Abstract
Objective:
Adverse childhood experiences are linked to the development of a number of psychiatric illnesses in adulthood. Our study examined the pattern of adverse childhood experiences and their relation to the age of onset of major psychiatric conditions in individuals from families that had ⩾2 first-degree relatives with major psychiatric conditions (multiplex families), identified as part of an ongoing longitudinal study.
Methods:
Our sample consisted of 509 individuals from 215 families. Of these, 268 were affected, i.e., diagnosed with bipolar disorder (n = 61), obsessive–compulsive disorder (n = 58), schizophrenia (n = 52), substance dependence (n = 59) or co-occurring diagnoses (n = 38), while 241 were at-risk first-degree relatives who were either unaffected (n = 210) or had other depressive or anxiety disorders (n = 31). All individuals were evaluated using the Adverse Childhood Experiences – International Questionnaire and total adverse childhood experiences exposure and severity scores were calculated.
Results:
It was seen that affected males, as a group, had the greatest adverse childhood experiences exposure and severity scores in our sample. A Cox mixed effects model fit by gender revealed that a higher total adverse childhood experiences severity score was associated with significantly increased risk for an earlier age of onset of psychiatric diagnoses in males. A similar model that evaluated the interaction of diagnosis revealed an earlier age of onset in obsessive–compulsive disorder and substance dependence, but not in schizophrenia and bipolar disorder.
Conclusion:
Our study indicates that adverse childhood experiences were associated with an earlier onset of major psychiatric conditions in men and individuals diagnosed with obsessive–compulsive disorder and substance dependence. Ongoing longitudinal assessments in first-degree relatives from these families are expected to identify mechanisms underlying this relationship.
Keywords
Introduction
Adverse childhood experiences (ACE) occur often (Kessler et al., 2010; Soares et al., 2016) and have been linked to a broad range of negative outcomes, both in terms of mental (Edwards et al., 2003; Van der Kolk, 2017) and physical health (Anda et al., 2006; Van der Kolk, 2017), as well as quality of life and life experiences (Chapman et al., 2011; Hillis et al., 2004). Overall, with respect to mental health, individuals who reported being physically and sexually abused as children were found to have more psychiatric conditions as adults (Jasinski et al., 2000; Leverich et al., 2002). Women who reported being victims of childhood sexual assault were found later to report greater levels of anxiety, anger regulation, paranoid ideation and obsessive–compulsive symptoms (Murphy et al., 1988).
In terms of specific psychiatric disorders, a number of ACE have been associated with the severity of specific disorders such as addiction, major depression and obsessive–compulsive disorder (OCD) (Brodsky et al., 2001; Dube et al., 2003; Lochner et al., 2002). The association between ACE and the onset of bipolar disorder (BD) and schizophrenia has also been previously investigated, with the presence of ACE being associated with an increased risk of psychosis (Etain et al., 2013; Read et al., 2005; Watson et al., 2014). Exposure to certain types of ACE, as well as ACE at a greater intensity, may be related to an earlier onset age of psychiatric conditions, such as substance dependence (SUD), depression and schizophrenia (Bernet and Stein, 1999; Etain et al., 2013; Li et al., 2012; Read et al., 2005), which may, in turn, indicate a more severe phenotype (Kessler et al., 2007). Early exposure to trauma also appears to increase the risk of psychotic symptoms in at-risk adolescents (Spauwen et al., 2006).
Overall, childhood trauma seems to contribute to the future occurrence of diverse symptom clusters and possibly to an earlier age of onset of illness. In this study, we examine the effect of ACE on the age of onset of different psychiatric syndromes. A trans-diagnostic approach could help us to understand the differential effect of ACE on the age of onset of such syndromes. In addition, evaluating individuals from multiplex families with a pre-existing genetic loading for psychiatric illness (see in ‘Methods’ section the source of data) might inform us about the role of ACE in increasing the risk for an earlier occurrence of illness in vulnerable individuals.
This study describes the pattern of ACE in affected individuals and at-risk first-degree relatives (FDRs) from multiplex families with major psychiatric disorders (schizophrenia, BD, SUD, OCD) and its relationship to the age of onset of the disorders. We hypothesised that ACE would influence the age of onset of different psychiatric syndromes in individuals from multiplex families.
Methods
Procedure
This study draws on data from the Accelerator program for Discovery in Brain disorders using Stem cells (ADBS) project, an ongoing longitudinal study at the National Institute of Mental Health and Neurosciences (NIMHANS), the National Centre for Biological Sciences (NCBS) and the Institute for Stem Cell Science and Regenerative Medicine (InStem), that began in 2016 (Viswanath et al., 2018). The project was reviewed and approved by the Institutional Ethics Review Board and written informed consent was obtained from all individuals who were recruited.
The ADBS project includes families in whom multiple members (at least two affected FDRs in a nuclear family) are diagnosed to have a major psychiatric disorder (schizophrenia, BD, OCD, Alzheimer’s dementia and SUD). As part of the first wave, participants enrolled undergo two levels of assessment – a brief assessment and the neurodevelopmental endo-phenotype (deep) assessment. The brief assessment consists of sociodemographic information, information about the onset and progression of the illness and physical examination to take note of any pre-existing medical conditions. It also includes measures for cognitive functioning and current illness severity. The psychiatric diagnoses are corroborated by two trained psychiatrists using the Mini International Neuropsychiatric Interview (MINI) (Sheehan et al., 1998), which is administered during the brief assessments. Further clinical evaluation, including assessment of temperament, personality, ACE, life events, handedness, global functioning and psychopathology specific scales, is done on participants who consent for deep assessments. Participants for whom deep assessments have been conducted are re-assessed 2 years after the baseline assessment as part of the second wave of the study.
