Abstract
This article is a detailed response to the criticisms levelled by the authors of an accompanying viewpoint, which claims that the positioning of repetitive transcranial magnetic stimulation (rTMS) in the 2020 Royal Australian and New Zealand College of Psychiatrists (RANZCP) clinical practice guidelines for the management mood disorders (MDcpg2020) is incorrect. We, the authors of the MDcpg2020, strongly refute these assertions and argue that first we have determined the positioning of rTMS using the same criteria as those applied to other treatments for depression. Second, in accordance with National Health and Medical Research Council (NHMRC) guidelines, the processes by which we have developed the MDcpg2020 have been guided by best practice and have been overseen throughout by the RANZCP. Third, our objective and detailed examination of the relevant research has shown that the evidence needed to support the positioning of rTMS alongside standard therapies for depression is severely deficient. And therefore, as a consequence, we set out clearly both our logic and reasoning with respect to interpreting rTMS data and outline our evidence-informed position in which rTMS remains a potential alternative therapy that can be considered in certain clinical circumstances once both suitable psychological and pharmacological treatments have been trialled. We also discuss why, until further research is conducted, rTMS is perhaps best regarded as an experimental therapy and an investigational tool, and to assist in this regard, we propose a framework for consideration by those conducting rTMS studies in the future. Thus, based on current knowledge, we conclude that rTMS does not have a sufficient evidence base to warrant recognition as a standard therapy for depression alongside established treatments such as psychological interventions, pharmacotherapy, and electroconvulsive therapy. Furthermore, there is no clinical profile for depressed patients that might benefit from rTMS and therefore tolerability alone is not good enough reason to promote rTMS in the management of major depression.
Introduction
The viewpoint by Fitzgerald et al. (2021) is highly critical of the recently published Royal Australian and New Zealand College of Psychiatrists (RANZCP) clinical practice guidelines for mood disorders (MDcpg2020) (Malhi et al., 2021). Unfortunately, the article itself is somewhat haphazard both in its construction and argumentation, and so, to address the concerns of the authors we have succinctly and clearly summarised the criticisms levelled against the authors of the MDcpg2020, and an accompanying editorial (Malhi, 2021) so that they can be properly understood. We have then provided a detailed response that provides specific rebuttal and delineates our reasoning. In our response, we challenge the many incorrect assertions made by the viewpoint and provide detailed explanation for our perspective on these matters – drawing on available evidence. Finally, in an effort to move the discussion forward, we conclude by outlining an agenda for future repetitive transcranial magnetic stimulation (rTMS) research and provide a suitable template.
Terminology and scope
To maintain clarity, we will refer to the article by Fitzgerald et al. as the
The viewpoint
It is important to note that while the Viewpoint is titled ‘
The crux
The main concern of the Viewpoint is the clinical positioning of rTMS in the treatment of major depression. In fact, essentially, this is their only concern. To appreciate this fully, it is important to understand the process by which any new potential treatment for depression, such as rTMS, is positioned in an existing therapeutic algorithm or model of care.
In the MDcpg2020, the management of major depression is summarised in Figure 26 (p. 64) and captured schematically by the MIDAS diagram 1 in Figure 33 (p. 87). The addition of a new intervention within this existing schema has to be determined on the basis of evidence. Such evidence should first establish the efficacy of the treatment and then, second, demonstrate its safety. Finally, the evidence should point to where, among existing treatments, the new treatment best fits. For instance, if in terms of clinical benefit, a new intervention is superior to established treatments, it may be validly considered ahead of these. But if, when compared with established treatments, its benefits are either unclear or inferior, then logically and ethically, the new treatment must be relegated to a later stage of management, or be considered an optional or experimental strategy, so as not to disadvantage patients and preclude them from receiving established optimal care.
In the case of rTMS, the focus is principally on
Professional Practice Guideline 162
This guideline was developed by the Section of Electroconvulsive Therapy and Neurostimulation Committee (SENC), which contains many of the same authors that have written the Viewpoint.
