Abstract
Introduction:
The inability to extinguish a conditioned fear is thought to be at the core of post-traumatic stress disorder. Eye movement desensitization and reprocessing therapy has been efficacious for post-traumatic stress disorder, but the brain mechanisms underlying the effect are still unknown. The core effect of eye movement desensitization and reprocessing therapy seems to rely on the simultaneous association of bilateral alternating stimulation and the recall of the traumatic memory. To shed light on how eye movement desensitization and reprocessing therapy functions, we aimed to highlight the structures activated by bilateral alternating stimulation during fear extinction and its recall.
Methods:
We included 38 healthy participants in this study. Participants were examined twice in functional magnetic resonance imaging, over 2 consecutive days. On the first day, they performed two fear conditioning and extinction procedures, one with and one without the bilateral alternating stimulation during the fear extinction learning phase in a counter-balanced order across the participants. On the second day, participants completed the fear extinction recall procedure, in the same order as the previous day. Statistical significance of maps was set at p < 0.05 after correction for family-wise error at the cluster level.
Results:
The analysis revealed significant activation with versus without bilateral alternating stimulation at the early extinction in the bilateral auditory areas, the right precuneus, and the left medial frontal gyrus. The same pattern was found in the early recall on the second day. The connectivity analysis found a significant increase in connectivity during bilateral alternating stimulation versus without bilateral alternating stimulation in the early extinction and recall between the two superior temporal gyri, the precuneus, the middle frontal gyrus and a set of structures involved in multisensory integration, executive control, emotional processing, salience and memory.
Conclusion:
We show for the first time that in the eye movement desensitization and reprocessing therapy the bilateral alternating stimulation is not a simple sensory signal and can activate large emotional neural networks.
Keywords
Introduction
The inability to extinguish a conditioned fear is thought to be at the core of post-traumatic stress disorder (PTSD; Milad and Quirk, 2012; VanElzakker et al., 2014) according to results from behavioral and neuroimaging experiments using fear conditioning and extinction protocols in both rodents and humans (LeDoux, 1998; Milad et al., 2005, 2006; Pitman et al., 2012; Wurtz et al., 2016). These studies have suggested that people with PTSD have faster fear conditioning and reduced fear extinction learning and retrieval (Blechert et al., 2007; Milad et al., 2009; Wurtz et al., 2016). The fear conditioning extinction is a learning process involving brain structures with a key role in memory (hippocampus), emotion regulation (insular cortex, anterior cingulate, dorsolateral prefrontal cortex, ventromedial prefrontal cortex, amygdala), multisensory integration (the precuneus, the thalamus, the visual associative cortex, the lateral cerebellum) and motor control (premotor cortex) (Fullana et al., 2018; Gold et al., 2011; Milad et al., 2007; Pitman et al., 2012; Shin et al., 2004; Shin and Liberzon, 2010).
Eye movement desensitization and reprocessing (EMDR) therapy is one of the most efficient therapeutic approaches to treating PTSD symptoms (Watts et al., 2013; World Health Organization (WHO), 2013). This psychotherapy is a structured eight-phase approach using standardized procedure to address the past, present and future aspects of a traumatic memory. EMDR consists of accessing cognitive, emotional and physical aspects of actual distress to traumatic event. Imaginative exposure to the traumatic event is then after proposed in association with bilateral alternating stimulations (BAS). This BAS can be auditory, visual or somesthetic and alternates between the two sides of the body (Servan-Schreiber et al., 2006). The major therapeutic action of EMDR is achieved by the association of the patient’s traumatic memory and the presentation of BAS resulting in a fast extinction of emotional responses elicited by the traumatic memory (Shapiro, 1996, 2014). This results in a change of cognitive processing of memory and cessation of trauma-related distress, while eliminating physical discomfort associated with the initial memory and establishing a positive cognition about the self (Shapiro, 1996). In PTSD individuals, the therapy improves fear extinction learning (Rousseau et al., 2019; Wurtz et al., 2016). More specifically, adding BAS during fear extinction learning therefore models the core feature of EMDR therapy. The approach has alleviated early extinction and long-lasting fear recovery in mice (Wurtz et al., 2016) and recently in humans (Boukezzi et al., 2017). As in EMDR therapy, there was the simultaneous experience of the feeling of fear and the sensation of BAS.
However, studies of BAS in EMDR therapy are sparse, and the specific implication of BAS in the recovery process of EMDR therapy needs further exploration. Only one neuroimaging study explored the brain mechanisms of the BAS action (Herkt et al., 2014). Participants were scanned while being confronted with blocks of neutral and disgusting visual stimuli. Functional magnetic resonance imaging (fMRI) analyses revealed a functional increase in the right amygdala activity and a functional decrease in the left dorsolateral prefrontal cortex activity with BAS added while viewing pictures. Still, the study included only women and did not explore the fear mechanisms that are disrupted in PTSD.
To explore the mechanisms underlying the effect of BAS in a proven model of PTSD and EMDR therapy, in this study, healthy participants underwent scanning while they performed a fear conditioning, extinction and extinction recall paradigm. We administered BAS or not during the fear extinction learning phase. We aimed to highlight the structures activated by the BAS during fear extinction and its recall. We also aimed to explore functional connectivity changes induced by the BAS. We hypothesized that the additional information provided by the BAS would activate the auditory network and also modify the fear extinction learning network, to facilitate fear extinction. EMDR seems to have a stable effect after desensitization of traumatic memories (Högberg et al., 2008), so the BAS should also modify the fear extinction recall network even when not applied during recording. A large amount of regions are involved in fear extinction learning and recall (Fullana et al., 2018; Sehlmeyer et al., 2009), and the BAS should modify the activity and connectivity in structures known to be disrupted in PTSD (Herringa et al., 2012; Koch et al., 2016; Milad et al., 2007, 2009) (i.e. structures involved in emotion regulation, memory and even multisensory integration).
Methods
Participants
We included 38 healthy participants. One of the participants did not complete the experiment because of panic attacks in the scanner. Participants did not have a neurological or psychiatric history. All participants were assessed by a psychiatrist for all mental health disorders with the structured Mini-International Neuropsychiatric Interview for Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (Lecrubier et al., n.d.). Participants were examined twice, over 2 consecutive days (Figure 1). On the first day, they performed two fear conditioning and extinction procedures, one with and one without the BAS in a counter-balanced order across the participants. On the second day, participants completed the fear extinction recall procedure, in the same order as the previous day.
