Abstract
Objective:
We evaluated the effects of bone marrow–derived mesenchymal stem cells in a model of Alzheimer’s disease using serial [18F]Florbetaben positron emission tomography.
Methods:
3xTg Alzheimer’s disease mice were treated with intravenously injected bone marrow–derived mesenchymal stem cells, and animals without stem cell therapy were used as controls. Serial [18F]Florbetaben positron emission tomography was performed after therapy. The standardized uptake value ratio was measured as the cortex standardized uptake value divided by the cerebellum standardized uptake value. Memory function and histological changes were observed using the Barnes maze test and β-amyloid-reactive cells.
Results:
Standardized uptake value ratio decreased significantly from day 14 after stem cell administration in the bone marrow–derived mesenchymal stem cells–treated group (n = 28). In contrast, there was no change in the ratio in control mice (n = 25) at any time point. In addition, mice that received bone marrow–derived mesenchymal stem cell therapy also exhibited significantly better memory function and less β-amyloid-immunopositive plaques compared to controls.
Conclusion:
The therapeutic effect of intravenously injected bone marrow–derived mesenchymal stem cells in a mouse model of Alzheimer’s disease was confirmed by β-amyloid positron emission tomography imaging, memory functional studies and histopathological evaluation.
Introduction
The prevalence of dementia in the elderly population is expected to steadily increase in the future, given the overall pattern of aging society (Brayne and Miller, 2017). Alzheimer’s disease (AD), the most common degenerative brain disease causing dementia, is accompanied by impairments in memory and language function, poor judgment, cognitive dysfunction and eventual loss of all functions of daily life. Deterioration associated with AD can lead to burdens for both patients and their families, resulting in social and economic losses (Hugo and Ganguli, 2014).
Currently, there is no fundamental treatment of AD. A treatment based on the cholinergic hypothesis has been used (Ferreira-Vieira et al., 2016), but with very limited effects. Recently, attempts have been made to use stem cells to treat intractable diseases. Importantly, unlike embryonic stem cells, the use of mesenchymal stem cells (MSCs) in clinical applications does not raise ethical problems, and they have been used to treat various central neurologic diseases (e.g. stroke, trauma, Parkinson’s disease and AD) (Guo et al., 2017; Kim et al., 2015; Kwak et al., 2018; Mendes Filho et al., 2018; Zheng et al., 2018). MSCs are known to secrete various types of cytokines and neurologic factors, and they may therefore have great therapeutic potential in treating neurological disorders (Kyurkchiev et al., 2014; Volkman and Offen, 2017).
Several investigators have studied the effect of MSC therapy on AD (Alipour et al., 2019; Babaei et al., 2012; Bae et al., 2013; Kang et al., 2016; Kim et al., 2015; Lee et al., 2009, 2010; Shin et al., 2014). However, these previous studies primarily focused on assessments of memory/learning functions or immunohistochemistry to reveal the effects of stem cells. Functional assessment can involve subjective factors, and immunohistochemistry is an invasive method based on biopsy with limited clinical applications. Recent advances in imaging technology have led to the use of tools that can visualize brain metabolism and function. In the field of nuclear medicine, positron emission tomography (PET), a non-invasive method for assessing brain β-amyloid deposition, has received much attention in the study of AD. However, evaluations of the efficacy of stem cell therapy using β-amyloid PET in animal models of AD have been rarely reported thus far (He et al., 2010).
We hypothesized that changes in β-amyloid deposits after stem cell therapy could be observed with PET imaging in a mouse model of AD. Therefore, we performed this study to assess the effect of MSC treatment on AD using β-amyloid PET images.
Methods
AD mice
Commercially available 3xTg-AD mice (B6;129-Psen1 tm1Mpm Tg(APPSwe, tauP301 L)1Lfa/Mmjax, MMRRC stock #34830, Jackson Laboratories, Bar Harbor, ME, USA) were used in this study. Details of this model were given in a previous report (Oddo et al., 2003). Briefly, this AD model has three allelic mutations including a homozygous Psen1 mutation and homozygous expression of co-injected APPSwe and tauP301 L transgenes (Tg(APPSwe, tauP301 L)1Lfa). All mice were housed with conditions that included a 12-hour light/dark cycle and free access to food and water. Fifteen-month-old 3xTg-AD mice were used in this study due to the time required to form sufficient β-amyloid plaques (Oddo et al., 2003).