The present analysis only includes data from the first wave of the study. Individuals for whom complete sociodemographic and clinical information (including age of onset of the syndrome, gender, maximum education attained and psychiatric diagnosis) and Adverse Childhood Experiences – International Questionnaire (ACE-IQ) form (World Health Organization [WHO], 2011) were available were included. Individuals affected with Alzheimer’s dementia were excluded from the study, as most were unable to complete the ACE-IQ form. A total of 509 participants (affected individuals and at-risk FDRs) met these criteria from the data available in the first wave of the ADBS project. For this study, we divided the affected individuals into five groups by diagnosis: BD, OCD, schizophrenia, SUD and co-occurring diagnoses group (American Psychiatric Association, 2000). Co-occurring diagnoses were defined as the presence of more than one lifetime Axis I diagnosis of BD, SUD, OCD or schizophrenia. At-risk FDRs consisted of individuals with no Axis I diagnoses, as well as individuals with Axis I diagnoses other than the diagnostic groups described above.
Measures
Age of Onset was defined as the age at which individuals fulfilled Diagnostic and Statistical Manual of Mental Disorders (4th edition, text revision; DSM-IV-TR) criteria for any of the four disorders, as determined by the psychiatrist, based on the information obtained from the patient and their family members. The information obtained was also verified from hospital records.
ACE were assessed using the WHO ACE-IQ questionnaire. It consists of 31 questions across 13 subdomains: physical abuse; emotional abuse; contact sexual abuse; alcohol and/or drug abuser in the household; incarcerated household member; household member with a psychiatric condition or suicidality; household member treated violently; one or no parents, parental separation or divorce; emotional neglect; physical neglect; bullying; community violence; and collective violence (WHO, 2011).
A total ACE exposure score was calculated by adding the total number of subdomains where any adversity was reported irrespective of the frequency at which it might have occurred. A total ACE severity score was also calculated by adding the total number of subdomains where adversity was reported to have occurred above a predefined frequency threshold. This threshold differed by the subdomain of ACE reported (e.g. contact sexual abuse only requires being touched sexually once, but emotional abuse requires being screamed at many times) (WHO, 2011).
The total ACE exposure score is sensitive and identifies the occurrence of any adversity. The total ACE severity score is specific and only detects more severe and potentially impactful adversity. Therefore, the total ACE severity score was used for this analysis. Since there were no major differences in the results noted in the analyses performed using the two different scores, the total ACE exposure score–related analyses are presented as supplementary information (Supplementary text, Supplementary Figures 1 and 2, Supplementary Tables 2 and 3).
Statistical analyses
Means, medians and proportions were used to describe the sociodemographic details and ACE scores of the study sample. Generalised linear mixed effects Poisson regression was used to examine group-by-gender interaction for ACE scores, after accounting for correlated ACE between family members.
We applied mixed effects survival analysis methodology to model the age of onset, using correlated frailty models to account for correlated ACE between family members. This was done by the use of family ID as a random effects confounding variable in the model. Family ID was a label used to denote all individuals from a single family. In the analysis, we allowed each family to have their own intercept, with the assumption that some families may have correlated ACE, while others may not. This family-wise effect was then controlled for in the analysis. This was done to ensure that there are no families that unduly over- or underestimate the overall effects of ACE on age of onset. In addition, we used a random effects model to reduce the effects of two other potential confounders of ACE on the age of onset of illness; these included stressor at the onset of illness (coded present–absent) and cohort effect (three groups based on the age of assessment distribution fractiles at 1/3 and 2/3 levels).
The survival analyses model age of onset as a function of time and takes into account that some records are uncensored reflecting the actual age of onset (affected individuals) and some are censored at the age last seen as unaffected (at-risk FDRs). The age last seen as unaffected was the age at which these individuals were seen and assessed as a part of the first wave of the study. Separate Cox mixed effects models were fit to examine the effects of ACE scores, ACE-by-Gender interactions and ACE-by-Diagnosis interactions on the age of onset of psychiatric illness. The hazard ratios (HRs) provide the instantaneous risk of becoming affected at age ‘t’, given that an individual was not affected at age (t − 1) as an effect of an increase in ACE scores. In each case, the models were also repeated after excluding individuals with age of onset <18 years.
All analyses were performed in the R environment for statistical computing, version 3.5.2, using the packages ‘lme4’ and ‘coxme’.
Results
Sociodemographic and clinical details
This study sample consisted of 509 individuals (43.2% females) from 215 different families. Of the participants, 268 were affected (36.6% females), while 241 were at-risk FDRs (50.6% females). Supplementary Table 1 describes the sociodemographic characteristics of the individuals in the sample. The mean age of onset for the whole sample was 31.8 ± 14.1 years. The mean age of onset for men was 29.8 ± 13.9 years and for women was 34.4 ± 14.0 years. Table 1 describes clinical details of the sample by diagnosis.
Clinical profile of the sample.