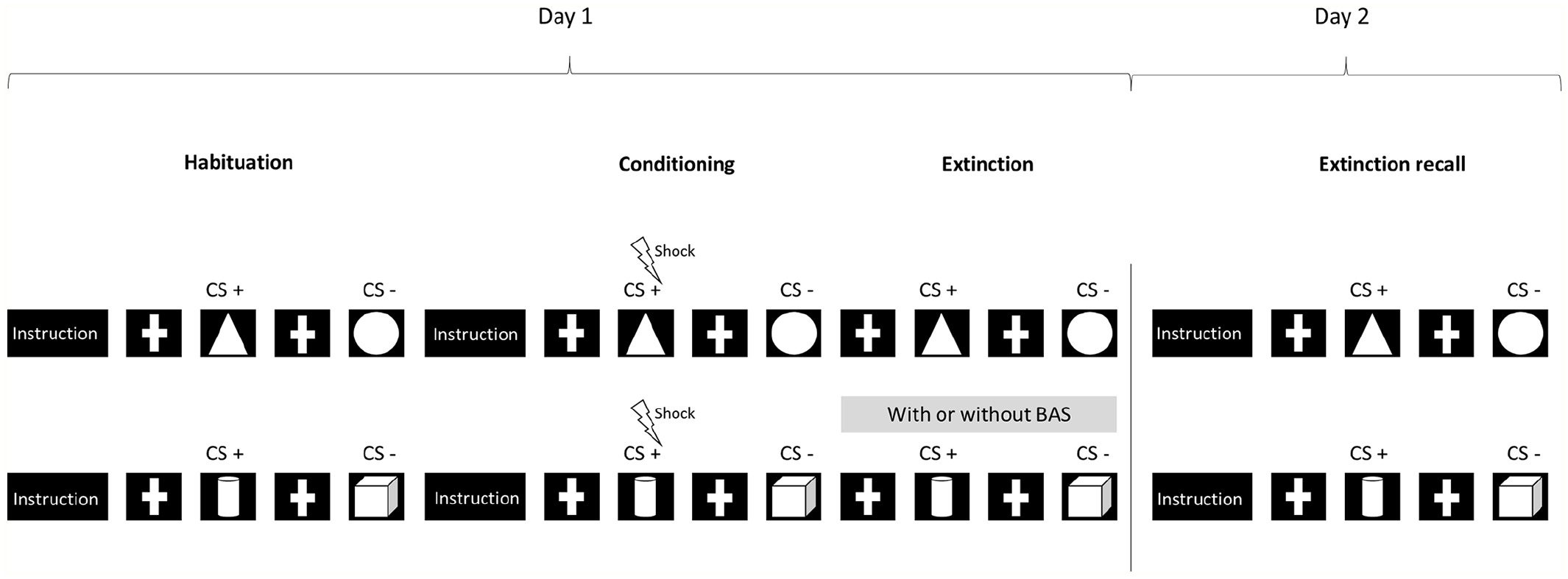
Illustration of the experimental protocol including habituation, fear conditioning, extinction and fear extinction recall with two conditions during the extinction phase, with and without auditory bilateral alternating stimulation (BAS).
Fear conditioning, fear extinction learning and fear extinction recall
We used the same protocol as in a previous behavioral study (Boukezzi et al., 2017) in which PTSD individuals underwent the experimental protocol of fear extinction and recall over 2 separate days (Milad et al., 2005, 2007, 2008, 2009). In our study, each participant performed two different fear conditioning tasks, with or without BAS, during extinction acquisition (Figure 1). The BAS involved auditory large-band tones similar to those used by many EMDR practitioners that were alternately delivered to the right and left ears at a frequency of 1 Hz by use of an MRI-compatible headset.
On the first day, participants underwent the habituation, conditioning and extinction phases in two recording sessions (Figure 1). The BAS was administered during one of the two extinction phases in a counter-balanced order. In one recording session, the stimulus shapes presented in picture form were a triangle and a circle against a dark blue background. The electric shock was accompanied by presentation of the triangle image, as a conditioned stimulus (CS) (CS+). The image of the circle shape (CS−) was never presented with the electric shock. In the second recording session, the stimuli were pictures of a cube and a cylinder against a dark red background. In this case, the CS+ was the cylinder picture and the CS− was the cube picture. For each trial, the CS+ and CS− pictures were presented for 3 seconds in a random order. On the second day, participants underwent the recall phases twice with the shapes used the day before (with or without BAS during fear extinction learning).
On day 1, the fear conditioning and extinction protocol consisted of three phases: habituation, conditioning and extinction. The electric shock was a 500-ms electrical stimulation delivered immediately after the end of the CS+. Electrical stimulation intensity was individually pre-set using an up–down staircase method to achieve a level of ‘unpleasant but not painful’ sensations. The electrical stimulation intensity was kept constant during the conditioning phase.
The habituation phase then started with written instructions announcing that two geometric shapes would appear on the screen and no electrical shock would be delivered. The habituation phase consisted of 12 trials, including 6 as CS+ and 6 as CS− in a random order.
During the conditioning phase, written instructions specified that two shapes would be shown on the screen, with only one occasionally followed by an electrical shock. The CS+ was paired with the unconditioned stimulus (US) at a partial reinforcement rate of 66.7%. The conditioning phase consisted of 24 CS+ and 24 CS− trials.
No instructions were given at the onset of the extinction phase. The extinction phase consisted of 24 CS+ and 24 CS− trials. The only difference between the conditioning and extinction phases was that the CS+ trial was not followed by electrical stimulation during extinction learning. Participants were not informed of the transition from the conditioning to the extinction phase.
Immediately after displaying the shapes on the screen, participants used a five-button keypad in the scanner to respond as quickly as possible to the following question: ‘How much do you expect this stimulus to be paired with an electrical stimulation: 0%, 25%, 50%, 75% or 100%?’ The participant’s responses were recorded for each trial.
On day 2, participants performed the extinction recall with the two series of stimuli used on day 1. Participants were exposed to 24 CS+ and 24 CS− trials as defined for day 1 (with BAS) and then 24 CS+ and 24 CS− trials (without BAS) in two successive scanning sessions, in the same order as for day 1. As soon as the shapes appeared, the participants responded via the five-button keypad as quickly as possible to the question: ‘How much do you expect this stimulus to be paired with an electric stimulation: 0%, 25%, 50% or 100%?’ During this phase, the CS+ trials were not followed by an electric shock and no BAS was delivered.
Electrical stimulation
We used a stimulator powered by a 12-V battery. Electrodes were attached to the participants’ left ankle and filled with isotonic electrode paste. The electrodes delivering the electric stimulation remained attached to the participant’s left ankle throughout the experiment. The US shock occurred for 500 ms immediately at CS+ offset.
