Abstract
Objective:
Given the fundamental emotional, social and physical development that occurs during the early years of life, childhood experiences are formative in shaping a person’s life trajectory. Childhood trauma is a prevalent, multifaceted issue with well-documented long-term adverse health effects in clinical populations however; the impact of childhood trauma in the community is less clear. To address this, this study investigated how childhood trauma may impact physical and psychological health, sleep quality and autonomic function in a non-clinical community sample of adults.
Method:
Participants completed questionnaires, an in-laboratory autonomic assessment (including stress reactivity to mental and physical stressors) and overnight autonomic and sleep monitoring. Overall childhood trauma and its subtypes (e.g. physical abuse, emotional neglect) were defined using the Childhood Trauma Questionnaire.
Results:
We identified 22 childhood trauma cases (total score > 36) and, of the 89 non-childhood trauma cases, some individuals also experienced significant levels of trauma in one or more of the childhood trauma subtypes. Childhood trauma and some trauma subtypes were significantly correlated with a myriad of negative physiological and physical health outcomes including elevated psychological distress, increased sleep disturbances, reduced emotional wellbeing and lower perceived social support. Autonomic dysregulation was found in those with high levels of childhood trauma, which was reflected in an increased stress response to laboratory tasks. Notably, the experience of physical abuse in childhood was significantly associated with alterations in nocturnal heart rate and heart rate variability.
Conclusion:
Together, these results highlight that childhood trauma can have lasting detrimental consequences on an individual’s emotional and physical health, sleep quality and stress reactivity.
Introduction
The alarmingly high prevalence of childhood trauma (CT) worldwide represents a largely unaddressed public health issue (Stoltenborgh et al., 2011). Given the fundamental emotional, social and physical development that occurs during the early years of life, childhood experiences are formative in shaping a person’s life trajectory. Trauma during this time can elicit neurobiological changes that may negatively impact an individual’s development and may increase later risk for psychopathologies (Pechtel and Pizzagalli, 2011). CT can be experienced in many forms including being subjected to, or witnessing, physical abuse (PA), sexual abuse (SA) or emotional abuse (EA) and the experience of emotional neglect (EN) or physical neglect (PN).
CT has been linked to a spectrum of adverse health consequences in clinical samples. Relationships have been found between the severity and number of adverse childhood experiences, and mental health issues including somatic disorders, anxiety, obsessive–compulsive disorders, suicide attempts and depression (Briggs and Price, 2009; Carr et al., 2013; Flaherty et al., 2009; Maaranen et al., 2004; Mersky et al., 2013; Schilling et al., 2007). While these clinical issues are indeed sobering, CT in the community is also likely associated with physical and psychological health outcomes that are not clinically recognized. Research in this population may enable early detection of those at higher risk of long-term adverse outcomes and aid in the development of targeted interventions.
It has been speculated that the long-term adverse effects of CT are driven by neurobiological changes in stress response systems, including the hypothalamic–pituitary–adrenal (HPA) axis and the sympathetic and parasympathetic nervous system (Agorastos et al., 2018; Heim et al., 2000). CT has been associated with stronger reactions to daily stressors in adulthood such as higher intensity negative emotional reactions to minor stressors and increased levels of perceived stress (e.g. Glaser et al., 2006; van Nierop et al., 2018). However, the literature to date has mostly assessed stress reactivity via self-report questionnaires with a lesser focus on objective physiological measures of stress reactivity. A careful assessment of the physiological processes that underlie the stress response such as via monitoring cardiac autonomic activity would permit clearer characterization of CT-related changes in stress reactivity and complement self-report outcomes.
Individuals who have experienced CT commonly report disturbed sleep. CT has been shown to increase the likelihood of sleep disturbance in adults characterized as difficulty falling asleep, waking throughout the night and experiencing unrefreshing sleep (Chapman et al., 2011; Kajeepeta et al., 2015). Poor sleep quality in those with CT is hypothesized to mediate some of the negative health outcomes associated with CT, such as stress sensitivity and psychological distress (Kajeepeta et al., 2015). Again, studies utilizing objective measures of sleep quality are lacking. Cardiac autonomic measures, including parameters of heart rate variability (HRV; the beat-to-beat variation in timing of consecutive heart beats), may offer a non-invasive option of such an assessment. Specific parameters of HRV (e.g. high-frequency normalized units [HFnu]) are widely recognized as valid and sensitive indices of parasympathetic activity, which drives vegetative and restorative functions (Camm et al., 1996). Autonomic de-arousal, reflected in a marked decrease in heart rate and a parallel increase in HRV, is essential for the induction and maintenance of deep recuperative sleep. High HRV reflects greater parasympathetic activation and thus contributes to sleep quality. In addition, it is strongly related to behavioural flexibility (including during executive cognitive performance) and adaptive responding to stressors (Burton et al., 2010; Cvejic et al., 2018). Conversely, low HRV (and high nocturnal heart rate) has been linked to superficial, hypervigilant sleep (Patel et al., 2013).
In summary, the generalizability of the CT literature has been limited by a strong focus on clinical and psychiatric samples and a scarcity of studies exploring the impact of CT in non-clinical, community samples. Moreover, methodological issues are prevalent in the literature, especially with respect to the definition of CT and potential screening methods with a lack of standardized and adequately validated tools for retrospective reporting of abuse and neglect. Finally, most research has focused on subjective and psychiatric reports to determine the impact of CT and few studies have concentrated on more objective measures of health.
This study addresses a number of these limitations. Specifically, we aimed to investigate the impact of CT on (1) self-reported measures of psychological and physical health, daily functioning and sleep quality and (2) autonomic function and stress reactivity by assessing cardiac autonomic responses during rest, exposure to mental and physical stressors, and during nocturnal sleep. The Childhood Trauma Questionnaire (CTQ) was utilized to determine overall CT and exposure to trauma subtypes. The CTQ has been extensively validated in both clinical and community populations and enables prompt screening of self-reported childhood maltreatment histories in a respectful and non-intrusive way (Bernstein et al., 2003).