Preparation and transplantation of mouse bone marrow–derived mesenchymal stem cells
B6129SF2/J mice (Jackson Laboratories), weighing 20–25 g, were housed in groups of two or three under environmentally controlled conditions at 23°C ± 2°C and 50 ± 10% relative humidity and given free access to food and water. All experimental procedures were approved by the Institutional Animal Care and Use Committee of Ajou University School of Medicine, Suwon, Republic of Korea (IACUC No. 2015-0068). The method for bone marrow–derived mesenchymal stem cell (BMSC) preparation from mice was based on the protocol published by Soleimani and Nadri (2009). Mouse BMSCs were isolated from the femurs of 6- to 8-week-old female B6129SF2/J mice. Both ends of the tibias and femurs were removed, and the remaining bones were centrifuged at 762g for 20 minutes. The supernatant was discarded, and the cell pellets were resuspended in phosphate-buffered saline (PBS). After additional centrifugation, the cells were resuspended and incubated in high-glucose Dulbecco’s modified Eagle medium containing 15% fetal bovine serum and 1% penicillin/streptomycin (Invitrogen, Carlsbad, CA, USA) at 37°C in a fully humidified 5% CO2 incubator. Non-adherent cells were removed 18 hours later by replacing the medium (passage 0). On day 10 of incubation, the cells were detached with 0.25% trypsin/0.1% EDTA (Sigma, St. Louis, MO, USA) and replated on 100 mm culture dishes (passage 1). In passages 2 and 3, when these primary cultures reached 80% confluence, the cells were harvested using 0.25% trypsin and subcultured, respectively. At passage 4, BMSCs were detached, collected and prepared for transplantation. Mouse BMSCs were characterized by immunofluorescence and fluorescence-activated cell sorter analysis (Park et al., 2008). BMSCs (5.0 × 105 cells/mouse) were suspended in 200 μL of normal saline and then transplanted via the tail vein into AD mice; these comprised the stem cell–treated group (n = 28). The amounts of injected cells were adapted from previously published studies (Goncalves Fda et al., 2014; Song et al., 2009). Twenty-five AD mice treated with 200 μL of saline administered via the tail vein were used as the sham control group.
[18F]Florbetaben PET imaging
A commercially available [18F]Florbetaben (BAY-949172, trade name: NeuraCeq, DuChemBio, Seoul, South Korea) was used to obtain PET images. The end of synthesis-specific activity averaged 774 GBq/μmol and the maximum mass dose was 30 μg for all image acquisitions. PET imaging studies were performed on the day of stem cell treatment as baseline (day 0) before cell administration and at 7, 14, 21 and 28 days after stem cell therapy in each mouse. [18F]Florbetaben PET was also performed on the control mice at the same time. Each mouse was anesthetized using isoflurane (vaporizer to 2% to 2.5%), and 22.2 MBq [18F]Florbetaben was injected intravenously. After 60 minutes of distribution, a brain image of the mouse was obtained using a high-resolution eXplore Vista PET scanner (GE Healthcare, Chicago, IL, USA). Each mouse was scanned for 30 minutes in the prone position with its brain centered in the horizontal field of view. PET images were reconstructed using a three-dimensional ordered subset expectation maximization algorithm with corrections for decay, detector dead time, scatter and random coincidences. The final image resolution in the central field of view was <1 mm at full-width half maximum (FWHM).
PET image analysis
PET data were analyzed with the PMOD Neuro Tool (PNEURO) software, version 3.802 (PMOD Technologies, Zurich, Switzerland). β-amyloid PET images were spatially normalized to the Mouse (Ma-Benveniste-Mirrione) volume of interest (VOI) atlas (Ma et al., 2005; Mirrione et al., 2007) provided by PNEURO. Subsequently, a 0.6-mm filter was applied for smoothing. Transformed PET images were automatically co-registered with the Mouse Brain Atlas T2-weighted magnetic resonance imaging (MRI) template, and segmentation was processed to set the VOIs. Representative images of fused PET and MRI template images and the drawn VOI are presented in Figure 1. Among the 20 VOIs provided by PNEURO (Ma et al., 2005), the main areas of interest were the cerebral cortex, which is the major deposition site of β-amyloid, and cerebellum, which was used as a reference based on previously published studies (Chiquita et al., 2019; Son et al., 2018). The standardized uptake value ratio (SUVR) was defined as the cortical standardized uptake value (SUV) divided by the cerebellar SUV.
