Abstract
Objective:
This study investigates (1) the proportion of people with psychosis who are on long-acting injectable antipsychotics; (2) the difference in the demographic, clinical, substance use and adverse drug reaction profiles of people taking long-acting injectables compared to oral antipsychotics; and (3) the differences in the same profiles of those on first-generation antipsychotic versus second-generation antipsychotic long-acting injectables.
Methods:
Data were collected as part of the Survey of High Impact Psychosis. For this study, participants with diagnoses of schizophrenia or schizoaffective disorder who were on any antipsychotic medication were included (
Results:
Nearly a third (31.5%) of people with psychosis were on long-acting injectables, of whom 49.7% were on first-generation antipsychotic long-acting injectables and 47.9% were on second-generation antipsychotic long-acting injectables. This contrasts with oral antipsychotics where there was a higher utilisation of second-generation antipsychotics (86.3%). Of note, compared to those on the oral formulation, people on long-acting injectables were almost four times more likely to be under a community treatment order. Furthermore, people on long-acting injectables were more likely to have a longer duration of illness, reduced degree of insight, increased cognitive impairment as well as poor personal and social functioning. They also reported more adverse drug reactions. Compared to those on first-generation antipsychotic long-acting injectables, people on SGA long-acting injectables were younger and had had a shorter duration of illness. They were also more likely to experience dizziness and increased weight, but less likely to experience muscle stiffness or tenseness.
Conclusion:
Long-acting injectable use in Australia is associated with higher rates of community treatment order use, as well as poorer insight, personal and social performance, and greater cognitive impairment. While long-acting injectables may have the potential to improve the prognosis of people with psychosis, a better understanding of the choices behind the utilisation of long-acting injectable treatment in Australia is urgently needed.
Introduction
Antipsychotic medications are the mainstay of treatment for schizophrenia (Galletly et al., 2016). Medication non-adherence can lead to relapse and functional decline (Novick et al., 2010). Nevertheless, antipsychotic non-adherence rates remain significant, ranging from 20% to 70% (Lacro et al., 2002; Novick et al., 2010). Long-acting injectable antipsychotics (LAIs) offer several advantages over oral antipsychotics, including improved adherence, reduced hospitalisation rates (Kishimoto et al., 2013; Taipale et al., 2018a) and reduced mortality (Taipale et al., 2018b).
First-generation antipsychotics (FGA) LAIs were introduced in the late 1960s (Brissos et al., 2014). By 1997, 25.6% of participants in the Low Prevalence (Psychotic) Disorders Study in Australia were on FGA LAIs (Jablensky et al., 2000). In the 2000s, second-generation antipsychotics (SGAs) were reformulated as LAIs and began to be more widely used. The 2010 Survey of High Impact Psychosis (SHIP) occurred on the cusp of the shift from FGA LAI to SGA LAI prescribing in Australia (Morgan et al., 2012). It provides a unique opportunity to explore aspects of experiences and characteristics of people using either FGA or SGA LAIs in routine clinical settings.
Using the data from the SHIP, we explored the following questions:
What proportion of people with schizophrenia or schizoaffective disorder are on LAIs?
Are there differences in the demographic, clinical, substance use and adverse drug reaction (ADR) profiles of people taking LAIs compared to oral antipsychotic medications?
Are there differences in the demographic, clinical, substance use and ADR profiles between those on FGA and SGA LAIs?
Methods
The SHIP was conducted within seven catchment sites across five Australian states, covering a population of some 1.5 million people aged 18–64 years, approximately 10% of the Australian population in this age range (Morgan et al., 2012). A two-phase design was followed. In Phase 1, screening for psychosis occurred in public specialised inpatient and community mental health services and in non-government organisations supporting people with mental illness in the census month (March 2010). A psychosis screener was used to identify individuals likely to meet criteria for formal diagnosis (Jablensky et al., 2000; Morgan et al., 2012). Administrative records were scanned to identify people with a recorded diagnosis of psychosis and in contact with public specialised mental health services in the 11 months prior to census but not in the census month. A total of 7955 people screened positive for psychosis and met eligibility criteria. In Phase 2, 1825 people who were screened positive were randomly selected, stratified by catchment site and age group (18–34 years and 35–64 years) and interviewed. The research protocol for the study was approved by all relevant institutional ethics committees at each catchment site and all participants gave written informed consent.
Demographic characteristics
Age and sex were determined by self-report. Country of birth was based on the question ‘What country were you born in?’ and responses were dichotomised into either born in Australia or not born in Australia. Marital status was divided into (1) separated, divorced or widowed; (2) single, never married; or (3) partnered (married or de facto). Post-school qualification was dichotomised into yes or no. Employment status was determined according to whether or not the participant was in any employment in the 12 months before the interview. Homelessness over the past 12 months was self-reported and included primary, secondary and tertiary homelessness. Life satisfaction was categorised into the following groups: (1) Mostly dissatisfied or worse (Terrible, Unhappy and Mostly dissatisfied); (2) Mixed (Mixed); and (3) Mostly satisfied or better (Mostly satisfied, Very pleased and Delighted). The Personal and Social Performance (PSP) Scale was used to estimate the personal and social functioning over the last 12 months (Morosini et al., 2000). The ratings were categorised into the following groups: (1) absence of disability or only mild difficulties, (2) varying degrees of disability and (3) poor functioning.
Clinical characteristics
For clinical variables examined, the age of onset for psychosis was determined by participants’ response to a series of questions including: ‘When did you first experience mental health problems?’, ‘When were you first admitted to hospital with a mental health problem?’ and ‘When did others first say that they thought you had a psychiatric problem?’. The duration of illness was derived from the age of onset. The type of psychotic disorder was determined according to Diagnostic Interview for Psychoses - Diagnostic Module (DIP-DM) (Castle et al., 2006). It uses prompts adapted from the Schedules for Clinical Assessment in Neuropsychiatry (SCAN) (World Health Organization, Division of Mental Health, 1994) to elicit signs and symptoms, then applies the Operational Criteria Checklist for Psychosis (OPCRIT) (McGuffin et al., 1991) algorithm embedded into a software package to generate diagnoses according to several classification systems including the Tenth Revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10) (World Health Organization, 1992). For this study, the diagnoses were then divided into either schizophrenia or schizoaffective disorder. The course of illness was assessed and rated by the interviewers capturing the number of episodes of mental illness that a person had experienced and the degree of recovery after each episode (Castle et al., 2006). The course was dichotomised into non-chronic (single episode with good recovery or multiple episodes with good or partial recover between) or chronic (continuous chronic illness with or without deterioration). The utilisation of community treatment order was categorised into (1) current, (2) not current but in last 12 months and (3) no. We examined binary responses (present or not present) for both lifetime and present state according to DIP-DM questionnaire regarding suicidal thought, hallucinations, delusions, subjective thought disorder, depressive symptoms and manic symptoms. Negative symptoms were assessed to be present if the interviewers rated presence of any of the following: restricted affect, poverty of speech, a diminished sense of purpose, social drive and interest. Insight was determined to be absent if the participant was unable to recognise that his or her experiences are abnormal, denied being currently or ever mentally unwell, or recognised that his or her experiences are abnormal but gave a delusional explanation. Two tests were used to assess cognitive symptoms. First, the National Adult Reading Test (NART-R) is a word-reading test widely used as an estimate of premorbid IQ (Schretlen et al., 2005). Based on summary NART-R scores, premorbid IQ was categorised into the following three levels: Below Average (>1 SD below sample mean), Average (within 1 SD of sample mean) and Above Average (>1 SD above sample mean). Likewise, the Digit-Symbol Coding task (DSCT) from the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) (Randolph et al., 1998), a test of speed of processing, was used to assess current cognitive ability. The lower the test score, the poorer a person’s speed-based performance. Based on summary DSCT scores, current cognitive ability was categorised into three levels: Below Average (>1 SD below sample mean), Average (within 1 SD of sample mean) and Above Average (>1 SD above sample mean).