Behavioral analysis
To analyze the behavioral results obtained from the response to the question ‘How much do you expect this stimulus to be paired with an electric stimulation: 0%, 25%, 50% or 100%?’, we averaged the percentages obtained for each CS type (CS+, CS−) for six consecutive trials, which resulted in four values for the conditioning phase (C1−C4) and the extinction phase (E1–E4) on day 1 and for the extinction recall phase (R1−R4) on day 2, for the two conditions (with or without BAS). The averaged values for CS+ were subtracted from CS− values. Percentage differences closer to 100 indicated that participants had learned that the electric shock would follow the image (i.e. conditioned fear is acquired), whereas differences closer to 0 indicated that participants no longer differentiated the two stimuli. These differences for the fear extinction and fear recall phases were separately analyzed by two-way repeated-measures analysis of variance (ANOVA) with Condition (with and without the BAS) as a between factor and Time (habituation, fear conditioning [C1−C4], fear extinction learning [E1–E4] and fear extinction recall [R1−R4]) as a within factor. When significant effects were obtained, t test or paired t test with Bonferroni corrections was used as a post hoc test to compare the fear expectation between the habituation and conditioning, conditioning and extinction and the beginning and end of the recall.
fMRI
Brain image acquisition
Data were acquired on a 3-T MEDSPEC 30/80 AVANCE imager (Bruker) at the fMRI center of Marseille, France. Head movements were restricted with foam cushions. After an initial localizing scan, a fieldmap was estimated (64 × 64 matrix; field of view [FOV] = 192 × 192 × 192; excitation pulse angle = 30; bandwidth = 50,000 Hz; echo time = 3.7 ms; repetition time = 0.03 seconds; number of echoes = 2; inter-echo time = 4.552 ms). Functional data were acquired with a T2*-weighted gradient echo-planar imaging (EPI) sequence (repetition time [TR] = 2530 ms, echo time [TE] = 30 ms; FOV = 19.2 × 19.2; 64 × 64 matrix; flip angle = 82.4; voxel size 3 × 3 × 3 mm³). Volumes comprising 38 interleaved axial slices were acquired along the anterior–posterior commissure plane with a continuous slice thickness of 3 mm to cover the whole brain. One functional run consisted of 205 volumes for day 1 and 92 volumes for day 2. After the fMRI scans, high-resolution images were acquired for anatomical identification with a sagittal T1-weighted magnetization-prepared rapid gradient-echo (MP-RAGE) sequence (TR = 9.4 ms; TE = 4.42 ms; inversion time [TI] = 800 seconds; 256 × 256 × 180 matrix; flip angle = 30; voxel size = 1 × 1 × 1 mm3).
Image preprocessing
We used the SPM12 software (www.fil.ion.ucl.ac.uk/spm/software/spm12) to analyze fMRI data. The first functional volumes, corresponding to signal stabilization, were discarded. For the functional images, we first estimated the magnetic field inhomogeneity map. Next, functional images were realigned to control motion effects and to estimate the six head motion parameters and unwarped according to the estimated fieldmap. A slice-timing correction algorithm was used to correct delay in acquisition of individual slices. The high-resolution structural T1-weighted image for each participant was co-registered to the EPI mean images and segmented into gray matter, white matter and cerebrospinal fluid (CSF). A DARTEL template (Ashburner, 2007) was created with the structural images from all participants. The functional volumes were normalized to the Montreal Neurological Institute (MNI) space at 3 × 3 × 3 mm3 voxel resolution with the group template. The resulting data were finally smoothed with a 6-mm-FWHM (full width at half maximum) isotropic Gaussian kernel.
Individual participant analysis
Data from CS+ and CS− trials were modeled separately with a canonical hemodynamic response function to form regressors. The six movement parameters were included in the analyses as regressors of no interest to model residual effects due to head motion. A 128-second high-pass filter was applied to the data to remove low-frequency noise. For each participant, the contrast of CS+ versus CS− was calculated for the four fear extinction periods (E1−E4) and fear extinction recall periods (R1−R4).
Group analysis
We performed a whole-brain analysis for each phase (extinction learning and extinction recall), investigated separately. Group fMRI brain activity was analyzed with a flexible factorial design in SPM with three factors: Participant, Condition (with or without BAS) and Time (E1−E4 for extinction; R1−R4 for extinction recall). We also evaluated the interaction between Condition and Time. Statistical significance of maps was set at p < 0.05 after correction for family-wise error (FWE) at the cluster level. If clusters were the same for the 2 days, a common mask was created using the image calculation facility in SPM12.
Connectivity analysis
After group analysis, we performed a connectivity analysis for the same time periods using the functional Connectivity Toolbox (Conn) for MATLAB (Whitfield-Gabrieli and Nieto-Castanon, 2012). The setup included a loading of functional and structural data and gray and white matter and CSF masks for each participant. The seeds were the significant clusters activated during fear extinction learning and recall. If clusters were the same in the two analyses, the mask of the common voxels served as a seed. Brain areas from the automated anatomical labeling atlas, implemented in the Conn toolbox, were the targets of interest. For each participant and session, we added as a covariate the six rigid-body realignment parameters characterizing the estimated motion as well as a covariate named ‘scrubbing’ that contained the individual images discarded because of excessive movements.
The de-noising step involved applying linear regression and band-pass filtering (0.008−0.09 Hz) to remove unwanted motion, physiological and other artefactual effects from the blood-oxygen-level-dependent (BOLD) signal before computing connectivity measures. The system starts the de-noising step with three different sources of possible confounders: BOLD signal from the white matter and CSF masks (five dimensions each), the within-participant covariates (realignment and scrubbing parameters) and the main conditioning effects (condition blocks convolved with hemodynamic response function).
Fist-level analysis involved correlating the time course from the seeds to whole-brain voxels, creating connectivity maps for each seed region, using bivariate correlations. These connectivity maps were then analyzed at the group level (region of interest [ROI] to ROI module). Statistically significant connectivity was set at pcorrected < 0.05 for false discovery rate (FDR), because FWE correction is not available in the Conn toolbox.
Results
Behavioral results
The mean (±standard deviation [SD]) age of the 37 participants was 32.6 (±1.7) years; 25 were males; the mean education level was 4.78 (±1.93) according to the International Standard Classification of Education standard; and four were left-handed.
Participants correctly completed the conditioning and extinction tasks. For the conditioning phase, whatever the condition, they exhibited greater fear during each conditioning phase (C1−C4) than during the habituation phase with and without BAS, F(1, 35) = 3.87, p < 0.005, and F(1, 35) = 3.71, p < 0.006; p < 0.01 for each phase in the post hoc test. Participants exhibited conditioned fear during fear acquisition, extinguished their acquired fear during extinction learning and recalled expressed extinction memory during extinction recall (Figure 2).