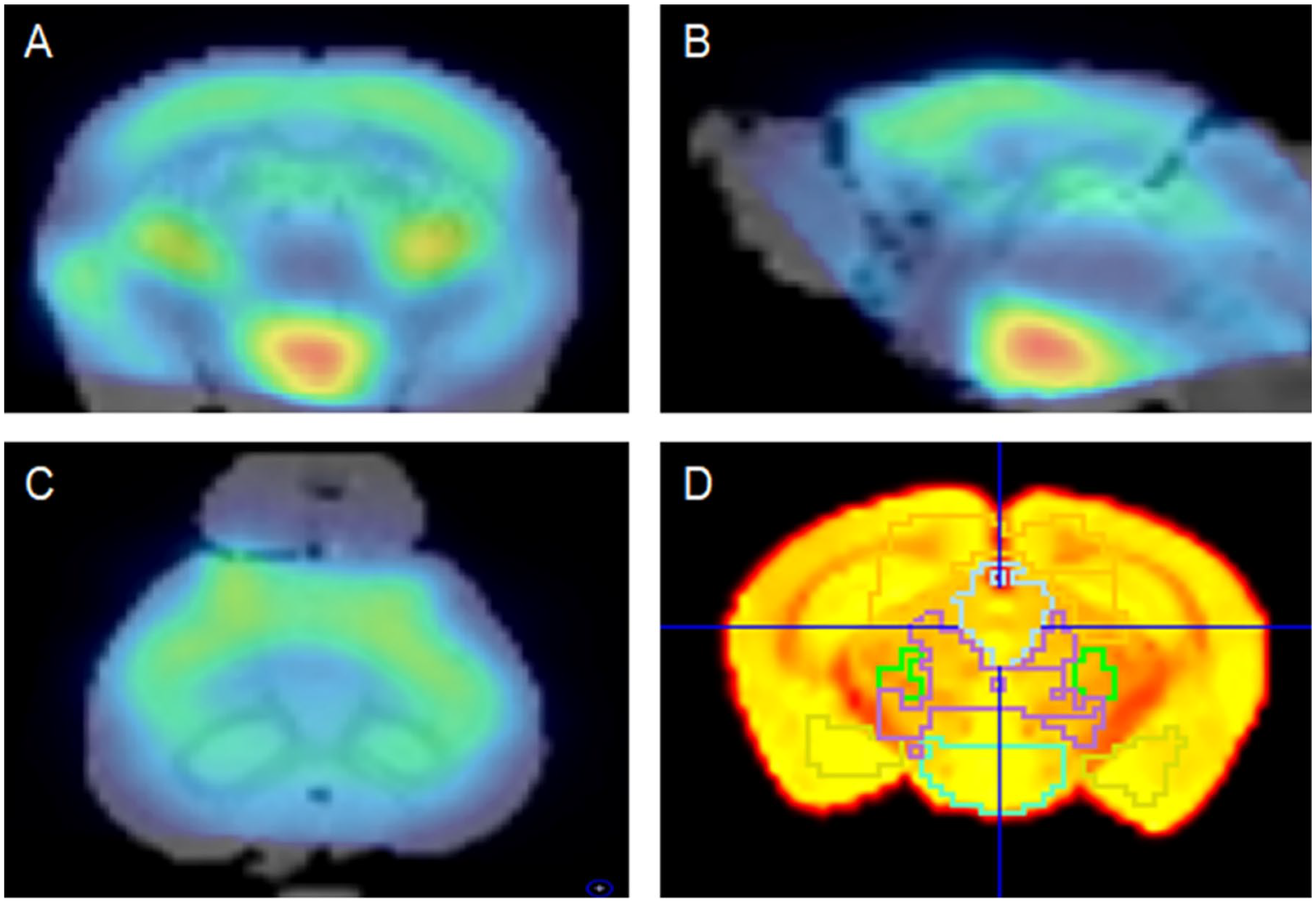
Representative images of fused PET/MRI and drawn VOIs. The [18F]Florbetaben PET image was co-registered with the T2-weighted MRI template: (A) coronal, (B) sagittal and (C) transaxial images. (D) Segmented VOIs were drawn with guidance by the PMOD Neuro Tool using the mouse (Ma-Benveniste-Mirrione) template.
Barnes maze test
To investigate whether stem cell therapy affected memory changes in AD mice, the statuses in each group at 4 weeks after stem cell treatment were evaluated by the Barnes maze test, which was used to measure cognitive and spatial memory function in the AD mice (Attar et al., 2013). The apparatus consists of a white acrylic circular disk with 20 equally spaced holes. One of the holes was connected to the escape box, a small dark chamber located under the platform. The circular disk was virtually divided into four zones, including the target quadrant with the escape hole and the opposite quadrant (Attar et al., 2013). During the adaptation period (day 0), the mouse was placed in a black start chamber for 10 seconds. Then, the chamber cover was lifted and the mouse was gently guided to the escape box. Once the mouse was inside the box, the light was turned off, and the mouse remained in the escape box for 2 minutes. During the spatial acquisition phase (days 1–4), the mouse remained in the black start chamber for 10 seconds and was then allowed to exit the chamber and explore the maze for 3 minutes. Immediately after the mouse entered the escape box, the light was turned off, and the mouse remained in the box for 1 minute. Each mouse completed four trials per day. On day 5, the probe trial was conducted to evaluate memory retention. The target box was removed, and the target hole was closed. Then, the mouse was allowed to explore the maze for 90 seconds after remaining in the black start chamber for 10 seconds. Latency to reach the target hole for the first time, number of entries in the platform zone and time spent in each quadrant were recorded.
