Abstract
Objective:
Poorer mental health in adulthood is associated with increased risk of cardiovascular disease and reduced life expectancy. However, little is known of the molecular pathways underpinning this relationship and how early in life adverse metabolite profiles relate to self-reported variation in mental health. We examined cross-sectional associations between mental health and serum metabolites indicative of cardiovascular health, in large Australian population-based cohorts at two stages of the life-course.
Methods:
We characterised cross-sectional serum nuclear magnetic resonance metabolite profiles of positively and negatively framed mental health in a large population-based sample of Australian 11- to 12-year-olds (n = 1172; 51% girls) and mid-life adults (n = 1322; mean age 45 years; 87% women). We examined multiple standard self-report mental health scales, spanning psychosocial health, general well-being, life satisfaction, and health-related quality of life. Linear regression was used to investigate the cross-sectional association between mental health and each metabolite (n = 73) in children and adults separately, unadjusted and adjusted for age, sex, socioeconomic position and body mass index.
Results:
Better child and adult mental health were associated with lower levels of the inflammatory marker glycoprotein acetyls, and a favourable, less atherogenic lipid/lipoprotein profile. Patterns of association in children were generally weaker than in adults. Associations were generally modest and partially attenuated when adjusted for body mass index.
Conclusions:
In general, metabolite profiles associated with better child and adult mental health closely aligned with those predictive of better cardiovascular health in adults. Our findings support previous evidence for the likely bidirectional relationship between mental health and cardiovascular disease risk, by extending this evidence base to the molecular level and in children.
Introduction
Poorer mental health in adulthood is associated with increased risk of cardiovascular disease (CVD) (Scott et al., 2016) and reduced life expectancy (Diener et al., 2017). However, little is known of the molecular pathways underpinning this relationship, its directionality, any mediating effects of social and environmental factors, and how early in life adverse metabolite profiles are associated with variation in mental health. Approximately half of mental disorders have their onset during adolescence (Jones, 2018), and for some mental disorders, such as anxiety, the median onset is younger still (Essau et al., 2018). To address the growing burden of mental illness, we need to address the wider mental health spectrum during key developmental windows and understand molecular mediators of the relationship with key co-morbidities, such as CVD, which may identify potential intervention points for multiple conditions. The first step towards understanding if these pathways may be causal is to establish whether or not cross-sectional relationships exist, and if so in what age groups.
Previous data suggest the relationship between mental health and CVD is partially mediated via chronic inflammation (Haapakoski et al., 2015). In addition, people with mental illnesses have been shown to have a unique biomarker profile. For example, in a large meta-analysis of nine Dutch cohorts, adults with depression were compared with those without depression and found to have less optimal metabolic profiles (Bot et al., 2020). Specific observed differences for adults with versus without depression included lower plasma apolipoprotein A1 (ApoA1), high-density lipoprotein (HDL) cholesterol and acetate; smaller HDL diameter; higher very low density lipoprotein (VLDL) cholesterol, apolipoprotein B (ApoB), glycerides, phospholipids, triglycerides, total and monounsaturated fatty acids (FAs), glycoprotein acetyls (glycA), tyrosine and isoleucine; and larger VLDL diameter and fatty acid chain length. Similar associations are also seen in other mental illnesses, such as schizophrenia and bipolar disorder (Quintero et al., 2019). Some blood biomarkers associated with mental illnesses are potentially relevant to CVD (Würtz et al., 2012), but studies examining mental health-related molecular profiles in population-based cohorts are sparse. Growing evidence in adults suggests an interaction between depression and metabolic syndrome (McIntyre et al., 2009), an early indicator for CVD risk at a molecular level (Han and Lean, 2016). It is unclear whether similar associations exist earlier in the life-course, or across the full mental health spectrum, from positive mental health and sub-clinical mental health problems, to overt mental illnesses such as depression, schizophrenia and bipolar disorder.
We examined cross-sectional associations between mental health and serum metabolites in a large Australian population-based cohort at two life stages, 11- to 12-year-olds and mid-life adults (parents of the children). We included measures of negatively framed and positively framed mental health, to span its full spectrum at different ages. We included serum metabolites with known CVD associations, as we were interested in whether patterns similar to those underlying CVD might emerge for mental health. This knowledge could potentially elucidate shared pathogenesis for mental illness and CVD. However, this analysis is exploratory; it simply seeks to establish whether metabolites known to be associated with CVD also share a relationship with mental health. It neither assumes a unidirectional path between mental health and cardiovascular health, nor precludes relationships with other physical diseases.
Methods
Study population
Data are drawn from the Longitudinal Study of Australian Children (LSAC)’s bio-physical module, the Child Health CheckPoint (CheckPoint). LSAC utilised a two-stage clustered random sampling design based on postcode (Soloff et al., 2005), to obtain a sample relatively representative of the Australian population. LSAC’s birth cohort recruited 5107 0- to 1-year-olds in 2004, with follow-up every 2 years (retention rate 74% in wave 6, 2014) (Edwards, 2014). CheckPoint comprised a once only physical health and biomarkers assessment nested between LSAC’s sixth and seventh waves, from February 2015 to March 2016, for 1874 birth cohort children and one on their parents (Clifford et al., 2019). Most families attended a ‘pop-up’ Assessment Centre in central locations across Australia, where families rotated through stations measuring specific health attributes. Families unable to attend an Assessment Centre were offered a home visit, but were not included in these analyses as venous blood samples were not collected at home visits. Of the 3764 families eligible for CheckPoint, 1874 families (50%) took part, of whom 1172 children and 1322 adults with metabolomic data and at least one mental health measure were included in our analyses (Supplemental Figure S1).