Substance use characteristics
Participants were asked to self-report lifetime and present use of alcohol, cannabis and amphetamine. The responses were dichotomised into either any or no use. The Fagerström test for nicotine dependence was used to categorise smoking into very low/low, moderate or high/very high (Heatherton et al., 1991).
ADRs
Medication use was based on self-report with participants being asked to bring their medication or medication list to the interview (Waterreus et al., 2012). Only medications taken for at least the last 4 weeks were recorded. Participants were asked about a range of ADRs. We categorised them into the following groups: sedation (drowsy or sleepy during the day), Parkinsonian symptoms (hands, arms, or legs shaking or trembling; muscle stiff/tense; unsteady walking/standing; slowing down of movement; shuffling along), akathisia (inner restlessness; inability to stand still, desire to move legs, pacing up and down; inability to relax), tardive dyskinesia (unwanted tongue movement), anticholinergic side effects (dry mouth or mouth more watery than usual; dizziness; trouble with eyesight; constipation), hormonal side effects (change in interest in sex; difficulty achieving orgasm; period change; swollen/tender chest) and weight changes (increased weight; decreased weight).
Data analysis
This study included 1049 participants who met the criteria for ICD-10 diagnosis of schizophrenia or schizoaffective disorder and who reported taking any antipsychotic medication in the 4 weeks before the survey. We first dichotomised the participants into two groups: people on LAI antipsychotic medication (LAI group) and people who were on only oral antipsychotic medication (non-LAI group). First, we conducted a series of logistic regressions to examine demographic, clinical, substance use and ADR profiles associated with the two groups. We calculated the odds ratios (ORs) with 95% confidence intervals (CIs) adjusted for age, sex and duration of illness to estimate the likelihood of participants being in the LAI group. We then conducted the same series of analyses comparing those on SGA LAI versus FGA LAI. All analyses were done in R (version 4.0.0) (R Core Team, 2020) using packages tidyr (Wickham and Henry, 2020) and dplyr (Wickham et al., 2020).
Results
The pattern of medication use in people with SHIP is summarised in Supplementary Table 1. Of 1049 participants included in the study, 81.9% (
Demographic characteristics of people on LAI compared to people not on LAI.
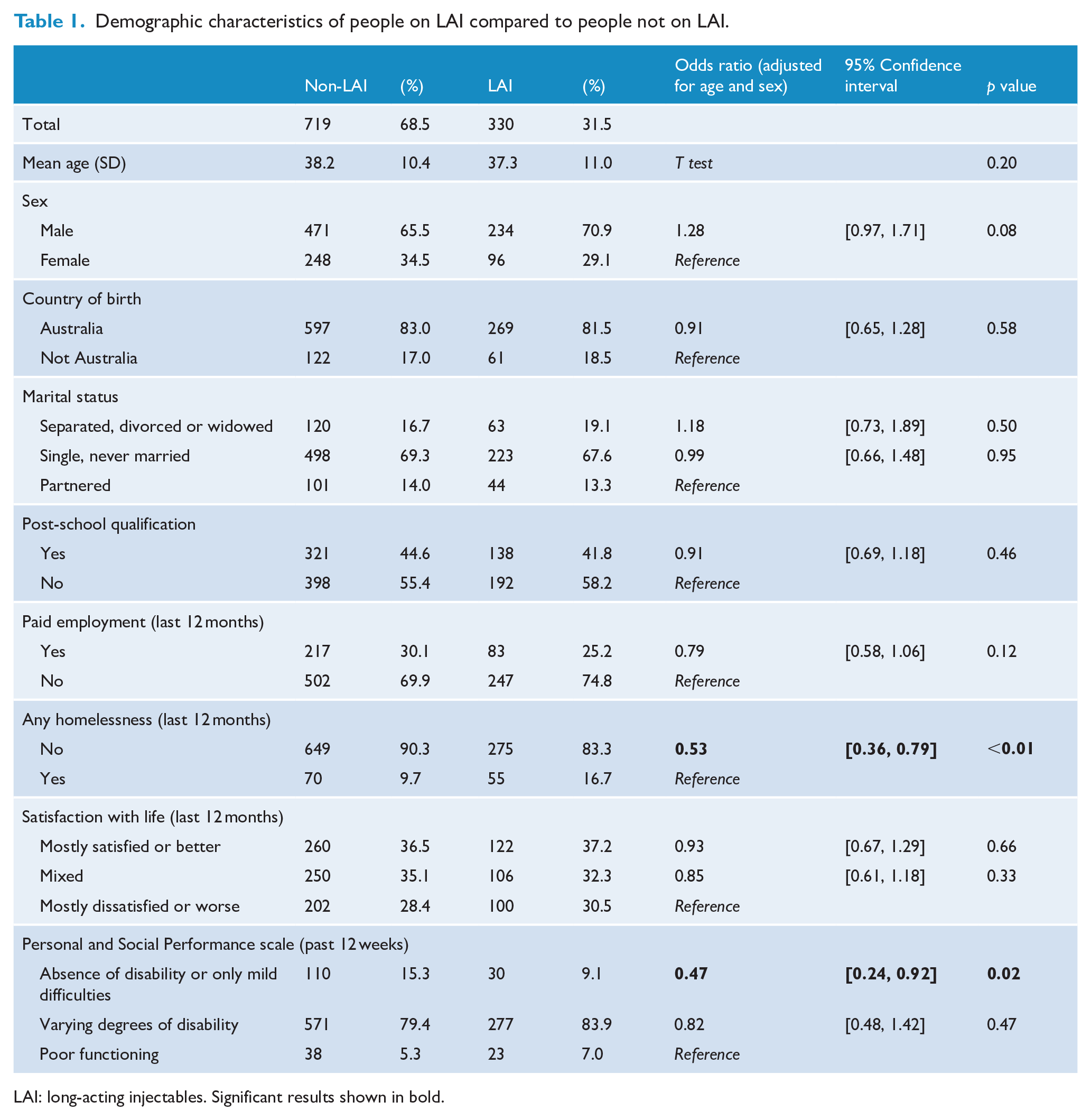
LAI: long-acting injectables. Significant results shown in bold.
Demographic characteristics
Demographic characteristics of people on LAIs compared to people not on LAIs are presented in Table 1. Out of 1049 participants, the majority (68.5%,
Clinical characteristics
Clinical characteristics of people on LAIs compared to people not on LAIs are summarised in Table 2. Compared to the non-LAI group, the LAI group were more likely to have a longer duration of illness (15.2 years vs 13.9 years,
Clinical characteristics of people on LAI compared to people not on LAI.