In the PPG section on ‘
The immediate question that arises, and one which has immense clinical salience, is how to determine who is suitable? Is the PPG suggesting that anyone who does not respond to a single medication trial can or should move to rTMS? The section on ‘
Instead, the PPG is littered with broad statements that are difficult to make sense of, for example, ‘
The lack of specificity concerning the indication for rTMS is further evident when considering ‘
Appraising the evidence
To meaningfully assess clinical trial evidence, several kinds of knowledge are necessary. First, familiarity with the illness being treated (depression); second, an understanding of the treatment being administered (rTMS); and third, proficiency in the interpretation of clinical trials, both methodologically and statistically. Collectively, this knowledge can be referred to as expertise in the field.
Upon appraising findings from research, the evidence can then be stratified according to the robustness of the studies that have been conducted and the significance of the reported findings. In other words, the ‘
However, it is important to note that ultimately, the significance of evidence is derived from the quality of the data. In other words, the raw findings from actual studies themselves are more important than any signal that is generated from subsequent derivative analyses. Furthermore, such secondary analyses (see below for further discussion) cannot close any inherent evidence gaps that may be present in the primary data, such as a lack of comparative studies or clinical equipoise studies. And it is the bridging of these evidence gaps that is key to determining the positioning of a new treatment such as rTMS in the treatment algorithm for depression.
According to the National Health and Medical Research Council (NHMRC), the development of a guideline 3 should be evidence-informed such that it will enable the extraction of meaningful information and the formulation of actionable recommendations. And hence this was the approach adopted by the MDcpg2020 committee, which carefully considered clinically important evidence gaps.
Additional viewpoint criticisms
As discussed, the main purpose of the Viewpoint is to lament the positioning (or more accurately the lack of positioning as part of routine standard of care) of rTMS within MDcpg2020 treatment algorithms. This is argued, unconvincingly, largely on the basis of assertions, and this is principally because unequivocal evidence in support of their position is lacking.
First, the Viewpoint stridently criticises the entire process by which the evidence has been synthesised, claiming that ‘
It is noteworthy that the Viewpoint also contains two tables; the first compares the recommendations of the current guidelines to the previous version (Malhi et al., 2015), and the second provides a summary of the College position statement 79, 4 the PPG 16, and the public summary from the Medical Services Advisory Committee (MSAC). 5 Interestingly, the abstract of the Viewpoint omits some key assertions made by the authors in the text and instead concludes by referring the reader to the PPG 16, which we have commented on above.
Fundamentally, the authors of the Viewpoint are unhappy with the positioning of rTMS within the current guidelines and therefore, they challenge the evidence that has been used to determine its positioning, the process by which the guidelines have been developed, and even the expertise of the people involved.
The response
The core purpose of the MDcpg2020 is to provide clinically meaningful guidance for clinicians managing depressive and bipolar disorders. It is an important document as it is likely to inform clinical decision-making, service design and delivery, and the provision of funding for clinical care. This is why a considered, balanced and comprehensive process was utilised for the development of the MDcpg2020, and experts with both broad and specialist experience rather than those with narrow or special interest constituted the mood disorders committee (MDC).
Furthermore, the recommendations and the advice within the guidelines have to be easily accessible and relevant to clinical practice. The MDC, based on their expertise and experience, and in this case previous performance, were entrusted with this task and executed it accordingly. The relevant literature was reviewed, synthesised and discussed in detail. Indeed, the number of committee meetings undertaken to discuss every facet of the guideline probably surpasses the commitment of most committees that have developed such guidelines internationally. In addition, the committee adopted a somewhat cautious approach, with the reasoning that any recommendations that inform clinical practice need to have robust support.
To understand how the recommendations have been derived, it is important to understand the different kinds of data and data analyses that are currently conducted to identify treatment effects. Primary studies which generate raw data, and primary findings, concern the implementation and testing of a therapy. This can be very basic and straightforward such as an open-label study, where there may not be any comparator; however, more often, there is a placebo control. The randomisation of subjects to the various arms of a study and procedures of blinding enhance the quality of the data by reducing the likelihood of bias. The other factor that is important is the population that is being tested, for example, its homogeneity and its representativeness – so that results from any clinical trial are generalisable. For example, the findings from a young group of patients may not be particularly useful for informing the management of a much older group of patients who are likely to have many more comorbidities. Thus, the
Schematic detailing the hierarchy of evidence synthesis methods.