Immunohistochemistry
Mice were sacrificed by cervical translocation. The brain was removed and then formalin-fixed and paraffin-embedded for immunohistochemical analysis. Briefly, immunohistochemistry was performed on 4-µm paraffin sections, in accordance with standard protocols. Immunoreactivity was enhanced by heating in 0.01 M citrate buffer (pH 6.0) in a microwave oven and pretreatment with 88% formic acid. The sections were treated with fetal calf serum prior to the addition of the primary antibodies to block non-specific binding sites. The sections were then incubated with primary antibody, rabbit monoclonal [mOC98] antibody to the 1-42-conformation-specific form of β-amyloid (CAT# ab201061, Abcam, Inc., Cambridge, MA, USA; 1:1500) for 15 minutes in a humid chamber at room temperature. Protein expression was then detected with secondary antibody, anti-rabbit immunoglobulin G conjugated to horseradish peroxidase (CAT# ab21488, Abcam, Inc.; 1:300). DAB was used as the chromogen. Counterstaining was performed with hematoxylin. After immunostaining, all slides were scanned using a ScanScope CS (Aperio Technologies, Inc., Vista, CA, USA) and processed with ImageJ freeware (Rasband, W.S., ImageJ, US National Institutes of Health, Bethesda, MD, USA, https://imagej.nih.gov/ij/, 1997-2018). β-amyloid-immunopositive plaques were visually quantified in three different fields (×200) of representative sections, including the cortex and hippocampus, and the total number of plaques were counted. When a plaque showed a strong immunostaining with roughly spherical extracellular deposit pattern, it was counted as a β-amyloid-immunopositive plaque. Areas which showed diffuse weak immunostaining were considered non-specific and not be counted as positive. Quantification was performed in a blinded manner. To reduce potential bias and interindividual variability, all slides underwent the staining protocol simultaneously, and a single investigator performed the quantification (J.-H.K.).
Statistical analysis
All data are presented as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) followed by the Tukey–Kramer post hoc test for pairwise comparisons of subgroups was used to evaluate changes in the SUVR. Independent samples t-tests were used for between-group comparison of data from the memory function test and the number of β-amyloid plaques on pathology analysis. MedCalc Statistical Software version 13.0 (MedCalc Software bvba, Ostend, Belgium) was used for all statistical analyses, and p-values < 0.05 were considered to indicate statistical significance.
Results
Change in β-amyloid plaque activity assessed using [18F]Florbetaben PET
The mean SUVR, used to assess β-amyloid plaque activity in AD mice treated with stem cells, was significantly reduced, from 1.05 ± 0.08 at baseline to 0.89 ± 0.05, 0.86 ± 0.04 and 0.84 ± 0.02 at 2, 3 and 4 weeks after therapy, respectively (ANOVA, p < 0.001; Table 1, Figure 2(A)). The mean SUVR was significantly higher 14, 21 and 28 days post-treatment compared to earlier timepoints. Furthermore, the day 28 post-treatment SUVR was significantly higher than observed at day 14 post-treatment (Tukey–Kramer post hoc method, p < 0.05; Figure 2(A)). In contrast, there were no significant changes from baseline in the mean SUVR in the control group (p = 0.075; Table 1, Figure 2(A)). Representative β-amyloid PET images from both groups are shown in Figure 2(B) and (C).
Serial changes in β-amyloid plaque activity in AD mice with and without stem cell therapy.
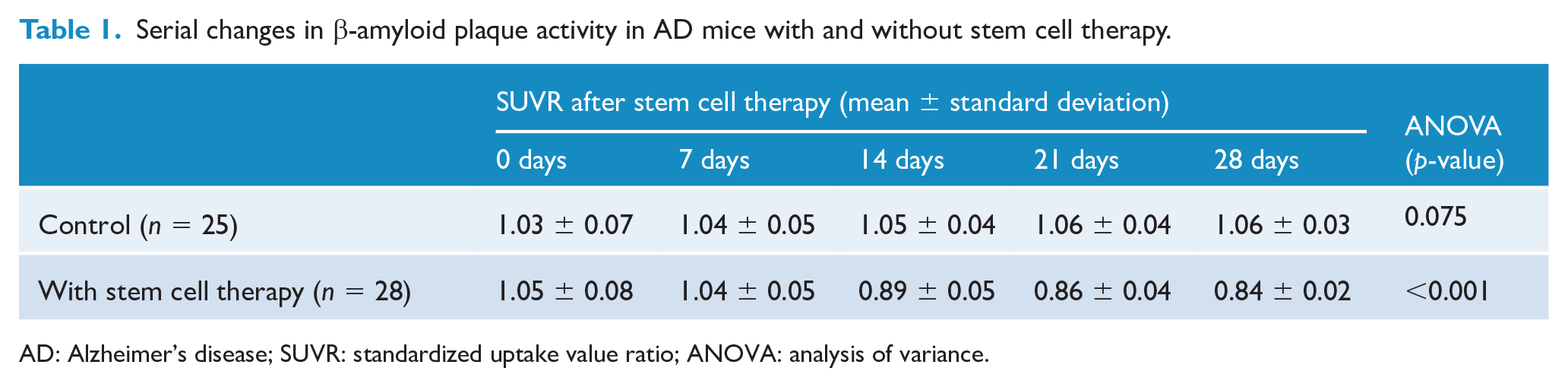
AD: Alzheimer’s disease; SUVR: standardized uptake value ratio; ANOVA: analysis of variance.