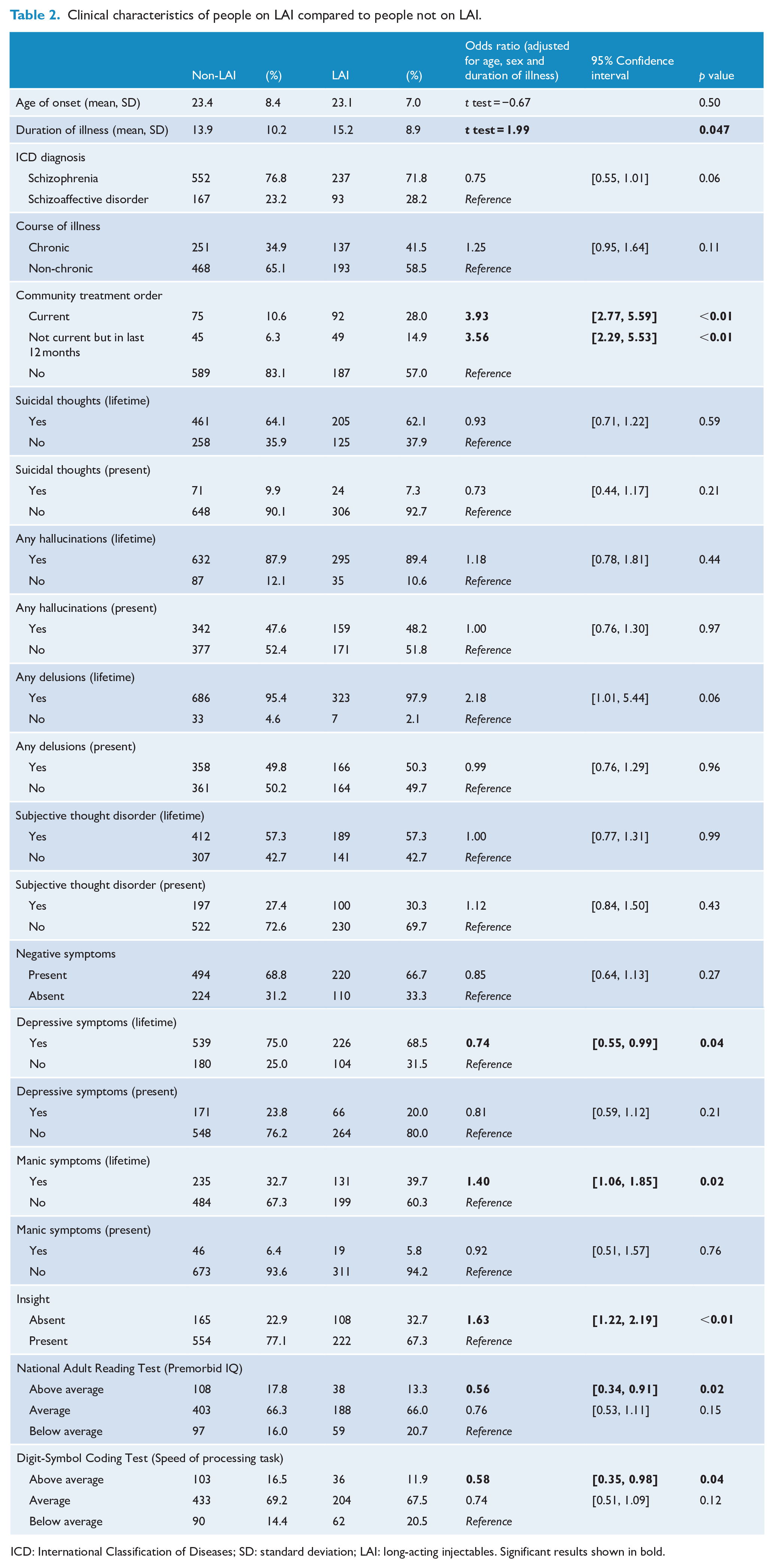
ICD: International Classification of Diseases; SD: standard deviation; LAI: long-acting injectables. Significant results shown in bold.
Substance use characteristics
Substance use characteristics of people on LAIs compared to people not on LAIs are shown in Table 3. Compared to the non-LAI group, the LAI group were more likely to have used cannabis in the past 12 months (OR = 1.79, 95% CI: [1.30, 2.48]), to have used amphetamine in lifetime (OR = 1.51, 95% CI: [1.10, 2.10]) and to have high/very high nicotine dependence (OR = 2.20, 95% CI: [1.66, 2.93]). There was no difference between the two groups in the following variables: alcohol use (both lifetime and past year), cannabis use (lifetime) and amphetamine use (past year).
Substance use characteristic of people on LAI compared to people not on LAI.
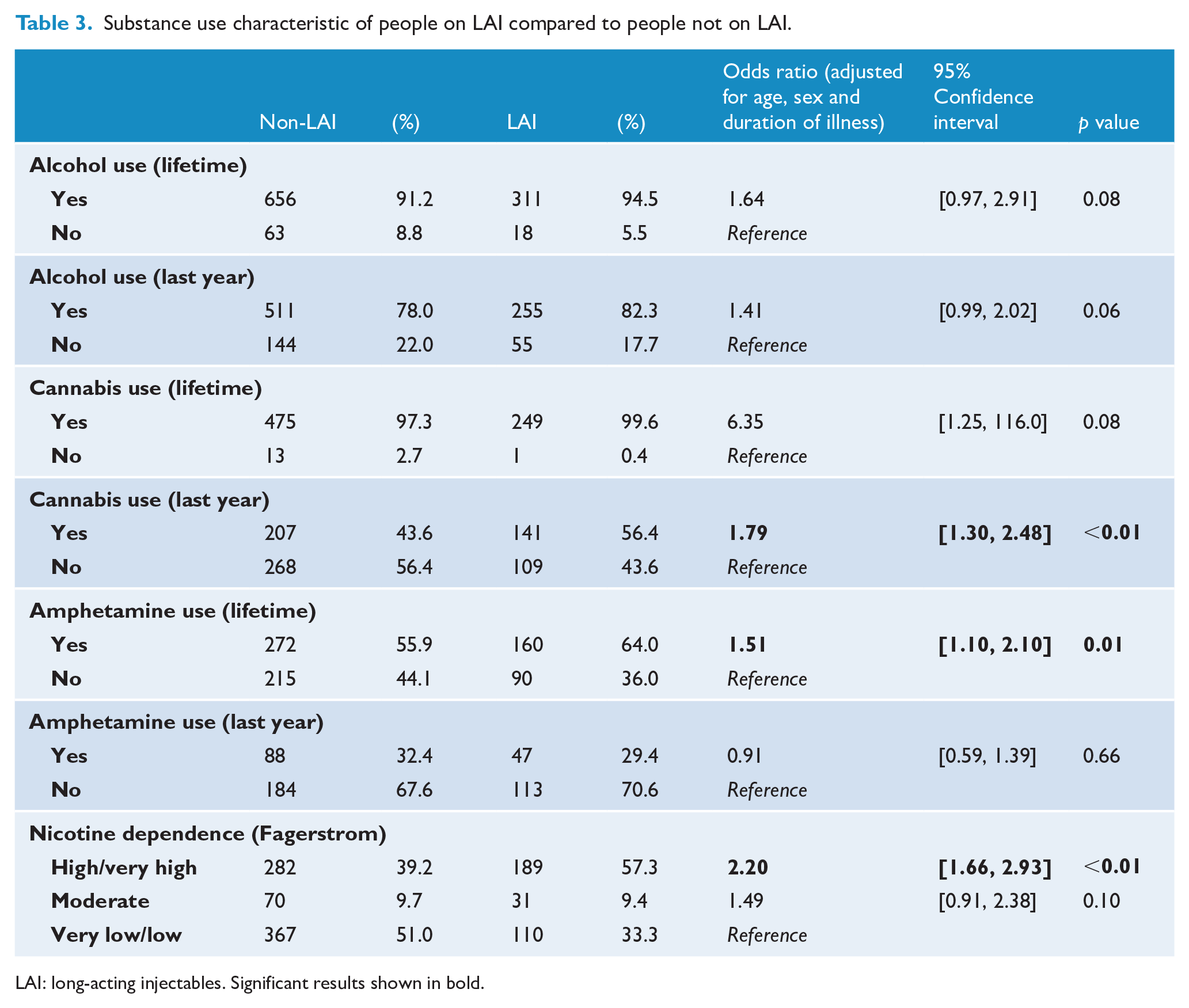
LAI: long-acting injectables. Significant results shown in bold.