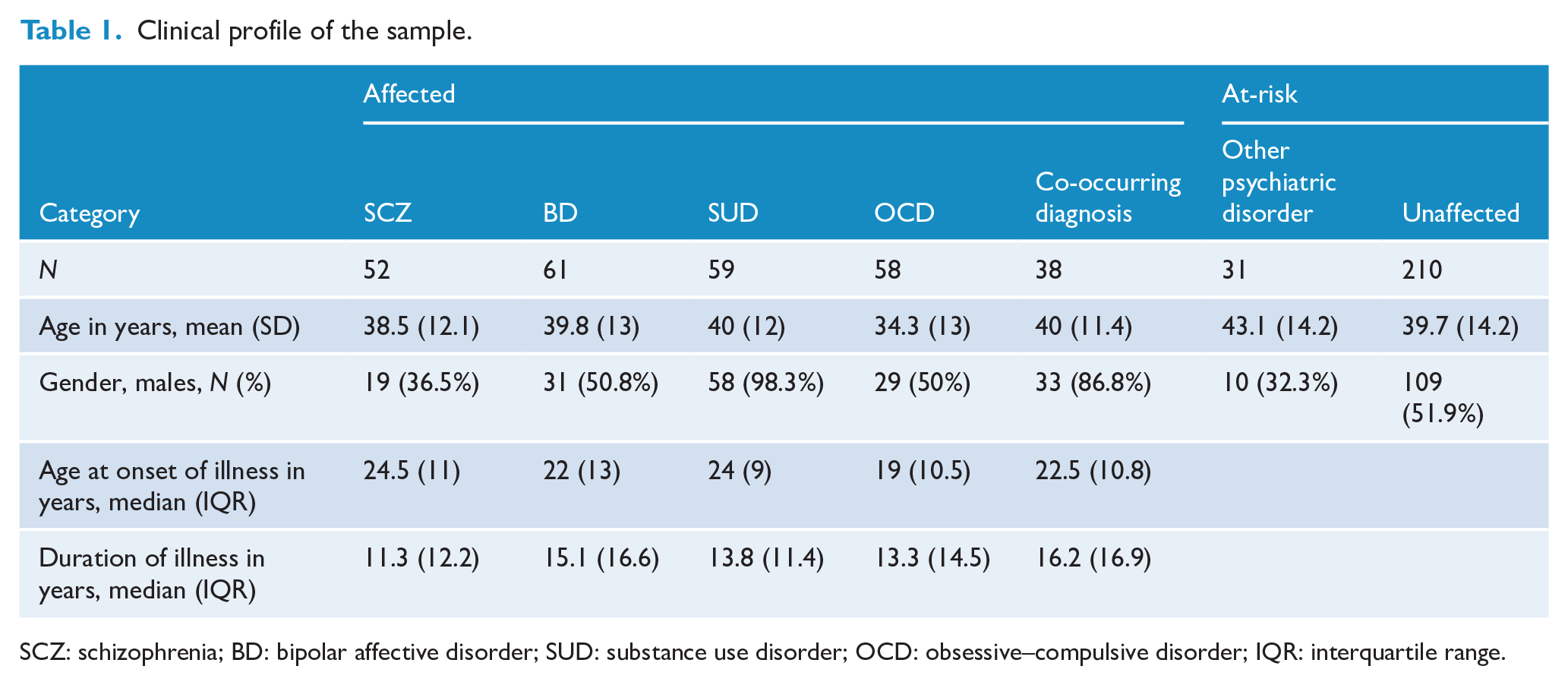
SCZ: schizophrenia; BD: bipolar affective disorder; SUD: substance use disorder; OCD: obsessive–compulsive disorder; IQR: interquartile range.
Description of total ACE severity score
A total of 404 out of 509 (79.4%) experienced at least one ACE above the pre-determined frequency threshold (i.e. total ACE severity score ⩾1) (Figure 1(A)). The mean (SD) total ACE severity scores for individuals with BD, schizophrenia, OCD, SUD and co-occurring diagnoses were 1.9 (1.6), 2.0 (2), 1.71 (1.4), 3.8 (2.5) and 2.73 (1.8), respectively. Figure 1(B) describes the distribution of total ACE severity scores in the affected group split by diagnosis.