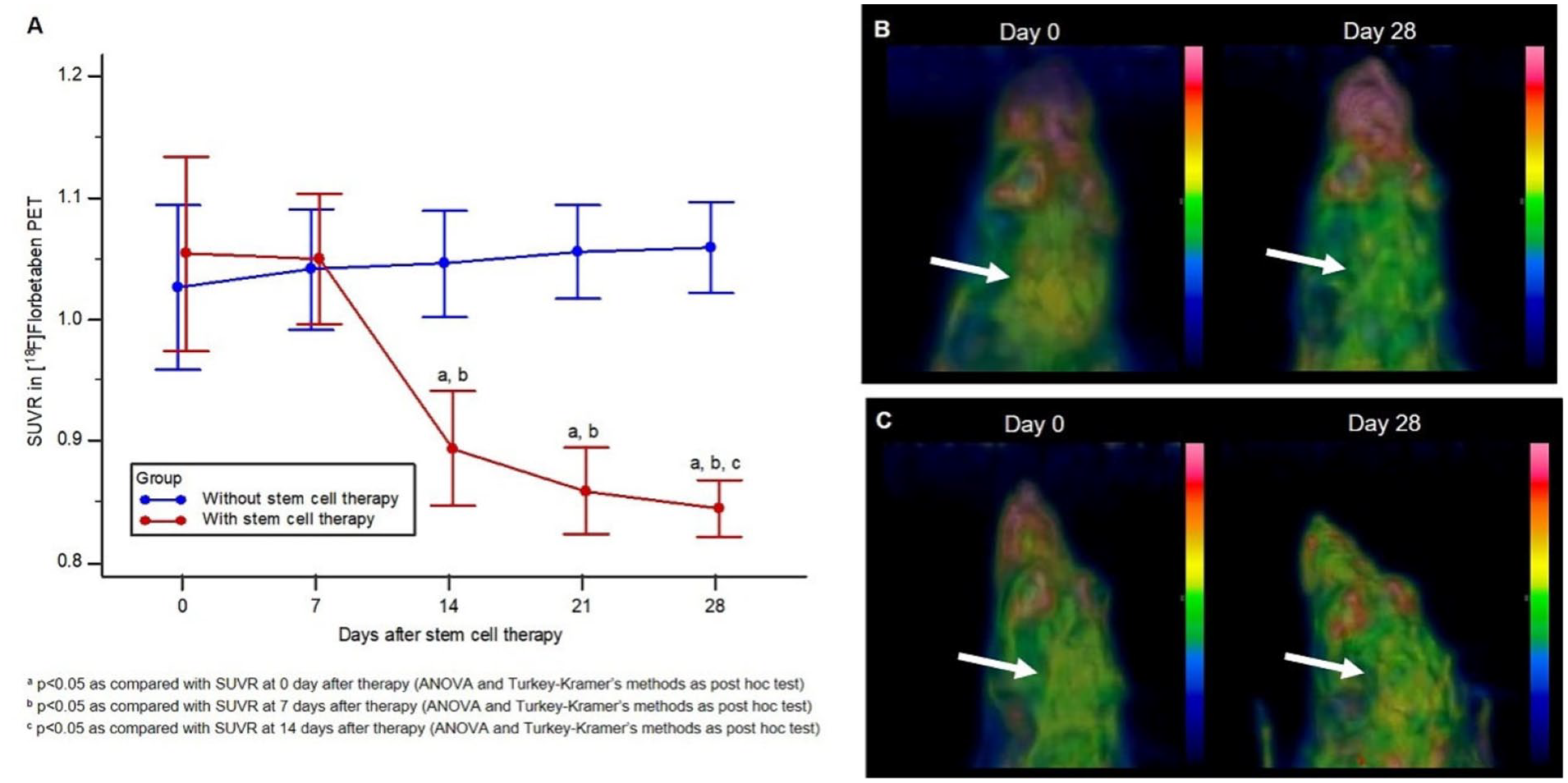
Serial changes in amyloid plaque activity represented by SUVR. (A) The stem cell therapy group exhibited significantly reduced SUVR in serial imaging over time (p < 0.001). SUVRs at 2, 3 and 4 weeks were significantly different from the uptake on day 0 (a) and day 7 (b) after stem cell therapy (p < 0.05, Tukey–Kramer post hoc test). In the graph, points indicate mean values, and error bars indicate standard deviation. (B, C) Representative β-amyloid PET images show β-amyloid deposition in cortex on day 0 (arrow in B and C) in both control and stem cell therapy groups. However, β-amyloid plaque activity was reduced by day 28 of stem cell therapy (arrow in B). There was no change in cortical activity in the control group (arrow in C).
Change in memory function assessed by Barnes maze test
The latency for the stem cell–treated group to reach the target hole (n = 6, 5.2 ± 3.2 seconds) tended to be shorter than that for the control group (n = 5, 11.6 ± 4.5 seconds), but the difference was not statistically significant (p = 0.215, Figure 3(A)). The time spent in the target quadrant tended to be longer in the stem cell–treated group (80.6 ± 17.8%) than in the control group (70.0 ± 21.8%), but this difference also failed to achieve statistical significance (p = 0.423, Figure 3(B)). The frequency of visits to the target hole by the stem cell–treated group (6.2 ± 0.8) was significantly greater than that by the control group (3.4 ± 1.6) (p = 0.010, Figure 3(C)).