ADRs
Table 4 presents the reported ADRs of people on LAIs compared to those who are not on LAIs. Compared to those in the non-LAI group, people in the LAI group were more likely to report muscle stiffness/tenseness (OR = 2.03, 95% CI: [1.50, 2.77]), inability to stand still, desire to move legs, pacing up and down (OR = 1.43, 95% CI: [1.05, 1.94]), unwanted tongue movement (OR = 1.99, 95% CI: [1.30, 3.06]) and trouble with eyesight (OR = 1.43, 95% CI: [1.03, 1.97]). There was no difference between the two groups in other reported medication side effects.
Reported adverse drug reactions with people on LAI compared to people not on LAI.
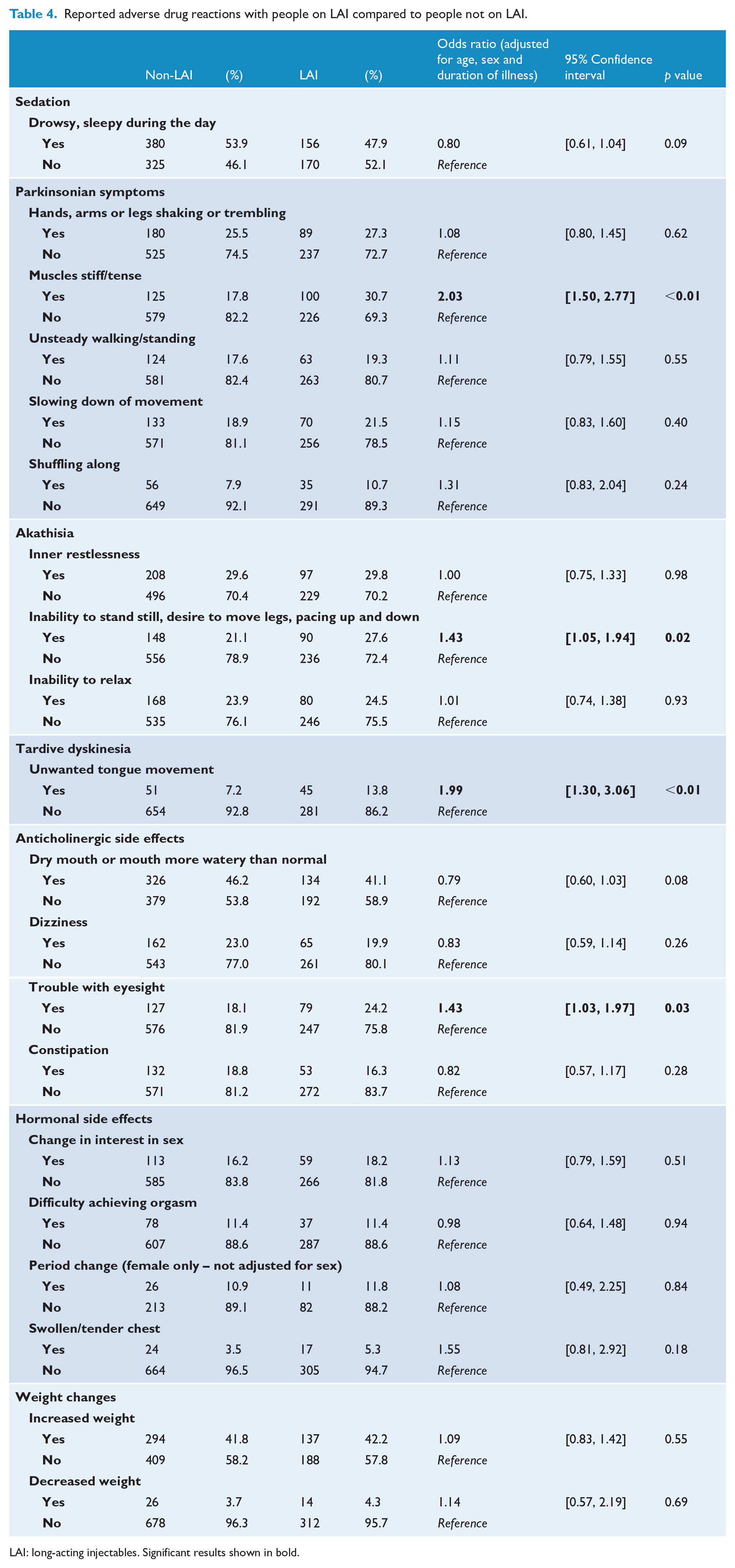
LAI: long-acting injectables. Significant results shown in bold.
Demographic characteristics among people on LAIs
Demographic characteristics of people on SGA LAIs compared to people on FGA LAIs are presented in Table 5. After removing eight participants who were on both SGA and FGA LAIs, 322 people were included in this section of analyses. Of 322 participants, there was almost an equal number of people on SGA LAIs (49.1%,
Demographic characteristics of people on SGA LAI compared to people who are on FGA LAI.