Materials and methods
Participants
Participants were recruited as part of a larger cohort study, that is, the Sydney Infections Outcomes Study (SIOS). The SIOS participants were recruited either via community advertisements seeking healthy adult volunteers, or through medical clinics where they were identified as suffering from a common infective illness (e.g. influenza). The healthy participants were re-assessed in the future when they became unwell, and the unwell participants were re-assessed over the course of their illness until they were medically healthy. All SIOS participants completed a questionnaire battery to evaluate their vulnerability to ill health. All participants included in this study (n = 111) were young, healthy adults that were free of any apparent infective illness at that time and with no known or disclosed medical conditions or medication use that may interfere with autonomic function. Informed written consent was obtained prior to assessment. This study was approved by the UNSW Human Research Ethics Committee (HREC Approval #10380).
Procedures
All participants attended a single laboratory visit between 8:00 and 11:00 a.m. to minimize the impact of diurnal variation on autonomic activity and were instructed to abstain from caffeine and alcohol on the morning of their visit. Participants completed self-report questionnaires, an in-laboratory autonomic assessment and overnight ambulatory autonomic and sleep monitoring at home.
Questionnaires
The Childhood Trauma Questionnaire Short Form (CTQ-SF; Bernstein et al., 2003) screened for a history of early abuse or neglect and derived scores on five subtypes of maltreatment (PA, SA, EA, PN and EN). This questionnaire is a reliable and valid self-report measure of CT in a community sample (Bernstein and Fink, 1998; Glaesmer, 2016; Scher et al., 2001). The 28 items are assessed on a scale from ‘never’ = 1 to ‘most of the time’ = 5. A CT case is defined by a total score greater than 36 and each subtype of trauma was categorized as a case if it was scored in the ‘slight’ to ‘extreme’ range (EA ⩾ 9, PA ⩾ 8, SA ⩾ 6, EN ⩾ 10 and PN ⩾ 8; Bernstein and Fink, 1998; Glaesmer, 2016). In addition, a three-item Minimization and Denial (MD) subscale is incorporated into the CTQ-SF to detect under-reporting of CT (MacDonald et al., 2015).
In addition, a number of self-reported questionnaires were utilized to assess potential domains which could be impacted by CT: current psychological distress (Kessler Psychological Distress Scale [K10]; Kessler et al., 2002), levels of perceived stress (Perceived Stress Questionnaire [PSQ]; Levenstein et al., 1993), emotional and physical wellness and role limitations (Medical Outcomes Short Form 36 [SF-36]; Ware and Sherbourne, 1992), physical ailments (Physical Symptom Check [PSC]; Vollmer-Conna et al., 2008), perceived level of social support (Duke-UNC Functional Social Support Questionnaire [DFSS]; Broadhead et al., 1988) and sleep quality (Pittsburgh Sleep Quality Inventory [PSQI]; Buysse et al., 1989). The Somatic and Psychological Health Report (SPHERE; Couvy-Duchesne et al., 2017) was also utilized to screen for psychological symptoms (PSYCH subscale) and non-specific somatic symptoms (SOMA subscale). Fulfilling case criteria for both PSYCH and SOMA is highly associated with mental health disorders or chronic fatigue syndrome diagnosis (Couvy-Duchesne et al., 2017; Hickie et al., 2001).
Laboratory autonomic assessment
Laboratory-based measures of heart rate (via three-lead electrocardiogram [ECG]) and respiration (strain gauge transducer; Pneumotrace; UFI, Morro Bay, CA, USA) were continuously recorded (PowerLab 16/30SP; ADInstruments, Bella Vista, Australia) while participants listened to a gentle nature soundscape for 10 minutes to establish their baseline autonomic activity, and during the completion of a mental and a physical stressor. The presentation of the mental and physical stressors was counterbalanced across participants.
A computerized version of the Stroop colour–word interference task (Lemon, 1990) was used to assess autonomic reactivity to a mental stressor (Kahneman, 1973). Three colour words (‘RED’, ‘GREEN’, ‘BLUE’) were presented in three different coloured ink (red, green or blue). The colour and the meaning of the word could be concordant (the word ‘RED’ written in red) or discordant (the word ‘RED’ written in blue). During discordant trials, participants have to actively inhibit the prepotent reading response to provide the correct colour name.
The Cold Pressor test (Mourot et al., 2009) was used as a physical stressor and involved complete submersion of the subject’s bare foot into an ice-bath (~3°C) until they could no longer tolerate the discomfort, or for a maximum of 2 minutes.
At the conclusion of each stressor, participants were instructed to sit back and relax for 2 minutes. The time taken for participants’ heart rates to recover to baseline levels was recorded. It is well documented that, following a stressor, the vagus nerve plays a major role in restoring heart rate to baseline values. Therefore, heart rate and heart rate recovery after stressors such as the Stroop and Cold Pressor test are considered valid measures of cardio-autonomic reactivity to, and recovery from, stress (Beaumont et al., 2012). HRV measures were derived from the R–R intervals of ECG recordings in 5-minute epochs using LabChart 8, which utilizes the Lomb–Scargle periodogram (Moody, 1993) to determine the HFnu spectral component (0.15–0.40 Hz), a well-validated marker of parasympathetic, vagal activity (Camm et al., 1996).
Overnight autonomic assessment
Continuous monitoring of autonomic activity and activity–rest behaviour was achieved via a lightweight ambulatory system (Equivital; Hidalgo Ltd, Cambridge, UK) which was worn from the end of the laboratory session until the following morning. This device, worn around the chest and fixed securely in place with a harness strap, consists of a two lead ECGs (sampled at 256 Hz), a respiratory belt and a tri-axis accelerometer (enabling the detection of movement and body position). Upon waking, participants completed a brief questionnaire assessing sleep and wake time and sleep quality. Processing of these recordings was analogous to the procedures used for laboratory-based autonomic recordings.