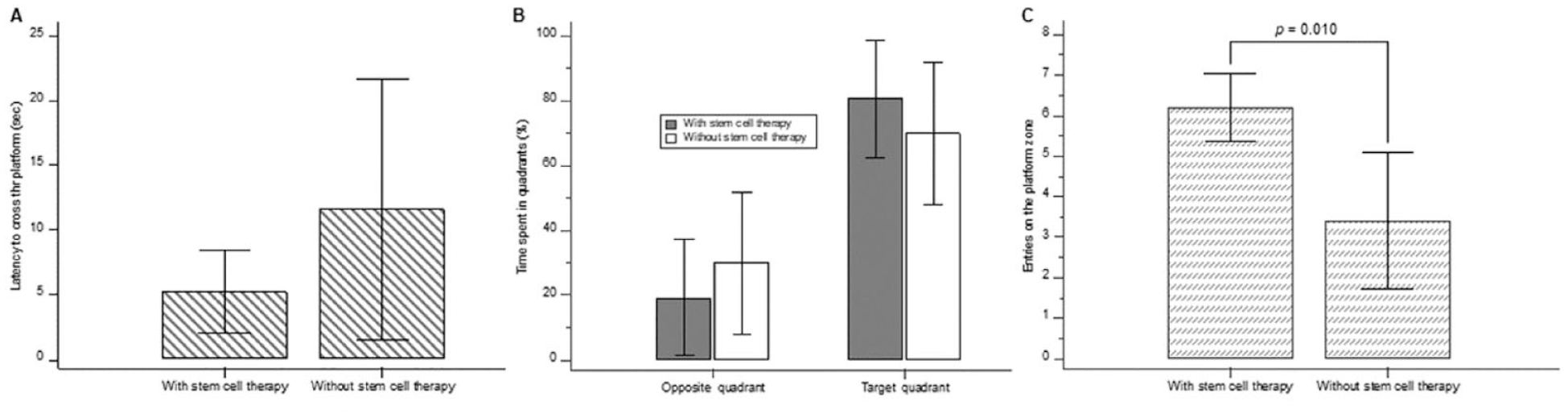
Memory function test results. (A) Latency to reach the target hole was shorter in the stem cell–treated group than in the control group, but the difference was not significant (p = 0.215). (B) Time spent in the target quadrant was longer in the stem cell–treated group than that in the control group, but this difference also did not achieve statistical significance (p = 0.423). (C) The frequency of visits to the target hole significantly differed between the two groups (p = 0.010).
Changes in β-amyloid deposits assessed by immunohistochemical staining
Among the control group (n = 6), five (83.3%) mice showed β-amyloid-immunopositive plaques (mean total number of β-amyloid-immunopositive plaques ± SD: 17 ± 39.8), whereas no β-amyloid plaques were found in the stem cell–treated group (n = 6) (p = 0.008). Representative staining images in each group are presented in Figure 4.
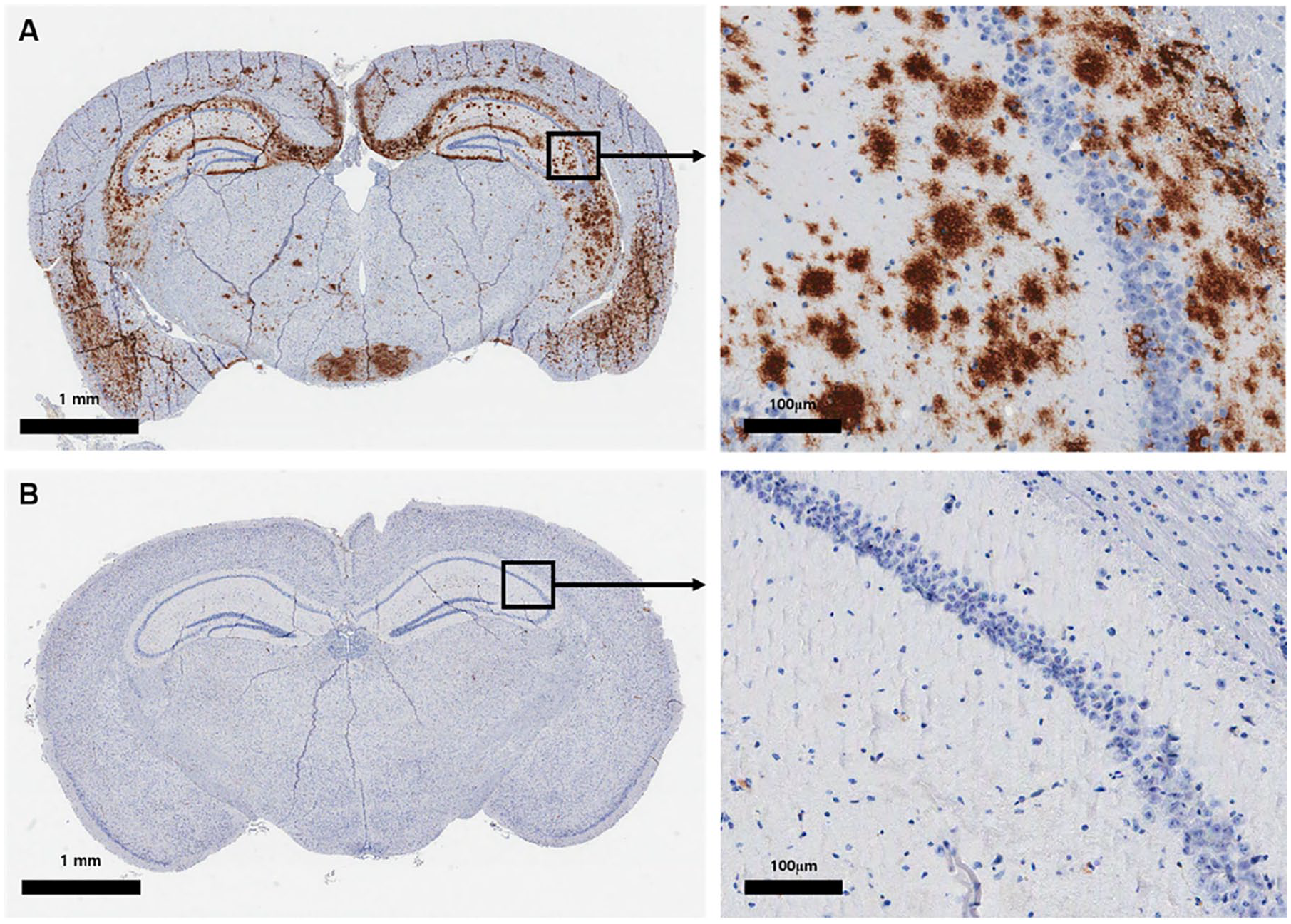
β-amyloid immunohistochemistry. (A) Abundant β-amyloid-immunopositive plaques were identified in the brain of the control group. (B) No β-amyloid-immunopositive plaques were present in the brain of the stem cell–treated group.
Discussion
MSCs have advantages such as easy access, relatively easy handling, possible passage through the blood–brain barrier, intravenous administration, low tumorigenicity and lower immune response compared to other stem cells; thus, they have attracted attention as a disease-modifying treatment method for AD (Kim et al., 2015; Kwak et al., 2018). Efforts have been made to demonstrate the efficacy of BMSCs in AD animal models, and several previous studies have been published (Babaei et al., 2012; Lee et al., 2009, 2010; Malm et al., 2005; Ruzicka et al., 2016; Salem et al., 2014; Shin et al., 2014). These studies report a reduced number of β-amyloid plaques in histopathological analysis after MSC treatment in AD animal models (Lee et al., 2009; Malm et al., 2005; Oddo et al., 2003; Salem et al., 2014; Shin et al., 2014) or recovery of memory and learning abilities after