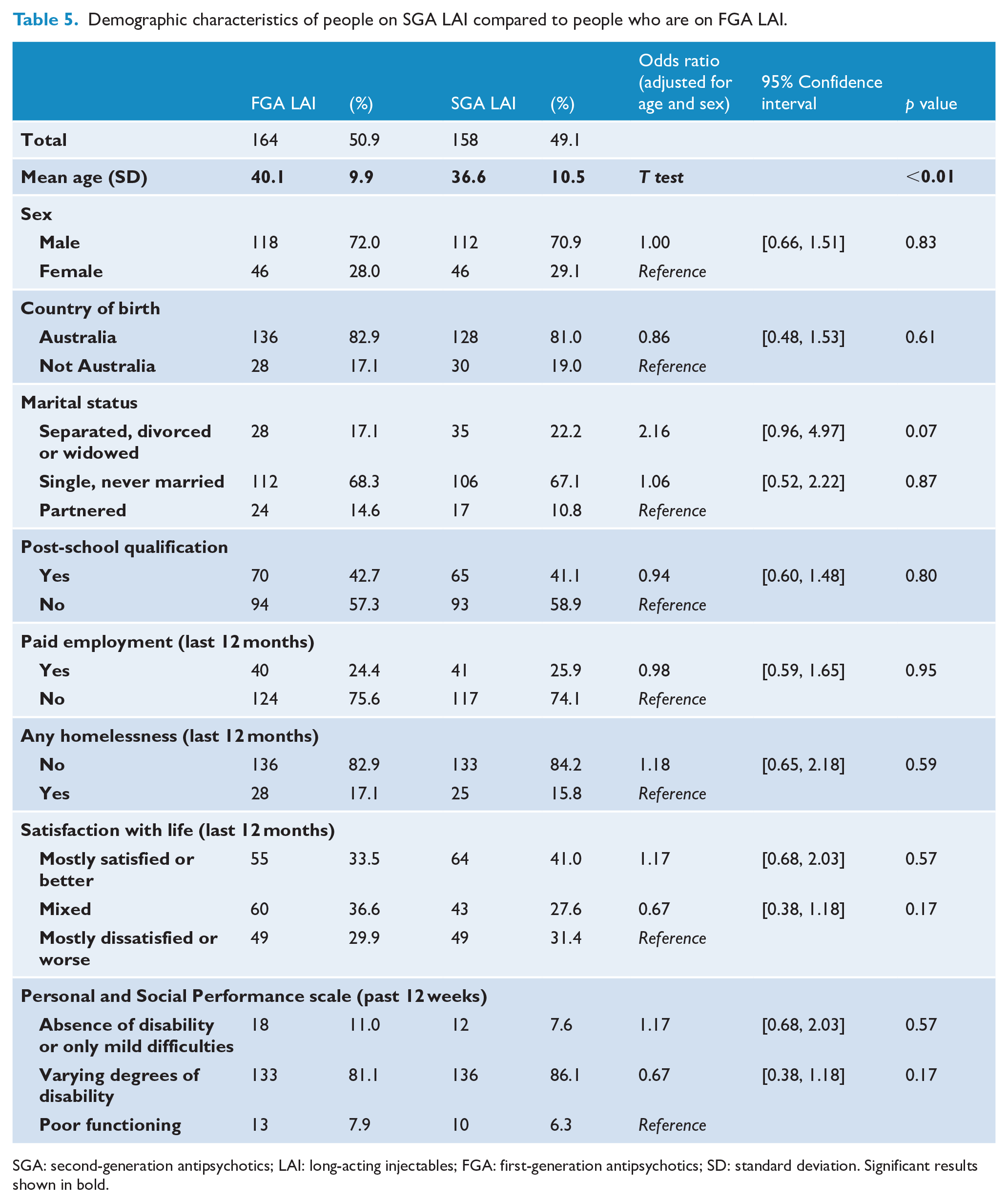
SGA: second-generation antipsychotics; LAI: long-acting injectables; FGA: first-generation antipsychotics; SD: standard deviation. Significant results shown in bold.
Clinical characteristics among people on LAIs
Clinical characteristics of people on SGA LAIs compared to people on FGA LAIs are summarised in Table 6. Compared to the FGA LAI group, the SGA LAI group were more likely to have a shorter duration of illness (12.7 years vs 17.5 years,
Clinical characteristics of people on SGA LAI compared to people who are on FGA LAI.
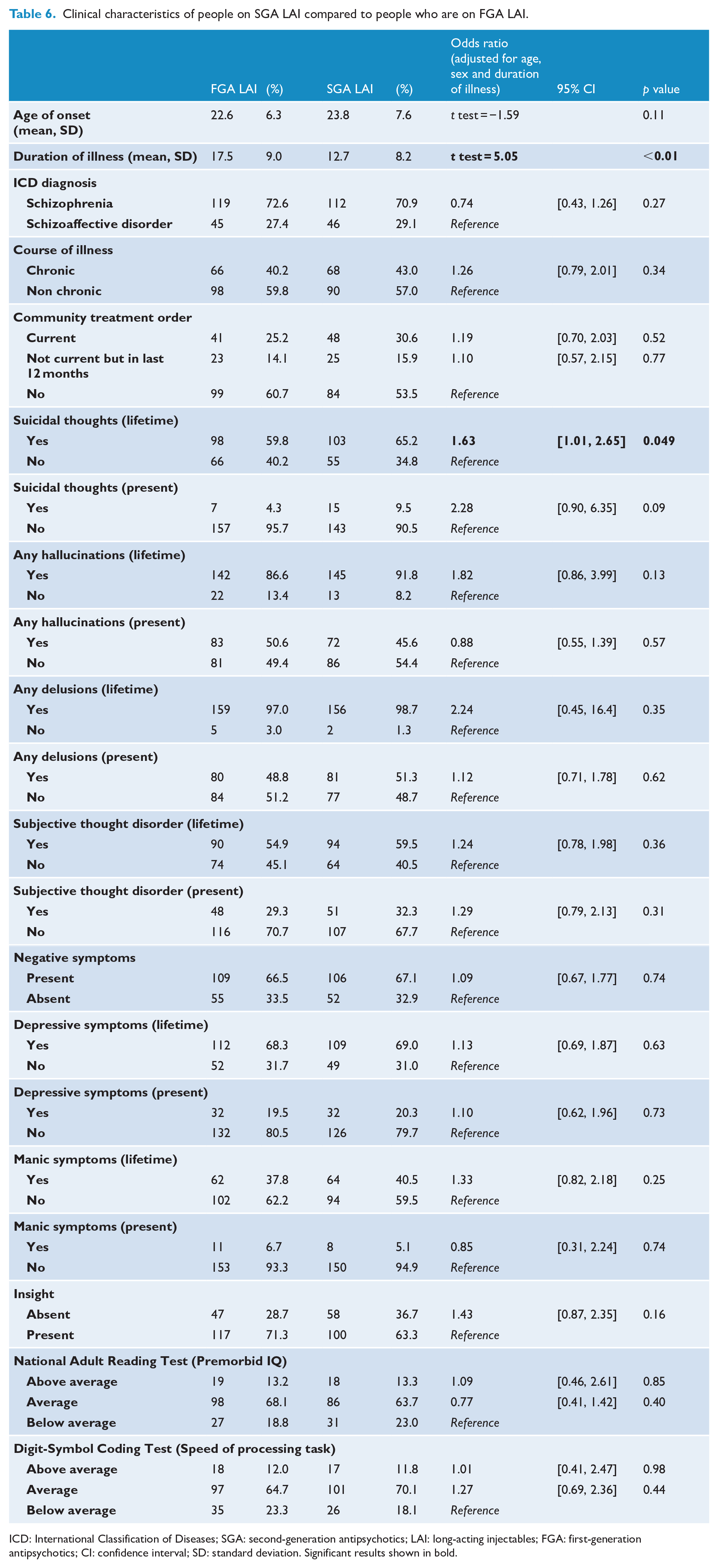
ICD: International Classification of Diseases; SGA: second-generation antipsychotics; LAI: long-acting injectables; FGA: first-generation antipsychotics; CI: confidence interval; SD: standard deviation. Significant results shown in bold.
