Abstract
There has been a revolution in the use of mobile health devices for monitoring physical health. There is more recent interest in whether these devices can also be used for monitoring symptoms of mental illness. This paper considers how stress increases risk of mental deterioration and individuals with mental illness are sensitive to the effects of stress. It discusses how an inexpensive mobile health device could be used for detecting physiological signs of stress: deviations in biometrics such as sleep, activity and arousal may reflect a stress response and increased risk of relapse. These biometrics can allow patients to self-monitor and clinicians to detect early warning signs. This paper reviews the measurement of electrodermal activity, actigraphy and heart rate to predict mental deterioration. It considers the advantages of continuous measurement and reviews studies using mobile health devices to monitor stress and psychosis. It describes the potential for using a mobile health device to manage and monitor severe mental illness in young adults. Finally, this paper considers challenges associated with this approach, particularly with regard to correctly interpreting the physiological data and integrating the mobile health device into clinical practice. This paper concludes a mobile health device has the potential to enhance care by improving detection of early warning signs and increasing the connection between clinicians and their patients.
Effective management of severe mental illness in the community faces several challenges: poor engagement between patient and clinician, patient’s being unaware of their symptoms and a slow response by clinicians when early warning signs emerge. The revolution in personal health monitoring now means a simple mobile health (mHealth) device can bridge the gap between patients and clinicians, assisting them in detecting early warning signs. These inexpensive devices can be worn on the body like a wristwatch or embedded within clothing. They have been effective in managing physical conditions, such as cardiovascular conditions, diabetes and obesity (e.g. Silvaa et al., 2015). More recently, there has been interest in how these devices detect the physiological effects of stress (e.g. Cella et al., 2019; Sano et al., 2018). A mHealth device can reliably record biometric data, including electrodermal activity (EDA; a measure of perspiration), actigraphy (rest and activity), heart rate and body temperature. Significant or prolonged deviations in these measurements could indicate a stress response associated with mental deterioration.
This biometric data could be returned to a treating clinician allowing them to assertively respond to emerging symptoms. For example, a clinician notified that a patient has not slept well on consecutive nights will know they are at higher risk. Biofeedback can also be sent to the patient’s mobile phone allowing them to self-monitor, which has shown to be a low intensity yet effective intervention. This is particularly important for patients with severe mental illness who are less aware of the relationship between their symptoms and their mental state. For example, a patient who notices higher arousal levels can implement strategies to reduce their stress. The patient and clinician can use the shared information to interact and manage symptoms, as the mHealth device potentially provides a digital interface enhancing their therapeutic alliance (e.g. Moore et al., 2020). The greater detection, self-monitoring and collaboration could potentially prevent a minor relapse from turning into a lengthy hospitalization (see Figure 1). As patient data becomes more detailed and richer, it is possible to develop models that predict and pre-empt a relapse using patient characteristics and continuous physiological data (e.g. Iniesta et al., 2016). For example, unusual sleep patterns and electrodermal hypoactivity are markers for depression (e.g. Harvey et al., 2011; Sarchiapone et al., 2018) and anxious states can be associated with increased EDA (Boucsein, 2012). The continuous data flow from the mHealth device allows the examination of trends and comparisons with the patient’s healthy state. Precise psychiatric treatments could be developed based on individual characteristics as measured by the device (Insel, 2017). What follows is an article examining the potential value and possible challenges of using a mHealth device to enhance treatment of mental illness. It also describes the use of a mHealth device to manage and monitor severe mental illness in young adults, a group who are more adept at using technology and are at higher risk for developing mental illness.
