Abstract
Objectives:
To review brief cognitive screening instruments for routine clinical monitoring in electroconvulsive therapy.
Methods:
Brief cognitive screening instruments specifically developed for electroconvulsive therapy and commonly used brief generalised cognitive screening instruments were reviewed with relative advantages and disadvantages highlighted.
Results:
Several brief cognitive screening tests designed for use in electroconvulsive therapy have been found sensitive for monitoring electroconvulsive therapy–related cognitive side effects. The choice of a brief generalised cognitive screening instrument for use in an electroconvulsive therapy clinical context comes with several pertinent considerations.
Conclusion:
Electroconvulsive therapy is a highly effective treatment for pharmacoresistant and severe neuropsychiatric illness although cognitive side effects can be a barrier for treatment. Routine monitoring using brief cognitive screening instruments has advantages in busy clinical settings and can assist with optimising patient outcomes. More detailed neuropsychological assessment is recommended if the results from brief cognitive screening raise concerns.
Introduction
Due to its high efficacy in pharmacoresistant major depressive disorder (MDD) and other severe neuropsychiatric illnesses, electroconvulsive therapy (ECT) remains a highly relevant and clinically important treatment (Malhi et al., 2015; National Institute for Health and Care Excellence (NICE), 2009; NSW Health, 2010; UK ECT Review Group, 2003; Weiss et al., 2019). Cognitive side effects that occur during the treatment course though remain a concern for patients and clinicians. In clinical settings, routine monitoring is warranted in order to address patients’ concerns, assist with identifying patients who are developing adverse outcomes, adjust treatment plans and identifying patients who require comprehensive clinical neuropsychological assessment.
Despite recommendations from many professional bodies (e.g. Weiss et al., 2019) and the recent reclassification of ECT devices by the United States Food and Drug Administration (US FDA, 2018), there remains no consensus regarding how best to routinely assess cognitive side effects in clinical practice. Unlike in research settings where detailed cognitive monitoring is typically undertaken, in busy clinical settings, clinicians are often faced with challenges (e.g. time, material resources, personnel) that can limit the capacity to conduct routine neuropsychological assessments. In this context, brief cognitive screening instruments have appeal as such tests require less time and expertise compared to traditional neuropsychological tests. Although some brief screening measures have been specifically developed for use in ECT, many others were developed for more general cognitive screening purposes (e.g. dementia).
Here, we provide a narrative review of currently available brief cognitive screening instruments and assess their advantages and disadvantages for use in ECT clinical settings. Factors relevant to the suitability of each test are described to assist clinicians in choosing measures appropriate for routine clinical monitoring. The review focuses on measures that primarily assess objective cognitive functioning, as subjective measures may be conflated by patients’ perceptions and show divergent results relative to objective outcomes (Berman et al., 2008; Kumar et al., 2016).
ECT-related cognitive side effects
Cognitive effects induced by a single ECT treatment and across multiple treatments have been well documented. While adverse effects have historically been considered as an unwanted, though unavoidable, consequence of treatment, significant advancements in treatment techniques have substantially minimised these while maintaining high efficacy. Nevertheless, even with the use of the unilateral electrode placement, empirical titrated dosing and modifications of the ECT stimulus waveform, cognitive side effects remain common and show substantial variability between patients (Loo et al., 2015; Sackeim, 2014; Semkovska et al., 2016).
Acute cognitive side effects
Acute cognitive effects refer to those that are temporally related to a single treatment. The most prominent acute cognitive side effect is post-ECT disorientation. With older forms of ECT (e.g. bitemporal electrode placement, sine wave pulse width), post-ECT disorientation often lasted for several hours, but with modern techniques (e.g. right unilateral electrode placement, ultra-brief pulse width), it usually resolves within approximately 30 minutes. Aspects of patients’ recovery of reorientation occur non-simultaneously, with information relating to person recovering first (e.g. name, age), followed by place (e.g. country, hospital) and then time (e.g. month, year) (Daniel et al., 1987). This pattern is similar to the temporal gradient of retrograde amnesia after acquired brain injury, where information that was most recently acquired is most vulnerable relative to memories that are more temporally distant (i.e. Ribot’s law; Ribot, 1882). When measured after every ECT treatment, the mean duration of post-ECT disorientation has been found to be moderately correlated with the magnitude of retrograde amnesia post treatment (Martin et al., 2015; Sobin et al., 1995). Post-ECT disorientation is additionally highly sensitive to variations in ECT technique, including dosing (McCall et al., 2000) and electrode montage (Sackeim et al., 2008), and patient factors including pretreatment global cognitive functioning (Sobin et al., 1995). These characteristics highlight the utility of routine monitoring of this acute side effect in ECT clinical practice.