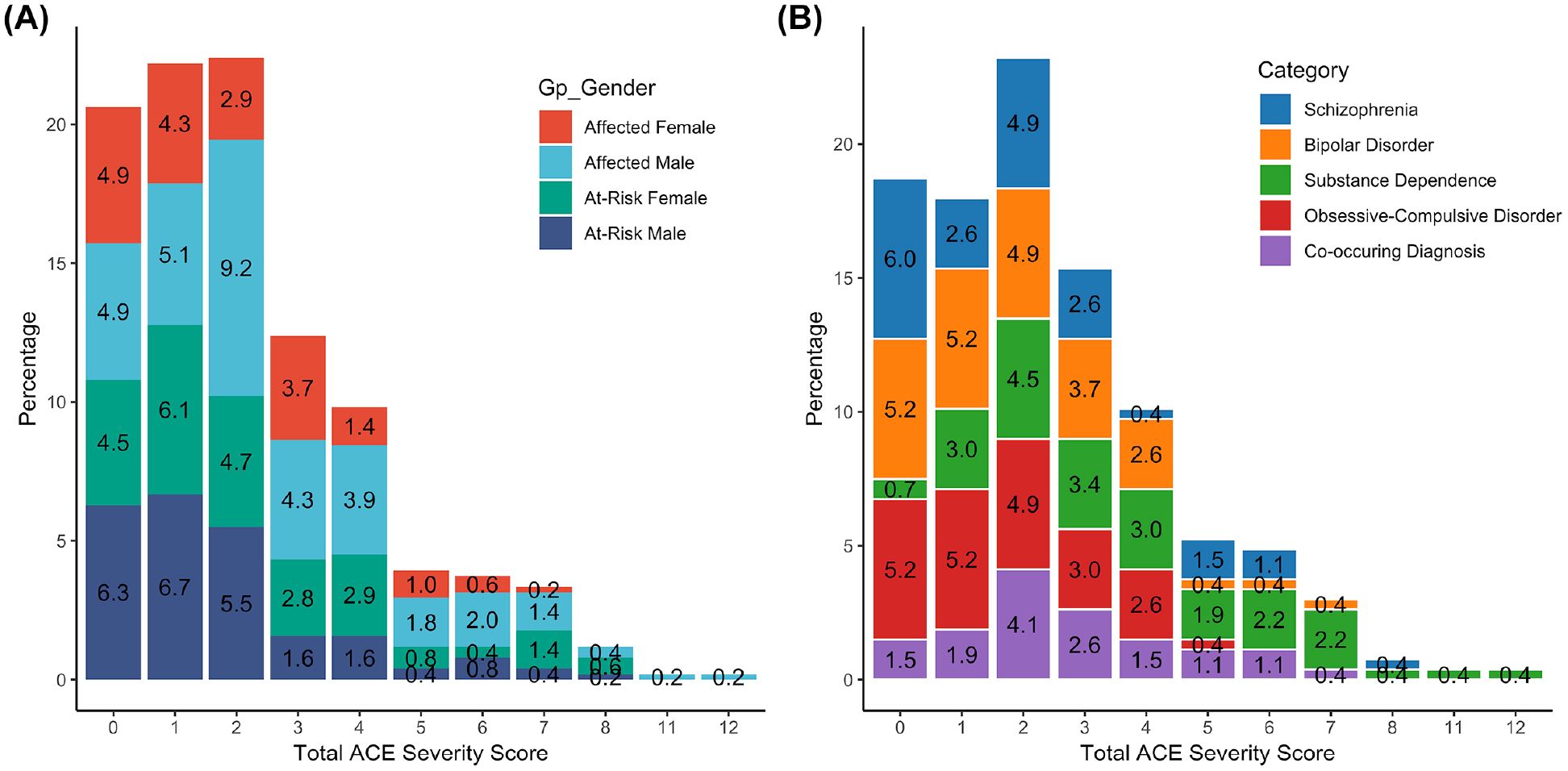
Distribution of total ACE severity scores for (A) affected individuals and at-risk first-degree relatives split by gender and (B) affected individuals split by diagnosis.
Incidence rate ± 95% confidence interval (CI) is back-transformed from the log scale and represented by dot and blue bars. The red arrows are for the post hoc tukey comparisons, with overlaps in the arrow indicating that the between-group difference for that comparisons are not significant.
Figure 2 describes the incidence rate (95% confidence interval [CI]) of total ACE severity score in affected and at-risk groups split by gender. Affected males had significantly greater total ACE severity scores compared to affected females, at-risk males or females.
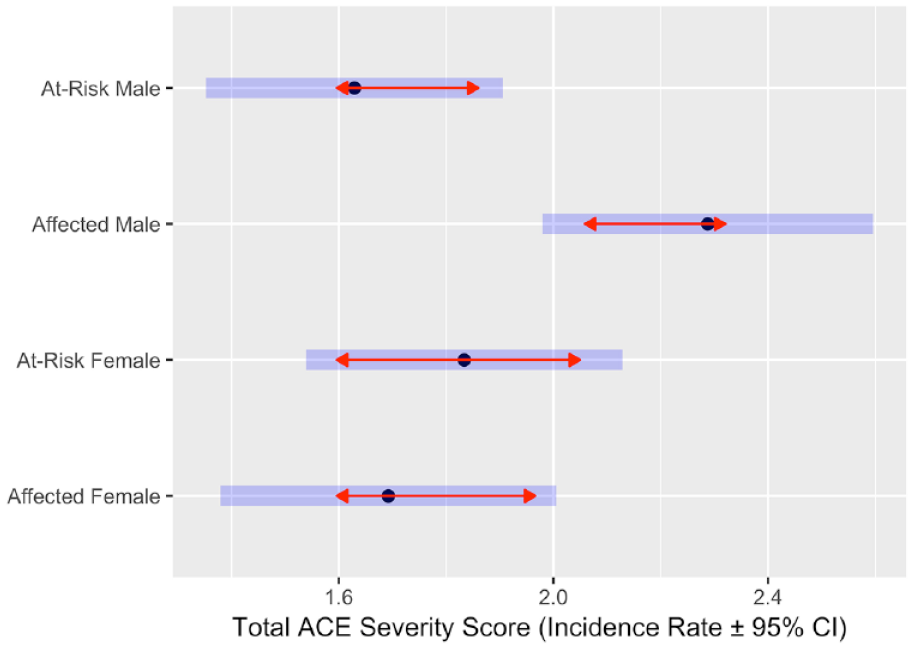
Incidence rate ± 95% confidence interval (CI) for the Group-by-Gender interaction for total ACE severity score. The results indicate significantly greater incidence rates of total ACE severity score for affected males.
Incidence rate ± 95% confidence interval (CI) is back-transformed from the log scale and represented by dot and blue bars. The red arrows are for the post hoc tukey comparisons, with overlaps in the arrow indicating that the between-group difference for that comparisons are not significant.