Data analysis
Statistical analyses were performed in SPSS version 24 (IBM, Chicago, IL, USA). Normality assumptions for all key variables were checked using Q–Q plots; variables that were not normally distributed were log-transformed to permit parametric analyses; or non-parametric analyses (chi-square test and Spearman’s rank-order correlation) were used to examine bivariate associations between key parameters. Simple group comparisons were made using t tests; and repeated-measures analysis of variance (ANOVA), or analysis of covariance (ANCOVA) as appropriate, was used to assess between-group differences in responses over time (across task duration or hours of sleep). Kaplan–Meier time-to-event analysis was used to assess differences in heart rate recovery time after exposure to the stressors between those with and without CT.
Results
Physical and psychological health
A total of 22 participants met overall CT case criteria (Table 1). Of those individuals, 11 experienced significant trauma in two or three subcategories and 11 experienced significant trauma in four or five subcategories. Additional non-CT individuals also experienced significant levels of trauma during childhood in one (n = 22) or two (n = 3) of the trauma subcategories. Overall, EA and EN were reported most often, with 20.7% and 26.1% of the sample experiencing these, respectively. Notably, all CT cases also fulfilled criteria for minimization and denial and there was a highly significant relationship between CT caseness and minimizing and denying trauma history (p = 0.002).
Childhood Trauma Questionnaire: total score and trauma subscale scores for the CT and non-CT cases.
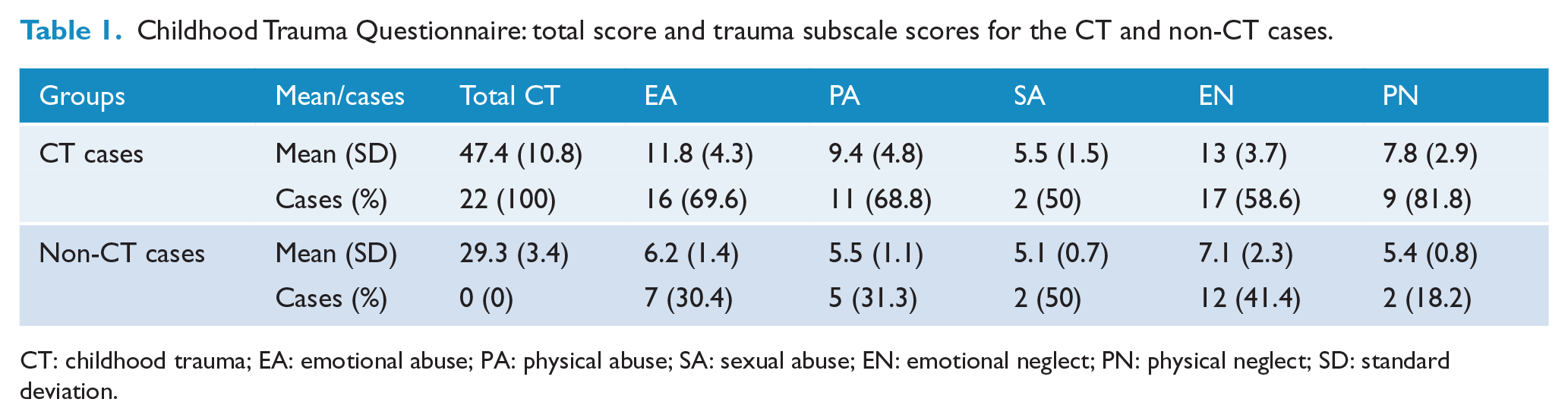
CT: childhood trauma; EA: emotional abuse; PA: physical abuse; SA: sexual abuse; EN: emotional neglect; PN: physical neglect; SD: standard deviation.
Table 2 summarizes basic demographic information and self-report questionnaire outcomes grouped by CT (n = 22) and non-CT (n = 89) cases. Age and sex were comparable between groups. The CT group reported significantly higher current psychological distress (t = –4.49, p < 0.001), perceived stress (t = –2.87, p = 0.005), physical (t = 3.639, p < 0.001), somatic (t = –4.84, p < 0.001) and psychological (t = –3.41, p = 0.001) symptoms and role limitation due to emotional problems (t = 2.59, p = 0.01). Moreover, the overall CT group reported poorer general health (t = 3.84, p < 0.001) and emotional wellbeing (t = 3.205, p = 0.002), poorer sleep quality (t = –4.53, p < 0.001) and lower energy (t = 3.705, p < 0.001) and perceived social support (t = 4.52, p < 0.001). There were 19 participants who qualified as cases for both PSYCH and SOMA, 41% of CT cases compared to 11% of non-CT cases. The experience of CT was significantly linked to comorbid PSYCH and SOMA caseness (χ2 = 10.95, p = 0.001). In fact, those who experienced CT were 5.5 times (confidence interval [CI]: [1.9, 16.0]) more likely to also fulfil case criteria for psychological and non-specific somatic symptoms which are closely related with a mental health diagnosis, or chronic fatigue syndrome diagnosis (Couvy-Duchesne et al., 2017; Hickie et al., 2001).
Participant demographic, clinical and lifestyle characteristics, grouped by CT caseness.