Acute retrograde amnesia for information acquired immediately prior to the administration of a single ECT treatment is also common. These side effects have been observed across multiple different memory domains, are typically large in magnitude (Martin et al., 2019) and are sensitive to ECT treatment factors, including electrode montage (Prudic et al., 1994), dose (Sackeim et al., 1993) and pulse width (Loo et al., 2015; Sackeim et al., 2008). While acute cognitive side effects other than memory have also been investigated (e.g. attention, verbal fluency), acute retrograde memory side effects are the most prominent (Prudic et al., 1994). Similar to recovery of orientation, these side effects involve dysfunction of memory retrieval processes. These may also then be considered an important indicator for adverse outcomes, although this has yet to be empirically demonstrated.
Long-term cognitive side effects
Long-term cognitive side effects instead refer to those that emerge after repeated treatments. These more persisting side effects can present as a barrier for continued treatment, even when this is not clinically preferable (i.e. in the case of a patient choosing to cease treatment prior to attaining clinical remission). Unlike acute side effects, long-term cognitive side effects tend to show much larger variability between individuals because they are measured following repeated treatments. Hence, factors such as patients’ level of pretreatment cognitive functioning (Sobin et al., 1995), electrode placement (Sackeim et al., 1993), dosing (McCall et al., 2000), use of medications during treatment (Sackeim et al., 2009), the number of ECT treatments (Sackeim et al., 2007), depression severity (Sackeim et al., 2007) and the time of testing relative to the last ECT treatment (Semkovska and McLoughlin, 2010) are important for interpretation. At the group level, research has shown that the majority of the long-term cognitive side effects are greatest in the first week following the acute treatment course, then resolve within a few weeks following the acute treatment course and even improve beyond pretreatment levels (Semkovska and McLoughlin, 2010). However, these outcomes do not always occur at the individual level. Reductions in anterograde memory, particularly delayed recall, can persist for a longer duration (Falconer et al., 2010; Semkovska and McLoughlin, 2010). The most prominent and persistent side effect is retrograde amnesia for personal and impersonal memories (Lisanby et al., 2000; Sackeim, 2014). Nevertheless, controversy surrounds these due to the challenges with measurement, the potential role of illness factors and lack of normative data for memory loss over time (Semkovska and McLoughlin, 2013).
ECT-specific brief cognitive screening measures
Assessment of recovery of orientation post ECT has particular utility as outcomes can assist with treatment decisions including dosing, spacing between treatments, and electrode placement. In research, the most common method for assessing this, developed by the Columbia University group (Prudic et al., 1994), involved asking patients from the time of eyes opening or ability to respond verbally, five simple questions in a continuous manner until the patient correctly answered four items. The time between eyes opened and when the patient has correctly answered the last question is defined as the time to orientation. An alternative questionnaire (10-item orientation questionnaire), recently developed and found to be feasible in the clinical setting, similarly assesses patients’ recovery of orientation though can be administered at a single time point 20–30 minutes post each ECT (Martin et al., 2018b).
Tracking patients’ cognition at several set time points during the acute treatment course has utility for monitoring emerging and potentially long-term cognitive side effects. Several instruments have been developed for this purpose, namely the Brief ECT Cognitive Screen (BECS; Martin et al., 2013), B4ECTrecode (Viswanath et al., 2013) and the Electroconvulsive Therapy Cognitive Assessment (ECCA; Zixun, 2015). While normative data is provided for the BECS for predicting cognitive changes following treatment, this data is currently lacking for B4ECTrecode and the ECCA. Notwithstanding, the latter instruments may have particular sensitivity for monitoring changes in retrograde memory with the inclusion of autobiographical memory items and informant or caregiver rated items, respectively. The Autobiographical Memory Interview Short Form (AMI-SF; McElhiney et al., 2001) which assesses retrograde autobiographical memory consistency though has the strongest research support for this domain (Sackeim, 2014). Table 1 shows a summary of brief cognitive screening instruments designed for ECT.
Summary of brief cognitive screening instruments designed for ECT.