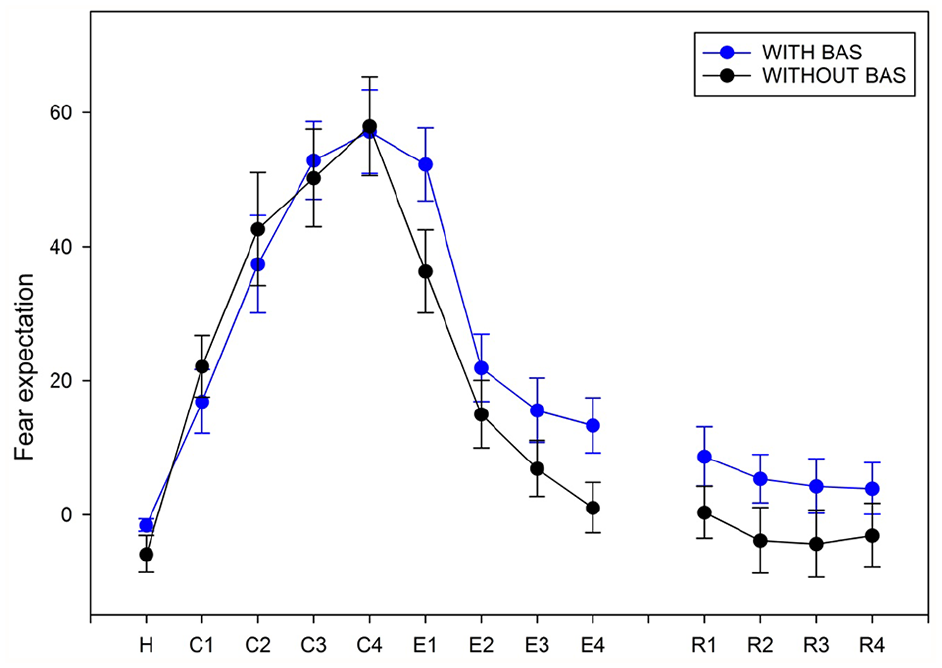
Behavioral responses for participants during the fear conditioning (C), extinction (E) and extinction recall (R) protocols.
During fear extinction, we found a significant effect of only Time, F(1, 35) = 44.16, p < 0.001, on fear expectation; the expectation of fear decreased over time: E1 > E2, E3 and E4 (p < 0.001) and E2 > E4 (p < 0.01).
During fear extinction recall, we found no significant effect of Condition and Time factors and no interaction on fear expectations.
fMRI results
We found no significant activation without versus with BAS for each time of the fear extinction phase (E1−E4). The factorial design analysis revealed some significant activation with versus without BAS only at E1. At time E1, we found significant activation in bilateral auditory areas (including the right and the left superior temporal gyrus) with versus without BAS (Table 1 and Figure 3) as well as increased activity in the right precuneus and the left medial frontal gyrus.
Characteristics of the significant clusters in the extinction (E) and recall (R) phases with versus without auditory bilateral alternating stimulation (BAS) for CS+ versus CS− (see Figure 1).
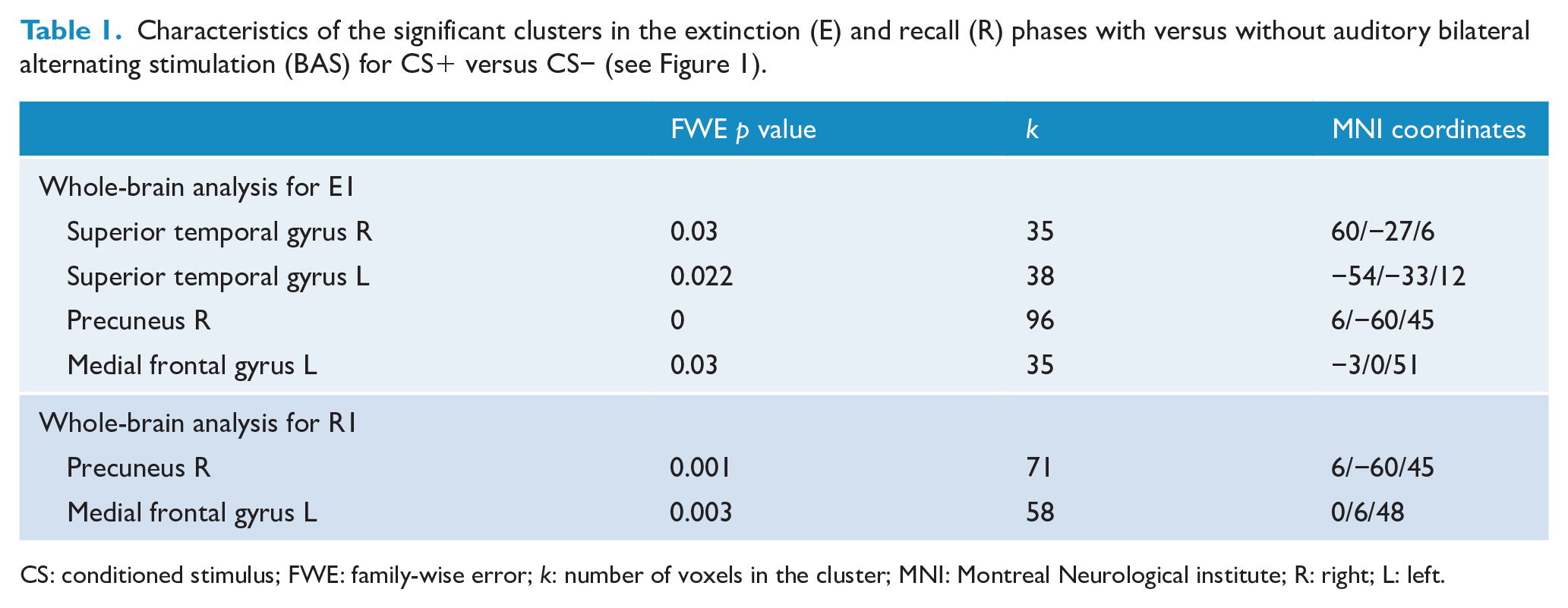
CS: conditioned stimulus; FWE: family-wise error; k: number of voxels in the cluster; MNI: Montreal Neurological institute; R: right; L: left.