Secondary analyses such as systematic reviews and meta-analyses are sometimes useful and provide a shortcut to confirm findings that are present in primary studies. In instances where the primary studies are equivocal, further analyses may provide an indication of where further research and refinement is necessary, and although they may provide new insights and an alternative perspective, they don’t generate new data per se. Hence, any systematic or umbrella review or meta-analysis is only as good as the quality of the data from primary studies and for that reason high-quality meta analyses should also critically appraise the quality of each study. Furthermore, it is important to note that such secondary analyses alone are not substantive enough to provide the basis for a clinical practice guideline. This is especially the case because of potential publication bias because negative studies are less likely to be published and can therefore slant meta-analyses towards positive findings (Amad et al., 2019a). This is an important limitation to bear in mind, even of the highest level of evidence such as meta-analysis of double blinded randomised controlled trials (RCTs).
The difficulty for rTMS is that primary data regarding its efficacy is weak and inconsistent – especially with respect to the kinds of depressed patients that have been tested. In addition, only some secondary data seem to show some modest effects and only in specific circumstances. As a consequence, the evidence does not meaningfully inform clinical practice and critically, cannot assist clinicians in making decisions regarding the management of their depressed patients. Furthermore, there is very little evidence as regards the comparative efficacy of rTMS, against psychological or pharmacological interventions in the treatment of depression. It is this lack of evidence in the extant literature regarding rTMS and the ongoing significant gaps in knowledge which preclude it from being incorporated into the routine treatment algorithm for mood disorders. We therefore briefly discuss this evidence base and the specific papers raised in the Viewpoint in more detail below.
The evidence: revisited
The Viewpoint states that ‘
Papadimitropoulou et al. (2017): First, it is important to note that the Papadimitropoulou et al. (2017) paper has a total of five authors, and that the first three are employed by MAPI as part of the MAPI group, a consultancy company engaged by Janssen-Cilag to write the paper. The remaining two authors are employees of Janssen Pharmaceutica who, through MAPI, ‘
The analyses in the paper compared the efficacy and tolerability of pharmacological and somatic interventions (physical treatments e.g. ECT and rTMS) and did so in the context of TRD. The authors defined TRD according to the Thase and Rush (1997) model that is based largely on pharmacotherapy, and considered this definition that was developed more than two decades ago, as robust, even though recent papers including those by Rush, have sought to update it because it is too narrow and poorly defined (see Dodd et al., 2020; McAllister-Williams et al., 2020; Malhi et al., 2019).
The only other significant results the paper reports in its abstract are that at later time points, 4, 6 and 8 weeks, augmentation with quetiapine and risperidone were the first- and second-best treatments, respectively, and that the most tolerable treatment was augmentation with lamotrigine. The authors concede that in their searches many publications might have been missed, and they attempt to explain why it is difficult to harmonise results from different studies over time because of gradually increasing placebo response rates and the fact that many primary studies did not use equivalent scales for assessing depression. However, while the paper recognises these limitations, it is also important to note that it only includes one ECT study (Rosa et al., 2006) and that the vast majority of the rTMS studies have extremely small numbers and no comparator studies with antidepressants.
Indeed, the primary data for rTMS in this review is remarkably poor. By their own account the authors note that the ‘
In addition to the primary data being weak, the other main problem this paper exemplifies is the difficulty of pooling data. The validity of pooling low, moderate and high intensities together is questionable despite the authors’ assertion. This is because clinical effects have been found to vary considerably depending on stimulation intensity (Lefaucheur et al., 2020; Trevizol et al., 2019).