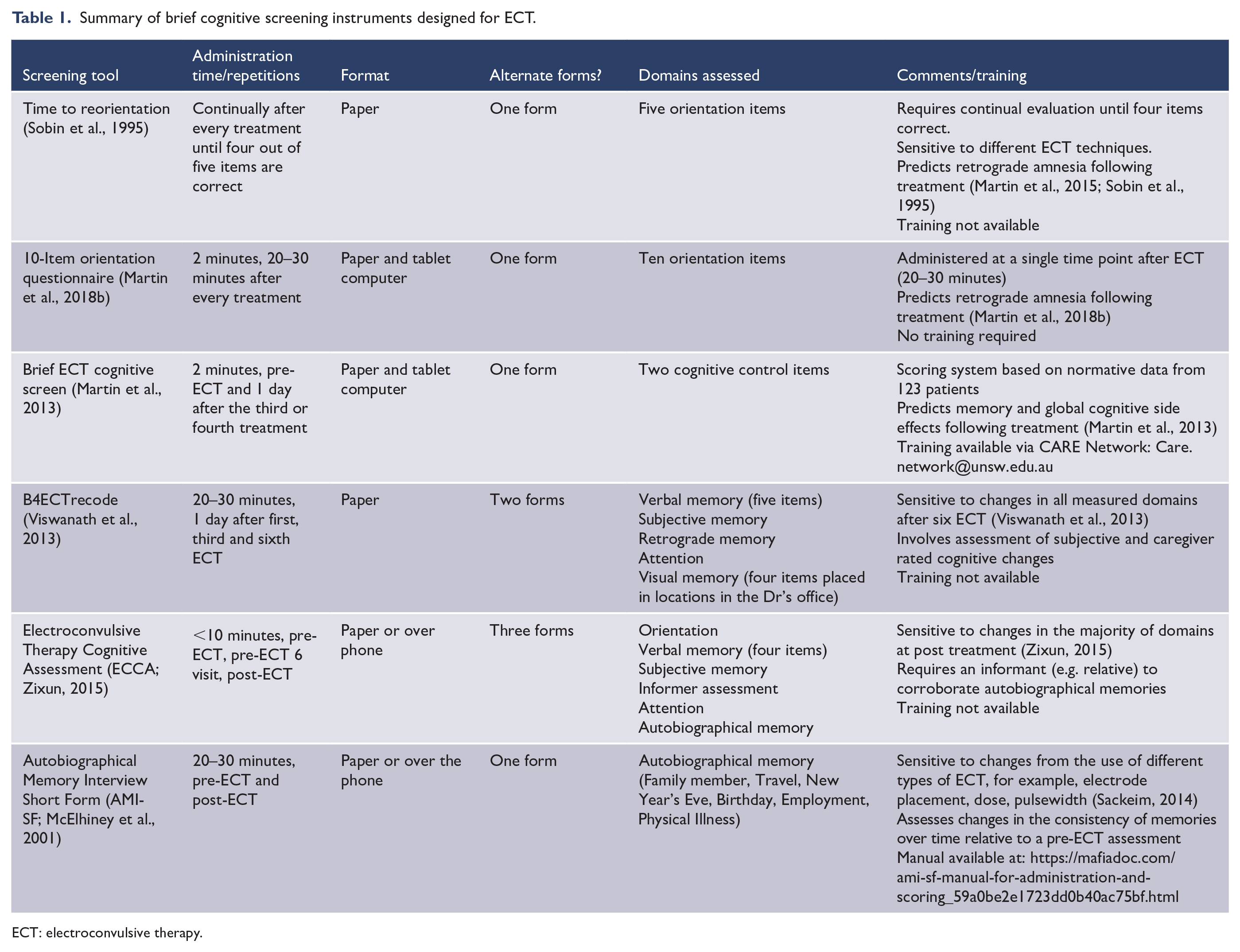
ECT: electroconvulsive therapy.
Brief generalised cognitive screening measures
Multiple generalised cognitive screening instruments have been developed to assist clinicians for screening for significant cognitive impairment. Unlike the ECT-specific measures, these instruments may lack sensitivity for detecting ECT-related cognitive side effects. Indeed, none of these measures has consistently shown sensitivity for detecting ECT-related cognitive side effects at the group level (see Table 2). Nevertheless, the use of brief generalised screening instruments has clinical utility for screening for cognitive impairment prior to treatment and for communicating patient’s clinical progress within the broader treating team. Some of these instruments are routinely used in clinical contexts, for example, during hospital admissions, which further provide the option for comparisons with prior assessments. Brief generalised cognitive screening instruments can also identify relative areas of cognitive weakness that can be followed up with more detailed neuropsychological assessments. An important caveat, however, is that they may only have sensitivity for detecting significant changes, and/or show ceiling effects in higher functioning patients. Relevant pertinent factors for the ECT clinical context are described below.
