Abstract
Deciding when and how to change treatment in patients with major depressive disorder who have inadequate response to initial antidepressant therapy is an important everyday clinical question. Here, we ask whether an early change of approach is superior to a delayed change. We consider the recommendations provided by recent guidelines, examine the evidence behind this guidance and suggest a decision tree to clarify treatment options and timing. Both the early and late-change strategies may have their place in clinical practice. However, we take the view that an earlier than currently usual change in antidepressant treatment should be considered more frequently in cases of non-response. Specific studies are needed to identify and to better understand predictors of early and late response.
For many patients with major depressive disorder (MDD), response to antidepressant treatment is not sufficient (Bauer et al., 2013; Rush et al., 2006). The Sequenced Treatment Alternatives to Relieve Depression (STAR*D) study showed that only 37% of adult patients with MDD reported remission of symptoms (Quick Inventory of Depressive Symptomatology–Self-Report [QIDS-SR16] score ⩽ 5, corresponding to a Hamilton Rating Scale of Depression17 score of ⩽7) with their first treatment step (Rush et al., 2006). And only 49% of patients achieved a response to first-line treatment, defined as a ⩾50% reduction from treatment step entry in QIDS-SR16 score.
Other studies indicate that at least 30% of patients will either not respond to or only partially respond to their initial treatment, irrespective of the antidepressant (Bauer et al., 2013).
Although there is agreement that a change of treatment benefits many patients, the optimal timing and nature of that change remain unclear. A recent systematic review showed no consensus among clinical practice guidelines with regard to the appropriate timing for determining inadequate response (MacQueen et al., 2017).
We considered the arguments for changing antidepressant treatment early versus delaying change for as long as possible in patients with inadequate response to initial therapy and examined the evidence behind both approaches.
As a consideration against early switching, it can plausibly be argued that lack of early improvement selects a subgroup of patients who are more difficult to treat and more likely to have a relatively poor prognosis irrespective of the timing of any change in therapy. And there are no compelling data to demonstrate that early changes in management improve outcome.
However, based on available data, which are admittedly incomplete and at times inconsistent, and on our clinical experience, we suggest that 2 weeks (with 4 as an absolute maximum) is an appropriate time at which to reconsider the treatment approach in the case of non- or partial response. There is general consensus that options following partial response include an increase in antidepressant dose, augmentation or combination therapy. In cases of non-response, clinicians should consider changing treatment to a different class of antidepressant. These options, and appropriate points at which to consider them, are shown in a decision tree (Figure 1).