treatment (Babaei et al., 2012; Lee et al., 2010; Ruzicka et al., 2016). Currently, there are ongoing clinical trials in Korea, China and in the United States that aim to assess the effect of stem cells treatment using Fluorine-18-based β-amyloid PET imaging (Kang et al., 2016; Kim et al., 2015); however, we are unable to identify any published results. Previous studies regarding the effect of AD stem cells on β-amyloid PET, as measured in the present study, were difficult to identify. We found a single publication, by He et al. (2010), that reported a change in C-11 Pittsburgh compound B (PIB) after direct administration of neural stem cells to the hippocampus in an AD rat model. The previous studies differed from ours in terms of animal model, stem cells and β-amyloid radiopharmaceuticals used, and it is thus difficult to compare their results with ours. Our study constitutes a groundbreaking investigation of the efficacy of MSCs in transgenic AD mice using β-amyloid PET images, validated with functional and histologic recovery.
Unfortunately, the mechanism by which MSCs exert this therapeutic effect in AD has not yet been fully elucidated, but various factors that are secreted by MSCs have been reported to exhibit effects in the AD brain (Alipour et al., 2019; Kwak et al., 2018). BMSCs are known to exert neuroprotective and immunomodulatory effects by secreting factors such as interleukin (IL)-6, IL-10, transforming growth factor-β and prostaglandin E2 (Aggarwal and Pittenger, 2005; Beyth et al., 2005; Ramasamy et al., 2007). They are also known to interfere with the function of monocyte-derived dendritic cells and to alter natural killer cell phenotypes (Ryan et al., 2007; Sotiropoulou et al., 2006). Previous studies suggested that MSCs can be effective in AD through activation of microglial function (Lee et al., 2009, 2010) and modulation of the autophagy pathway (Shin et al., 2014). Although the efficacy of MSCs in our study likely resulted from these mechanisms, further investigations are warranted in the future to establish a clear mechanism.