The study protocol was approved by The Royal Children’s Hospital Melbourne Human Research Ethics Committee (33225D) and Australian Institute of Family Studies Ethics Committee (14-26). The attending parent or guardian provided written informed consent for them and their child to participate in the study.
Measures
Supplemental Table S1 provides details of validated instruments used to collect mental health, venous blood and covariates, which are summarised below.
Mental health
Participants self-reported mental health on iPad-based questionnaires. Children completed two negatively framed measures, capturing psychosocial health (Paediatric Quality of Life 4.0: Psychosocial sub-scale [PedsQL-PS]; Varni et al., 2001) and overall health-related quality of life (HRQoL; Child Health Utility 9-Dimensions [CHU9D]; Stevens, 2011), and two positively framed measures, capturing general well-being (Paediatric Quality of Life 4.0: General Well-being Scale [PedsQL-GW]; Varni et al., 2001) and life satisfaction (International Survey of Children’s Well-being Brief Multidimensional Students’ Life Satisfaction Scale [ISCWeb]; Seligson et al., 2003).
Adults completed one negatively framed measure capturing HRQoL (CHU9D; Stevens, 2011, adapted for adults), and an overall measure of positive and negative psychosocial quality of life (QoL; Assessment of Quality of Life 8-Dimensions: Psychosocial sub-scale [AQoL8D-PS]; Richardson et al., 2014). To assess the impact of item framing in adults, we additionally conducted analyses using two reduced versions of the AQoL8D-PS, comprising unweighted scores of the positively framed (happiness, coping skills, self-worth and relationships) and negatively framed (mental health, self-worth and relationships) items separately. However, as associations were very similar for these scores compared with the overall AQoL8D-PS, only the validated AQoL8D-PS results are presented. All mental health cumulative scores are scaled to a 0–1 or 0–100 scale, such that a higher score indicates better mental health.
Metabolic measures
Metabolites were analysed in serum from semi-fasted venous blood samples (children 50 minutes to 20 hours, median 4.1 hours; adults 30 minutes to 22.6 hours, median 2.9 hours). Blood samples were collected by a single venepuncture from the non-dominant arm of semi-reclining participants, processed on-site into six serum aliquots generally within an hour (median 53 minutes), and stored at –80°C. Samples were shipped on dry ice to the long-term storage facility, and one aliquot per participant shipped on dry ice to Nightingale® Health (Helsinki, Finland). Metabolite profiles were measured by nuclear magnetic resonance (NMR) spectroscopy on the Nightingale® metabolomics platform. Data described here were derived using the facility’s 2016-version quantification algorithm. Details of this platform have been described elsewhere (Soininen et al., 2009), including distributions in CheckPoint participants (Ellul et al., 2019). Briefly, NMR-based high-throughput quantification measured the absolute concentration and ratios of 228 metabolites simultaneously from a single 0.35 mL serum aliquot. This included standard lipids, lipoprotein subclass distributions, particle size and composition, fatty acids (FAs), and other low molecular weight metabolites, such as amino acids and glycolysis-related metabolites.
We have previously established that many metabolites correlate substantially within adults and children (Ellul et al., 2019). Thus, we focused on an informative subset of 73 metabolites that capture the majority of variation within the dataset (Supplemental Table S2). These can be grouped as (1) lipids, cholesterol and triglycerides, (2) FAs and cholines, and (3) other metabolites (including apolipoproteins, amino acids, ketone and glycolysis factors, fluid balance and inflammation markers). For different size lipoprotein particles, figures only show the most representative of similar associations to reduce repetition, including VLDL lipids (large), low-density lipoprotein (LDL) lipids (medium), HDL lipids (medium), and HDL cholesterol (HDL1 cholesterol).
Related measures
Our analyses accounted for age, sex, socioeconomic position (SEP), body mass index (BMI), puberty and pre-existing mental health conditions, since these could affect associations between mental health and metabolite profiles. Certain metabolites measured on the NMR platform show clear sex differences (e.g. higher leucine and valine in males and higher LDL particle diameter in females), and more variation in profiles and larger sex differences with increasing age (Ellul et al., 2019). Date of birth and sex were obtained from Medicare records for children and self-reported for adults. In addition, socioeconomic gradients have been documented in mental health (Stewart-Brown et al., 2018) and metabolic profiles (Slopen et al., 2013). SEP was obtained from LSAC’s wave 6 data (preceding CheckPoint, at child age 10–11), incorporating parent-reported education, income and occupation at wave 6, and internally standardised (mean 0, standard deviation [SD] 1) (Blakemore et al., 2009). BMI (kg/m2) was included in some analyses as the relationship between BMI, mental health and serum metabolites is unclear (Luppino et al., 2010). We corrected for known variation in BMI across age and sex in children by using the Centers for Disease Control and Prevention z-score (Ogden et al., 2002). We assessed pubertal status in children using CheckPoint’s 5-item self-report Pubertal Development Scale (Petersen et al., 1988), as pubertal timing can affect mental health (Oldehinkel et al., 2011). Pre-existing mental health and related conditions were drawn from LSAC’s waves 1–6 data collected over the 10 years prior. Adults self-reported in alternating waves if they had ever experienced a mental condition (including depression, schizophrenia, bipolar disorder, or other mental illness excluding post-natal depression). For children, parents proxy-reported each wave if their child had an ongoing mental condition (including anxiety, depression, attention deficit hyperactivity disorder and autism spectrum disorder) (Edwards, 2014).