Thus, it is concerning to us that this paper is cited as evidence for the efficacy of rTMS. The analysis in the paper has many significant limitations with respect to pooling rTMS data, not least because of the poor quality of the primary data in many of the included rTMS studies. Notably, only one study involved ECT, and yet findings are reported as relating to somatic treatments as a whole. Even so, the paper states quite emphatically that ‘
Additional literature: Equally of concern is the accusation by the Viewpoint authors that the ‘
For example, one of the largest sham-controlled studies of rTMS (O’Reardon et al., 2007) with 301 participants failed to show any statistical differences on the primary outcome. Similarly, a recently published randomised sham-controlled trial of 164 subjects with TRD failed to show any difference in remission rates with active rTMS treatment (Yesavage et al., 2018). Even as an add-on to antidepressant treatment, rTMS has failed to show an effect in a large active versus sham-controlled study (
Extrapolation upon extrapolation: Even though the effects of HF rTMS have yet to be established, there is a quest to make rTMS faster. As a consequence, some studies have looked at protocols that deliver HF rTMS more frequently over a shorter period of time (Fitzgerald et al., 2018), and others have investigated the effects of more intense forms of rTMS (Blumberger et al., 2018). The interesting aspect of these endeavours is that neither has tried to compare to sham or other treatments. Instead, they have adopted a non-inferiority study design, which is predicated on the assumption that rTMS is effective. And so, all that any new version of rTMS treatment has to demonstrate is that it is no worse than standard rTMS! What this approach discounts is the very likely possibility that the different forms of rTMS are ‘
Recommendation 1.7 (Box 1. MDcpg2020)
Criticism is then made of the second CBR, in which quite reasonable and sensible advice is offered to moderate the expectations of patients as regards likely treatment outcomes, as gains are probably modest at best, given the response and remission rates for rTMS and its overall effect size compared with sham. This is contested by the Viewpoint, once again on the basis of exaggerated evidence. Furthermore, they complain that ‘
In fact, the side-effects of medications are discussed repeatedly throughout the guideline, and in many instances, the recommendations for various medications have been downgraded because of their side-effect profile. However, instead of dismissing them and disregarding their role, the guideline states quite clearly ‘
Clinical profiling
The Viewpoint also complains that rTMS does not feature on the main summary diagrams, for example, in the management of major depression (Figure 26, MDcpg2020), a point to which we responded when communicating through the feedback process via the College. The reason for this is that evidently, rTMS is not yet an equivalent option to psychological interventions, pharmacotherapy or ECT. It not only lacks undisputed equipoise efficacy, but equally importantly, has not identified a clinical profile to which it is best suited. This is an important point. When the MDcpg2020 discusses indications for treatment, the MDcpg2020 doesn’t simply mean the broad diagnostic category. The MDcpg2020 actually means the clinical profile, the subtype, the kind of illness and its context. At a general level, this means whether it is being used for acute treatment or long-term therapy. But beyond this, the indication of treatments and interventions is for specific clinical symptoms and patterns of illness, in other words, the clinical profile of patients and their depression. For example, patients with negative cognitive schema generally do well with cognitive behavioural therapy (CBT), and patients with certain depressive symptoms are likely to do well with particular antidepressant therapies (see Table 1, adapted from Table 9, p. 48, MDcpg2020); for instance, for melancholia – tricyclic antidepressants, for psychotic depression – antipsychotics in combination with antidepressants, and for anxious depression – selective serotonin reuptake inhibitors (SSRIs). Similarly, as mentioned previously, for psychotic depression and melancholia with psychomotor features, ECT is the treatment that is most likely to be effective.
Pharmacological treatment based on clinical profile (Adapted from Table 9; MDcpg2020).
