Abstract
Background:
Up to one in five women meet diagnostic criteria for an anxiety disorder during the perinatal period (i.e. pregnancy and up to 1 year postpartum). While psychotropic medications are effective, they are associated with risks for mothers and babies. There is a growing demand for evidence-based non-pharmacological treatments for perinatal anxiety.
Objective:
To evaluate the effectiveness of a cognitive behavioral group therapy protocol for perinatal anxiety.
Methods:
In total, 96 women were randomized to cognitive behavioral group therapy or waitlist at a clinic specializing in women’s mental health. Participants were 22–41 years of age, pregnant or up to 6 months postpartum and had an anxiety disorder with or without comorbid depression.
Results:
Compared to waitlist, participants in cognitive behavioral group therapy reported significantly greater reductions in the primary outcome of anxiety (State-Trait Inventory of Cognitive and Somatic Anxiety, η2p = .19; Hamilton Anxiety Rating Scale, η2p = .16), as well as in secondary outcomes including worry (Penn State Worry Questionnaire, η2p = .29), perceived stress (Perceived Stress Scale, η2p = .33) and depressive symptoms (Edinburgh Postnatal Depression Scale, η2p = .27; Montgomery–Åsberg Depression Rating Scale, η2p = .11). Maternal status (pregnant, postpartum) and medication use were unrelated to treatment outcomes. All gains were maintained, or continued to improve, at 3-month follow-up.
Conclusion:
Cognitive behavioral group therapy was effective in improving anxiety and related symptoms among women with anxiety disorders in the perinatal period.
The perinatal period is a time of vulnerability for mental health problems in women (Dennis et al., 2017; Giardinelli et al., 2012; Wenzel et al., 2005). Most research has focused on postpartum depression, which affects 13–19% of women in the first year after delivery (O’Hara and McCabe, 2013). Anxiety disorders during the perinatal period are now known, however, to be at least as common, affecting up to 15% of women in pregnancy and up to 20% postpartum (Goodman et al., 2016; Grant et al., 2008). Despite their high prevalence, anxiety disorders in the perinatal period have received considerably less research and clinical attention. This is problematic given the substantial adverse impact anxiety disorders can have on mothers and their infants (e.g. more frequent obstetric complications, poor neonatal and psychosocial outcomes, disruption in bonding and increased healthcare costs; Alder et al., 2007; Andersson et al., 2004; Banhidy et al., 2006; Bauer et al., 2016; Berle et al., 2005; Grigoriadis et al., 2018; Putnam et al., 2017).
Anti-depressants are effective for anxiety and depression in pregnant and postpartum women (MacQueen et al., 2016; Marchesi et al., 2016). However, some medications have been associated with risks for the fetus and infant (Grigoriadis et al., 2013; for example, poor neonatal adaptation syndrome, persistent pulmonary hypertension of the newborn). Best practice guidelines in Canada (MacQueen et al., 2016), Australia (Andrews et al., 2018), the United States (American Psychiatric Association [APA], 2010) and the United Kingdom (McAllister-Williams et al., 2017) recommend non-pharmacological interventions as first-line treatments for depression or anxiety during pregnancy and the postpartum, while psychotropic medications are only recommended for severe symptoms. Moreover, many women prefer non-pharmacological interventions during this period (Dennis and Chung-Lee, 2006). Empirically supported non-pharmacological interventions are therefore imperative for this population.
Cognitive behavioral therapy (CBT) is considered the first-line psychological treatment for anxiety and depressive disorders in the general population (McAllister-Williams et al., 2017). Studies with non-perinatal samples show that CBT is more effective than waitlist or placebo conditions (Cohen’s d = 0.82–1.26) and, in at least some studies, is more effective than other active interventions (d = 0.69–1.20; Covin et al., 2006; Gould et al., 1997; Otto, 2005). Furthermore, gains can be maintained for at least 1 year following treatment for anxiety (DiMauro et al., 2012) and depression (Wiles et al., 2016) and up to 8 years following treatment for worry (Covin et al., 2006; Durham et al., 2003).
A small number of studies have examined the effectiveness of CBT for anxiety symptoms in pregnant or postpartum women with evidence that CBT can be effective (see Goodman et al., 2016; Maguire et al., 2018, for reviews). In a recent meta-analysis, however, Maguire et al. (2018) reported a small between-group effect size (Cohen’s d = 0.49) across studies comparing CBT for perinatal anxiety to control conditions. Moreover, considerable heterogeneity in effect sizes across studies was noted. These findings are somewhat at odds with the strong empirical support for CBT for anxiety disorders in the general population. Moreover, several important limitations were identified, including a reliance on open trials (e.g. Kim et al., 2014; Lilliecreutz et al., 2009; Milgrom et al., 2015; Nieminen et al., 2016), small samples (e.g. Green et al., 2015; Karamoozian and Askarizadeh, 2015; Kim et al., 2014) and a focus on a narrow range of anxiety symptoms and corresponding interventions (e.g. Fathi-Ashtiani et al., 2015; Karamoozian and Askarizadeh, 2015; Nieminen et al., 2016). Given these limitations, the existing empirical support for non-pharmacological interventions for perinatal anxiety must be considered preliminary at best (Goodman et al., 2016; Maguire et al., 2018).