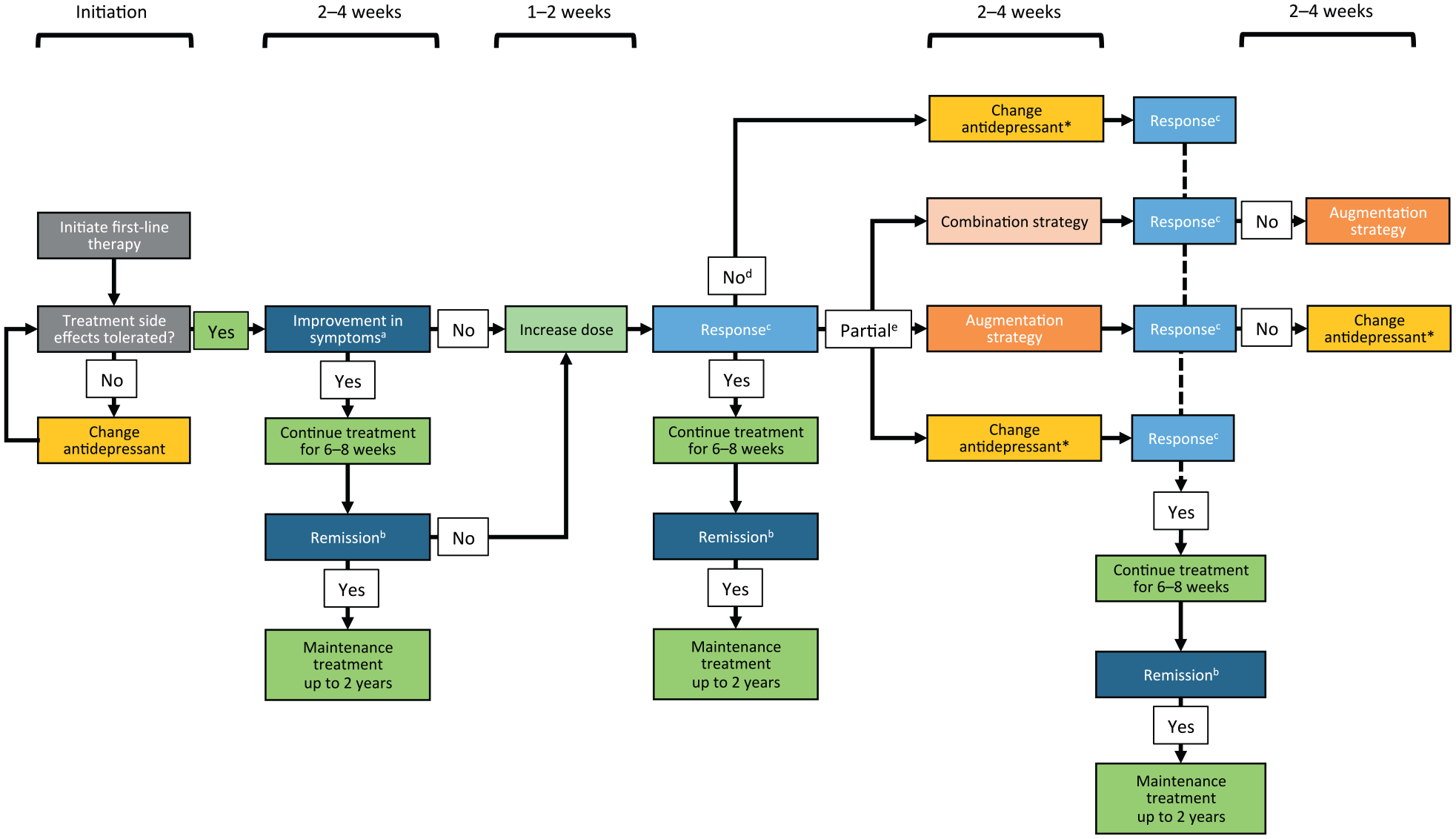
Decision tree for changing treatment in depression.
When and how to change first-line therapy remains an important everyday clinical dilemma. Recently, Malhi et al. (2020a) have proposed a Windows of Antidepressant Response Paradigm (WARP) which conceptualizes three distinct periods – immediate (1–2 days), fast (up to 1 week) and slow (1 week onwards) – which can be used to categorize response to therapy and aid the development of more effective agents. A subsequent editorial in this Journal advocated a change in perspective so that we focus not on treatment resistance but on response and acknowledge that the overall presentation of depression can be made up of many different components which may respond differentially to a variety of different treatment modalities.
It is clear that how best to approach these issues continues as a subject for active discussion (Malhi et al., 2020b) and debate and that is the context in which we contribute this Viewpoint.
Changing depression therapy in good time
The STAR*D study showed that success rates decline with each successive step of treatment (Rush et al., 2006). This emphasizes the need to determine the optimal therapy for individuals as early as possible.
The 2013 World Federation of Societies of Biological Psychiatry (WFSBP) guidelines recommend strategies for patients with a partial response to initial therapy. This is defined as a decrease in symptom severity compared to baseline of 26–49% in those who tolerated treatment for 2–4 weeks at adequate dosage (Bauer et al., 2013). These recommendations include the use of augmentation, or, for patients who initially receive a selective serotonin reuptake inhibitor (SSRI), SSRI combination therapy with a presynaptic alpha2-autoreceptor and presynaptic inhibitory auto- and heteroreceptor agents (e.g. mirtazapine, mianserin) and then augmentation, if the patient does not respond to combination. According to these guidelines, change to an antidepressant with evidence of superior efficacy should be considered, if the patient does not show adequate response to augmentation (Bauer et al., 2013).
These recommendations for patients with a partial response to initial therapy are corroborated by a review of studies investigating early treatment change. This review concluded that the majority of patients who did not show an improvement on a depression rating scale (i.e. less than 20% reduction from baseline) within 4 weeks would not show a response by 8 weeks and would therefore likely benefit from a change in management (Kudlow et al., 2014). We would suggest that the same could be said of non-response after 2 weeks. However, evidence is conflicting with regard to early versus no change of antidepressant.