Statistical analyses
Unadjusted and adjusted linear regression models were used to examine associations between mental health and serum metabolites for complete cases, in children and adults separately. We used a Student’s t-test to confirm there was little evidence of a difference between complete cases and those with missing data across mental health measures, age, sex and BMI. However, complete cases had a higher socioeconomic position that those missing data (see limitations). Scatterplots of metabolites and mental health measures were examined to check for outliers and to ensure assumptions for linear regression models were met. No outliers were identified beyond the expected variation within a population-based cohort. Results are presented in figures using internally standardised variables (mean = 0, SD = 1), with mental health as the independent variable. However, results on the original scale and considering both model directions are available in Supplemental Tables S4 and S8. We assessed additional models including (1) age, sex and SEP as covariates, and (2) age, sex, SEP and BMI as covariates. Given the similarity of results from unadjusted and model (1), we present only unadjusted and model (2) results in figures and tables. We conducted additional secondary analyses with model (1) also adjusted for puberty in children, stratified model (2) by sex to examine any potential sex differences, and excluded those with a diagnosed mental health condition to examine the sensitivity of findings to the effects of clinically significant mental illness. Given the descriptive aims of the paper, we report all standardised estimates with associated 95% confidence intervals (CIs) and focus on the pattern of associations, rather than p-values or specific results in isolation. Although some metabolites demonstrated skewed distributions, log transformation did not significantly alter the patterns of association, so we have reported results without log transformation for ease of interpretability. Statistical analyses were conducted using Stata v14.2.
Results
Sample characteristics
Table 1 describes sample characteristics. Children (mean age 12.0 [SD 0.4] years) included similar numbers of boys and girls, but most adults (mean age 44.5 [SD 5.2] years) were female (87%). BMI was in line with national Australian averages, with mean child BMI z-score 0.3 and mean adult BMI 27.8 kg/m2. Ever experiencing a mental health or related condition was parent-reported in 10% of children (equally anxiety and cognitive conditions), and self-reported in 66% of adults (predominantly depression). The SEP of participants included in our analyses was about a quarter of a standard deviation above, and had a smaller variance than, the main LSAC Birth cohort at wave 1 (Blakemore et al., 2009).
Sample characteristics.
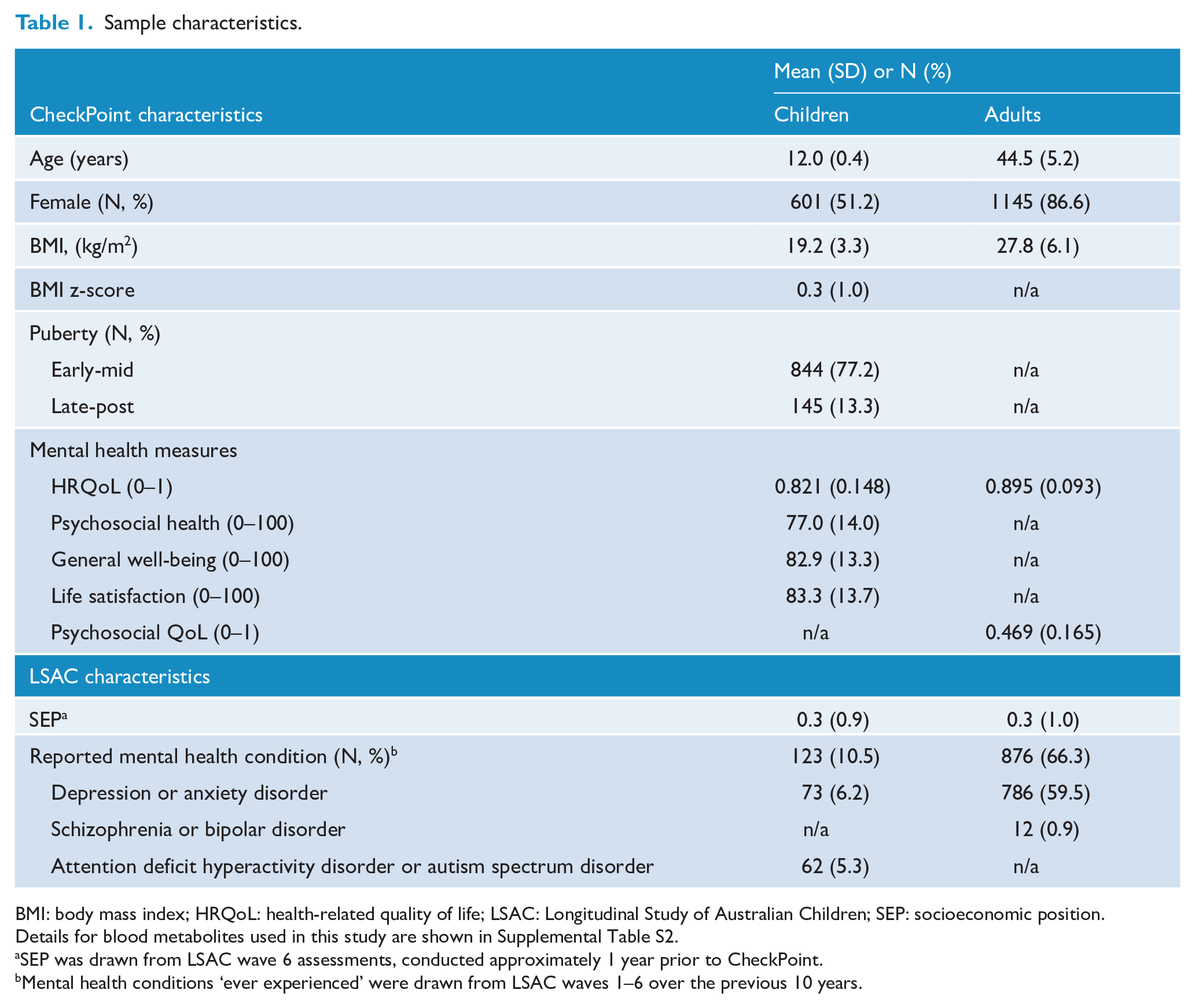
BMI: body mass index; HRQoL: health-related quality of life; LSAC: Longitudinal Study of Australian Children; SEP: socioeconomic position.