Summary of brief generalised cognitive screening instruments.
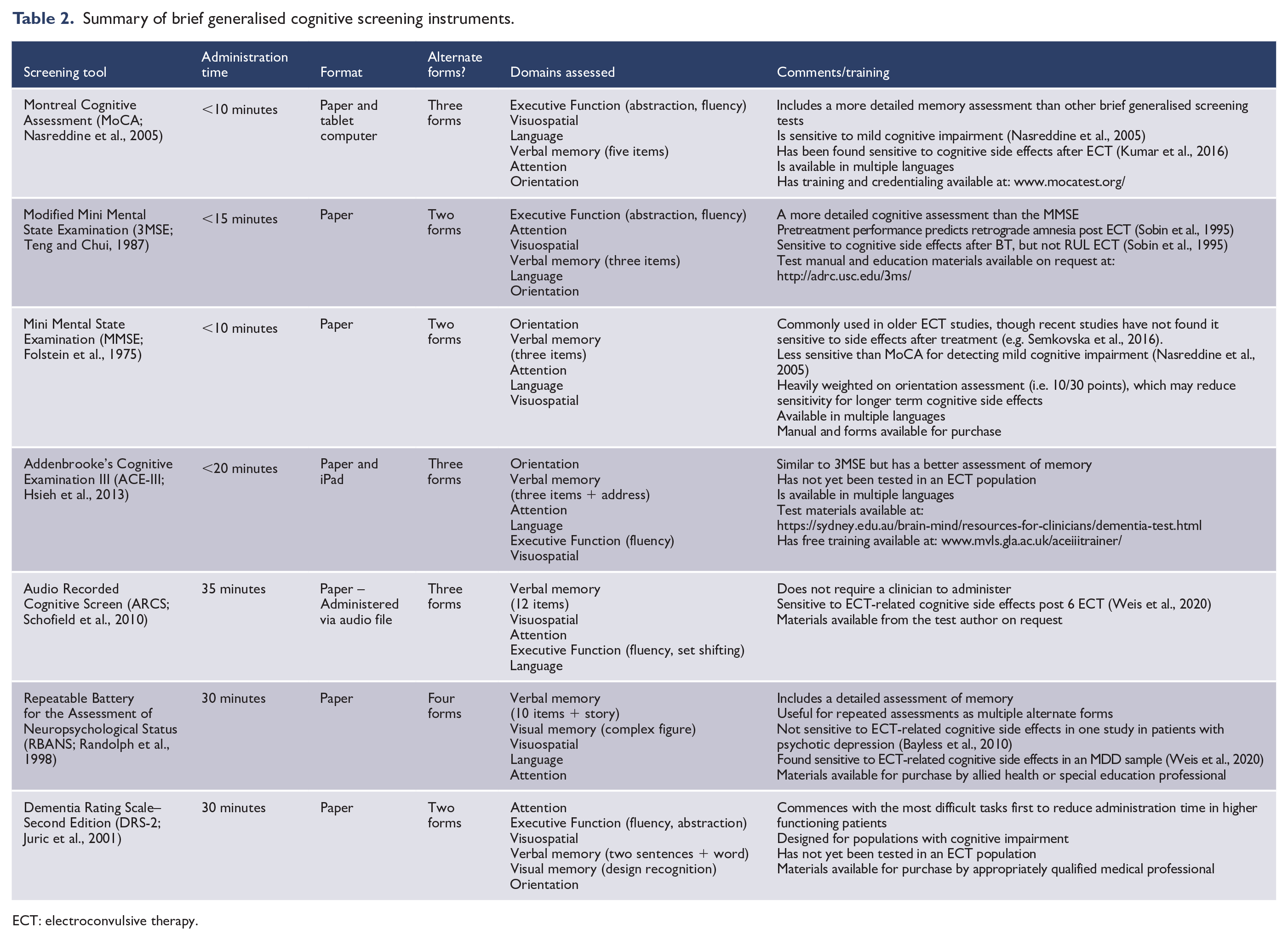
ECT: electroconvulsive therapy.
Administration time
ECT patients are often severely ill and may have limited tolerability for extensive testing. Even in highly selected research participants, recent large research trials have reported a considerable proportion of missing cognitive data (e.g. Kellner et al., 2010; Semkovska et al., 2016). Administration time is additionally relevant from a service perspective due to staffing priorities and/or logistical issues (e.g. rapid intake or discharge).