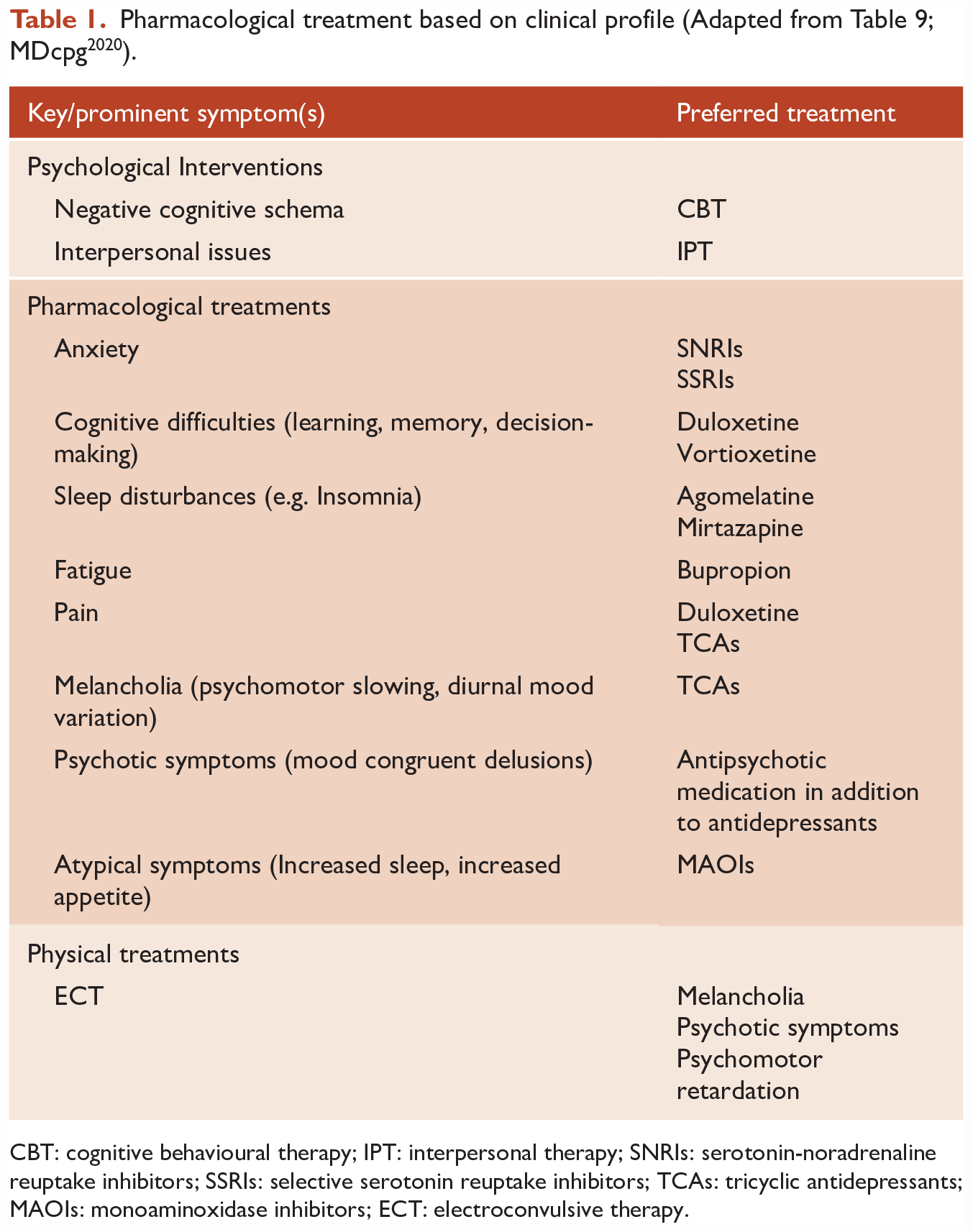
CBT: cognitive behavioural therapy; IPT: interpersonal therapy; SNRIs: serotonin-noradrenaline reuptake inhibitors; SSRIs: selective serotonin reuptake inhibitors; TCAs: tricyclic antidepressants; MAOIs: monoaminoxidase inhibitors; ECT: electroconvulsive therapy.
As a consequence, rTMS sits among non-specific alternatives, and, since there are many non-standard therapies, for which the evidence base is either unsubstantiated or weak, the MDcpg2020 did not emphasise these options and nor did they feature on the management figures. The authors of the Viewpoint clearly believe otherwise and make the wholly unsubstantiated assertion that rTMS is part of the ‘
The authors of the Viewpoint go on to complain that ‘
A framework for future rTMS research?