Details for blood metabolites used in this study are shown in Supplemental Table S2.
SEP was drawn from LSAC wave 6 assessments, conducted approximately 1 year prior to CheckPoint.
Mental health conditions ‘ever experienced’ were drawn from LSAC waves 1–6 over the previous 10 years.
Associations with metabolite groups
Lipids, cholesterol and triglycerides
Within lipids, cholesterols and triglycerides, better mental health in children and adults was most strongly associated with lower VLDL components (VLDL lipids, cholesterol and triglycerides, and lower mean diameter) and lower triglycerides (total, VLDL and HDL triglycerides) (Figure 1, with numeric values in Supplemental Table S3). For example, in the fully adjusted model, 1SD unit higher life satisfaction for children and psychosocial QoL for adults was associated with −0.12SD units (95% CI −0.17, −0.06) and −0.07SD units (−0.11, −0.02) VLDL lipids, respectively. Better mental health was also associated with higher HDL (lipids and cholesterol, and higher mean diameter) in children and adults, and lower LDL and intermediate density lipoprotein (IDL) triglycerides and higher mean LDL diameter in adults only. Associations with mental health were less pronounced for non-HDL cholesterol, phosphoglycerides, and LDL and IDL lipids.
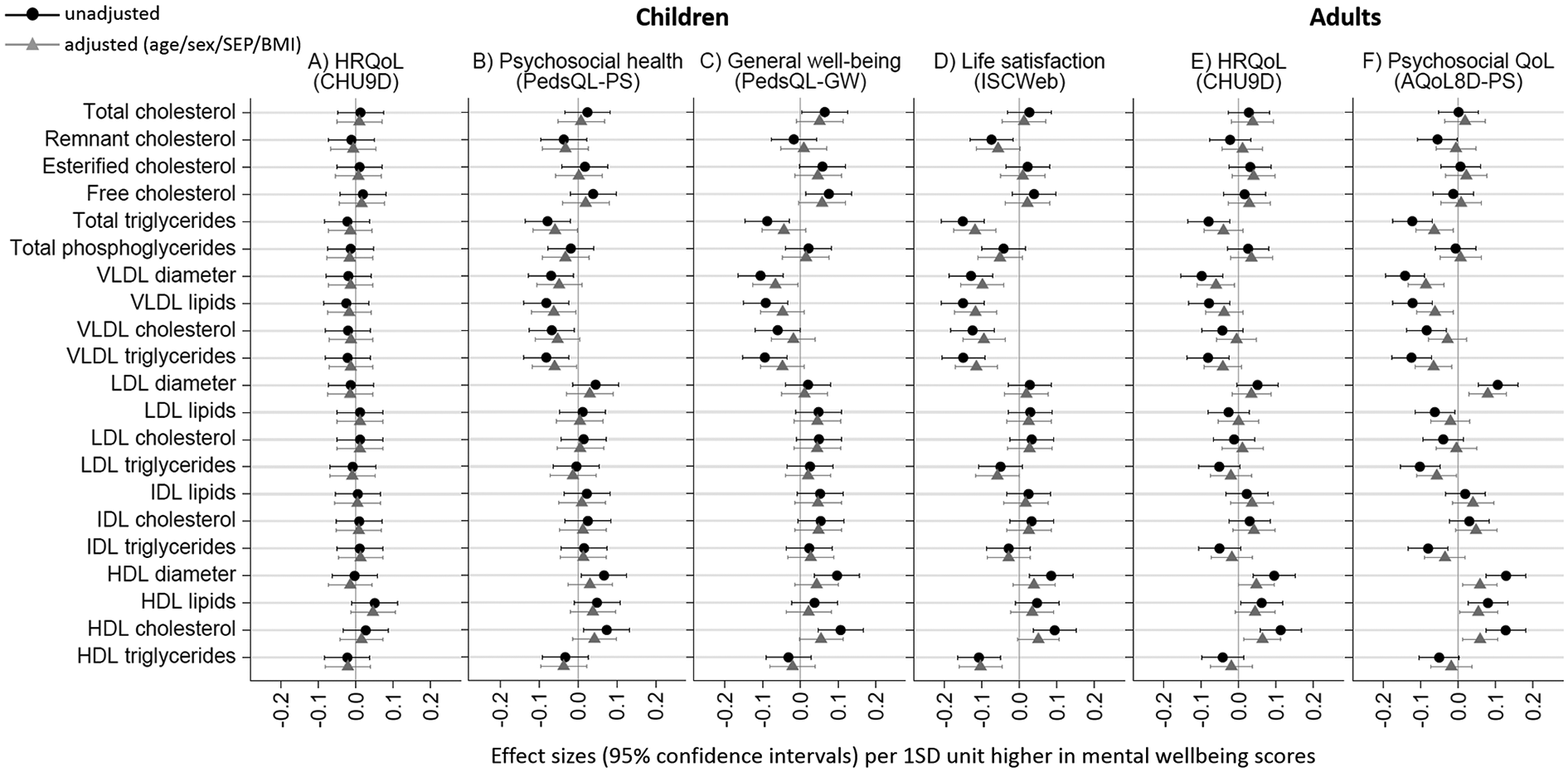
Associations of mental health with lipid, cholesterol and triglyceride profiles in children (A–D) and adults (E–F).