Substance use characteristics among people on LAIs
Substance use characteristics of people on SGA LAIs compared to people on FGA LAIs are shown in Table 7. Compared to the FGA LAI group, the SGA LAI group were significantly less likely to have high/very high nicotine dependence (OR = 0.52, 95% CI: [0.31, 0.87]). There was no difference between the two groups in any other substance use characteristics including alcohol use (both lifetime and past year), cannabis use (both lifetime and past year) and amphetamine use (both lifetime and past year).
Substance use characteristic of people on SGA LAI compared to people who are on FGA LAI.
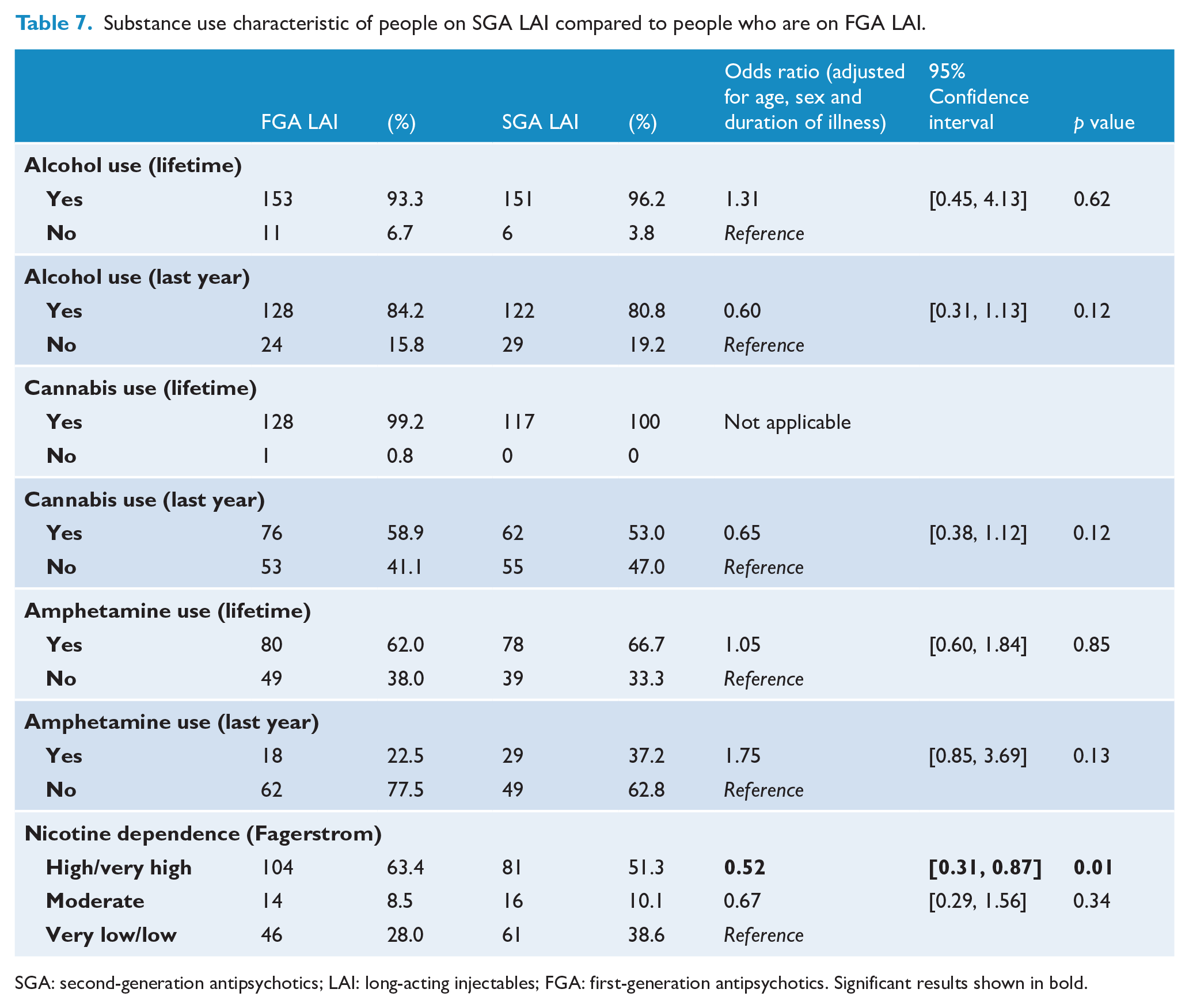
SGA: second-generation antipsychotics; LAI: long-acting injectables; FGA: first-generation antipsychotics. Significant results shown in bold.
ADRs among people on LAIs
Table 8 presents the reported ADRs of people on SGA LAIs compared to those who are on FGA LAIs. Compared to those in the FGA LAI group, people in the SGA LAI group were more likely to report dizziness (OR = 1.94, 95% CI: [1.08, 3.54]) and weight gain (OR = 1.76, 95% CI: [1.09, 2.84]), but less likely to report muscle stiffness/tenseness (OR = 0.55, 95% CI: [0.33, 0.91]). There was no difference between the two groups in other reported medication side effects.
Reported adverse drug reactions with people on SGA LAI compared to people who are on FGA LAI.
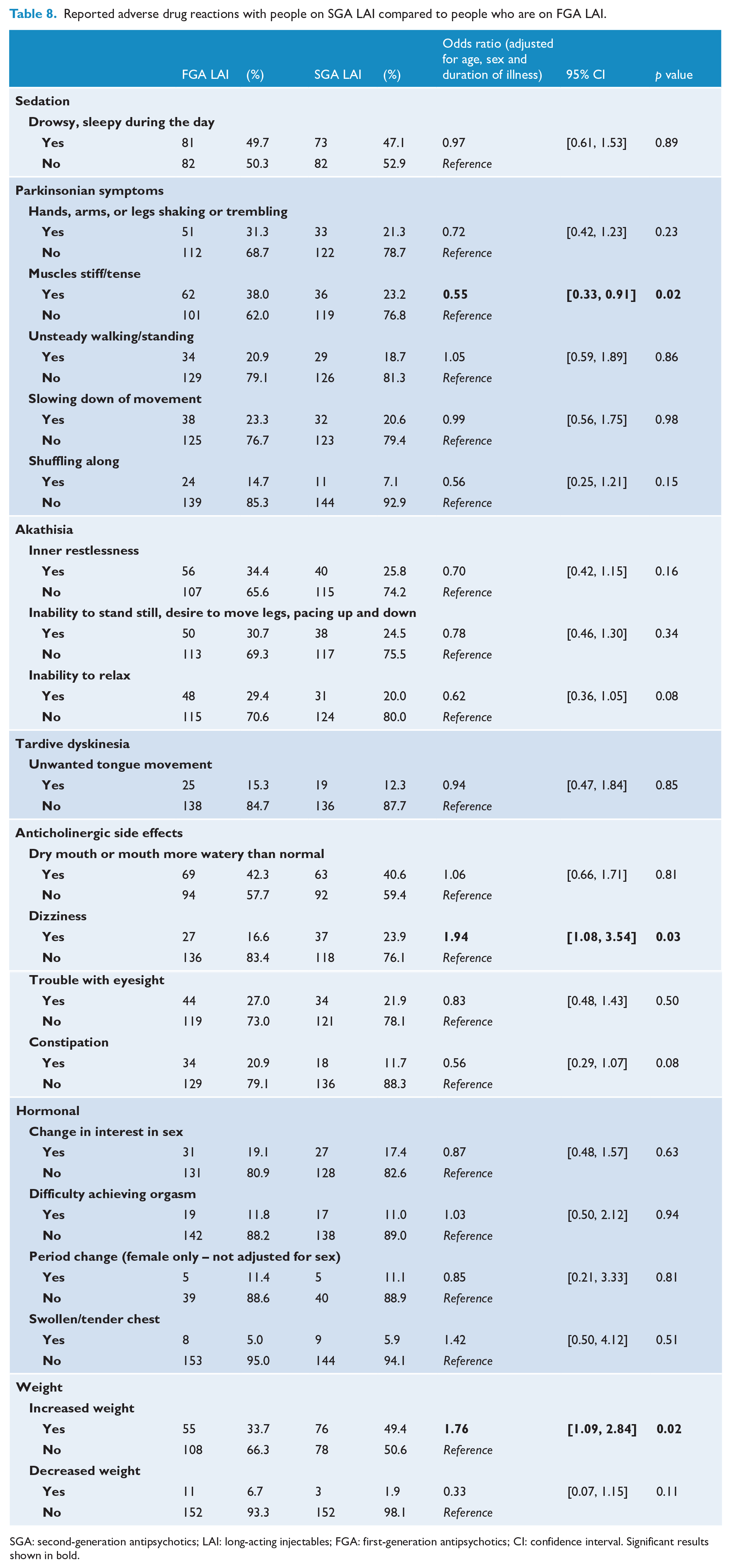
SGA: second-generation antipsychotics; LAI: long-acting injectables; FGA: first-generation antipsychotics; CI: confidence interval. Significant results shown in bold.