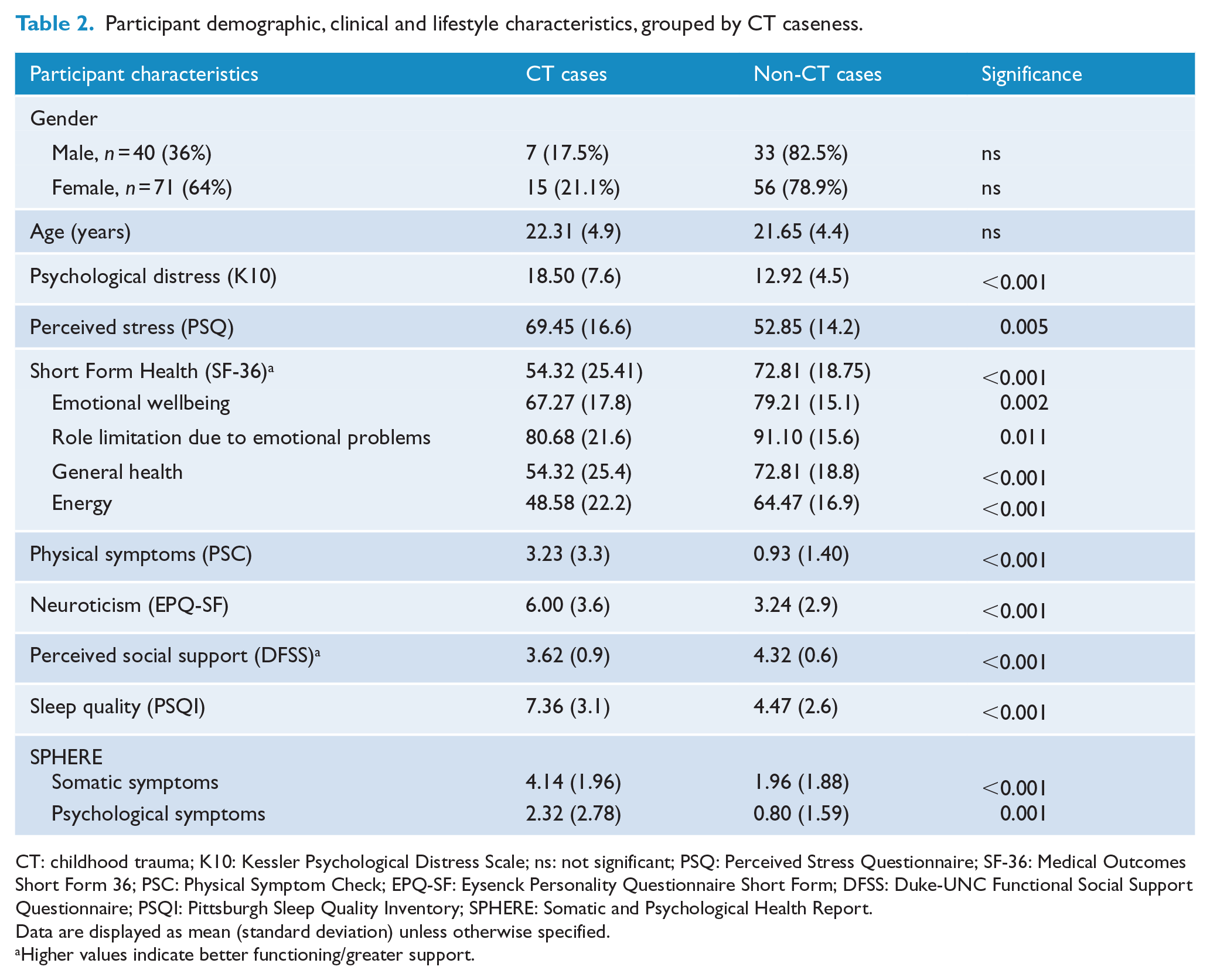
CT: childhood trauma; K10: Kessler Psychological Distress Scale; ns: not significant; PSQ: Perceived Stress Questionnaire; SF-36: Medical Outcomes Short Form 36; PSC: Physical Symptom Check; EPQ-SF: Eysenck Personality Questionnaire Short Form; DFSS: Duke-UNC Functional Social Support Questionnaire; PSQI: Pittsburgh Sleep Quality Inventory; SPHERE: Somatic and Psychological Health Report.
Data are displayed as mean (standard deviation) unless otherwise specified.
Higher values indicate better functioning/greater support.
Psychological and physical correlates of the CT subtypes
Table 3 highlights that the experience of EA, EN and PN subtypes of CT significantly correlated with key measures of psychosocial health including elevated psychological distress (all p’s ⩽ 0.001) and perceived stress (all p’s ⩽ 0.001), reduced emotional wellbeing (all p’s < 0.01) and reduced social support (all p’s ⩽ 0.001). However, correlations between SA, PA and psychosocial outcomes were not substantive. With respect to physical health outcomes, EA, EN and PN were negatively associated with energy (all p’s ⩽ 0.001) and general health (all p’s ⩽ 0.001) and were positively correlated with poorer sleep quality and worse physical health (all p’s ⩽ 0.05). SA and PA, although showing some significant associations (e.g. with scores on the physical symptom checklist), overall had fewer significant physical health correlations.
Total childhood trauma and subtypes’ association with psychological and physical health outcomes.