FAs and cholines
Within FAs and cholines, better mental health in children and adults was most strongly associated with lower total, saturated and monounsaturated FAs, and higher polyunsaturated FAs and degree of fatty acid unsaturation, up to 0.11SD units (95% CI 0.04, 0.15) for child life satisfaction and up to 0.10SD units (0.02, 0.13) for adult HRQoL utility (Figure 2, with numeric values in Supplemental Table S3). Better mental health in children and adults was also associated with higher ratios of omega 3 FAs (primarily 22:6, docosahexaenoic acid) and omega 6 FAs (including 18:2 linoleic acid) relative to total FAs, but associations were less pronounced for the absolute serum levels of these FAs. Less prominent associations with better mental health were seen for higher sphingomyelins and lower cholines (including phosphatidylcholine) in children and adults.
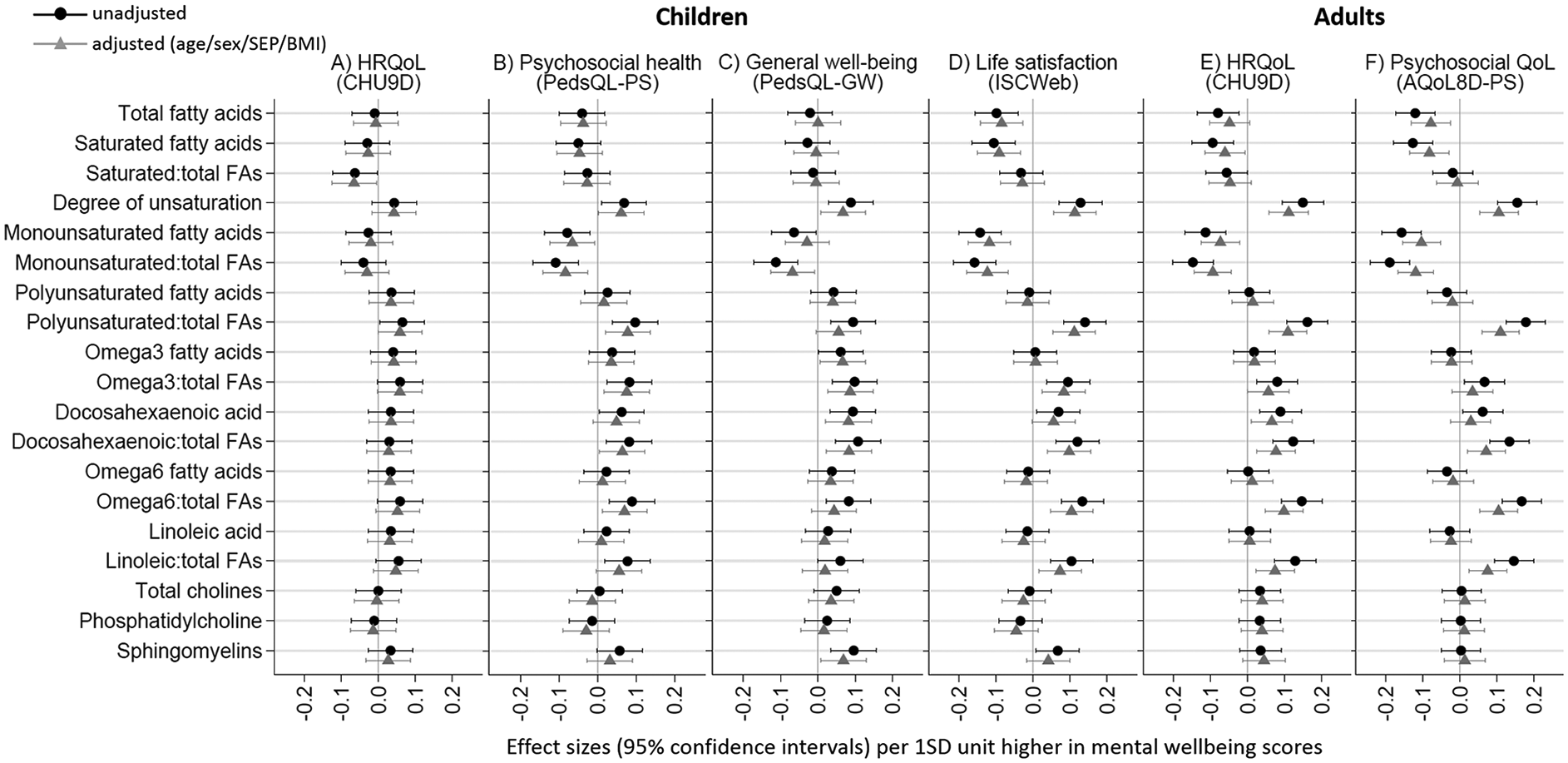
Associations of mental health with fatty acid and choline profiles in children (A–D) and adults (E–F).