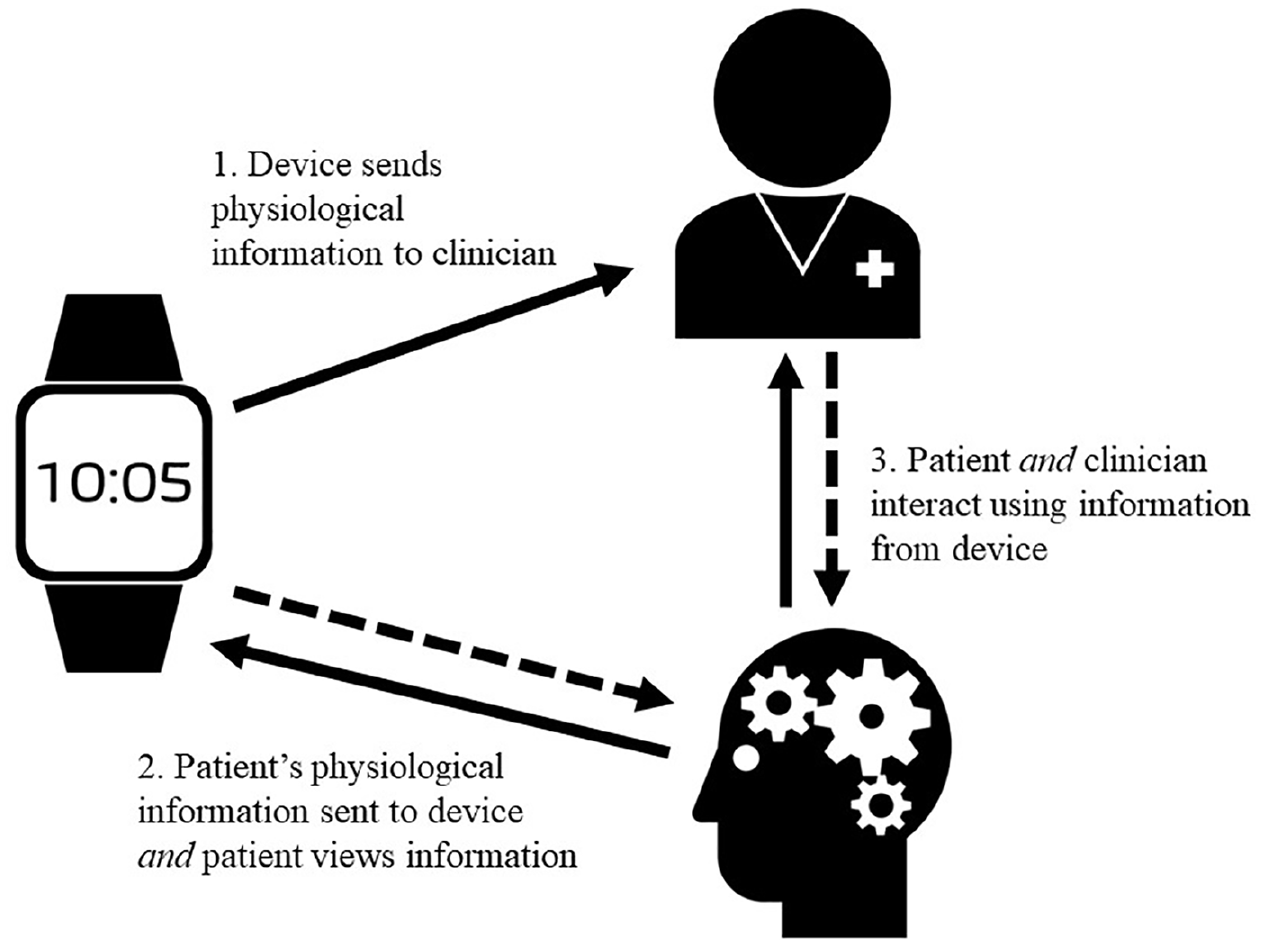
Information flow between patient, clinician and mHealth device.
Physiological stress as a signal of mental deterioration
The human body has a natural homeostatic state, which can be temporarily altered or disrupted by the effects of stress; patients with mental illness are more sensitive to the effects of stress (e.g. Shah and Malla, 2015). In healthy adults, environmental challenges are countered by a physiological stress response. The sympathetic response increases heart rate, dilates pupils and increases sweating. At the level of neurophysiology, stress is associated with neurochemical changes that enhance how the individual responds to adversity. For example, stress activates a complex neurohumeral response that helps the body adapt to challenges (De Kloet et al., 2005). Yet chronic or acute levels of stress can disrupt the body’s natural state, causing alterations to functioning, including changes in autonomic activity, circadian rhythms and physical activity. Ongoing stress can also increase vulnerability to mental health problems like psychosis, anxiety and depression (De Kloet et al., 2005). Stress changes neurotransmitter function, such as dopaminergic transmission, which is associated with positive symptoms of schizophrenia (Remington et al., 2014). The diathesis stress model posits that an underlying genetic predisposition interacts with environmental stress to cause mental illness.
Emotional states can also generate a stress response that is associated with physiological changes. The emotion does not need to be negatively valenced to have a physiological effect. For example, high levels of excitement or joy can elicit significant physiological changes. Emotion dysregulation can temporarily disrupt the body’s natural rhythms and resting state as a means of preparing for threats or challenges (see Figure 2). Being able to regulate emotion and the stress response, either psychosocially or neurochemically, is vital for remission. For example, arousal can be reduced by psychological means (e.g. relaxation techniques) or neurochemically (e.g. with tranquilizing medications). The speed with which the patient returns to their original non-stressed state, the more likely their mental health will be restored.