Our research team recently developed a new cognitive behavioral group therapy (CBGT) protocol for perinatal anxiety tailored to meet the unique needs of this population. This protocol (Green et al., 2019) was developed to be transdiagnostic to target any anxiety disorder (e.g. generalized anxiety disorder, social anxiety disorder), with or without comorbid depression, to reach as many women as possible. The protocol includes a range of cognitive and behavioral interventions, all of which have empirical support in the non-perinatal literature. Furthermore, the content of our CBGT protocol targets common anxiety-related beliefs, expectations, predictions and associated behaviors that women report during pregnancy or postpartum and that are likely to occur in the context of the significant life-changing transitions and challenges encountered during this period (e.g. changes in relationships, roles, responsibilities, lifestyles). Finally, the protocol was designed as a six-session treatment to make participation feasible for this population. A single-sample pilot study provided initial empirical support with significant reductions in anxiety symptoms and depressive symptoms as well as high treatment satisfaction (Green et al., 2015). The aim of this study was to validate CBGT for perinatal anxiety in a randomized controlled trial (RCT). We hypothesized that CBGT would be associated with greater reductions in anxiety, as well as secondary outcomes (i.e. worry, depressive symptoms perceived stress), compared to a waitlist. Finally, we expected that the benefits of CBGT would be maintained 3 months post-treatment. Client satisfaction was also assessed.
Methods
A single-blind RCT (Clinicaltrials.gov ID NCT02850523) was conducted between September 2016 and January 2019 at a university-affiliated teaching hospital clinic that specializes in assessment and treatment of women’s mental health during reproductive milestones. The study protocol was approved by the Hamilton Integrated Research Ethics Board in Hamilton, Ontario. All participants provided written informed consent before study entry.
Participants
An initial sample of 96 women was recruited from the regular clinic patient flow. Eligibility criteria were as follows: (1) 18–45 years old; (2) pregnant or within the first 6 months postpartum; (3) a principle diagnosis of an anxiety disorder as per the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) (First et al., 1994) with or without comorbid depression (all diagnoses were checked against Diagnostic and Statistical Manual of Mental Disorders [5th ed.; DSM-5; APA, 2013] diagnostic criteria and, where any discrepancies occurred, DSM-5 criteria were followed); (4) not taking psychotropic medication or, if taking medication, no change in dose and type for a minimum of 6 weeks prior to baseline assessment; (5) no changes in psychotropic medication during 6-week CBGT or 6-week waitlist; (6) no concurrent psychological treatment; (7) fluent in English. Exclusion criteria were as follows: (1) active suicidal ideation and (2) current psychosis or substance use disorder. Screening, eligibility and participation are shown in Figure 1.
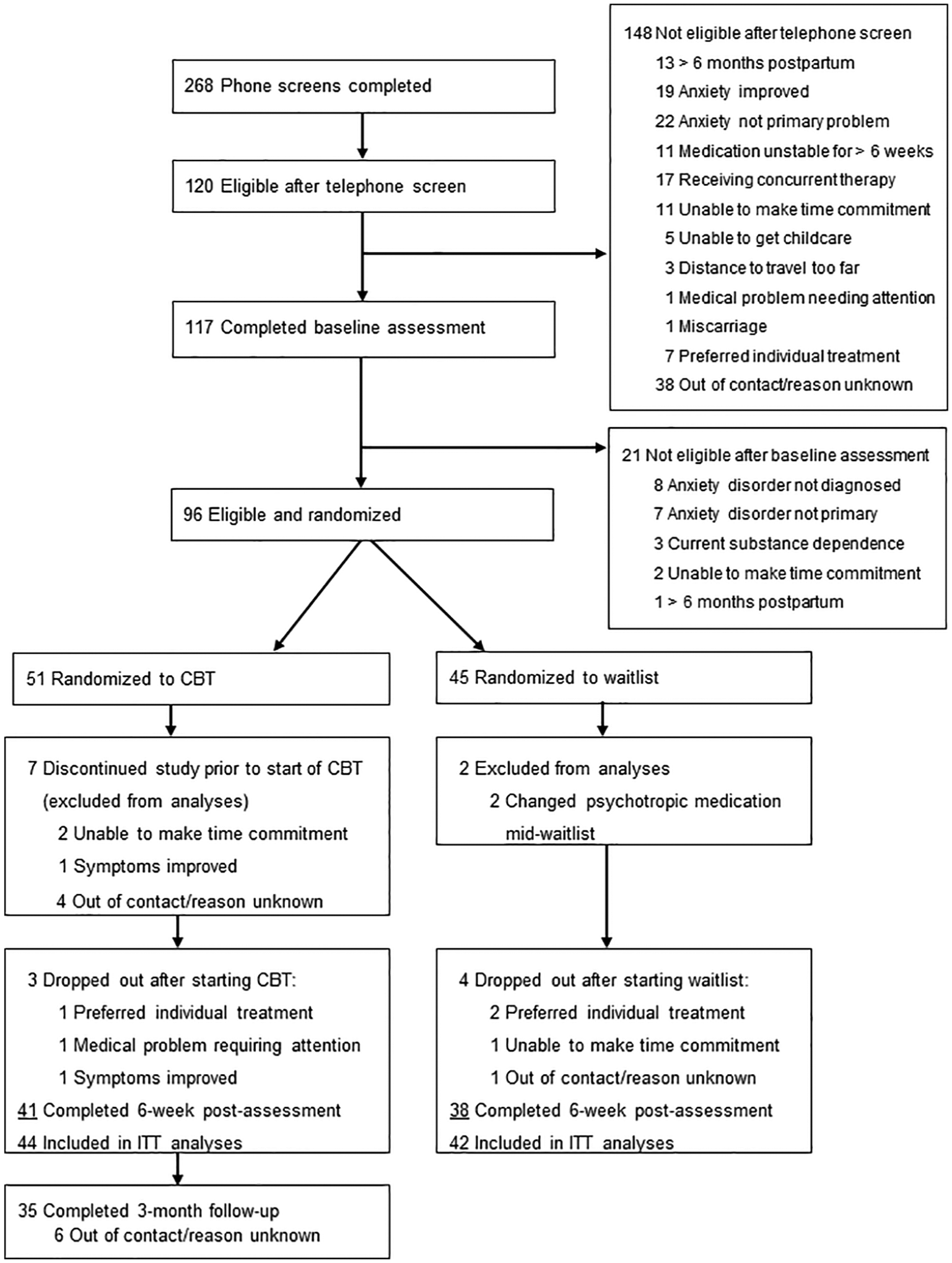
Flowchart of participants.