Other metabolites
Better mental health in children and adults was associated with higher apolipoprotein A1 (ApoA1), albumin and creatinine, and with lower GlycA, apolipoprotein B (ApoB), and pyruvate, around –0.08SD units for children and –0.06SD units for adults per SD unit higher mental health in the fully adjusted model (Figure 3, with numeric values in Supplemental, Table S3). In children, better mental health was associated with lower glutamine, aliphatic (alanine, glycine, isoleucine and leucine), and aromatic amino acids (phenylalanine and tyrosine) (–0.08 to –0.10SD units), while associations for valine and glycerol were inconsistent between child mental health measures. Associations were less pronounced for amino acids and glycerol in adults. Better mental health was associated with higher citrate and ketone body metabolites (acetate, acetoacetate and 3-hydroxybuturate) in children, but lower levels in adults.
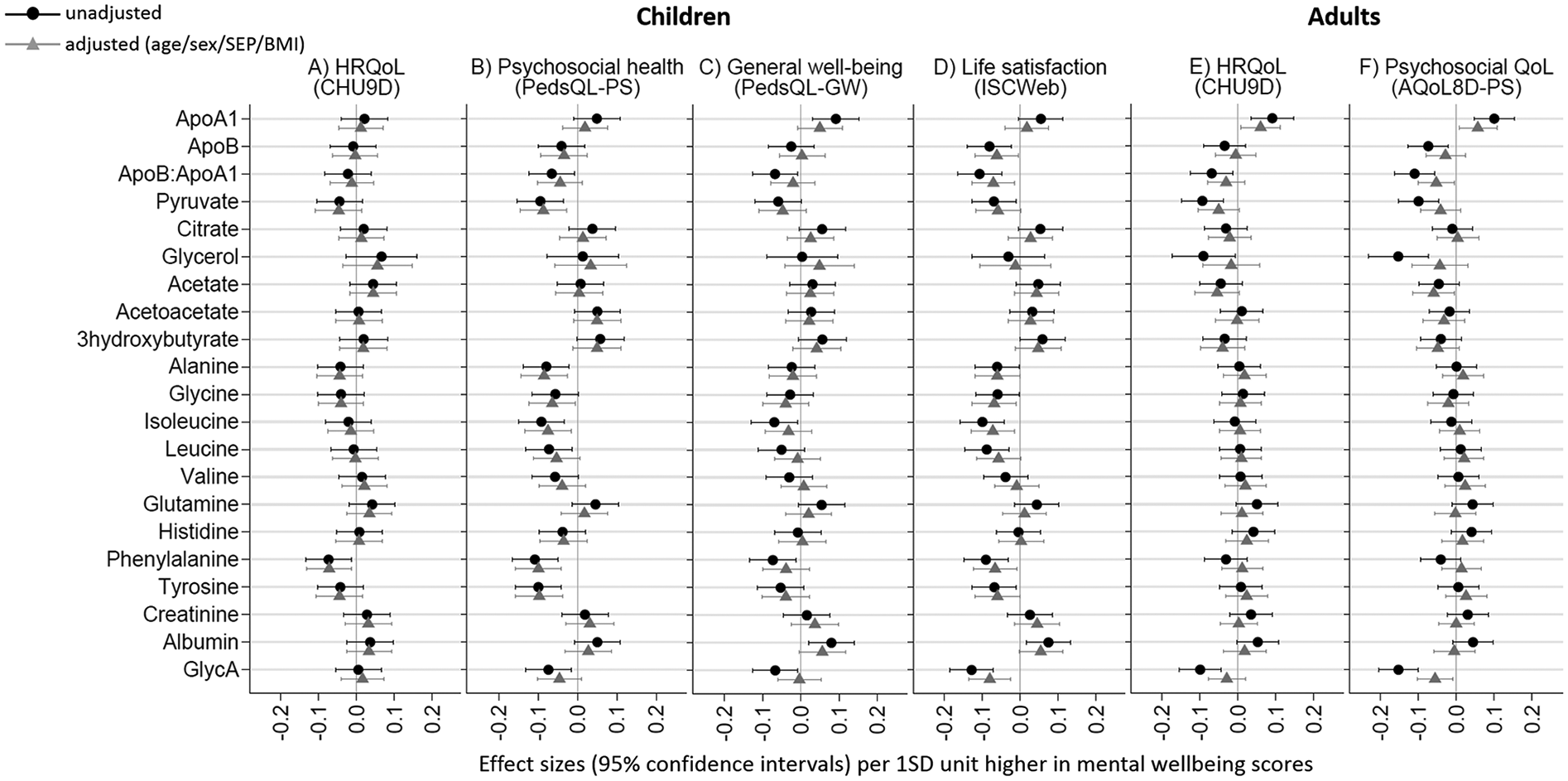
Associations of mental health with other metabolite profiles (apolipoproteins, ketone and glycolysis metabolites, amino acids, inflammation and fluid balance factors) in children (A–D) and adults (E–F).