Association between age of onset and total ACE severity score
Model 1 investigated the effects of ACE on the age of onset of psychiatric illnesses and found that greater total ACE severity score (HR = 1.076, 95% CI = 1.017, 1.138) significantly increased the risk for an earlier age of onset across all disorders (Table 2). As the ACE-IQ form pertains to incidents occurring below the age of 18, we also repeated the analysis after excluding participants with an onset age ⩽18 years. This was to adjust for the influence of ACEs that may have occurred as a consequence of the illness. The associations remained significant for the total ACE severity scores (HR = 1.115, 95% CI = 1.043, 1.193).
Results from mixed effects Cox models (1 and 2) to examine the relationship between ACE scores, ACE-by-Gender interactions on the Age-at-Onset (AAO) of psychiatric illness.
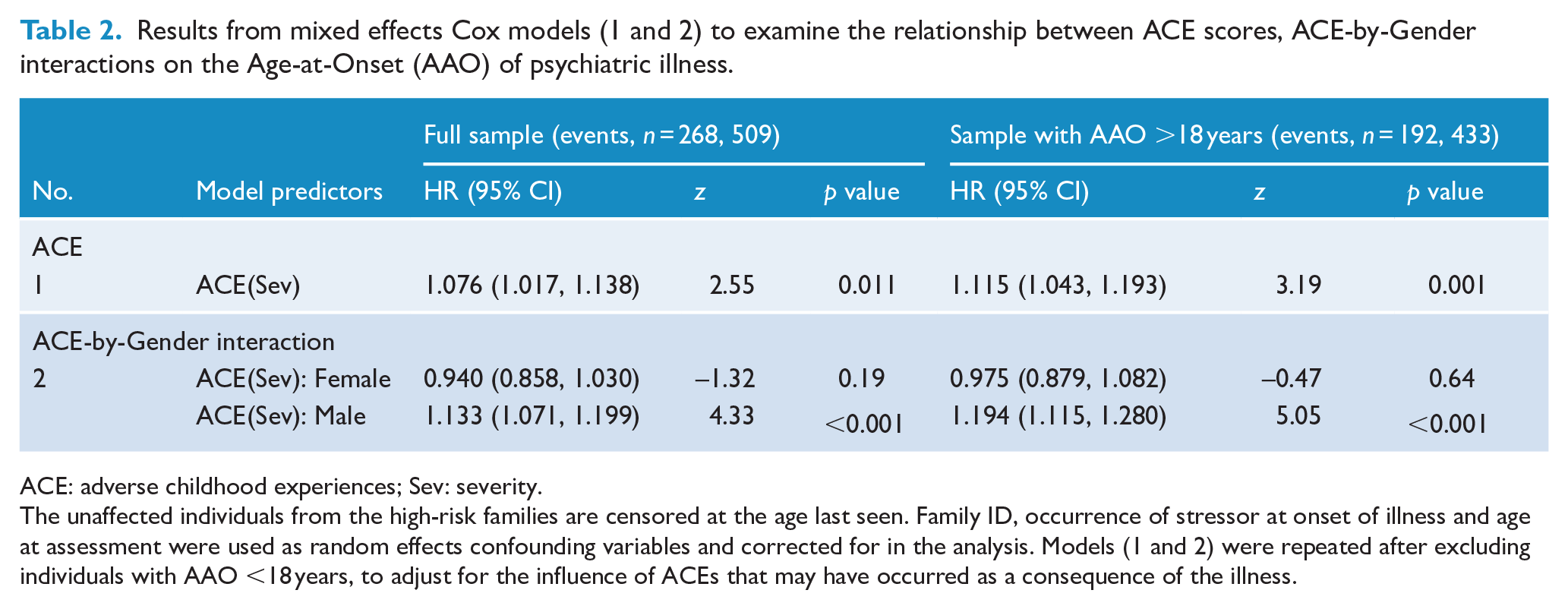
ACE: adverse childhood experiences; Sev: severity.
The unaffected individuals from the high-risk families are censored at the age last seen. Family ID, occurrence of stressor at onset of illness and age at assessment were used as random effects confounding variables and corrected for in the analysis. Models (1 and 2) were repeated after excluding individuals with AAO <18 years, to adjust for the influence of ACEs that may have occurred as a consequence of the illness.
Association between age of onset and ACE-by-Gender interactions
Model 2 investigated the effects of ACE-by-Gender interactions on age of onset of psychiatric illness and found that greater total ACE severity scores (HR = 1.133, 95% CI = 1.071, 1.199) significantly increased the risk for an earlier age of onset only in males and not in females (Table 2). The same held true even on excluding individuals with onset age ⩽18 years. As there was a clear gender skew in individuals with SUD, this analysis was redone after excluding the SUD group; the gender difference continued to persist.
Association between age of onset and ACE-by-Diagnosis interactions
Model 3 investigated the effects of ACE-by-Diagnosis interactions on the age of onset of psychiatric illness (Table 3). It was found that greater total ACE severity score (HR = 1.262, 95% CI = 1.102, 1.446) significantly increased the risk for an earlier age of onset in individuals with OCD both in the full sample and after exclusion of individuals with onset age ⩽18 years. This finding was also seen in individuals with co-occurring diagnoses (HR = 1.125, 95% CI = 1.010, 1.254) but only in the full sample. In individuals with SUD, greater total ACE severity scores (HR = 1.089, 95% CI = 1.006, 1.180) predicted earlier age of onset in only in those with an onset age >18 years and not in the full sample.