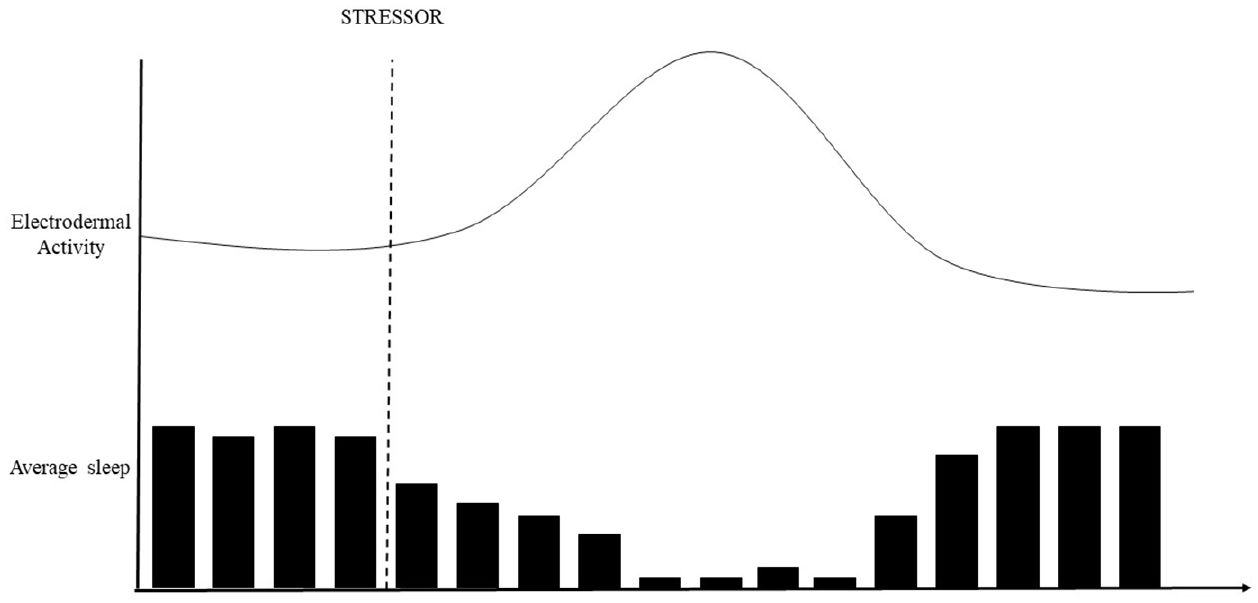
Visual representation of a temporary average increase in EDA and decreased sleep in response to a stressor.
Activity as a marker of mental deterioration
Activity monitoring has become commonplace among the health conscious general public. It has also been used for psychiatric patients who are vulnerable to weight gain due to their lifestyle and medication side effects (Naslund et al., 2016). Actigraphy, the measurement of rest and activity, is a commonly monitored biophysical marker that can be associated with changes in emotional state. mHealth devices typically come with an accelerometer which detects acceleration across three axes. This movement data can be transformed into relevant variables, such as steps counts and periods of rest. Changes in movement and activity may reflect changes in mental state. For example, depressive states are often characterized by periods of inactivity and slowed movement.
Stress can also disrupt sleep patterns and circadian rhythms. While the relationship between sleep and mental state is complex and bidirectional, sleep disruption is associated with a variety of mental health problems (e.g. Harvey et al., 2011). Sleep quality is reliably associated with mental illnesses such as depression, anxiety, trauma and bipolar disorder (Harvey et al., 2011). Examining resting time and physical activity in combination may be particularly informative. For example, manic states are associated with hyperactivity and reduced need for sleep. Depression can be associated with hypersomnia and reduced activity
EDA as a marker of mental deterioration
The autonomic nervous system (ANS) maintains the body’s homeostatic state, with the sympathetic and parasympathetic systems interacting to modulate arousal. Alterations in ANS functioning are indicative of stress and can be associated with mental illness (e.g. Shah and Malla, 2015). Stress on the ANS causes the secretion of small amounts of perspiration on the skin from the eccrine sweat glands. This perspiration has a direct relationship to the sympathetic nervous system which controls the fight or flight response and enables thermoregulation during high stress events (Boucsein, 2012). EDA is a peripheral measure of the electrical conductivity of the skin, which is modulated by autonomic arousal and regulated by the hypothalamus (Boucsein, 2012). It can be measured by placing two electrodes between emotionally sensitive body locations, applying a constant low voltage and measuring the electrical current between the body parts. There are two types of EDA measurements that are mediated by different neural structures (1) tonic: slow moving baseline levels of EDA, and (2) phasic: fast changing skin conductance responses due to external stimuli, such as noises.