There are inconsistent results from randomized controlled trials (RCTs) comparing an early change of antidepressant against titration of first-line treatment or later change (Bose et al., 2012; Nakajima et al., 2011; Romera et al., 2012; Tadic et al., 2016). In an open-label study, 41 patients who did not show an adequate response (⩾20% reduction in Montgomery-Åsberg Depression Rating Scale; MADRS) after 2 weeks of sertraline were randomized to early change to paroxetine or sertraline titration (Nakajima et al., 2011). Early change was superior to titration in rates of response (⩾50% reduction in MADRS: 75% vs 19%; p = 0.002) and remission (MADRS ⩽ 10: 60% vs 14%; p = 0.004). In contrast, a large double-blind RCT in patients who did not respond adequately to escitalopram (Bose et al., 2012), which did not meet its primary outcome (difference in time to all-cause discontinuation), indicated superiority of dose increase over early change of treatment to duloxetine in both change on MADRS at week 8 and rates of remission (MADRS ⩽ 10: 54% vs 42%; p = 0.013). However, this analysis showed no difference in rates of response (⩾50% reduction in MADRS).
In another large RCT, non-responders to escitalopram, defined as those with a <30% reduction on the Hamilton Depression Rating Scale (HAM-D17) at 4 weeks, were randomized to early change of treatment to duloxetine or continuation on initial therapy for a further 4 weeks (Romera et al., 2012). Although the study did not meet its co-primary endpoints (time to confirmed response and remission, i.e., HAM-D17 ⩽ 7), early change was superior to continuation of initial therapy in terms of remission (43% vs 36%; p = 0.048), while no difference was observed in response rates.
An open-label study in 192 patients without improvement after 2 weeks of initial escitalopram compared a two-step early change/augmentation strategy (non-responders 4 weeks after change to venlafaxine received lithium) versus continuation on escitalopram, with treatment change after 4 weeks in those still not responding (Tadic et al., 2016). In contrast to the results of Romera et al. reported above, there was no significant difference in remission rates between the two strategies.
Opportunities in changing antidepressant treatment early
Increasing evidence suggests that many patients with MDD begin to show an improvement in symptoms within 2 weeks of starting treatment (Bauer et al., 2013; Stassen et al., 2007; Taylor et al., 2006). In a study of 2848 patients, the mean time to onset of improvement was 13 days and the mean time to response was 19 days (Stassen et al., 2007). Similarly, a meta-analysis showed that clinical improvements are seen by the end of the first week with an SSRI (Taylor et al., 2006). Improvements continue over time, but at a decreasing rate. Hence, we suggest that clinicians should think about the possible need to change treatment as early as 1 week, and certainly consider it after 2 weeks of non-response. Waiting for 4 weeks is, in our opinion, too long.
In a study of 2848 patients treated with one of seven antidepressants or placebo, those with an early response were at least three times more likely than others to become sustained responders (Stassen et al., 2007). Response usually precedes remission (broadly defined as the absence of symptoms) (Bauer et al., 2013) and remission is highly predictive of good functional recovery and lower relapse rates (Rush et al., 2006). Therefore, early response might be expected to lead to early remission and improved outcomes.
Recent guidelines are consistent with this early response time frame. The Canadian Network for Mood and Anxiety Treatments (CANMAT) recommend an initial increase in dose, if no improvement is seen after 2–4 weeks of first-line treatment. If response remains inadequate, then a change of antidepressant or, for patients with early treatment resistance, adjunctive medication should be considered (Kennedy et al., 2016).
The Treatment Algorithm Group from Australia and New Zealand recently suggested that patients showing <20% improvement after 3 weeks, following partial or no response at 2 weeks, require an escalation in dose or change of antidepressant (Boyce et al., 2019). Both the WFSBP and CANMAT guidelines recommend changing antidepressant, if initial therapy is not tolerated.
Kudlow et al. (2014) evaluated studies that used early improvement to predict outcome. Only approximately 20% of patients without improvement at 2–4 weeks became stable responders at 8 weeks. Since lack of early response is predictive of poor response later, and since poor response adversely affects disease course and can result in chronicity (Judd et al., 2000; Whiteford et al., 2013), lack of improvement within 2–4 weeks should alert us to the need for prompt change in therapy (Kudlow et al., 2014). A number of earlier guidelines agree that insufficient response can be defined within 2–4 weeks of initiating treatment (MacQueen et al., 2017).