Results from mixed effects Cox model 3 to examine the relationship between ACE-by-Diagnosis interaction on the Age-at-Onset (AAO) of psychiatric illness.
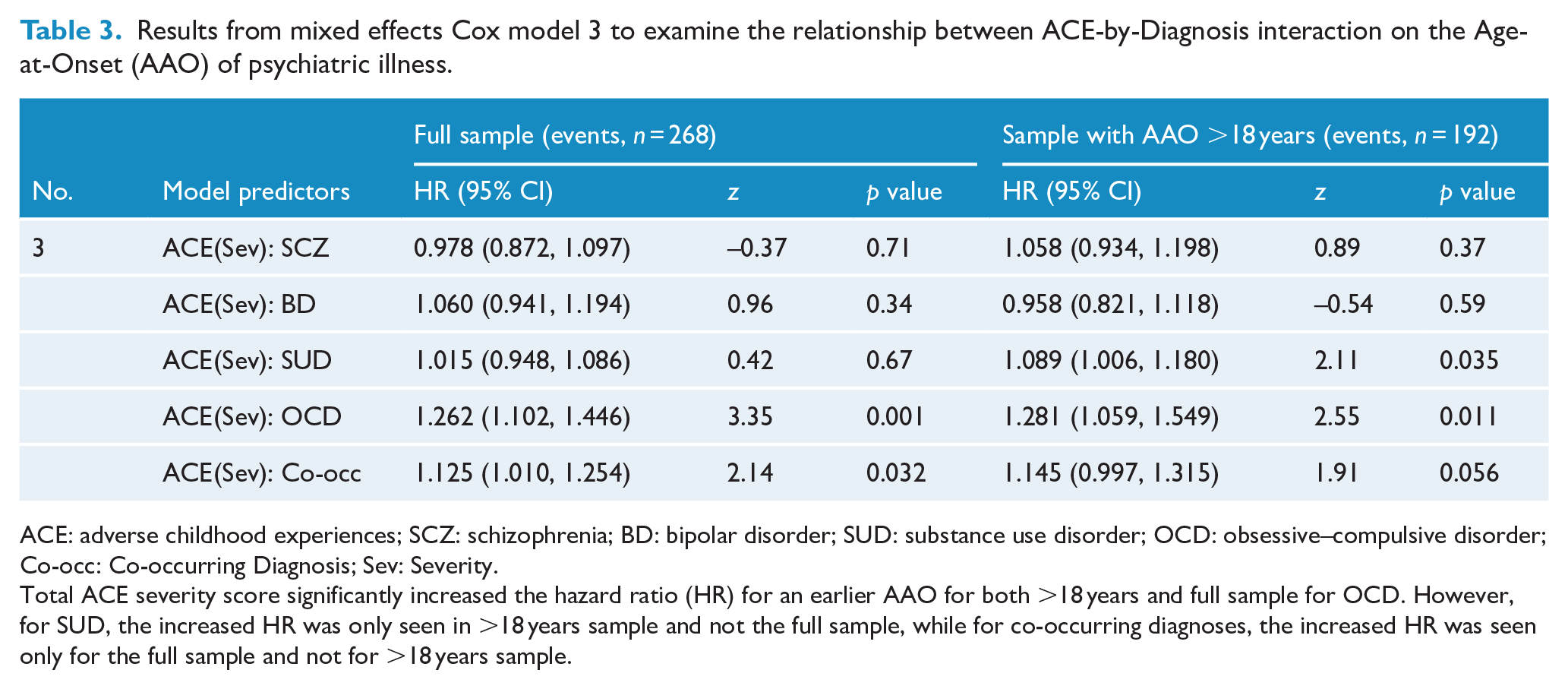
ACE: adverse childhood experiences; SCZ: schizophrenia; BD: bipolar disorder; SUD: substance use disorder; OCD: obsessive–compulsive disorder; Co-occ: Co-occurring Diagnosis; Sev: Severity.
Total ACE severity score significantly increased the hazard ratio (HR) for an earlier AAO for both >18 years and full sample for OCD. However, for SUD, the increased HR was only seen in >18 years sample and not the full sample, while for co-occurring diagnoses, the increased HR was seen only for the full sample and not for >18 years sample.
Discussion
Our study evaluated the pattern of ACE in affected and at-risk FDRs from multiplex families across diagnostic groups. Furthermore, we investigated the relationship between ACE and the age of onset of psychiatric illness. Our primary findings are that ACE were associated with an earlier onset of major psychiatric conditions in men and specifically in individuals diagnosed with OCD and SUD.
Both a greater exposure to and severity of ACE increased the risk for an earlier age of onset of psychiatric illness in our sample. Although these findings are in line with previous research, our findings assume importance because most previous studies have used the presence or absence of illness as the variable in question (Bernet and Stein, 1999; Edwards et al., 2003; Felitti et al., 2019; Van der Kolk, 2003). Age of onset represents a more complex phenotype than merely the presence or absence of the illness. Also, our study is one of the first studies to examine the impact of ACE on mental illness emerging from the context of South Asia.