While EDA can be increased by physical exertion, it can also be triggered by high levels of emotion, such as excitement or fear (Boucsein, 2012). For example, sweat glands on the palms and soles respond almost exclusively to emotional stimuli associated with fear or anxiety and they are mediated by their relevant brain structures (Boucsein, 2012). Because EDA has a direct relationship with the sympathetic nervous system, it can measure emotional response, yet it cannot be consciously faked or altered. While results are not equivocal, different types of psychopathology are associated with different EDA profiles. Psychotic disorders are typically overly sensitive to the effects of stress and result in difficulty modulating arousal (e.g. Remington et al., 2014). A potentially useful EDA marker of psychotic disorders is nonhabituation following a stimulus (see Boucsein, 2012). Affective disorders may also be associated with differing EDA profiles. For example, depressive disorders have been shown to be associated with EDA hypoarousal (Vahey and Becerra, 2015). Sarchiapone et al. (2018) also found EDA hypoactivity was a reliable feature of depression and suicide risk. By contrast, there is evidence fear and anxiety can be associated with increased EDA (Boucsein, 2012). While EDA is associated with mental state, it is not necessarily causal and can be influenced by other factors including startle or physical activity. This highlights the importance of considering EDA in the context of other measurements and self-report.
Heart rate and skin temperature as physiological markers
Similar to EDA, the functioning of the heart reflects the functioning of the ANS. Heart rate varies in response to stressors and their associated emotions such as fear or excitement. An important biomarker for mental illness is heart rate variability (HRV), which describes variation in time between heartbeats. HRV represents the parasympathetic nervous system’s ability to respond to physiological and environmental stimuli. Low HRV indicates impaired homeostatic and regulatory ANS function (Beauchaine and Thayer, 2015). It is a marker for psychopathology and poor stress response (Beauchaine and Thayer, 2015). A mHealth device can also measure body temperature. While there are fewer studies examining the relationship between skin temperature and mental state, this could be informative in its relationship to other biometrics, such as levels of sleep and activity.
Continuous measurement using a mHealth device
There are several advantages to taking patient measurements continuously rather than during discrete periods. First, patient data can be compared and tracked when they are well versus unwell, so in terms of measurement, patients can act as their own control. Absolute scores and group difference are less important than changes within an individual. Second, the high volumes of data from continuous measurement provide a closer approximation to true scores. Inaccuracy can be partially overcome by large data sets that self-correct (Iniesta et al., 2016). For example, machine learning, a subfield of artificial intelligence, can be used to detect mental deterioration using subtle changes in physiological cues (e.g. Sano et al., 2018). Finally, the data are ecologically valid and measured in real time. Clinicians can potentially observe the effects of stress as it occurs in the patient’s daily life. Data can be validated against ecological momentary assessments where a mHealth device gathers subjective ratings of a participant’s mental state in real time (e.g. Bell and Alvarez-Jimenez, 2019).
Continuous activity and EDA data from wearable devices has shown promise in predicting stress and mental deterioration. Devices can also potentially take ongoing passive measurements, such as phone calls, location, social media usage, characteristics of voice and speech to predict relapse (e.g. Insel, 2017). Sano et al. (2015) found sensors from a wearable device (e.g. actigraphy and skin conductance) and mobile phone data (e.g. phone calls and screen time) predicted high versus low academic performance, sleep quality, mental health and perceived stress with 67–92% accuracy. More recently, Sano et al. (2018) examined whether physiological and behavioural measures collected by wearable sensors and mobile phones could predict stress and poor mental health, this time in a larger student sample (
mHealth devices have also been used to detect mental deterioration in clinical samples. For example, Cella et al. (2018) used a wearable device to compare heart rate, EDA and movement for 30 participants with schizophrenia and 25 controls. Compared to controls, participants with schizophrenia showed lower HRV, movement and functioning, consistent with ANS dysregulation. The participant’s positive symptoms were also correlated with parasympathetic deregulation. Cella et al. (2019) then asked 15 participants with first-episode psychosis to wear a mHealth device continuously for 10 days. They measured HRV and EDA while participants self-reported psychotic symptoms on a mobile phone application. Participants reported higher EDA levels during periods when they were experiencing distressing psychotic symptoms. These studies generally indicate the approach is feasible and provide some evidence physiological measures are associated with mental deterioration.