General patterns
Patterns of associations were highly consistent across mental health measures in children and adults. The exception was children’s HRQoL utility, which demonstrated a less pronounced pattern of metabolite associations compared with other measures. Similar patterns of association were seen for positively and negatively framed measures in children and adults, although associations were generally more pronounced for positively framed scales in children. Results were similar when adjusted for age, sex, SEP and puberty (for children; Supplemental Table S5). For some scales, associations for VLDL and HDL diameter, HDL lipids, HDL cholesterol, ratio of Omega 3 to total FAs, sphingomyelins, ApoA1 and albumin were slightly attenuated towards to null, while associations for total and saturated FAs were slightly more substantial, when additionally adjusted for puberty (additionally to age, sex and SEP) in children. However, addition of BMI to the adjustment set slightly to moderately attenuated most results towards the null (adjusted models in Figures 1–3 and Supplemental Table S3). Associations were similar but generally less pronounced when excluding participants reporting ever experienced any mental health condition (Supplemental Table S6). The largest difference between those with and without ever experiencing any mental health condition were seen for HDL diameter, HDL cholesterol, degree of unsaturation, Omega 3 FAs, ApoA1, ApoB, glycerol, alanine, histidine, tyrosine and glycA in adults, while no substantial changes were seen in children with the smaller number of excluded children with mental health conditions. Associations were less robust (larger CIs) when stratified by sex, especially for the small number of men (Supplemental Table S7). Some minor differences were seen between boys and girls, with higher mental health scores associated with lower acetate, glutamine, albumin and phenylalanine in boys, and higher histidine and lower glycerol in girls. In adults, metabolic profiles for women mirrored those for all adults (reflecting the large proportion of mothers in our sample), while higher mental health in men was associated with higher cholesterol, triglycerides, LDL and IDL lipids, cholines, tyrosine and albumin, and lower citrate, alanine, isoleucine, leucine, glutamine, histidine and creatinine. Patterns of association were similar when the direction of the model was reversed (Supplemental Table S8).
Discussion
Principal findings
The metabolite profiles associated with better mental health in children and adults aligned with those previously associated with better cardiovascular health (Ritchie et al., 2015; Russo, 2009), consistent with previous evidence for substantial co-morbidity of mental illnesses and CVD in adults (Cohen et al., 2015). A ‘better mental health’ profile showed lower levels of the inflammatory marker GlycA, the glycolysis factor pyruvate, and a favourable lipid profile (including lower VLDL, triglycerides, ApoB, and saturated and monounsaturated FAs, and higher HDL, omega 3, omega 6, and polyunsaturated FAs). Better mental health in children was associated with lower levels of aromatic (phenylalanine and tyrosine) and aliphatic amino acids (alanine, glycine, isoleucine and leucine), previously linked to increased risk of CVD (Magnusson et al., 2013) and type 2 diabetes (Würtz et al., 2013).
Consistency of findings
Patterns of associations were consistent across positive and negative mental health measures, in adults and children, supporting robustness of the metabolic profiles across the mental health spectrum. Associations for children were more pronounced in positively framed than negatively framed measures. This may relate to the length of recall time or breadth of the measures used. In addition, associations between mental health and metabolites were generally more pronounced and consistent in adults than children. This may relate to cumulative exposures over time and known associations between metabolites and overt disease, or potentially differences in emotional expression with age.
The addition of BMI generally attenuated results, although less so in adults. Combined with modest associations, this suggests potential confounding or mediation by BMI, or a more complex relationship between mental health, metabolism, obesity and cardiovascular risk. As the relationship between BMI and mental health may be bidirectional (Luppino et al., 2010), the additional model with BMI as a covariate is included with caution. Longitudinal analyses are warranted to establish the role of BMI in the causal pathway, and any opportunity to inform optimal targets and timing for prevention efforts. As our aim was to establish patterns of association rather than pursue causality, we did not examine the potential for residual confounding here. As such, we have also not included other potential confounders in the relationship between mental health and CVD, such as medications, smoking and physical activity, as this type of investigation of causality was beyond the focus of this study.
Findings in light of previous research
Our results for triglycerides and HDL cholesterol were consistent with previous results from an elderly Taiwanese population-based cohort (Seplaki et al., 2004), with slightly smaller associations in our mid-life adults. Our results in a population-based study of children and adults were also consistent with many previous studies in adults with depression. This included better mental health associated with lower VLDL cholesterol and diameter, ApoB, triglycerides, total and monounsaturated FAs, and glycA, and higher linoleic acid, ApoA1, HDL cholesterol and diameter, and omega-3 FAs (Bot et al., 2020; Ding et al., 2014; Ditzen et al., 2011; Lu et al., 2014; Mischoulon and Fava, 2000). The positive association of mental health with acetate, and the negative association of mental health with tyrosine, glycine, alanine and isoleucine, in children has been previously associated with symptoms of depression and anxiety in some adults (Altmaier et al., 2013; Bot et al., 2020; Ding et al., 2014).
Our results, and those in a recent study of depressed adults (Bot et al., 2020), for GlycA support evidence of inflammation mediating the relationship between mental health and CVD (Haapakoski et al., 2015). Similarly, the less atherogenic lipid/lipoprotein profile supports potential mediation by metabolic syndrome (McIntyre et al., 2009). Genome-wide association studies have identified an enrichment of inflammatory (Kao et al., 2012) and metabolism (Hebebrand et al., 2018) genetic variants associated with mental health related phenotypes. As many mental health traits are moderately heritable (Bartels, 2015), this may indicate potential complex gene–environment interactions underlying our associations. However, higher monounsaturated FAs may have protective effects in some contexts, such as diabetes, metabolic syndrome and CVD (Gillingham et al., 2011; Maedler et al., 2003; Ros, 2003). The negative relationship seen with mental health here and previously in depressed adults (Bot et al., 2020) may suggest nuances in the relationship of monounsaturated FAs with mental and physical health.