Randomization and blinding
Participants were randomly assigned to CBGT or waitlist using block randomization, a 1:1 allocation ratio and a computer-generated assignment sequence prepared prior to the study. A semi-structured diagnostic interview, self-report measures and clinician-rated measures were administered at baseline. All measures, with the exception of the diagnostic interview, were re-administered at 6 weeks post-baseline to all participants and at 3 months post-treatment for participants randomized to CBGT. Assessors were blind to treatment condition and timing for all assessments.
Intervention
The CBGT treatment, based on a published manual (Green et al., 2019), was designed to be transdiagnostic to target a range of anxiety symptoms as well as comorbid depressive symptoms. The treatment involved 6-weekly 2-hour sessions in a small-group format (up to six participants per group; range = 4–6). Session content was tailored to address the unique challenges experienced by women with perinatal anxiety and depression (see Table 1 for details). Learning was reinforced with weekly assigned exercises and participant progress was reviewed each week in group.
Six-week CBT protocol for perinatal anxiety session content.
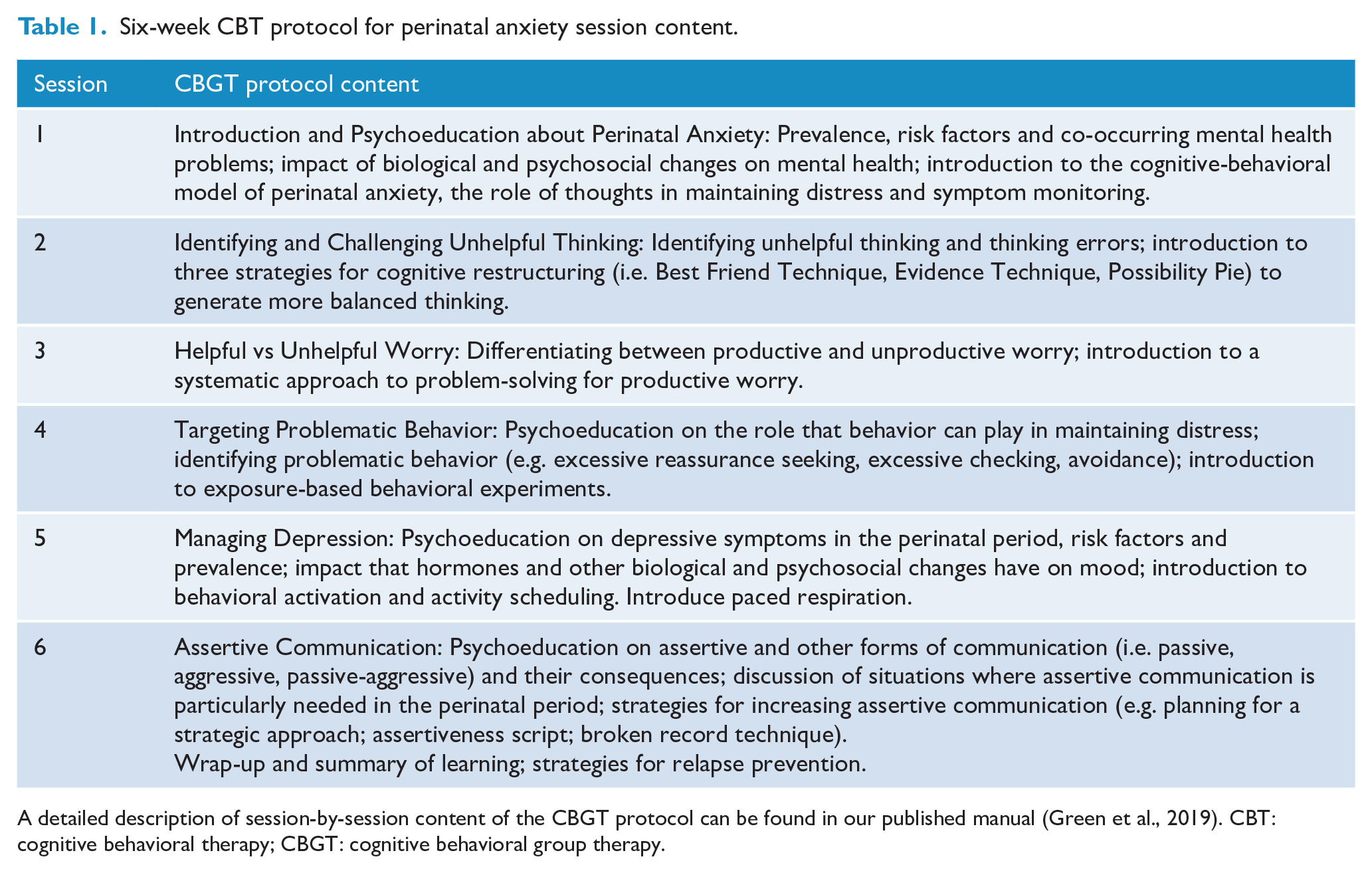
A detailed description of session-by-session content of the CBGT protocol can be found in our published manual (Green et al., 2019). CBT: cognitive behavioral therapy; CBGT: cognitive behavioral group therapy.