Specifically, the relationship between severity of ACE and risk for an earlier age of onset was seen in individuals with OCD, where the risk was 26.2%, co-occurring diagnoses where the risk was 12.5% and SUD where the risk was 8.9% (when the age of onset of dependence was >18 years). The relationship between ACE and OCD is important because the literature in this area is scarce and inconclusive (Selvi et al., 2012). Some studies report no association between ACE and OCD and few others support higher ACE in those with OCD, but possibly mediated by coexisting affective, anxiety, substance use and eating disorders (Benedetti et al., 2014; Briggs and Price, 2009; Visser et al., 2014). In our study, the association with age of onset survived even after controlling for early onset OCD, implying the effect of ACE on the onset of OCD was irrespective of age of onset. The relationship between ACE and SUD is better examined and our findings confirm the observations made in previous studies (Anda et al., 2006; Dube et al., 2003, 2006), which demonstrate a clear link between adversity and development of SUD as well as earlier age of onset. However, in contrast to OCD in individuals with SUD, the relationship was seen only in individuals who had an age of onset of dependence after 18 years of age. The above findings make sense given that anxiety, obsessionality and addiction are known to have a complex pattern of clustering in families, with non-genetic and learning factors, apart from genetic factors, being strongly implicated (Craske et al., 2017; Mataix-Cols et al., 2013; Merikangas et al., 1998).
Interestingly, the risk for an earlier age of onset attributable to ACE was not seen in individuals with schizophrenia and BD. This is in contrast to previous literature where childhood trauma is associated with greater illness severity in individuals with psychosis and BD (Etain et al., 2013; Read et al., 2005). We know that the heritability estimates in schizophrenia (Hilker et al., 2018) and BD (McGuffin et al., 2003) are greater than those for OCD (Browne et al., 2014) and SUD (Ducci and Goldman, 2012). We speculate that in our sample of individuals from densely affected families, the impact of ACE in influencing the age of onset may be greater in OCD and SUD, which are disorders where genetic factors overall may play a smaller role in comparison with disorders such as schizophrenia and BD. However, it was also noted that in individuals with a very early onset of SUD (<18 years of age), the experience of adverse events was not a significant contributor. One may speculate that this subset of individuals may have stronger genetic underpinnings, and in that respect may be more similar to individuals with schizophrenia and BD. ACE also predicted earlier age of onset in individuals with co-occurring diagnoses, but this did not persist after the exclusion of individuals with an age of onset before 18 years, indicating that greater ACE may have been a consequence, rather than antecedent, of the illness.
Our study observed that exposure to and severity of ACE reported were higher for affected individuals than for at-risk individuals, an observation which is in line with previous studies (Dube et al., 2005; Felitti et al., 2019; Leverich et al., 2002). However, all these studies were case–control studies, unlike our study which is based on multiple affected families. Given that both affected and at-risk individuals in our study shared similar family environments, this difference is of clear interest, as it indicates that ACE may have additive interactions with genetic risk in such families. It could also be attributable to a greater recall of ACE by affected individuals. It may also have been because individuals who develop psychiatric illness in adulthood may manifest pre-clinical symptoms in childhood and adolescence, which exposes them to a greater risk of experiencing adversity (Howes and Murray, 2014).
Gender differences in the prevalence of ACE were also noted in our sample, with affected men reporting a greater exposure to and severity of ACE than affected women. Gender differences in the types of ACE experienced have been previously described, with women more often reporting sexual abuse and household violence and men reporting physical abuse (Edwards et al., 2003). However, socio-cultural factors may also explain some of the differences seen in our sample, as it is known that women in the Indian context often do not reveal traumatic experiences owing to stigma and may instead express the same through somatic symptoms, which is a cultural idiom of distress (Desai and Chaturvedi, 2017). The association between age of onset and ACE in our sample was also seen in men but not in women. A study on a community sample found that males who underwent ACE were more likely to develop antisocial behaviours, including substance use, in adulthood as compared to females for whom affective illnesses were more common (Schilling et al., 2007). This could be related to the fact that males with genetic loading for externalising disorders are known to present with more behavioural deviance, and therefore more chastisement by family and alienation from peers, as compared to females who are more likely to develop internalising disorders (Cameron et al., 2017). Since depression and dysthymia were not among the conditions under study in our sample, we were concerned that these differences may have reflected a selection bias. However, the gender differences in age of onset persisted even upon the exclusion of individuals with SUD.
The ACE seen in affected individuals in our sample drawn from multiplex families is similar to the ACE reported by affected individuals in previous literature (Anda et al., 1999; Chapman et al., 2004; Kiburi et al., 2018; Mersky et al., 2013; Whitfield et al., 2005), which was somewhat unexpected. This indicates that affected individuals from multiplex families perhaps experience levels of adversity which are no different when compared to affected individuals from the general population. This suggests the crucial role that ACE may play, as an ‘extra hit’ in individuals from multiplex families, and hints to the interplay between environmental adversity during development and pre-existing vulnerability that may be mediated by gene–environment interactions. The pathways by which ACE influence the development of disease phenotypes, including epigenetics, immune-inflammatory and neuroendocrine mechanisms, need further exploration. This could have potential implications for mental health prevention in individuals from multiplex families.
Our study has some limitations. In our study, the ACE data are collected retrospectively, and thus may have introduced recall bias. While studies that have compared prospective and retrospective data collection of ACE data do not report any major differences (Patten et al., 2015), we would acknowledge this as a limitation. In addition, the possibility of cohort effects influencing the age of onset also cannot entirely be ruled out, although an attempt was made to control for the same in the analysis. There is also a lack of information in a cultural context as the ACE-IQ scale, while intended for international application, does not address critical dimensions of adversity (such as poverty and malnutrition) that may be more country-specific. In addition, the findings from multiplex families may not be generalisable to individuals without a family history of psychiatric illness.