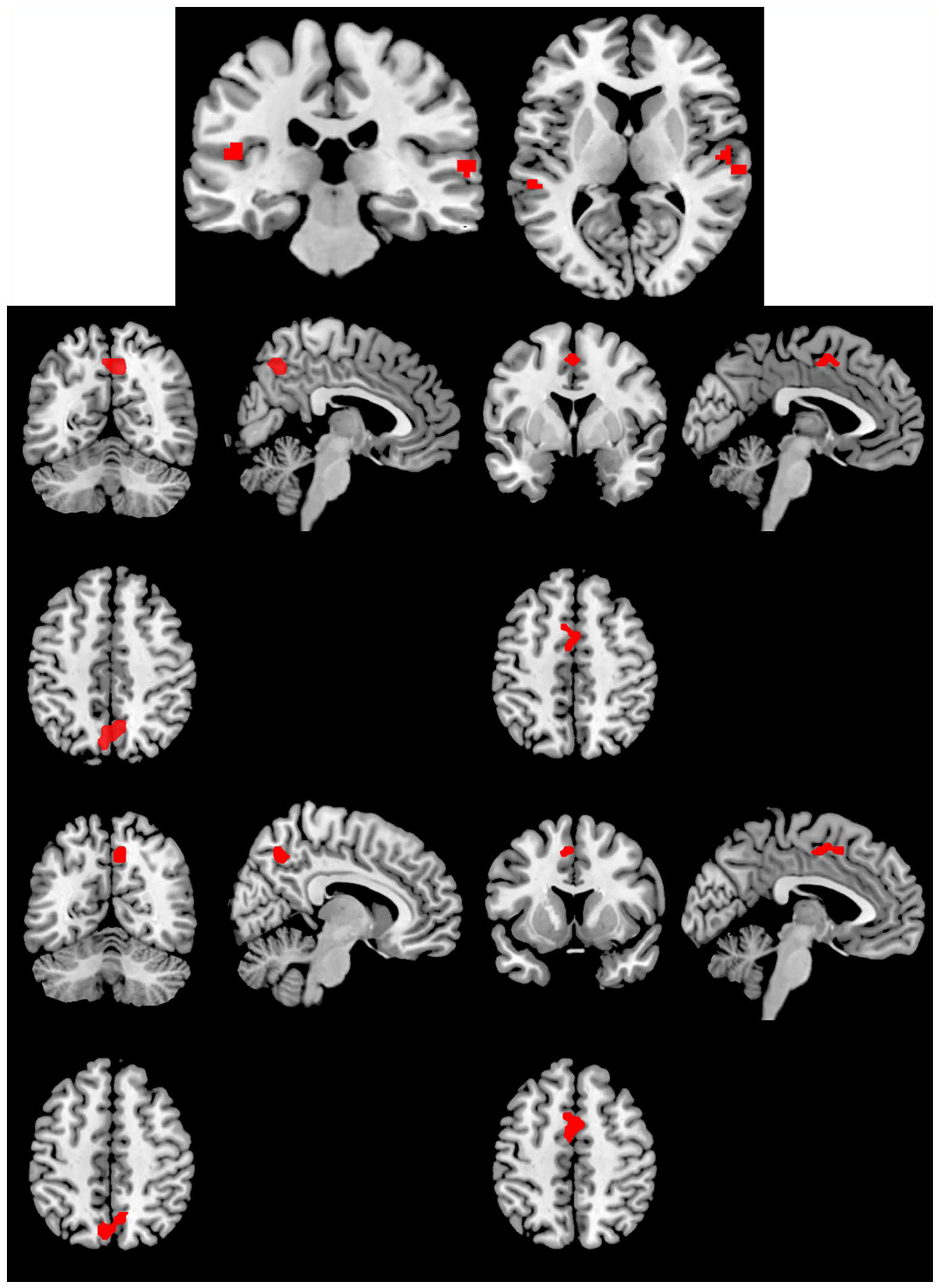
Significantly activated clusters during the extinction (E) and extinction recall (R) phases for the contrast with > without BAS.
We found no significant activation without versus with BAS for each time of the recall phase (R1−R4). The factorial design analyses exhibited significant cluster activations with versus without BAS only at R1 (Table 1 and Figure 3). We also found increased activity in the right precuneus and the left medial frontal gyrus, the same areas activated during E1.
Because two similar clusters of activation were found in both fear extinction learning and recall, we created a common mask. For the precuneus, the common mask shared 97.1% and 58.6% of voxels with the activation cluster for extinction learning and extinction recall, respectively. For the medial frontal gyrus, the common mask shared 67.7% and 91.5% of voxels with the activation cluster for extinction learning and extinction recall, respectively.
We extracted parameter estimates and time courses of response from these common masks for the CS+ and CS− trials for each phase of extinction learning (E1−E4) and extinction recall (R1−R4) and found similar patterns of response for these time courses during fear extinction learning and recall (Figure 4). Results for CS+ versus CS− were driven by the CS+ effect.
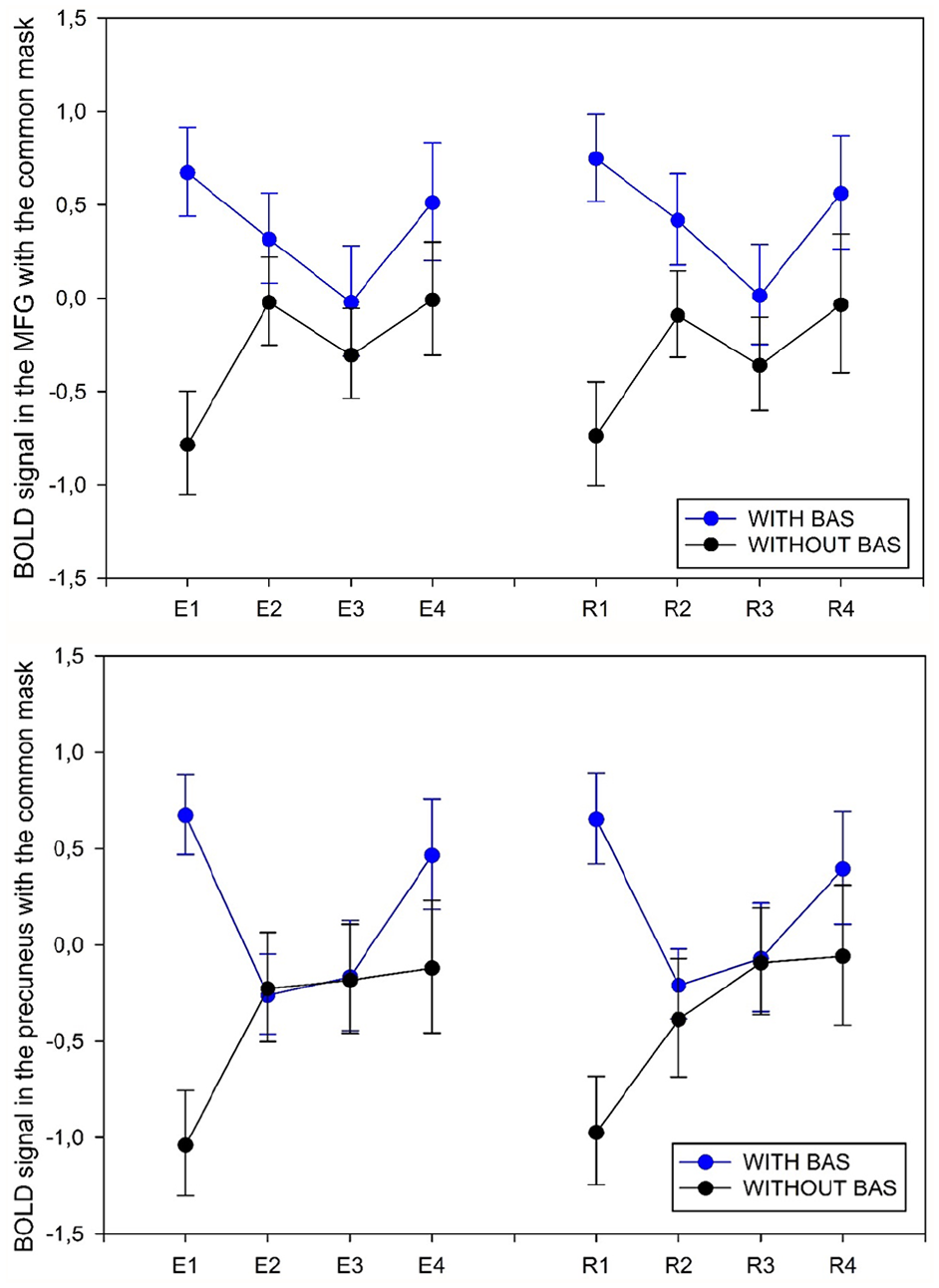
Time course of the BOLD signal during the fear extinction learning (E) and fear extinction recall (R) phases in the common mask for the two significant clusters: the medial frontal gyrus (MFG) and the precuneus. Data are mean ± SD.