Training, supervision and treatment fidelity
Assessors were graduate-level students in clinical psychology, trained in conducting diagnostic assessments. Each treatment group was led by a PhD-level licensed clinical psychologist and a graduate-level psychology trainee. A third staff member (e.g. registered nurse) served as an observer, completing weekly checklists to monitor therapist adherence to the protocol. Supervision for assessments and therapy was provided by licensed clinical psychologists (Drs Green and Donegan).
Measures
Baseline characteristics
Sociodemographic variables collected at baseline included age, ethnicity, education, marital status, perinatal status (pregnant, postpartum) and psychotropic medication use.
Primary outcome
The State-Trait Inventory for Cognitive and Somatic Anxiety, Trait Version (STICSA; Grös et al., 2007) was used to assess self-reported anxiety. The STICSA has excellent psychometric properties and has been validated in a clinical sample of adults with anxiety disorders (score range = 21–84; clinical cut-off ⩾43; Grös et al., 2007). As with all measures in this study, higher scores indicated greater symptom severity. The internal reliability was α = .89 in the study sample.
Secondary outcomes
The Penn State Worry Questionnaire (PSWQ; Meyer et al., 1990) assesses the general tendency to worry, including its excessiveness and uncontrollability (range = 16–80; clinical cut-off ⩾60). The Perceived Stress Scale (PSS-14; Cohen et al., 1983) measures perceived stress over the past month. Scores indicate low (0–13), moderate (14–26) or high (27–40) stress. The Edinburgh Postnatal Depression Scale (EPDS; Cox et al., 1987) assesses depressive symptoms in the past week (range = 0–30; clinical cut-off ⩾13). The EPDS has been validated to assess depressive symptoms in pregnant and postpartum women (Kozinszky and Dudas, 2015). Two clinician-rated measures were also administered: the Hamilton Anxiety Rating Scale (HAM-A; Hamilton, 1959) assesses anxiety symptoms (mild 17, moderate 18–24, severe ⩾25) and the Montgomery–Åsberg Depression Rating Scale (MADRS; Montgomery and Åsberg, 1979) assesses depressive symptoms (mild 7–19, moderate 20–34, severe ⩾35). The measures selected for secondary outcomes are commonly used, with adequate validity and internal reliability (Cohen et al., 1983; Cox et al., 1987; Fantino and Moore, 2009; Hamilton, 1959; Kozinszky and Dudas, 2015; Maier et al., 1988; Meyer et al., 1990; Montgomery and Åsberg, 1979).
Client satisfaction with treatment
The Client Satisfaction Questionnaire (CSQ; Larsen et al., 1979) has adequate validity and internal reliability (Attkisson and Zwick, 1982) and was administered immediately following CBGT to assess the acceptability and perceived benefits of treatment.
Statistical analysis
Participants in CBGT and waitlist were compared on baseline clinical and demographic characteristics with independent-sample t tests or chi square (χ2). An intention-to-treat (ITT) approach and mixed effects analyses of covariance (ANCOVAs) were used to compare the effectiveness of CBGT to waitlist from baseline to 6 weeks post-baseline. Covariates were (1) medication use (taking or not taking medication for anxiety or depressive symptoms) and (2) perinatal status (pregnant or post-partum). A last-observation-carried-forward approach was used for missing data due to drop-outs at 6 weeks post-baseline. Within-group comparisons and the maintenance of gains among treatment completers in CBGT by 3-month follow-up were assessed with paired-samples t tests. Partial eta-squared (η2p) and Cohen’s d values (post-treatment mean − 3-month follow-up mean/pooled standard deviation) were computed to estimate effect sizes. Statistical analyses were conducted using IBM SPSS Statistics (Release 25).