Integrating a mHealth device into a youth mental health team
Severe mental illness such as schizophrenia, bipolar disorder and borderline personality disorder are chronic, highly disabling and have their peak period of onset during early adulthood. While it is important to engage every age group, younger patients who are technologically savvy and at higher risk may particularly benefit from using a mHealth device. The majority of online interventions use treatments like cognitive behaviour therapy, social networking sites and/or apps to influence behaviour. The integration of data from mHealth devices will allow more active use of technology in care. For example, recent studies using a mHealth device to measure activity, sleep and EDA demonstrated the feasibility and acceptability for monitoring severe mental illness both in the short term (Cella et al., 2018) and in the long term (Byrne et al., 2019). The latter study showed over three quarters of the participants wore a mHealth device for clinically meaningful periods over the 5-month pilot phase (i.e. 15 out of 19 wore the device reliably for greater than 2 weeks).
Challenges facing this approach: implementation and analysis
mHealth devices can collect large volumes of physiological data, but making this data clinically relevant is vital. For example, while the body and mind are connected, physiological stress will not always predict mental deterioration. Physical activity, time of day, the use of illicit or prescribed drugs can also influence mHealth biometrics. For example, sleep can also be disrupted by physical health problems or pain. Learning how to manage, process and then analyse the volumes of data for markers is important. For example, Cella et al. (2018) investigated the autonomic signature associated with schizophrenia to examine if there were markers specific to this group. Ideally, there should be a period of data collection to determine the profile of an individual, as well as the incorporation of individual difference variables (e.g. Iniesta et al., 2016). The development of phenotypes using physiological data could allow for more precise psychiatric treatments; however, considerable work is required to achieve valid and clinically useful profiles (e.g. Insel, 2017). Ecological momentary assessments that use the patient’s subjective estimation of their mental state in real time or brief standardized measures could complement physiological measures, providing straightforward and accurate data. Establishing normative data in healthy versus mentally ill populations will be the gold standard; however, descriptive data such as sleep and arousal levels will also be of clinical value.
Another consideration is whether this approach will be readily adopted and adhered to by clinicians, patients and health services. The lag between research developments and their clinical uptake typically takes several years: clinicians and patients have set routines that are not easily altered. For example, patients will need to remember to wear and charge the device, which may be more difficult if they become unwell. Clinicians may feel overwhelmed by the amount of information that a mHealth device provides, so further processing to reduce it to a manageable and essential level will be needed. Yet the technical challenges of mHealth, though essential to surmount, are secondary to broader organizational and social aspects of introducing new treatments. An understanding of workplace culture is essential for the integration of new methods of care. Making sure all age groups and demographics have access to this type of intervention is another important issue. Clear leadership and support are necessary though not sufficient for engaging patients and staff in the design and implementation of new treatments.
The use of a mHealth device also raises issues regarding privacy and clinical responsibility, which could alter the relationship between clinician and patient. For example, patients may be concerned that information collected could result in an unwanted intervention; clinicians may be unsure if clinical information warrants action. Patients and clinicians will also need to feel confident that data are private and secure, so health services will need to have systems to implement this approach (e.g. Touros and Roberts, 2017). Clinicians will need to ensure that appropriate consent is gained to use a mHealth device and their therapeutic goals are aligned with their patients (Touros and Roberts, 2017).
Conclusion
Physiological indicators of stress can provide valuable insights into the unwell mind. Continuous measurement with a mHealth device now allows clinicians to monitor and interpret physiological changes which may be associated with mental deterioration. There is good evidence that this approach is feasible and preliminary findings suggest mHealth devices can measure effectively. There may be value in further testing this paradigm using single case designs with healthy participants under controlled conditions, similar to Sano et al. (2018). The use of a mHealth device may be particularly valuable in certain vulnerable populations, such as acutely unwell inpatients needing close observation or young adults who are tech savvy. Managing and meaningfully interpreting the large data sets will be vital, as will incorporating it into clinical practice. Ethical issues regarding consent, privacy and alignment of therapeutic goals between patient and clinician are vital to consider. Yet mHealth devices clearly have potential to improve care by enhancing early intervention and better connecting patients and clinicians. While the use of mHealth devices to manage mental illness is in its infancy, the approach has potential and should be thoroughly investigated.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: A.H. has received consultancy fees from Janssen Australia, Lundbeck Australia and Seqirus. He has received payments for educational sessions run for Janssen Australia and Lundbeck Australia. He has developed educational material for Servier. He is the recipient of an investigator-initiated grant from the Balnaves Foundation and Takeda Pharmaceutical Company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project was provided by the Balnaves Foundation and Western Sydney Local Health District.