Initial treatment selection should take into account depression history and severity, comorbidities, concomitant medications and patient preferences, as well as drug efficacy, safety and tolerability (Bauer et al., 2013; Kennedy et al., 2016). The range of effective and well-tolerated antidepressants (Cipriani et al., 2018) provides many options. First-line antidepressants differ in efficacy across symptom dimensions (mood, cognition, activity, sleep, anxiety and anhedonia) and hence offer further grounds for choice (Baune et al., 2019).
However, there is less consensus on the timing of treatment change should first-line treatment not result in adequate response (MacQueen et al., 2017). Although rating-based criteria should be used, in talking to patients it may become evident that some are experiencing helpful changes in some relevant aspects of their illness – even if thresholds for overall reduction of symptoms are not met.
Alternatives to an early change of antidepressant
For patients with inadequate response, the alternatives to an early change of antidepressant are dose increase, combination with another antidepressant and augmentation/adjunctive therapy (Kennedy et al., 2016). It is important to note that standardized stepwise drug treatment algorithms, when compared with computerized medication choice guidance or treatment as usual, reduce time to remission following non-response after 4 weeks’ monotherapy (Adli et al., 2017). This supports the view that using a drug treatment algorithm might be an easy way to distinguish responders from non-responders at an early stage.
A dose increase is a way forward for patients put on insufficient doses at the start of treatment (Adli et al., 2005). Serum drug level can identify rapid metabolizers and demonstrate the need for dose increase in this group (Bauer et al., 2013). Also, different agents have different dose–response relationships: data suggest a potential benefit of high-dose treatment with tricyclic compounds and some irreversible monoamine oxidase inhibitors, while no increased efficacy is seen with high-dose SSRIs (Adli et al., 2005).
There is also a case for augmentation with agents other than conventional antidepressants. A number of augmentation agents are significantly more effective than placebo in patients with partial response to antidepressant therapy, with the most robust evidence – though this is drawn from a study of treatment-resistant depression – seen among the atypical antipsychotics (Zhou et al., 2015). The issue of augmentation was also considered in the Veterans Administration Augmentation and Switching Treatments for Improving Depression Outcomes (VAST-D) RCT (Mohamed et al., 2017) which included 1522 patients unresponsive to at least one course of antidepressant. Augmentation with aripiprazole was associated with a modest but statistically significant increase in the chances of remission over 12 weeks when compared against bupropion monotherapy, although this was at the expense of a higher rate of adverse events. Agents which are not conventional antidepressants may be significantly less well tolerated than placebo, highlighting the need to carefully consider potential side effects alongside efficacy.
The STAR*D study supports the combination of certain classes of antidepressants (Trivedi et al., 2006). The addition of a norepinephrine/dopamine-reuptake inhibitor or a serotonin receptor agonist to an SSRI resulted in response and remission rates of ~30% in patients who had not achieved remission with an SSRI alone. In addition, the combination of a serotonin-noradrenaline reuptake inhibitor (SNRI) with an α2-antagonist was superior to monoamine oxidase inhibitor monotherapy. However, this regimen may have worse side effects (Bauer et al., 2013; Rush et al., 2006). Evidence also supports the combination of SSRIs with α2-antagonists. These findings are still preliminary since a continuation of monotherapy as a control group was absent.
In a clinical scenario of partial response to an SSRI or SNRI, there may be additional options for change to another class. The REVIVE study showed that treatment with vortioxetine vs agomelatine improved clinical outcomes in patients with partial response after one course of an SSRI or SNRI (remission rate 55.2% at week 12) (Montgomery et al., 2014).
Despite the possible successful strategies using two lines of treatments, there remains a clinical need for additional effective options in difficult-to-treat patients with severe depression. Another change of antidepressant or add-on therapy should be considered in case of partial response, and new combination therapies, electroconvulsive therapy or repetitive transcranial magnetic stimulation should be considered as fourth-line treatments and beyond.