Results
Participant characteristics at baseline
Following randomization (but before starting treatment), seven participants assigned to CBGT declined further participation or became ineligible and were excluded from analyses (see Figure 1). This resulted in an ITT sample of 86 participants (Mage = 31.91 years, SD = 3.61, range = 22–41). One-third of participants (n = 31; 36.0%) were pregnant and two-thirds (n = 55; 64.0%) were within the first 6 months postpartum. Approximately one-third of participants (n = 33; 38.4%) were taking psychotropic medication for anxiety or depressive symptoms. All participants met DSM-5 diagnostic criteria for at least one anxiety disorder. Principal anxiety disorders were generalized anxiety disorder (n = 73; 84.9%), social anxiety disorder (n = 8; 9.3%), panic disorder (n = 2; 2.3%) and other specified anxiety disorder (n = 3; 3.5%). About one-third of participants (n = 31; 39.7%) met diagnostic criteria for at least one secondary anxiety disorder or obsessive-compulsive disorder, illness anxiety disorder or post-traumatic stress disorder. Approximately one-third of participants (n = 25; 29.1%) met diagnostic criteria for a secondary depressive disorder (e.g. major depressive disorder). There were no significant between-group baseline differences on demographic or clinical variables (Table 2).
Baseline demographic and clinical characteristics by intervention group.
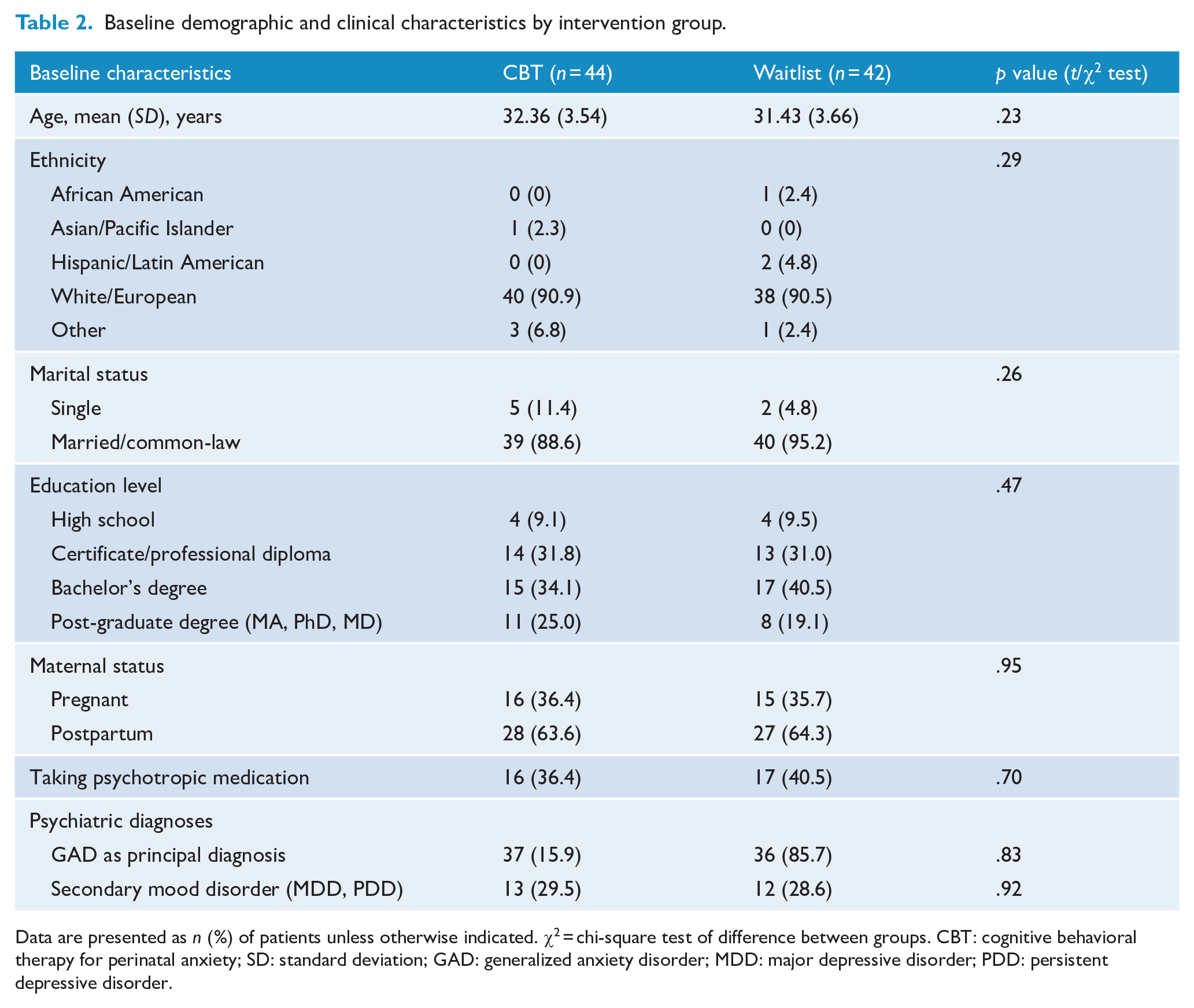
Data are presented as n (%) of patients unless otherwise indicated. χ2 = chi-square test of difference between groups. CBT: cognitive behavioral therapy for perinatal anxiety; SD: standard deviation; GAD: generalized anxiety disorder; MDD: major depressive disorder; PDD: persistent depressive disorder.
Intervention engagement, treatment discontinuation and therapist adherence
Participant drop-outs from CBGT (n = 3) had completed only one to four treatment sessions or did not provide 6-week post-baseline data. Participant drop-outs from waitlist (n = 4) did not provide 6-week post-baseline data. The difference in participant drop-outs between groups was not statistically significant, χ2 (1) = 0.18, p = .67. In each CBGT session, raters assessed therapist adherence to the protocol using a pre-determined content checklist. Therapist adherence was high, with therapists delivering 98.7% to 100% of prescribed content across sessions.