Given our data are cross-sectional, we do not imply directionality of the associations observed. Associations between better mental health and a cardio-protective metabolic profile could arise from a number of factors, such as the bidirectional relationship between mental health and dietary intake (Arvidsson et al., 2017), the influence of social patterning on mental health (Huurre et al., 2003) and dietary intake (Gasser et al., 2017), or shared genetic determinants (Mulle and Vaccarino, 2013). Similarly, analogous molecular profiles could have arisen from a common biological vulnerability or similar physical consequences rather than a direct causal pathway between mental health and CVD. Thus, longitudinal analyses are necessary to establish the sequence of events between mental health and changes in cardio-protective metabolic factors (directionality). Such studies could also help establish whether analogous metabolite associations suggest any common causal pathways or common consequences between mental health and CVD that could inform future intervention efforts.
Strengths and limitations
Our study builds on previous research by examining the mental health spectrum in population-based cohorts at two life stages. The LSAC study represents a carefully designed population-based cohort, but participation in CheckPoint assessment centres was influenced by regional remoteness, single-parent status, extracurricular activities, after-school care, parental education, SEP, and support service use (Ellul et al., 2018). The SEP of participants included in our analyses were also about a quarter of a standard deviation above participants missing serum metabolite data, due to SEP differences between those with and without blood samples collected. Our sample is therefore not representative of more disadvantaged families in Australia, but within population associations are likely to be preserved.
The benefit of our parent–child design is that it eliminates many sources of heterogeneity seen in other cross-cohort studies. However, this also brings small correlations in metabolites for parent–child pairs as previously published (Ellul et al., 2019), likely from both genetic and shared environmental influences. Parent–child correlations may slightly exacerbate the level of similarity between adult and child metabolic profiles of mental health. However, this does not invalidate our aim to show if, by what age group, and how strongly, patterns between mental health and metabolites emerge at the population level. The adult population are also predominantly mothers. Although the stratified sample sizes for male adults were too small to draw any robust conclusions, for completeness we include the stratified results. In addition, the NMR platform is a reproducible, widely used platform, but is largely focused on metabolites associated with cardiometabolic risk. A more detailed metabolomics and biomarker profile, incorporating other approaches (such as hormones, lipidomics, and proteomics) is needed to more fully explore the molecular underpinnings of mental health.
Our findings are interpreted in light of previous studies in mental disorders (predominantly depression) with caution. Our epidemiological measures span the mental health spectrum, with most individuals in the ‘normal’ range for each measure. Such measures do not parallel diagnostic categories, but are in line with the emerging notion of an overarching spectrum of psychopathology (Caspi and Moffitt, 2018). This would suggest that, if causally related, similar metabolite patterns could underlie multiple mental disorders as well as physical health conditions such as CVD.
Conclusion
We identified cross-sectional metabolic profiles of mental health in children and their parents, which are largely similar to those previously associated with cardiovascular health. This alignment of molecular profiles strengthens previous evidence for a relationship between mental health and CVD, by expanding this evidence base to the blood metabolite level. The next step would be longitudinal analyses to establish the directionality of this relationship, and whether these metabolites indicate common causal pathways or common consequences. If evidence suggests a causal direction, the metabolites highlighted here may suggest novel intervention points through which to address the higher incidence of CVD and increased mortality risk of mental health conditions.
Supplemental Material
Supplementary_Material – Supplemental material for Cross-sectional metabolic profiles of mental health in population-based cohorts of 11- to 12-year-olds and mid-life adults: The Longitudinal Study of Australian Children
Supplemental material, Supplementary_Material for Cross-sectional metabolic profiles of mental health in population-based cohorts of 11- to 12-year-olds and mid-life adults: The Longitudinal Study of Australian Children by Katherine Lange, Kate Lycett, Susan Ellul, Richard Saffery, Fiona Mensah, John Carlin, Lisa Gold, Ben Edwards, Peter Azzopardi, Michael Sawyer, Markus Juonala, David Burgner and Melissa Wake in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
This paper uses unit record data from the Longitudinal Study of Australian Children. The study is conducted in partnership between the Department of Social Services (DSS), the Australian Institute of Family Studies (AIFS) and the Australian Bureau of Statistics (ABS). The findings and views reported in this paper are those of the author and should not be attributed to DSS, AIFS or the ABS.
Data Sharing
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Health and Medical Research Council (NHMRC) of Australia (1041352, 1109355); the Royal Children’s Hospital Foundation (2014-241); the Murdoch Children’s Research Institute; The University of Melbourne, the National Heart Foundation of Australia (100660); Financial Markets Foundation for Children (2014-055, 2016-310); and the Victoria Deaf Education Institute. The following were supported by NHMRC: Senior Research Fellowships MW (1046518) and DPB (1064629); Principal Research Fellowship MW (1160906); Career Development Fellowship FKM (1111160); Early Career Fellowships KLy (1091124) and LG (1035100). The following were supported by the National Heart Foundation of Australia: Honorary Future Leader Fellowship DPB (100369); Postdoctoral Fellowship KLy (101239). The following was supported by the Royal Children’s Hospital Foundation: Postdoctoral Fellowship KLa (2018-984).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