Given the many gaps in the evidence for rTMS efficacy (see ‘
The two key aspects of rTMS that need to be established unequivocally are its efficacy in comparison to placebo and its efficacy relative to other established treatments (comparative efficacy) such as psychological therapies and medications.
Establishing efficacy
To determine the efficacy of rTMS in the treatment of depression, robust, methodologically sound studies need to be conducted in large numbers of patients. These studies should mirror the trials conducted for antidepressant medications and preferably use a clinical equipoise design such as that employed in the STAR*D study (Rush et al., 2004). The studies should also ideally be multi-centred and make use of a wide range of clinical measures emulating, where possible, clinical practice. Insights regarding design can be gained from landmark pharmacotherapeutic studies. For rTMS efficacy research, both the acute and long-term effects should be investigated and the duration of studies should be sufficient to capture and map the pattern of any changes that might occur over time. Where possible, rTMS studies should make use of effective sham, blinding and randomisation, and to ensure valid comparisons can be made across studies, stimulation parameters and sites of stimulation used for rTMS should be made consistent. Only if a sufficient number of high-quality studies with large numbers of patients are conducted, and the findings robustly corroborated, can rTMS hope to achieve equivalence with other kinds of treatments for depression in terms of efficacy.
Determining comparative efficacy
In addition to testing whether rTMS has meaningful clinical efficacy, its effects in patients need to be compared with existing treatments. The therapy used for comparison will depend on where within existing algorithms rTMS is thought to have potential benefit. However, to begin with, it should be compared against standard therapies, and this should include psychological interventions if it is to be used early in the course of depression. Its effects should also be directly compared with antidepressants in different settings. Only then will a clear picture emerge of its relative efficacy that may be able to meaningfully inform its positioning in the management of depression. Among physical treatments, it is already clear that rTMS lacks by a significant margin the efficacy of ECT, but it remains to be tested whether it benefits those that either choose not to have ECT, or are ineligible because of a contraindication, for example.
Such comparative clinical research is essential, first, to determine whether rTMS works, and then, second, to identify whom it benefits most. To position rTMS, it is essential that the latter question regarding the clinical profile of a depressed patient is clear. At present, there are no indicators as to what kind of depression and indeed what kind of depressed person is likely to benefit from rTMS. In addition, longitudinal research should be conducted to assess the long-term effects of rTMS – to determine, for example, whether any changes are sustained or whether patients only partially respond and then quickly relapse. Ideally, there should also be a rigorous economic analysis of the procedure that takes into account the opportunity cost of rTMS.
Mechanism of action research
In conjunction with clinical research, it is important that theoretical and technical research is undertaken to better understand the neural mechanisms that underpin the putative effects of rTMS. The reproducibility of the procedure needs to be ensured along with a deeper understanding of the downstream neurophysiological effects, and whether this leads to clinical benefit. As an experimental and investigational probe, TMS is an excellent tool; however, its transposition to the clinical arena, and in particular its positioning as a therapy, still requires substantive further research.
Conclusion
In this Response, we have tried to address some of the many concerns raised by the Viewpoint. We have demonstrated that rTMS does not have sufficient evidence to support a robust claim of efficacy in the routine management of depression compared with established treatments such as psychological interventions, pharmacotherapy and ECT. And that while it may be readily tolerated, there is limited and as yet unconvincing evidence that it is an effective therapy. For rTMS to be a part of routine care, it has to show meaningful efficacy and do so reliably in a defined population of depressed patients. Hence, the rTMS section of the MDcpg2020 has been written with the practising clinician in mind who, when managing patients suffering from depression, has to make decisions in their best interest and may occasionally have to consider alternatives.
While efficacy is clearly the basic consideration, there are many other issues that are relevant when making clinical management decisions concerning an individual patient. The MDcpg2020 therefore discusses a range of issues such as cost, and the inconvenience involved in daily treatment for a period of 4 to 6 weeks; problems in interpreting the results from research studies; the high degree of heterogeneity of rTMS techniques and treatment parameters within those studies and importantly the many deficiencies in the evidence base.