Primary outcome
A mixed-effects ANCOVA was conducted on the STICSA from baseline to 6 weeks post-baseline, with medication use and perinatal status as covariates. All ANCOVA results are reported in Table 3. There was a significantly greater reduction on the STICSA in CBGT compared to waitlist, F(1, 78) = 18.25, p < .001, η2p = .19. There was also a significant interaction between medication use, maternal status and anxiety symptoms over time, F(1, 78) = 4.15, p = .045, η2p = .05, such that women who were pregnant and taking medication were more anxious at baseline than pregnant women who were not taking medication, mean difference = 9.38, p = .03. However, neither medication use nor maternal status was a significant moderator of the effect of condition. There were no other statistically significant interactions with covariates (all p > .05). Finally, when mean STICSA scores were directly compared at 6 weeks post-baseline, participants in CBGT reported significantly lower anxiety symptoms compared to participants in waitlist, t(84) = 2.39, p = .02, d = 0.52.
Mixed effects ANCOVAs comparing CBGT (n = 44) to waitlist (n = 42) on outcomes from baseline to 6 weeks post-baseline.
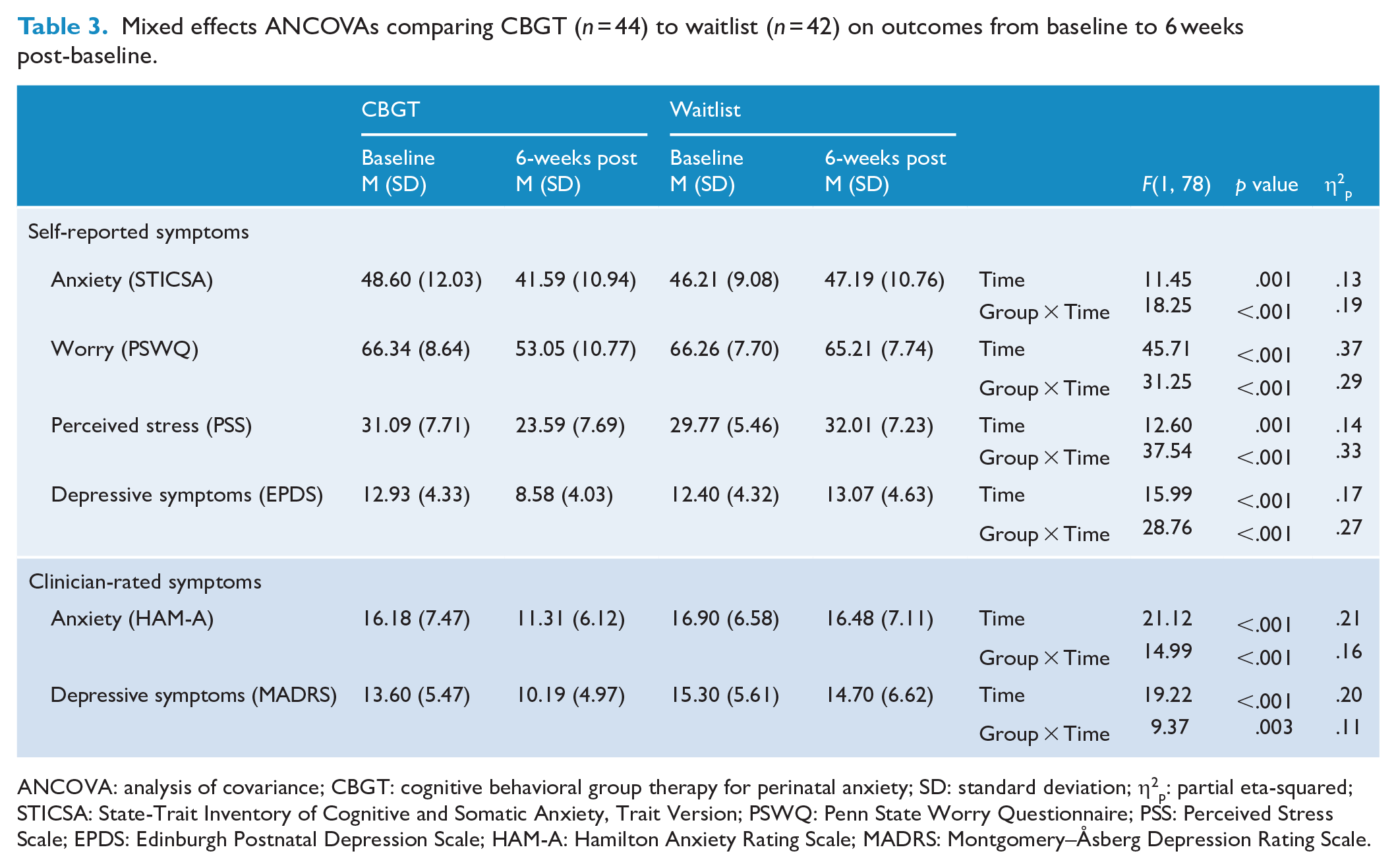
ANCOVA: analysis of covariance; CBGT: cognitive behavioral group therapy for perinatal anxiety; SD: standard deviation; η2p: partial eta-squared; STICSA: State-Trait Inventory of Cognitive and Somatic Anxiety, Trait Version; PSWQ: Penn State Worry Questionnaire; PSS: Perceived Stress Scale; EPDS: Edinburgh Postnatal Depression Scale; HAM-A: Hamilton Anxiety Rating Scale; MADRS: Montgomery–Åsberg Depression Rating Scale.