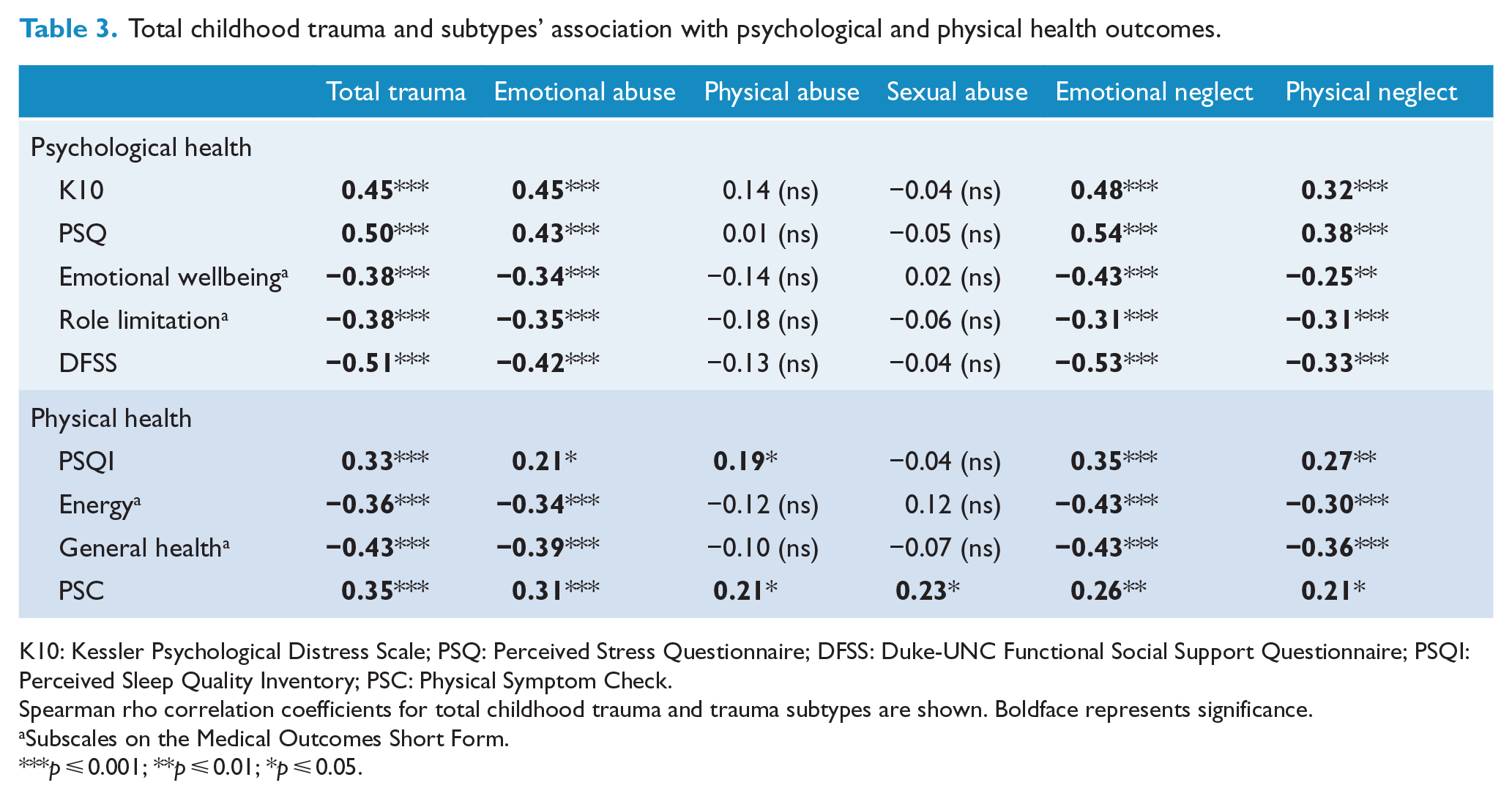
K10: Kessler Psychological Distress Scale; PSQ: Perceived Stress Questionnaire; DFSS: Duke-UNC Functional Social Support Questionnaire; PSQI: Perceived Sleep Quality Inventory; PSC: Physical Symptom Check.
Spearman rho correlation coefficients for total childhood trauma and trauma subtypes are shown. Boldface represents significance.
Subscales on the Medical Outcomes Short Form.
p ⩽ 0.001; **p ⩽ 0.01; *p ⩽ 0.05.
Autonomic function, stress reactivity and CT
Laboratory assessment
Laboratory-based autonomic assessment revealed that those who fulfilled case criteria for CT had higher resting HR than those who did not (Figure 1(A); t = 2.54, p = 0.012), but HRV (as HFnu) was comparable between groups (t = 0.55, p = 0.585). In relation to stress reactivity during the Stroop task, trend analysis using repeated-measures ANCOVA (with task performance included as a covariate) showed significance difference between groups in the trajectory of the HR response across the 5 minutes of the Stroop task (cubic trend, F = 6.3, p = 0.014). This is illustrated in Figure 1(B) where the cardiac stress response (as HR) in CT cases is consistently higher and additionally increases as the task continues, whereas the HR of non-CT cases levels out after the initial increase, likely indicating habituation to the stressor after an initial period of stress. In addition, compared to those who did not fulfil case criteria for CT, CT cases took longer to return to their resting HR after both stressors. This was highly significant for the physically stressful Cold Pressor task (Figure 2(B); χ2 = 6.75, p = 0.009); but was not significant for the Stroop task (Figure 2(A); p = 0.33).
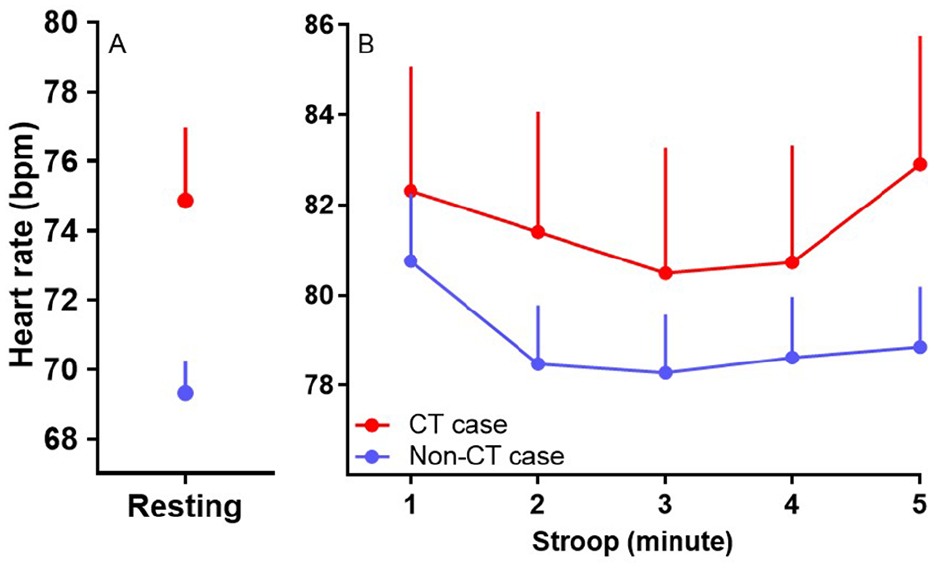
Heart rate at rest (A) and during the Stroop task (B) for participants with a history of childhood trauma (CT, red circle) and those with no history of CT (blue circle). Data are expressed as mean ± SEM.