Administration format
While many commonly available brief generalised cognitive screening instruments are only available in pen and paper format (see Table 2), this is changing, with popular screening measures such as the Montreal Cognitive Assessment (MoCA; Nasreddine et al., 2005) now available on a computer tablet. Electronic or computer-administered tests have relative advantages for clinical settings including rapid scoring and improved standardisation for test administration. In contrast, limitations include the lack of necessity of a test administrator and potential issues with electronic data storage, privacy, security and cost. The presence of a test administrator can also have relevance for testing validity as ECT patients may lack computer literacy or require additional support or assistance when completing computerised tasks.
Alternate forms
Repeated testing is important to gauge potential treatment-related changes. Practice effects, which refer to improved performance due to familiarity with the test stimuli, can be problematic as these can mask meaningful declines in scores (e.g. if improvement from practice occurs on some items and decline occurs on other items). Research has shown that when alternate forms are not used, practice effects on neuropsychological tests can be significant (Beglinger et al., 2005; Woods et al., 2006). For this reason, validated alternate forms are important for cognitive monitoring with ECT.
Domains assessed
The domains of cognitive function most affected by ECT have been well described (Ingram et al., 2008). Particular domains are more susceptible than others, including verbal and visual anterograde memory, executive function and attention/processing speed (Semkovska and McLoughlin, 2010). Others are not so relevant, such as language (e.g. naming, articulation) or writing. For the most relevant domains, the level of assessment is important, as more cursory assessment may lack sensitivity for detecting subtle cognitive changes. Notably, retrograde amnesia is not assessed by any of the currently reviewed generalised screening measures.
Training
An essential requirement for the administrator of any cognitive instrument is appropriate knowledge/training in the test’s administration, scoring and interpretation. Lack of proficiency can lead to errors that result in compromised test reliability and validity. Similarly, lack of knowledge can lead to errors in interpretation that can potentially cause harm to patients. For these reasons, some measures (e.g. Repeatable Battery for the Assessment of Neuropsychological Status [RBANS], Dementia Rating Scale–Second Edition [DRS-2]) require formal qualifications and/or professional registration to access. Other measures have resources available for appropriate training and credentialing (e.g. MoCA and Addenbrooke’s Cognitive Examination III [ACE-III]).
Conclusion
The use of brief cognitive screening instruments has advantages in ECT clinical settings due to enhanced patient tolerability from test brevity and relative requirements for staff time and expertise. The choice of what screening instrument(s) to use and when, however, comes with several considerations.
For monitoring during the acute treatment course, routine assessment of recovery of orientation after every ECT treatment can provide useful information for informing treatment decisions during acute treatment (e.g. dosing adjustments, spacing of treatments). This side effect is particularly important as it is the strongest predictor of retrograde amnesia (Martin et al., 2015; Sobin et al., 1995). Additional assessment, however, can also have utility for identifying patients at increased risk for developing adverse outcomes and those who may want to cease treatment prematurely because of such concerns. Several ECT-specific screening measures have been developed for this purpose. The choice of instruments during the ECT course and when to administer them are nevertheless dependent on balance of the utility of the clinical information gauged and the test administration requirements. As anterograde memory and retrograde memory tend to be most affected, these are the focus of the majority of these ECT-specific measures. From a psychometric perspective, however, the role of practice effects and repetition of task items (e.g. for autobiographical memories), the validity and sensitivity of task items and domains, and the potential confounding of subjective and objective memory items are relevant considerations.
Of the measures currently reviewed, the Clinical Alliance for Research in ECT (CARE) Network, which involves over 40 hospitals in Australia and overseas, has recommended a battery of brief screening tests and assessment time points for routine patient monitoring (Martin et al., 2018a). For this network, the MoCA was chosen as a brief generalised cognitive screening measure based on its relative advantages. The MoCA has similarly been recommended in the clinical guidelines for ECT monitoring in the Netherlands (Verwijk et al., 2019) and by the Global ECT-MRI Research Collaboration (GEMRIC) international consortium. Alternate measures that may have similar utility include the Modified Mini Mental State Examination (3MSE), Audio Recorded Cognitive Screen (ARCS) and ACE-III. It is important to note that none of these measures were specifically designed for use with ECT, and they are cognitive screening instruments and not standardised neuropsychological tests. Thus, interpretation of results from cognitive screening instruments should always be made with caution. Further investigation with comprehensive clinical neuropsychological evaluation is recommended in the case of clinical concerns.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