CBGT and waitlist were also compared on the percentage of participants whose scores fell above the clinical cut-off on the STICSA (⩾43; Grös et al., 2007). The majority of participants in CBGT (n = 30, 68.2%) and waitlist (n = 25, 61.9%) had scores above the clinical cut-off at baseline (χ2(1) = 0.37, p = .40). At 6 weeks post-baseline, only 36.4% (n = 16) of participants in CBGT fell above the clinical cut off, whereas the majority in waitlist (n = 25, 59.5%) remained above. This difference was statistically significant (χ2(1) = 4.62, p = .03).
Secondary outcomes
Mixed effects ANCOVAs were conducted on secondary outcomes from baseline to 6 weeks post-baseline, with medication use and perinatal status as covariates. There were significantly greater reductions in CBGT in self-reported worry (PSWQ), F(1, 78) = 31.25, p < .001, η2p = .29; depressive symptoms (EPDS), F(1, 78) = 28.76, p < .001, η2p = .27; and perceived stress (PSS), F(1, 78) = 37.54, p < .001, η2p = .33, compared to waitlist. There were no significant interactions with maternal status or medication use (all p > .05). When directly compared to waitlist participants at 6 weeks post-baseline, participants in CBGT reported significantly lower levels of worry, PSWQ, t(84) = 6.0, p < .001, d = 1.30; depressive symptoms, EPDS, t(84) = 4.80, p < .001, d = 1.03; and perceived stress, PSS, t(84) = 5.23, p < .001, d = 1.13.
CBGT and waitlist were compared on the percentage of participants who were above the clinical cut-off on the EPDS (⩾13; Cox et al., 1987) at baseline and 6 weeks post-baseline. At baseline, half of participants in CBGT (n = 22, 50.0%) and on waitlist (n = 22, 47.6%) had scores above the clinical cut-off, χ2(1) = 0.05, p = .83. However, at 6 weeks post-baseline, only 15.9% (n = 7) in CBGT versus 54.8% (n = 23) in waitlist were above the clinical cut-off. This difference was statistically significant, χ2(1) = 14.28, p < .001.
With respect to clinician-rated anxiety (HAM-A), there was a significantly greater reduction in anxiety symptoms in CBGT compared to waitlist, F(1, 78) = 14.99, p < .001, η2p = .16. There was also a significant interaction between medication use, maternal status and anxiety symptoms over time, F(1, 78) = 5.60, p = .02, η2p = .07. Specifically, at baseline, women who were pregnant and taking psychotropic medication were more anxious than pregnant women who were not taking medication, mean difference = 6.87, p = .02. However, as with the STICSA, neither medication use nor maternal status was a significant moderator of the effect of condition (all p > .05). Participants in CBGT were also rated by clinicians as having significantly lower anxiety scores on the HAM-A compared to waitlist participants at 6 weeks post-baseline, t(84) = 3.62, p < .001, d = 0.78.
In terms of clinician-rated depressive symptoms (MADRS), participants in CBGT experienced a significantly greater reduction in depressive symptoms than participants in waitlist, F(1, 78) = 9.37, p = .003, η2p = .11. Neither maternal status nor medication use was a significant moderator of condition (all p > .05). Participants in CBGT were also rated by clinicians as having significantly lower depressive symptoms on the MADRS compared to waitlist participants at 6 weeks post-baseline, t(84) = 3.59, p = .001, d = 0.77.
Treatment satisfaction
Participants’ satisfaction with CBGT was assessed at post-treatment. All participants rated the treatment as ‘excellent’ (95.7%) or ‘good’ (4.3%), reported that treatment helped them cope ‘better’ (30.4%) or ‘a great deal better’ (69.5%) with their problems, were ‘very satisfied’ (87.0%) or ‘mostly satisfied’ (8.7%) with treatment and all reported that they would recommend the treatment to others.
Three-month follow-up
Among women who completed CBGT, gains were maintained or further improvements observed, at 3-month follow-up. Specifically, there was a significant mean reduction in self-reported anxiety (STICSA; MPost = 42.33, M3mo = 38.78; t(34) = 2.58, p = .02, d = 0.33) from post-treatment to 3-month follow-up. Gains achieved during treatment were maintained in all other outcomes, including worry (PSWQ: MPost = 52.93, M3mo = 50.59; t(34) = 1.42, p = .17, d = 0.19), depressive symptoms, (EPDS: MPost = 8.89, M3mo = 7.52; t(34) = 1.78, p = .09, d = 0.21) and perceived stress (PSS: MPost = 23.10, M3mo = 22.14; t(34) = 0.76, p = .46, d = 0.11) as well as clinician-rated anxiety (HAM-A: MPost = 11.07, M3mo = 9.04; t(34) = 1.75, p = .09, d = 0.35) and depressive symptoms (MADRS: MPost = 9.50, M3mo = 7.79; t(34) = 1.35, p = .19, d = 0.31).