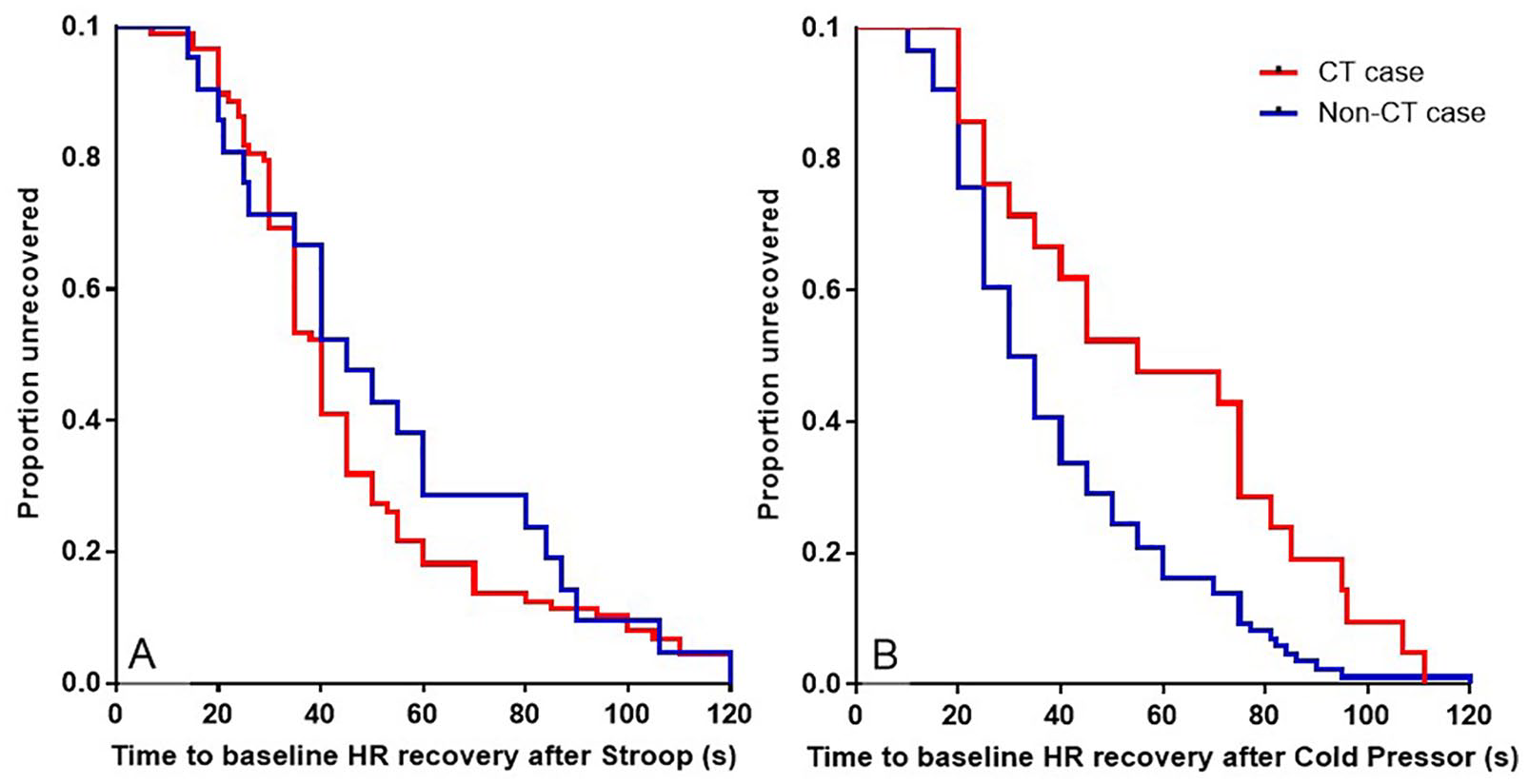
The proportion of participants with a history of childhood trauma (CT, red line) and those with no history of CT (blue line) who returned to resting heart rate (HR) during the recovery period after being exposed to a cognitive (A; the Stroop task) and a physical (B; Cold Pressor test) stressor.
Sleep quality and overnight autonomic assessment
CT cases reported significantly poorer sleep quality than those who had not experienced CT (t = –4.53, p < 0.001). Specifically, 72.7% of CT cases scored above 5 on the PSQI, indicative of sleep difficulties, compared to only 24.7% of participants who had not experienced CT (χ2 = 15.01, p < 0.001). In fact, those who experienced CT were 6.8 times (CI: [2.4, 19.4]) more likely to have sleep difficulties. With respect to trauma subtypes, EA (r = 0.21, p = 0.03), EN (r = 0.35, p = 0.001), PN (r = 0.27, p = 0.005) and PA (r = 0.19, p = 0.05) were all significantly correlated with poorer sleep quality. Initial inspection of differences in nocturnal HR and HRV in CT cases compared to non-CT cases revealed higher HR and lower HRV throughout the night; however, this was not statistically significant. Exploration of such differences in the trauma subcategories revealed significant differences in nocturnal cardiac autonomic responses for those who had experienced PA. Specifically, repeated-measures ANOVA produced a significant between-group difference between the PA cases and those that had not experienced any abuse in the HR response throughout the night (Figure 3(A); F = 4.15, p = 0.046). In addition, trend analysis showed a significant between-group difference in the trajectory (cubic trend) of the HRV response (Figure 3(B); F = 4.45, p = 0.039), such that PA cases, although initially increasing HRV as expected during the transition from wakefulness to sleep, then showed reduced HRV (i.e. parasympathetic drive) across the early hours of sleep struggling to maintain a level of HRV that is conducive to deep sleep. When the total CT score was considered, this cubic trend was also evident, albeit it was not statistically significant (F = 3.43, p = 0.068).