Finally, we performed connectivity analysis of the time periods with significant contrasts (i.e. E1 and R1 during the CS+ with vs without BAS) and found a significant increase in connectivity for fear extinction and recall (Table 2 and Figure 5). BAS mainly elicited an increase in functional connectivity between the two superior temporal gyri, the precuneus, the middle frontal gyrus and a set of structures involved in multisensory integration (the cerebellum, the thalamus and the occipital cortex), in executive control such as the frontal operculum cortex or the inferior frontal gyrus, in emotional processing and salience such as the anterior cingulate cortex and the insula, and in memory or containing the frontal eye field such as the middle frontal gyrus.
Characteristics of significant connectivity increases in the extinction and recall phases.
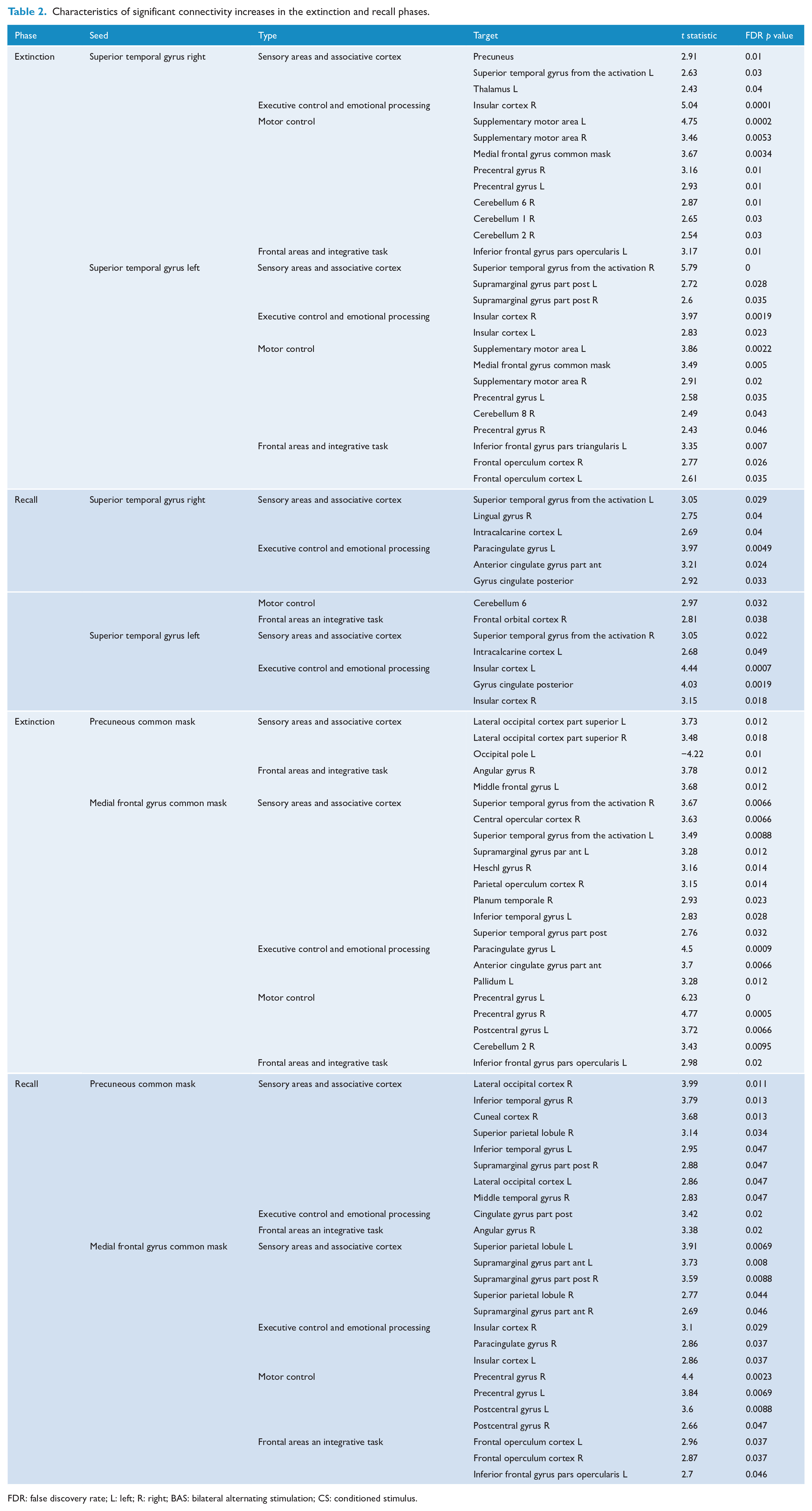
FDR: false discovery rate; L: left; R: right; BAS: bilateral alternating stimulation; CS: conditioned stimulus.