Discussion
In this study, using a large cross-sectional national dataset, we compared a wide range of variables between people with psychosis who were on LAI and those who were on oral antipsychotic medication. We also had the opportunity to further examine the differences between those on SGA LAI and FGA LAI. Compared to people on the oral formulation, those on LAI were more likely to be treated under a community treatment order and were also more likely to have a longer duration of illness, poorer personal and social performance, more limited insight, and greater cognitive impairment, as well as more reported side effects. We also found that people on SGA LAI were younger and had a shorter duration of illness than those on FGA LAI. In terms of ADRs, SGA LAIs were more likely to experience dizziness and increased weight, but less likely to experience muscle stiffness or tenseness. There were few other clinical differences between the FGA LAI group and the SGA LAI group.
What proportion of people with schizophrenia or schizoaffective disorder is on LAIs?
We found that 31.5% of people with psychosis were on LAI, of whom 49.7% were on FGA LAIs and 47.9% were on SGA LAI. A small proportion (2.4%) of people were on both. The proportion of people who were on LAIs had increased from the national survey 13 years earlier (25.6%) (Jablensky et al., 2000). While it is difficult to determine the precise rate of LAI use internationally, the recently reported rates of patients prescribed LAIs range from 10% to 50% (Brissos et al., 2014). For example, lower rates are reported in countries such as the United States of America and New Zealand, and higher rates are reported in countries such as Austria and the United Kingdom (Brissos et al., 2014). Thus, the estimate of approximately 31.5% lies somewhere in the middle of the international range. The current SHIP study also presented a unique strength in the timing of this survey collection. There was a balance between FGA LAI and SGA LAI use (50.9% vs 49.1%), allowing a robust comparison between the two groups. The proportion of people on SGA LAI in Australia has likely increased significantly over the past 10 years. For instance, a recent multi-site European study (Llorca et al., 2018) found that paliperidone LAI was the most commonly prescribed LAI with over a third (204 out of 572) of the participants recruited between 2013 and 2014 on it. At the time of the SHIP recruitment, none of the participants were on paliperidone LAI.
Are there differences in the demographic, clinical, substance use and ADR profiles of people taking LAIs compared to oral antipsychotic medications?
When compared to people on oral antipsychotic medication, people on LAIs were more likely to have a longer duration of illness. We also found that people on LAIs were more likely to lack insight and show more cognitive impairment compared to those on oral antipsychotic medication. In other words, as a group, they were probably more likely to lack the capacity to consent to treatment. Accordingly, we found that people on LAIs were much more likely to be under a community treatment order both currently (OR = 3.93, 95% CI: [2.77, 5.59]) and in the last 12 months (OR = 3.56, 95% CI: [2.29, 5.53]). This is consistent with previous studies. An Australian study conducted in Melbourne between 1998 and 2002 found that people under community treatment orders were more than three times as likely to be on LAI compared to voluntary patients (Lambert et al., 2009). Another study from Adelaide found that nearly 90% of people who were on community treatment orders in a community mental health service were on LAIs (Suetani et al., 2014). Similarly, a study from the United Kingdom found that, at the commencement of a community treatment order, people were significantly more likely to be on LAI than on oral antipsychotic medication (60.6% vs 39.4%) (Patel et al., 2013). We also found that people on LAIs were more likely to have used a range of substances including cannabis, amphetamine and nicotine. They also scored significantly lower on the PSP scale, indicating poor personal and social performance functioning. These differences may suggest increased clinical complexity of this particular patient cohort. Combined, we postulate that being on LAI may be a measure of treatment resistance for a sub-group of patients with psychosis, similar to those who are on clozapine (Siskind et al., 2017).
On the other hand, the reduced risk of homelessness among the LAI group probably reflects a practical consideration for LAI treatment. We suspect that it is more likely that people on LAIs are less likely to be homeless because it is challenging to administer LAIs on patients with no fixed aboard. In addition, we found that people on LAIs were more likely to have experienced lifetime manic symptoms but less likely to have experienced lifetime depressive symptoms – but these symptoms did not lead to a significant difference in the diagnosis between affective psychosis and non-affective psychosis in the two groups. We also found that people on LAIs had increased reporting of a range of ADRs (muscle stiffness, akathisia, tardive dyskinesia and anticholinergic side effects). We postulate that the increased reporting is likely to be due to (1) the difficulty associated with changing the LAI doses quickly to adjust for these side effects once they emerge and (2) increased compliance with the medication.
Are there differences in the demographic, clinical, substance use and ADRs between those on FGA and SGA LAIs?
The SGA LAI group in our study was significantly younger than the FGA LAI group. They also had a shorter duration of illness. This is likely due to the timing of the SHIP study (circa 2010). At the time, the SGA LAIs were still relatively new, and those who had been stable on LAIs for some time were much more likely to be on an FGA LAI than on an SGA LAI. Given the broader utilisation of SGA LAIs since then, the differences in the age and duration of illness may not have persisted. It is also interesting to note that SGA was widely utilised in the oral formulation at the time of the SHIP recruitment already (86.2%), but there was a delay in SGA LAIs being utilised more. Other than these factors, we found little clinical differences between the two groups. Consistent with the oral formulation, people on SGA LAIs were more likely to experience dizziness and increased weight, but less likely to experience muscle stiffness or tenseness (i.e. extrapyramidal side effect).