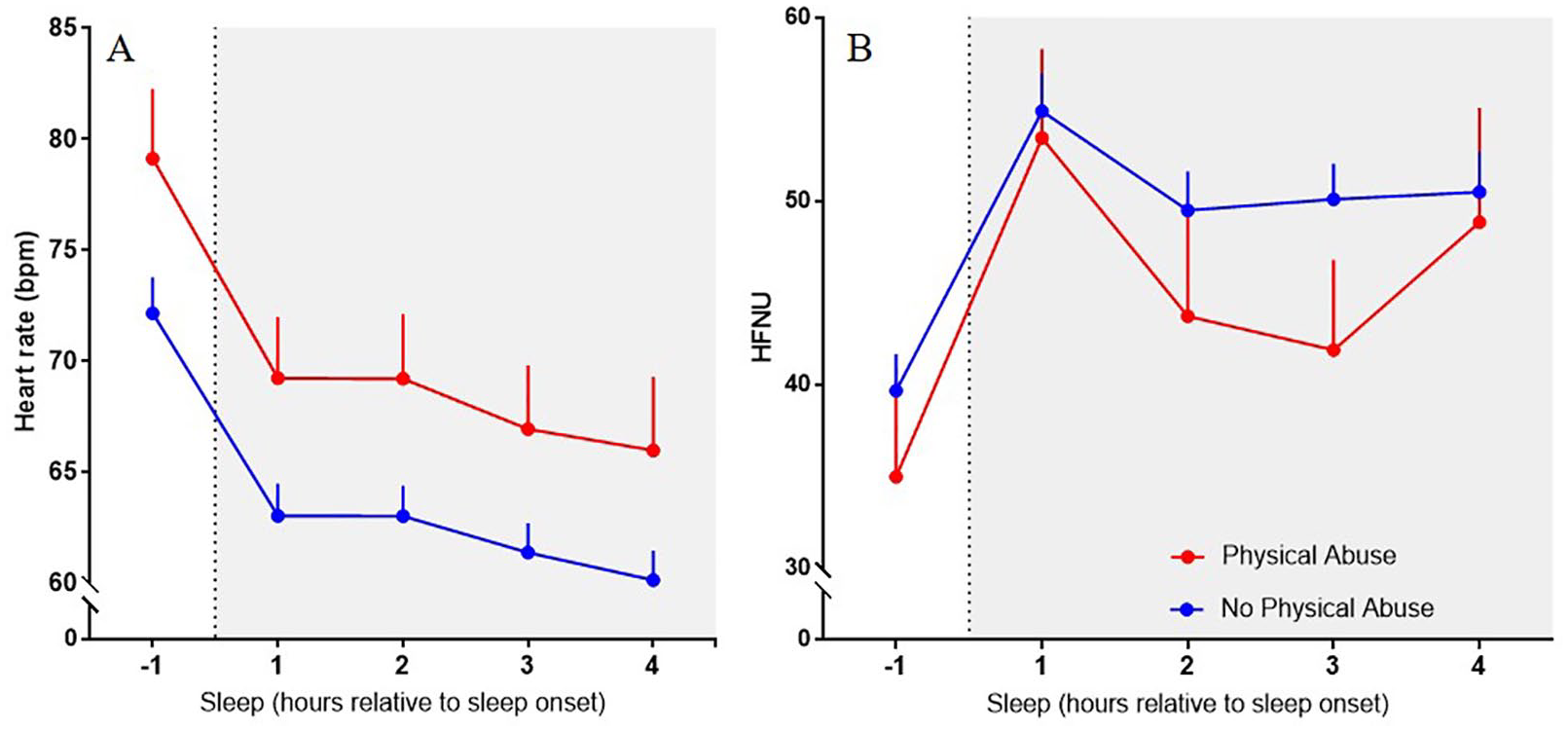
Heart rate (HR; A) and heart rate variability (HRV; B) during the night in physical abuse (PA) cases (red circle) and non-PA cases (blue circle). Data are expressed as mean ± SEM.
Discussion
This study of healthy young adults affirmed that the experience of trauma during childhood is associated with a spectrum of adverse health outcomes later in life including significant reductions in perceived physical and psychological wellness, increased distress, poor sleep and heightened stress reactivity. In addition, substantive autonomic perturbations were evident in participants who reported significant CT. These novel findings extend the current literature on CT by including objective physiological assessments and by highlighting the adverse impact of CT in a non-clinical, community sample.
Poor psychosocial outcomes in adulthood were strongly associated with the total CT and CT subtype (EA, EN and PN) scores, including a heightened perception of stress, reduced emotional wellbeing and decreased social support. Notably, more than 40% of the self-reported CT cases (with no formal psychiatric diagnoses) fulfilled case criteria for mental health problems on the SPHERE questionnaire. Those with CT were in fact 5.5 times more likely to fulfil case criteria for both PSYCH and SOMA caseness, which has been strongly linked to mental health disorders such as anxiety and depression, and with chronic fatigue states (Bennett et al., 2014; Hickie et al., 2001). Taken together, these results suggest that those who have experienced CT and have psychological disorders may not be seeking medical treatment. Screening for CT therefore may assist in identifying at-risk individuals and create an opportunity to advocate for, and provide, appropriate referrals and/or interventions.