This study aimed to investigate the efficacy of MSC treatment in a mouse model of AD with β-amyloid PET imaging. We focused on PET imaging because, although current AD stem cell treatment is at an early stage for clinical applications in patients, we believe that non-invasive PET imaging may be ideal for evaluation of stem cell treatment responses in future patient applications. Clinically, early PET radiopharmaceuticals for imaging β-amyloid plaques most commonly include PIB. Some studies have reported successful PET studies using PIB in AD animal models (Manook et al., 2012; Snellman et al., 2013), but PIB deposition reportedly may not occur in transgenic AD mice (Klunk et al., 2005), resulting in poor availability for imaging. In addition, PIB has a short half-life (20 minutes) because it is labeled with C-11 (20-minute half-life), so it can be used only in institutions with a cyclotron. Therefore, radiopharmaceuticals for β-amyloid deposition images labeled with F-18, which have a relatively longer half-life (110 minutes), have begun to be used in clinical practice; moreover, pre-clinical studies involving animal models of AD have been reported in which [18F]Flutemetamol and [18F]Florbetaben were used (Snellman et al., 2014; Son et al., 2018; Stenzel et al., 2019). In particular, Son et al. (2018) reported that [18F]Florbetaben exhibited more prominent β-amyloid deposits than did [18F]Flutemetamol in transgenic AD mice, confirming that the selection of radiopharmaceuticals was appropriate in the AD mice used in this study. In our study, the cortical SUVR obtained from [18F]Florbetaben PET in 3xTg-AD mice showed an average value of 1.04, which is higher than the average value of 0.82 observed in normal non-transgenic mice (C57BL/6 mice) and that previously reported by Son et al. (2018). Collectively, these findings support the decision to use the [18F]Florbetaben in our study and future investigations.
In this study, stem cell transplantation was performed via intravenous injection. Many stem cells administered intravenously become trapped in the lungs (Fischer et al., 2009), raising concerns that intravenously administered stem cells may not reach the brain. Therefore, we studied the biodistribution of intravenously injected MSCs in 3xTg-AD mice before this study and found that the intravenously transplanted MSCs could reach the brain in the AD mouse model (Park et al., 2018). We administered stem cell therapy intravenously because it was less invasive than administration via intracranial or intraarterial routes and may facilitate use in future clinical applications. In addition, several previous articles have reported the effectiveness of intravenously administered stem cell therapy in AD (Kim et al., 2012, 2013; Yun et al., 2013), and the future of intravenous stem cell treatment in AD appears optimistic.
Among the various AD animal models (LaFerla and Green, 2012), the 3xTg-AD mice used in this study are known to have very similar pathology with respect to β-amyloid plaques, compared to those in AD patients (Oddo et al., 2003). Our β-amyloid immunohistochemistry and Congo red and Bielschowsky staining of 15-month-old 3xTg-AD brain tissue confirmed the validity of 3xTg-AD mice as an AD animal model. We observed an abundance of amyloid plaque and senile plaque in 3xTg-AD mouse brain (Supplementary Figure 1). Also, 3xTg-AD mice have been used successfully in other stem cell therapy studies (Ager et al., 2015; Blurton-Jones et al., 2009; Ruzicka et al., 2016), supporting the use of this model in this study.
This study had some limitations. First, we attempted to demonstrate the therapeutic effect of MSCs in AD using β-amyloid PET as well as functional and pathological assessments; however, the memory function tests and histopathology assays included a small number of mice (Button et al., 2013) and did not include appropriate baseline data. This study mainly focused on β-amyloid imaging, and evaluations of functional and behavioral pathological changes were limited due to research costs and research conditions. Future studies that include a larger sample size and baseline functional and pathological assessments are needed to validate our results. Second, β-amyloid plaques were absent in one control subject and all mice that received stem cell treatment. This surprising result may be related to the presence of lesions in other sections in the mice. Whereas previous studies evaluated at least four sections per mouse (Malm et al., 2005; Shin et al., 2014), our β-amyloid plaque counts were conducted in a single section due to research constraints. Further studies addressing this deficit are needed to clarify the present results. Third, we did not obtain anatomic images, such as magnetic resonance imaging (MRI) or computed tomography (CT), of the mouse brain. If MRI or CT had been obtained with images, we might have obtained better-quality PET fusion images. However, the PET image data this study were sufficient for quantitative analysis, indicating that they posed no critical problem in the study results. Finally, this study did not elucidate a clear causative mechanism for the treatment effect. Here, we focused on visually demonstrating the therapeutic effect of MSCs on β-amyloid PET images. As noted above, further research regarding this issue is needed in the future.
Conclusion
In the transgenic AD mouse model, the reduction of β-amyloid deposits during BMSCs treatment could be confirmed by PET, which could serve as the basis for stem cell therapy in AD patients.
Supplemental Material
Supplementary_AnYS – Supplemental material for Therapeutic effect of mesenchymal stem cells in an animal model of Alzheimer’s disease evaluated by β-amyloid positron emission tomography imaging
Supplemental material, Supplementary_AnYS for Therapeutic effect of mesenchymal stem cells in an animal model of Alzheimer’s disease evaluated by β-amyloid positron emission tomography imaging by Bok-Nam Park, Jang-Hee Kim, Tae Sung Lim, So Hyun Park, Tae-Gyu Kim, Bok Seon Yoon, Keoung Sun Son, Joon-Kee Yoon and Young-Sil An in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a National Research Foundation of Korea (NRF) grant, funded by the Korean government (MEST) (NRF-2015R1C1A2A01052005).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