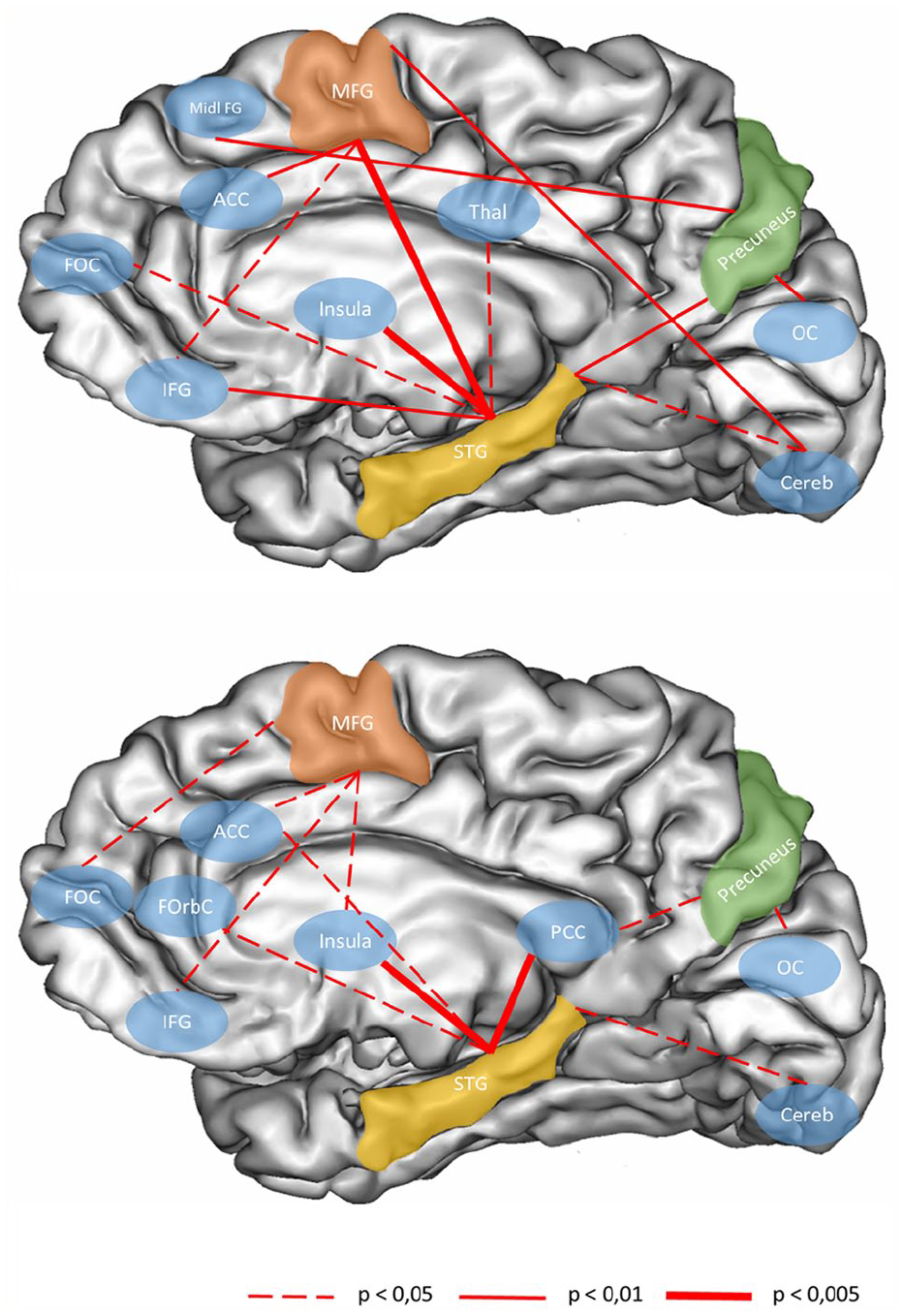
Illustration of the significant connectivity increases during the onset of the fear extinction learning (E1) on the top and the onset of the fear extinction recall phase (R1) below.
Some structures displayed similar increases in connectivity during fear extinction learning and recall. These shared connectivity increases were between the superior temporal gyri, the insular cortex and the cerebellum, between the precuneus and the occipital cortex and finally between the medial frontal gyrus and the inferior frontal gyrus.
Discussion
The core effect of EMDR therapy for PTSD seems to rely on the simultaneous association of BAS and recall of the traumatic memory. To shed light on the EMDR therapy functioning, we investigated the structures activated by BAS during fear extinction and recall in healthy participants. Our participants correctly performed the fear conditioning, extinction learning and extinction recall protocol with and without BAS. However, we did not replicate the facilitating effect of BAS on fear extinction learning found at the behavioral level in humans (Boukezzi et al., 2017). The MRI constraints (lying supine, loud noises in the scanner, anxiety being in a confined space) may explain this discrepancy in part. Further experiments should address the reproducibility of the BAS facilitation effect on fear extinction learning under the scanner conditions at the behavioral level. However, using objective measures, we clearly demonstrated a difference in brain activity with and without BAS.
Our results demonstrate for the first time that BAS during fear extinction activated the auditory cortex, as expected for acoustic stimuli, and also the precuneus and medial frontal gyrus. We found the same activation pattern during extinction and recall. BAS also elicited increased functional connectivity between the two superior temporal gyri, the precuneus, the medial frontal gyrus and a set of structures involved in multisensory integration, executive control and emotional processing.
Our results confirm that BAS activated brain structures and increased connectivity between structures known to be involved in fear extinction and extinction memory recall, as mentioned in two recent meta-analyses based on more than 1300 healthy volunteers (Fullana et al., 2018; Greco and Liberzon, 2016). More specifically, the precuneus has been identified as a key structure in the recall of context-dependent episodic memory (Bonnì et al., 2015), multisensory integration (Freton et al., 2014) and self-perception (Freton et al., 2014; Sartory et al., 2013). This observation may explain why EMDR therapy, by acting on such an integrative structure, can alter the trauma memory. Indeed, BAS involves sounds that do not cause fear and are not intended to elicit amygdala activation and therefore should be interpreted at the brain level as a neutral stimulus. BAS activating the medial frontal gyrus could be related to the latter’s function in integrating sensory information for making decisions, particularly go/no-go types of decisions (Talati and Hirsch, 2005). In our task, participants had to make decisions about the emotional valence of each stimulus and indicate their feelings using the keypad. Therefore, integrating neutral stimuli may add a safe dimension to the trauma memory (Losecaat Vermeer et al., 2014; Orsini et al., 2018) and may also modify decision-making about fear expectations. However, contrary to our hypothesis, the amygdala, a key structure in the pathophysiology of PTSD and in fear extinction (Pitman et al., 2012), did not show any change related to BAS, possibly because the amygdala is not the primary target of the BAS effect on fear extinction.
Our data also show that adding BAS during fear extinction learning and recall increases functional connectivity between the two superior temporal gyri, the precuneus and the medial frontal gyrus on one hand with structures involved in sensory, memory and emotional functions on the other. These functional connectivity changes could be at the core of the EMDR therapy mechanism (i.e. the desensitization of emotions, perceptions, sensations and negative cognitions related to the trauma memory; Shapiro, 1989).