In the current sample, PA and SA were not significantly associated with psychological health outcomes. Previous research has shown that childhood physical and sexual abuse is often linked to a broad spectrum of emotional and mental health disorders, including posttraumatic stress disorder (PTSD), personality disorders, anxiety and depression (Briggs and Price, 2009; Carr et al., 2013; Flaherty et al., 2009; Maaranen et al., 2004; Mersky et al., 2013; Schilling et al., 2007). There are several potential explanations as to why this was not observed in our community sample including an unpowered sample size (n = 16), under-reporting or denial of CT, and/or that individuals in the community may find it easier to disclose physical symptoms than mental health issues due to fears of stigmatization.
The experience of CT can be difficult to process, and initial reactions can involve many different emotions, ranging from anger and sadness to confusion and hopelessness. As individuals learn to develop coping mechanisms in response to their trauma, some may choose to deny, rather than report, or perhaps minimize their trauma experience. This was clearly evident in this study with all those fulfilling case criteria for CT also qualifying as minimization and denial cases. Minimization and denial was additionally apparent in a large number of participants who did not qualify as CT cases (n = 62) suggesting that the prevalence of CT may be higher than reported, or that people often idealize their childhood on reflection. These results are consistent with a previous study which found elevated minimization and denial scores in a community sample compared to a patient group (MacDonald et al., 2016). Possible reasons for these differences include that those from the community are less likely to discuss their trauma compared to clinical samples and that clinical samples may also undergo additional, more rigorous clinical interviews and assessment.
The experience of CT, and its subtypes, significantly affected the likelihood of reporting physical health symptoms including sleep disturbances, fatigue and poorer general health. These associations are consistent with the literature on CT in clinical samples which have well-documented links between CT (including emotional, physical and sexual) and somatic symptoms in adulthood (Edwards et al., 2003; Springer et al., 2007). This connection between traumatic experiences in early life and poor physical health in adulthood is thought to be related to a spectrum of potential predisposing vulnerabilities. These may include an enhanced risk of developing obesity and metabolic syndrome (Gunstad et al., 2006; Lee et al., 2014), as well as maladaptive coping behaviours such as disordered eating and alcohol and substance abuse (Lotzin et al., 2016; Moulton et al., 2015). The well-documented health consequences of early-life trauma may vary by the nature of the trauma (with especially strong relationships identified for EA), the victim’s sex and the coping mechanisms that he or she employs (Lee et al., 2014; Moulton et al., 2015).
In addition, early trauma can lead to significant alterations in the stress response, including increased sympathetic signalling leading to elevated HR, autonomic hypervigilance and poor physiological adaptations to stressors more generally (van der Kolk, 1989). This provides an opportunity to objectively assess stress reactivity. In our community sample, while resting HRV was comparable between those who experienced significant CT and those who did not, resting HR was significantly elevated in the cases of CT suggestive of heightened autonomic activation; this difference was not explained by differences in body mass index. Examining individuals’ responses to cognitive and physical stressors in the laboratory provided further evidence of autonomic disturbance in the CT sufferers. The Stroop task requires executive functioning, specifically the inhibition of a pre-programmed reading response, and is widely utilized as a reliable mental stressor (Chan et al., 2008). In those with significant CT, this task elicited a sustained elevation in HR revealing substantively greater stress reactivity than that observed in those without CT experience. CT cases also took longer to recover from this stressor, although this was not statistically significant. In contrast, following experience of a physical challenge – the Cold Pressor test – sufferers of CT showed a very significant delay in the time taken to return to their resting HR, again indicative of an imbalance in autonomic function towards sympathetic dominance (Beaumont et al., 2012).
Autonomic function was further examined via monitoring HR and HRV during nocturnal sleep. Sleep plays an important biological role in recuperation, allowing for optimal daytime functioning. Appropriate autonomic de-arousal prior to and during sleep is essential for good-quality sleep (Burton et al., 2010; Cvejic et al., 2018). Differences in nocturnal HR and HRV between those with the experience of CT and those without failed to reach significance; however, when examining trauma subtypes, a history of PA was significantly linked to higher HR and reduced HRV compared to those with no trauma history. These results suggest that the experience of PA may lead to hypervigilant sleep, with higher levels of sympathetic signalling persisting even during the night (Gillie and Thayer, 2014). Consistent with this is our finding of self-disclosed poorer sleep quality in sufferers of CT, including PA. Although there has been limited investigation on the effects of CT (and specifically PA) on nocturnal HRV, there are well-documented reductions in HRV and nocturnal HRV in clinical groups across a spectrum of disorders including depression, anxiety and PTSD (e.g. Green et al., 2016; Paniccia et al., 2017).
To gain further insight into the impact of CT, future studies would benefit from employing a longitudinal, prospective design. Longitudinal cohorts would permit better examination of the full extent and the time course of adverse effects resulting from the experience of CT. For instance, consistently elevated HR may increase an individual’s risk of developing cardiovascular diseases such as high blood pressure and heart disease later in life (Thayer et al., 2010). In addition, a larger study sample size would improve statistical power to conduct further subgroup analyses for the different types of CT, especially for PA and SA.
Overall, our results highlight that individuals functioning in the community who have experienced CT are vulnerable to negative health outcomes that are similar to those reported in clinical samples. The evidence presented in this study corroborates with the current literature that the experience of CT negatively impacts psychological and physical health, and especially levels of perceived stress, emotional wellbeing, sleep quality and physical symptoms. Furthermore, our novel findings document that CT can have an enduring impact on autonomic function that is evident in healthy adults when dealing with everyday stressors and even during their sleep. It is sobering that the experience of trauma in childhood has such a far-reaching impact on an individual’s ability to cope with everyday challenges and his or her emotional and physical wellbeing into adulthood.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Mason Foundation National Medical Program (Grant No. #13072; 2012-2018).