Discussion
Best practice guidelines (e.g. McAllister-Williams et al., 2017; MacQueen et al., 2016) recommend non-pharmacological interventions as first-line treatments for women with perinatal anxiety disorders. Despite a small number of promising studies demonstrating that CBT can be effective in reducing anxiety among pregnant and postpartum women, limitations in the existing literature suggest that the evidence thus far must be considered preliminary (Goodman et al., 2016; Maguire et al., 2018). The aim of this study was to add to the emerging treatment outcome literature by evaluating a new CBGT protocol for women with perinatal anxiety disorders with or without comorbid depression. The findings indicate that women in CBGT experienced significantly greater reductions in both self-reported and clinician-rated anxiety symptoms compared to women on the waitlist. Furthermore, a majority of participants in CBGT (but not in waitlist) scored below the clinical cut-off on self-reported anxiety symptoms (STICSA). These gains were maintained at 3-month follow-up with one notable exception: women who completed CBGT showed continued reductions in self-reported anxiety in the 3 months following treatment. This indicates that women can continue to benefit from the CBGT intervention once treatment has ended. Finally, women participating in CBGT experienced these benefits regardless of maternal status (pregnant, postpartum) and psychotropic medication use.
CBGT was also effective in reducing secondary symptoms, including worry and perceived stress, again regardless of maternal status or medication use. Worry is a common symptom across anxiety disorders (Drost et al., 2014) and therefore an important target in a transdiagnostic treatment. Importantly, the magnitude of the between-group difference in worry at 6 weeks post-baseline was comparable to studies evaluating CBT for anxiety disorders in non-perinatal samples (Covin et al., 2006; Gould et al., 1997; Otto, 2005). In addition, the positive impact of CBGT on stress is encouraging given that stress is associated with adverse outcomes in the perinatal period (Navarro et al., 2008).
The high comorbidity between anxiety and depression in the perinatal period makes it imperative that treatments target both in an effective manner. This is particularly true given that depression is also associated with adverse perinatal outcomes (Alder et al., 2007; Andersson et al., 2004; Berle et al., 2005). To our knowledge, our CBGT protocol is one of the first protocols to target anxiety disorders as principal diagnoses while also including components that directly target depressive symptoms. With that, CBGT was found to be significantly more effective in reducing both self-reported and clinician-rated depressive symptoms. Furthermore, only a minority of participants who completed CBGT (15.9%) were in the clinical range for depressive symptoms (EPDS) by post-treatment.
Strengths
This study aimed to address several limitations in the existing literature on CBT for perinatal mental health problems using an RCT design, an adequate sample size, a range of outcomes and CBT interventions relevant to perinatal mental health conditions and a protocol that directly targets both anxiety and depressive symptoms. The between-group effect sizes reported in this study ranged from medium (STICSA, HAM-A and MADRS) to large (PSWQ, PSS, EPDS) in magnitude. These effect sizes are well within the range of between-group effect sizes reported in the small number of RCTs that have been conducted on CBT for perinatal mental health problems to date (see, for example, Maguire et al., 2018, for a review) and are also within the range of the effect sizes reported in meta-analyses evaluating CBT protocols for adults with anxiety disorders in the non-perinatal literature (e.g. Covin et al., 2006; Gould et al., 1997; Otto, 2005). Moreover, this study demonstrated the benefits of CBGT even when controlling for maternal status (i.e. pregnancy, postpartum) and pharmacotherapy use.
Limitations
Limitations to this study include a relatively short (3-month) follow-up. Other studies evaluating CBT protocols for perinatal mental health problems have used longer follow-up periods (e.g. 6–9 months; Milgrom et al., 2015; Misri et al., 2004). A longer follow-up (even 1 or 2 years in duration) would help determine whether the benefits of CBGT last beyond the postpartum period. Furthermore, this study did not investigate the potential impact of CBGT on infant developmental outcomes. Future studies should examine outcomes associated with infant development and well-being (e.g. cognitive or emotional development) in both short- and long term. Furthermore, future studies would benefit from a comparison of CBGT with other active interventions.
Conclusion
Despite evidence that CBT is effective for anxiety disorders in non-perinatal populations, little research has examined the benefits of CBT for pregnant and postpartum women with principal anxiety disorder diagnoses. This RCT provides empirical support for a new group-based CBT protocol (CBGT), tailored to meet the unique needs of women with perinatal anxiety. This brief transdiagnostic protocol is effective for women who are pregnant or postpartum, with or without comorbid depression, and regardless of whether or not they are taking pharmacotherapy, and therefore has broad practical and clinical utility.
Footnotes
Acknowledgements
The authors would also like to extend our sincere gratitude to additional staff members who helped with this project, including Peggy Carter-Arrowsmith (RN), Dan-Bi Cho (RN) and Christelle Tshilenge (RSW), and our team of psychology research assistants, residents and practicum students at the Women’s Health Concerns Clinic, St. Joseph’s Healthcare Hamilton.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was funded by a grant from the 2016 Teresa Cascioli Charitable Foundation Research Award in Women’s Health, Research Institute of St. Joseph’s Healthcare, awarded to Dr. Sheryl Green (PI). The funding source had no role in the design, analysis, interpretation or publication of this study.