To conclude, the findings of our study provide an understanding of the profile of childhood adversity in individuals from multiplex families from a South Asian context. They highlight how exposure to childhood adversity is associated with a younger age of occurrence of particular psychiatric conditions such as OCD and SUD in these individuals. Future analysis in our longitudinal cohort is expected to identify mechanisms underlying this relationship, specifically in individuals who are at risk for developing mental illness.
Supplemental Material
Description_of_Total_ACE_Exposure_Score – Supplemental material for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses
Supplemental material, Description_of_Total_ACE_Exposure_Score for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses by Amala Someshwar, Bharath Holla, Preeti Pansari Agarwal, Anza Thomas, Anand Jose, Boban Joseph, Birudu Raju, Hariprasad Karle, M Muthukumaran, Prabhath G Kodancha, Pramod Kumar, Preethi V Reddy, Ravi Kumar Nadella, Sanjay T Naik, Sayantanava Mitra, Sreenivasulu Mallappagiri, Vanteemar S Sreeraj, Srinivas Balachander, Suhas Ganesh, Pratima Murthy, Vivek Benegal, Janardhan YC Reddy, Sanjeev Jain, Jayant Mahadevan and Biju Viswanath in Australian & New Zealand Journal of Psychiatry
Supplemental Material
Supplementary_Figure_1 – Supplemental material for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses
Supplemental material, Supplementary_Figure_1 for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses by Amala Someshwar, Bharath Holla, Preeti Pansari Agarwal, Anza Thomas, Anand Jose, Boban Joseph, Birudu Raju, Hariprasad Karle, M Muthukumaran, Prabhath G Kodancha, Pramod Kumar, Preethi V Reddy, Ravi Kumar Nadella, Sanjay T Naik, Sayantanava Mitra, Sreenivasulu Mallappagiri, Vanteemar S Sreeraj, Srinivas Balachander, Suhas Ganesh, Pratima Murthy, Vivek Benegal, Janardhan YC Reddy, Sanjeev Jain, Jayant Mahadevan and Biju Viswanath in Australian & New Zealand Journal of Psychiatry
Supplemental Material
Supplementary_Table_2 – Supplemental material for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses
Supplemental material, Supplementary_Table_2 for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses by Amala Someshwar, Bharath Holla, Preeti Pansari Agarwal, Anza Thomas, Anand Jose, Boban Joseph, Birudu Raju, Hariprasad Karle, M Muthukumaran, Prabhath G Kodancha, Pramod Kumar, Preethi V Reddy, Ravi Kumar Nadella, Sanjay T Naik, Sayantanava Mitra, Sreenivasulu Mallappagiri, Vanteemar S Sreeraj, Srinivas Balachander, Suhas Ganesh, Pratima Murthy, Vivek Benegal, Janardhan YC Reddy, Sanjeev Jain, Jayant Mahadevan and Biju Viswanath in Australian & New Zealand Journal of Psychiatry
Supplemental Material
Supplementary_Table_3 – Supplemental material for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses
Supplemental material, Supplementary_Table_3 for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses by Amala Someshwar, Bharath Holla, Preeti Pansari Agarwal, Anza Thomas, Anand Jose, Boban Joseph, Birudu Raju, Hariprasad Karle, M Muthukumaran, Prabhath G Kodancha, Pramod Kumar, Preethi V Reddy, Ravi Kumar Nadella, Sanjay T Naik, Sayantanava Mitra, Sreenivasulu Mallappagiri, Vanteemar S Sreeraj, Srinivas Balachander, Suhas Ganesh, Pratima Murthy, Vivek Benegal, Janardhan YC Reddy, Sanjeev Jain, Jayant Mahadevan and Biju Viswanath in Australian & New Zealand Journal of Psychiatry
Supplemental Material
Supp_table_1 – Supplemental material for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses
Supplemental material, Supp_table_1 for Adverse childhood experiences in families with multiple members diagnosed to have psychiatric illnesses by Amala Someshwar, Bharath Holla, Preeti Pansari Agarwal, Anza Thomas, Anand Jose, Boban Joseph, Birudu Raju, Hariprasad Karle, M Muthukumaran, Prabhath G Kodancha, Pramod Kumar, Preethi V Reddy, Ravi Kumar Nadella, Sanjay T Naik, Sayantanava Mitra, Sreenivasulu Mallappagiri, Vanteemar S Sreeraj, Srinivas Balachander, Suhas Ganesh, Pratima Murthy, Vivek Benegal, Janardhan YC Reddy, Sanjeev Jain, Jayant Mahadevan and Biju Viswanath in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors are grateful to all the patients and their family members who participated in the study.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The authors assert that all procedures contributing to this work comply with the ethical standards laid down by the National Institute of Mental Health and Neurosciences (NIMHANS) institutional ethics committee who have approved the study protocol and with the Helsinki Declaration of 1975, as revised in 2008.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research is funded by the Accelerator program for Discovery in Brain disorders using Stem cells (ADBS) (jointly funded by the Department of Biotechnology, Government of India, and the Pratiksha trust; Grant BT/PR17316/MED/31/326/2015).
Informed Consent
Written informed consent was obtained from all the participants in the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