Although dose increase, augmentation and combination therapy are recommended in some guidelines (Bauer et al., 2013; Kennedy et al., 2016), there is also evidence for negative effects. A meta-analysis of augmentation with atypical antipsychotics showed that discontinuation rates due to adverse events were fourfold higher than with placebo (odds ratio = 3.91; p < 0.00001) (Nelson and Papakostas, 2009). Despite such adverse event rates, an RCT showed lower patient retention rates at 12 weeks among those who changed antidepressant (69.1%) than in those assigned to either of two augmentation groups (80.4% and 74.7%) (Mohamed et al., 2017). Furthermore, withdrawal due to lack of response or adverse effects was highest in the change of antidepressant group. In an RCT of 567 patients with chronic or recurrent MDD, antidepressant combinations were associated with higher self-reported non-adherence than monotherapy (Warden et al., 2014).
Limitations of this viewpoint
Determining the need for change in antidepressant treatment and the nature and timing of that change remain major clinical challenges.
On one hand, the overwhelming evidence that a longer duration of illness is associated with poor clinical and psychosocial outcomes suggests an urgent need for early efforts to maximize the efficacy of treatment. On the other hand, there is lack of consistency in evidence from RCTs that early change in management benefits long-term outcome overall when compared with delayed change. It is plausible that lack of early improvement identifies a subgroup of poor-prognosis patients who will remain unresponsive to therapy whatever the changes introduced. It must also be acknowledged that the benefits for some patients that flow from early switching, combination or augmentation may be balanced by increased risk of adverse outcomes in others. These include side effects, higher rates of discontinuation and the drug interaction risks associated with polypharmacy.
We note that pharmacokinetic differences between drugs (such as the long period before dothiepin levels reach steady state) and differences between patients in drug metabolism may play a part in slowing therapeutic response. These factors should be taken into account when any change of therapy is contemplated. We also note that patients considered to have MDD who in fact have bipolar disorder may show lack of response – or even deterioration – when started on antidepressant medication. Accuracy in diagnosis is therefore important.
An individualized approach to treatment selection and strategy for changing therapy is therefore required. Guidelines do not provide consistent advice on when to add or change treatment in the event of inadequate response (Bauer et al., 2013; Kennedy et al., 2016; MacQueen et al., 2017). Current evidence suggests that changing antidepressant early or delaying change for as long as possible may both have a place in clinical practice. However, we do not have enough evidence to make an informed decision early in treatment which patient requires which option.
Conclusion
The identification of predictors of response (clinical, patient characteristics or biological) should be a high research priority. However, clinicians cannot wait until such predictors and possible decision-assisting tools become available. There is robust evidence that sustained remission is more likely in patients with early response, and recommendations by groups such as CANMAT and the Treatment Algorithm Group from Australia and New Zealand are moving towards earlier evaluation of the efficacy of initial therapy. This Viewpoint suggests that an earlier than currently usual change in antidepressant treatment should be considered more frequently in cases of non- and partial response based on clinical judgement and/or standardized ratings. Our view is intended as a contribution to ongoing debate. If a patient does not feel any benefit from an antidepressant therapy within a period of 2 weeks, the need for a change in treatment regimen should be taken into account.
Footnotes
Acknowledgements
This article is based on a debate presented by the authors and sponsored by H. Lundbeck A/S during the 31st European College of Neuropsychopharmacology Congress 2018, in Barcelona, Spain. Editorial support, including drafting and editing the manuscript and figure under the direction of the authors, was provided by Phillippa Curran and Rob Stepney of Insight Medical Writing and funded by H. Lundbeck A/S.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: B.T.B. has consulting roles with the National Health and Medical Research Council, Australia. He has received honoraria from AstraZeneca, Bristol-Myers Squibb, Lundbeck, Pfizer, Servier, Wyeth and Otsuka; research grants from private industries or non-profit funds from AstraZeneca and Sanofi-Synthélabo; and research grants from the National Health and Medical Research Council, Australia. He has served on advisory boards for Janssen-Cilag and Lundbeck and received funds from the Fay Fuller Foundation and James & Diana Ramsay Foundation, Adelaide. P.F. has received research support and honoraria from AstraZeneca, Bristol-Myers Squibb, GE Healthcare, GlaxoSmithKline, Janssen, Eli Lilly, Essex, Lundbeck, Otsuka, Pfizer, Servier and Takeda. He has also received grants from the German Ministry of Health, German Ministry of Science and the German Science Foundation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by H. Lundbeck A/S.