LAIs utilisation in people with psychosis
The current Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines (Galletly et al., 2016) acknowledge that LAI is an important treatment option for people with psychosis. The guidelines recommend the LAI treatment specifically for those patients who prefer LAI over oral formulation, or for the management of non-adherence. Given the high rates of non-adherence to antipsychotic medications and associated negative impact on the prognosis of people with psychosis (Leucht and Heres, 2006), the LAI treatment has a potential to improve the illness course significantly (Correll et al., 2016). There is an increasing call to utilise LAI earlier in the clinical course to improve outcomes for people with psychosis (Catts and O’Toole, 2016). Rather than waiting for intentional or non-intentional non-adherence to occur, LAIs may be a useful first-line treatment option that should be offered in those in early stages of psychosis (Lo et al., 2016). For instance, the Prevention of Relapse in Schizophrenia (PRELAPSE) trial, a cluster randomised clinical trial consisting of nearly 500 participants in the United States, recently found that LAI treatment (aripiprazole) in people with early-stage psychosis significantly delayed time to hospitalisation (Kane et al., 2020). There are considerations other than efficacy that need to be taken in determining the use of LAI such as patient choice or preference, self-determination and shared decision-making (Siskind and Dark, 2017), but the investigators of the PRELAPSE trial argue that, with adequate training, clinicians can communicate potential advantages of LAI over oral antipsychotic medication even among those with early-stage psychosis (Kane et al., 2020). It should be noted, however, that the PRELAPSE trial was funded by Lundbeck and Otsuka Pharmaceuticals, makers of aripiprazole LAI. In order to optimise the medication treatment for people living with psychosis, we need to further explore and examine why people – including patients, their families and clinicians – may prefer LAI (enablers) or avoid LAI (barriers). As an example, there is a common clinical perception that LAI treatment and treatment orders come together. The findings from our study support this notion. The use of LAIs under a community treatment order is often seen as a ‘clinical double-edged sword’. While it may convince some patients to continue LAI after the expiration of their treatment order (i.e. it works as a temporary ‘enabler’ of LAI use), the presence of the treatment order may also have patients associate LAI treatment with coercion, thus associating the negative feeling towards the treatment order with the LAI treatment, leading to discontinuation once the treatment order expires (i.e. it becomes a ‘barrier’ to LAI use) (Suetani et al., 2014). With more effective communication, clinicians could help reduce negative feelings towards LAI treatment for those treated under treatment orders. Despite the clinical relevance, the association between LAI use and treatment orders has not been sufficiently explored in Australasia (Kisely et al., 2020). Future research should explore in more detail how community treatment orders may impact on LAI treatment for our patients and clinicians, both in quantitative and qualitative manners. Finally, there is an accumulating evidence base to suggest that LAI treatment reduces morbidity and mortality among people with psychosis in the long term (Taipale et al., 2018b; Tiihonen et al., 2017). We need to further explore factors associated with appropriate LAI use (both from patient and clinical points of view) beyond non-adherence regardless of their treatment order status. Clinicians need to better communicate advantages and disadvantages of LAI use so that it becomes a genuine treatment choice rather than something that is forced under a treatment order. The principles of shared decision making may be useful in guiding our conversation about antipsychotic medication choices. The shared decision making model builds on long-standing ideas of patient-centred care and is central to the recovery model (Alguera-Lara et al., 2017). This approach aims to facilitate collaborative work between patients and care providers, by providing sufficient and appropriate information, reducing potential patient, clinician and systematic barriers, as well as utilising decision aids such as discrete choice experiments. A recent pilot study conducted in Melbourne, for instance, was able to explore the differences in decision-making process for antipsychotic medication – including attitudes to LAIs – among doctors and patients using a discrete choice experiment (Yeo et al., 2019). We believe that improved shared decision-making process regarding antipsychotic medication choices will foster patient autonomy and empowerment, thus improving their prognosis long term.
Limitations
A major strength of this study was that we were able to examine a wide range of variables in a large, nationally representative sample of people with psychosis. However, there are several limitations, and the interpretation of our findings requires caution. First, the main limitation of the study is the age of the data. Although there is no similar recent study, the current LAI utilisation pattern in Australia may be different compared to 2010. Of note, since the SHIP study was conducted, paliperidone LAI and aripiprazole LAI have become available and widely used in Australia. Although the ADRs of paliperidone are likely to be close to those of risperidone, other practical differences such as the more extended intervals (either 4 weekly or 12 weekly) between administrations and storage consideration (paliperidone does not need to be refrigerated) may have significant impacts on utility and experiences of SGA LAIs (Mathews et al., 2019). Also, the cross-sectional nature of the data prevented us from making any inferences about causation between LAI use and the examined variables. In addition, as the study was conducted in Australia, the findings may not be globally generalisable. Finally, even though the SHIP sample is representative of people receiving support from public mental health services and non-government organisations, it may not be representative of those in the private mental health services or those who have dropped out of treatment altogether.
Conclusion
There is a strong association with LAIs and community treatment orders among people with psychosis. Given its potential to significantly improve the prognosis of people with psychosis, we need to explore more nuanced factors (both enablers and barriers) associated with the utilisation of LAI treatment in Australia.
Supplemental Material
sj-docx-1-anp-10.1177_00048674211009602 – Supplemental material for Characteristics of people on long-acting injectable antipsychotics in Australia: Data from the 2010 National Survey of High Impact Psychosis
Supplemental material, sj-docx-1-anp-10.1177_00048674211009602 for Characteristics of people on long-acting injectable antipsychotics in Australia: Data from the 2010 National Survey of High Impact Psychosis by Shuichi Suetani, Dan Siskind, Andrea Phillipou, Anna Waterreus, Vera A Morgan and David Castle in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
This publication is based on data collected in the framework of the 2010 Australian National Survey of High Impact Psychosis. The members of the Survey of High Impact Psychosis Study Group at the time of the survey were V. Morgan (National Project Director), A. Jablensky (Chief Scientific Advisor), A. Waterreus (National Project Coordinator), R. Bush, V. Carr, D. Castle, M. Cohen, C. Galletly, C. Harvey, B. Hocking, A. Mackinnon, P. McGorry, J. McGrath, A. Neil, S. Saw and H. Stain. Ethics approvals for the study were obtained from relevant institutional human research ethics committees. This report acknowledges, with thanks, the hundreds of mental health professionals who participated in the preparation and conduct of the survey and the many Australians with psychotic disorders who gave their time and whose responses form the basis of this publication.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: S.S. has received Honoraria from Seqirus. D.C. has received grant monies for research from Eli Lilly, Janssen Cilag, Roche, Allergen, Bristol-Myers Squibb, Pfizer, Lundbeck, Astra Zeneca and Hospira; Travel Support and Honoraria for Talks and Consultancy from Eli Lilly, Bristol-Myers Squibb, Astra Zeneca, Lundbeck, Janssen Cilag, Pfizer, Organon, Sanofi-Aventis, Wyeth, Hospira, Servier and Seqirus; and is a current Advisory Board Member for Lu AA21004: Lundbeck; Varenicline: Pfizer; Asenapine: Lundbeck; Aripiprazole LAI: Lundbeck; Lisdexamfetamine: Shire; Lurasidone: Servier; Brexpiprazole: Lundbeck; Treatment Resistant Depression: LivaNova; Cariprazine: Seqirus. He is founder of the Optimal Health Program, currently operating as Optimal Wellness, and is part owner of Clarity Healthcare. He is on the board of The Mental Health Foundation of Australia. He does not knowingly have stocks or shares in any pharmaceutical company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was funded by the Australian Government Department of Health and Ageing.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
