is because of weight gain, with more than 50% accumulating between 10 and 12 kg, and a few much more [48, 49]. Some studies report considerable weight gain in more than half of those taking sodium valproate long-term. However, unlike lithium, sodium valproate associated weight gain does not seem to relate to gender, pretreatment body weight or the dose of the drug [50, 51]. Instead, it seems to be associated principally with the duration of treatment, with the proportion of patients experiencing weight gain increasing markedly after 2 years [52]. Weight gain occurs to a much lesser extent with carbamazepine than with lithium or sodium valproate, with one in three patients gaining 5 kg or more [34, 53, 54]. Although gabapentin also causes weight gain, the proportion of patients affected varies considerably [55, 56]. Clinically, weight gain does not seem to be an issue with lamotrigine. With topiramate, there is evidence that it may in fact reduce weight and it is often used clinically in the USA in combination with other mood stabilizers for this very purpose [57]. The weightreducing mechanism of action of topiramate is unclear. In animal studies changes in food intake, energy expenditure, or body composition have been described. If this effect is borne out in controlled trials, then this may be of considerable benefit to the many bipolar patients who face the dilemma of mood stability at the expense of weight gain.
Neuroleptics
Antipsychotic drugs, both old and new, often have to be taken for many months or years. Weight gain associated with their use is a considerable clinical impediment to compliance [58]. The atypical antipsychotics have gained favour because of their better side-effect profile in terms of extrapyramidal side-effects; however, some, in particular clozapine and olanzapine, can produce marked weight gain [59].
Clozapine is reported to produce a 10% increase in body weight in the first 6 months and double this when used long-term for several years [60]. Such marked and significant weight gain is not seen with the older neuroleptics such as haloperidol [61], but it is a problem with olanzapine, which produces comparable weight gain to clozapine in 33% of patients on long-term treatment [62].
Weight gain is also associated with the other atypical antipsychotics such as risperidone, sertindole, ziprasidone and quetiapine, although it is not as marked as that with clozapine and olanzapine [63]. Among the typical antipsychotics, chlorpromazine and thioridazine are associated with the greatest weight gain. For chlorpromazine, at least, this is likely to be dose-dependent [24, 64]. The exact mechanism by which the atypical antipsychotics produce weight gain is as yet unclear, but it is likely that 5-HT2c and H1 receptor interactions play an important role. It is therefore interesting to note that both clozapine and olanzapine have significantly greater affinity for these receptors than other atypical or typical antipsychotics [65, 66].
Preventing weight gain
After reaching adulthood, a weight gain of 10 kg or more is associated with increased middle-age mortality [67]. As patients become overweight and progressively develop mild, moderate and then severe obesity their risk of developing many diseases increases markedly [3]. Many psychiatric patients smoke, which also increases the risk of developing weight-related diseases [12]. Clinically, weight gain in psychiatric patients occurs for a variety of reasons. Psychiatric illness is often associated with changes of sleep, appetite and daily habit. Those patients who are employed may stop work. Many are hospitalized and restricted in activity, both by admission to hospital and by pharmacotherapy.
It is also worthwhile noting that the issue of weight gain is mainly pertinent to those in whom treatment is effective. Furthermore, it is not inevitable and for many psychiatric patients weight gain is minimal.
For those patients in whom weight gain occurs, practical means of managing the problem need to be instituted as soon as the problem is identified. In psychiatric disorders and treatment regimens where weight gain is clearly anticipated, the emphasis should perhaps be on prevention. Patients can be advised at the outset of suitable weight-limiting measures such as changes to their diet and the need for adequate levels of physical exercise [68]. Educating patients about adverse effects is essential and acknowledging the potential problem is likely to encourage compliance and enhance rapport. The likelihood of weight gain, however, needs to be balanced against the need for treatment. In cases where weight gain is related to drug dose the latter can be tailored so as to maintain therapeutic effect, but minimize weight gain. If this is not applicable (or not possible) then the agent can be substituted with another suitable psychotropic less likely to increase weight. In conjunction with this, the patient can be involved in a suitable weight loss program. It is important therefore, before embarking on a treatment plan, to gauge the patient's motivation, their cognitive capacity and their medication needs. A longterm treatment plan should articulate the necessary changes in diet and physical activity and include regular means of monitoring changes in weight. In patients who are already overweight or obese, the decision to lose weight should be made jointly. If the patient is reluctant to agree, then the aim can be altered to the prevention of further weight gain. Changes in lifestyle, although difficult to institute, can be very effective and should be tried, initially, for a period of at least 6 months before considering pharmacotherapy.
The majority of successful weight loss occurs because of reduced caloric intake. Excluding dietary fat alone will be unrewarding unless total caloric intake falls. Sustained physical activity is a good predictor of successful weight maintenance. It is an important component of weight loss programmes as it builds body mass, increases energy expenditure and decreases the chances of coronary-related illness. Successful weight loss also requires emotional and moral support, which can be provided via regular contact with relevant health-care professionals. However, medical supervision may also be necessary and various strategies addressing behaviour, including self-monitoring, stress management and cognitive restructuring can be used [69]. If, after 6 months, overweight or obese patients fail to lose any weight then pharmacotherapy may be considered. A variety of drugs are available with differing mechanisms of action. Sibutramine, an effective agent, is a derivative of the SSRIs that lacks any antidepressant effect. It is a serotonin and noradrenaline reuptake inhibitor that acts centrally to reduce appetite and energy intake and produce a ‘thermogenic effect’. It should not be used in patients with hypertension or heart disease [70]. Orlistat, a lipase inhibitor, blocks the gastrointestinal absorption of fat and is effective in combination with controlled dieting [71]. However, these treatments are to be used as a last resort and even then should be used in conjunction with lifestyle and behavioural methods. In severe cases surgical intervention is sometimes needed and energy intake is limited by gastric stapling.
Psychotropic medication-associated weight gain is generally easy to monitor and often predictable. It is therefore imperative that it not be ignored or overlooked, particularly as weight gain and obesity are associated with considerable health risks and even moderate weight loss enhances treatment compliance. A loss of 10% of body weight reduces the blood pressure of patients with hypertension, lowers the cholesterol levels in those with hyperlipidaemia and improves the control of blood sugar in those with NIDDM [72]. Weight gain in patients with psychiatric disorders is therefore an issue that requires awareness from the outset, close monitoring and, when necessary, appropriate clinical intervention. If this is done, then it is likely that it can be successfully managed in the majority of cases.
Footnotes
Acknowledgements
We thank Kerrie Eyers, Dusan Hadzi-Pavlovic and other members of the Mood Disorders Unit for their assistance.