We propose that adding neutral stimuli during the CS+ presentation gradually changed the valence assigned to the stimuli from dangerous to neutral via the brain structures activated by BAS. These changes implicate structures that are known to participate in trauma memory, including the prefrontal cortex, the precuneus, the insula and the sensory cortices (Fullana et al., 2018; Greco and Liberzon, 2016). The insula seems to be of particular importance in BAS action because its increased connectivity with the superior temporal gyri occurred during both extinction learning and extinction recall. Given that this structure is involved in emotion regulation for threat-related stimuli and in salience and monitoring of internal bodily states (Pitman et al., 2012; Veit et al., 2012), BAS could associate the CS+ with the absence of objective external danger felt during the fear extinction learning and recall. In contrast, individuals with PTSD exhibit greater insular cortex activation, possibly reflecting heightened detection of bodily arousal, during the anticipation of aversive images and in response to fearful facial expressions, painful stimuli and traumatic memories (Aupperle et al., 2012; Simmons et al., 2008; Strigo et al., 2010).
What is remarkable is that the day after the fear extinction learning, during recall, we found that activity again increased in the medial frontal gyrus and the precuneus as well as many functional connectivity changes, despite no BAS present. The effects of BAS persisted from one day to the next. This finding suggests that the BAS action was memorized, which could explain in part the lasting effect of EMDR therapy (Bryant et al., 2000; Högberg et al., 2008). Thus, BAS may interact with extinction memory.
Whether the remission from PTSD is due to the effect of treatment on fear reconsolidation (blockade) or extinction (facilitation) is debated. On one hand, the theory of reconsolidation proposes that an emotional reaction pattern is never permanently stored. For instance, when the fear-driven memory is recalled, it can be weakened or completely nullified through psychological or chemical interventions (Lee et al., 2017). One argument is that PTSD may be treated by altering fear memories during its reconsolidation. On the other hand, the theory of extinction suggests that extinction (or the gradual decline in response to a CS+ by the absence of anticipated reinforcement) involves new associative learning that inhibits or interferes with the original memory. For example, fear extinction memory has often been reported to compete with the original fear memory, which demonstrates that both memories co-exist (Garcia, 2002). Another argument is that some PTSD treatments, such as exposure therapy, may therefore work by interfering with memory for trauma-related information. However, whether BAS has an inhibitory effect on reconsolidation or a direct facilitation effect on extinction is unclear.
Given that both reconsolidation and extinction took place during re-exposure to the CS+ (in the absence of the US), during fear extinction training, the first presentation of the CS+ may induce reconsolidation. In line with our findings, in which the BAS effects on brain activity appeared at the beginning of extinction training and the beginning of the extinction recall, during reconsolidation, BAS may induce brain activity that may disrupt reconsolidation, therefore altering a future expression of fear memory. Of note, extinction training during the reconsolidation window can disrupt reconsolidation (Agren et al., 2012), particularly long term, up to 18 months later (Björkstrand et al., 2015). BAS application and extinction training during the reconsolidation window may have similar long-lasting effects, which may explain the persistence of EMDR benefits on PTSD symptoms over 35 months (Högberg et al., 2008). Moreover, in this study, during BAS presentation, extinction did not rely on US omission alone. Indeed, BAS could have represented a novel stimulus presentation during fear extinction learning. In rats and humans, the presentation of a novel non-aversive outcome during extinction training can facilitate a fear extinction memory (Dunsmoor et al., 2015). However, whether structures that are involved in these phenomena, that is, novelty-facilitated extinction (Dunsmoor et al., 2015) and counterconditioning-facilitated extinction (Kang et al., 2018), are similar to those activated by BAS as we identified needs further study.
BAS seems to have a maximal effect on brain activation at the beginning of fear extinction training (when fear is high). However, at the end of fear extinction training (when fear is significantly decreased), BAS had no effect on brain activations. During extinction recall, the same activation pattern was observed for the precuneus and the medial frontal gyrus, with high and low activation at the beginning and end of the recall, respectively. Thus, the BAS effect may depend on the negative emotional load of the memory as if the memory network were more labile when emotions were felt.
One limitation in interpreting these data stems from the lack of the facilitative effect of BAS on fear extinction. Also, we cannot extrapolate these data to PTSD given that we included only healthy participants in our experiment. However, we can make some strong assumptions because we previously demonstrated the effect of EMDR therapy on fear extinction learning in PTSD individuals (Boukezzi et al., 2017; Wurtz et al., 2016). A future study including individuals with PTSD would be informative. Another limitation is the difference between fear conditioning and extinction and the PTSD itself, and BAS administration during the only extinction phase. We administered BAS in the extinction learning phase to mimic what happens during EMDR therapy, and in line with our result in mice and human, showing that BAS performed during fear extinction learning phase alleviated early extinction and long-lasting fear recovery (Boukezzi et al., 2017; Wurtz et al., 2016). Moreover, if BAS had been delivered in all the protocol phases (habituation, conditioning and extinction), we would not have been able to attribute the effect of BAS on brain activity during the extinction phase. However, BAS may induce adaptive reactions at the early extinction that cannot be distinguished from the BAS effect, but could participate to the BAS effect on extinction. BAS may also act as any other extraneous stimulus even if BAS than continuous or intermittent bilateral stimuli and further experiments should focus on this issue. The other limitation is related to the type of paradigm used and its transposition to PTSD. For many years, the fear conditioning paradigm has been successfully used to mimic PTSD (Blechert et al., 2007; Milad et al., 2009; Rousseau et al., 2019). Alterations in fear conditioning, extinction learning and extinction retention are likely to be involved in the development and maintenance of PTSD (Pitman et al., 2012). In addition, PTSD remission parallels fear conditioning and extinction back to normal (Rousseau et al., 2019; Wurtz et al., 2016). Another point is that the paradigm only investigates one dimension of the PTSD. In reality, patients cannot control the intensity of the distress they are feeling and disturbing emotions are not just about fear. We have therefore strong arguments in favor of a likely transposition of our results to PTSD patients, but that should be submitted to experimentation and taken cautiously.
To conclude, our work demonstrates an effect of BAS on brain activity and connectivity that is not limited to sensory activation during fear extinction learning and recall. BAS may facilitate fear extinction in a lasting manner via its effect on multisensory integration, memory formation and emotion processing. Finally, the BAS impact on brain functioning appears to be limited to the period of high fear (at the beginning of extinction learning and the beginning of extinction recall). These results need to be reproduced to confirm and extend them to PTSD individuals and to BAS with sensory modalities other than auditory.
Footnotes
Author Contributions
P.-F.R., S.B. and S.K. collected the fMRI data during the study. P.F.R. and T.C. analyzed the data. All the authors contributed to writing the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