More specifically, there are two major knowledge gaps that are of most importance to the clinician, and these are first, identifying what illness and patient characteristics might predict response or non-response, and second, the lack of data comparing rTMS to other treatments, in particular to further trials of medication and especially to the combination of medication and psychological treatment. Without this information, it is extremely difficult for any clinician to judge the likelihood of rTMS being of benefit for a particular patient, or whether another treatment alternative would be a better option. These considerations were key when deliberating the positioning of rTMS.
Clearly, the evidence base for rTMS is complex, and interpreting the results of primary and, in particular, secondary studies such as meta-analyses is challenging. Hence, we have examined this further in this Response and pointed out that while statistically significant superiority of rTMS over sham may be reported in reviews, the effect sizes vary considerably and are often reported in forms that are unfamiliar to most clinicians. Furthermore, the issue of clinical significance, as opposed to statistical significance, has not been well explored in the literature and as we noted in the MDcpg2020, the Ontario Health Review (2016) reported that effect sizes did not reach pre-determined criteria for clinical significance. Hence, we argue that clinical significance is of far more relevance to clinicians than statistical significance and that again this is an area that needs considerable further research.
Indeed, in this regard, clinicians are more likely to be interested in response and remission rates and, possibly, numbers needed to treat (NNT). These give a much better idea of the probability of response than effect sizes alone. The MDcpg2020 discusses this aspect of reported research findings at some length, pointing out that pooled response and remission rates compared with sham are modest overall and problematic as clearly evidenced in our discussion of the Papadimitropoulou paper. This knowledge would give most clinicians (and the patient) a better understanding of the outcome their patient might expect, and which may be well below their expectations, hence our recommendation 1.7 (MDcpg2020) in which we state that these data and patient expectations should be discussed as part of the consenting process. We therefore stand by this statement.
Finally, the CPG points out that there is insufficient evidence to be able to confidently recommend at which point in our current treatment algorithm rTMS is best utilised. The only thing that is clear is that the evidence for psychological interventions and pharmacotherapy gives these treatments primacy. And hence why we have adopted the former into
In contrast, the RANZCP Professional Practice Guideline (PPG, 16) (2018) recommends that rTMS can be used after one failure of adequate antidepressant treatment, with no mention of psychological treatment. This would be a very large group of potential patients. Interestingly, the MSAC has recommended that patients who have failed two adequate courses of antidepressants from two different classes
Glossary of acronyms utilised in the Viewpoint and the Response.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.S.M. has received grant or research support from National Health and Medical Research Council, Australian Rotary Health, NSW Health, American Foundation for Suicide Prevention, Ramsay Research and Teaching Fund, Elsevier, AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier; and has been a consultant for AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier. D.B. has received funding to host webinars by Lundbeck. P.B. has received research support from the National Health and Medical Research Council, speaker fees from Servier, Janssen and the Australian Medical Forum, educational support from Servier and Lundbeck, has been a consultant for Servier, served on an advisory board for Lundbeck and has served as DSMC Chair for Douglas Pharmaceuticals. R.M. has received support for travel to education meetings from Servier and Lundbeck, speaker fees from Servier and Committee fees from Janssen. M.H. has received grant or research support in the last 5 years from the National Health and Medical Research Council, Medical Research Future Fund, Ramsay Health Research Foundation, Boehringer-Ingleheim, Douglas, Janssen-Cilag, Lundbeck, Lyndra, Otsuka, Praxis and Servier; and has been a consultant for Janssen-Cilag, Lundbeck, Otsuka and Servier. A.B.S. has shares/options in Baycrest Biotechnology Pty Ltd (pharmacogenetics company) and Greenfield Medicinal Cannabis, and has received speaking honoraria from Servier, Lundbeck and Otsuka Australia. R.J.P. has received support for travel to educational meetings from Servier and Lundbeck and uses software for research at no cost from Scientific Brain Training Pro. The authors E.B., Z.M. and B.L. declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
