Abstract
Objective:
Impairment in psychosocial function is common in schizophrenia. Long-acting injectable atypical antipsychotics are thought to enhance psychosocial function by boosting adherence. However, no systematic review has examined the effects of long-acting injectable atypical antipsychotics on psychosocial function in clinical trials.
Methods:
We searched major databases including Medline/PubMed, PsychINFO, EMBASE, CINAHL, Scopus, Web of Science, Cochrane Central Register of Controlled Trials and Clinical Trial Registries for randomised controlled trials that compared long-acting injectable atypical antipsychotics to placebo, oral antipsychotic medications or long-acting injectable atypical antipsychotics for all years till 2018, with no language limits. We performed a systematic review of findings on change in psychosocial function and its predictors in the included reports. Data on change in psychosocial functioning were meta-analysed using a random-effects model.
Results:
A total of 26 studies were included in systematic review, and 19 studies with 8616 adults, 68.1% males were meta-analysed. Long-acting injectable atypical antipsychotics were superior to placebo (standardised mean difference = 0.39; 95% confidence interval = [0.32, 0.47];
Conclusion:
Long-acting injectable atypical antipsychotics are beneficial for recovery of psychosocial function in comparison with placebo, but the magnitude of superiority over oral antipsychotic treatment was small. Severe psychopathology at baseline predicted poor psychosocial function. Future effectiveness trials in which post-randomisation involvement is kept to a minimum, and psychosocial function is included as primary outcome a priori, are needed to capture the real-world impact of long-acting injectable atypical antipsychotics and to address methodological biases.
Keywords
Introduction
Functional impairment is a major problem in schizophrenia. Globally, the disorder was estimated to account for 222.3 million disability-adjusted life years (DALYs) in 2016 (GBD 2016 DALYs and HALE Collaborators, 2016; GBD 2016 Disease and Injury Incidence and Prevalence Collaborators, 2016). Functional deficit is a core feature in the diagnosis and course of schizophrenia which can persist beyond hallmark clinical symptoms and negatively impact prognosis (Mueser and McGurk, 2004; Van Os and Kapur, 2009). While there is strong evidence for the efficacy of antipsychotic medications in treating psychotic symptoms, functional improvement is more variable and does not necessarily require complete symptom remission (Jaaskelainen et al., 2013).
Medication adherence is key to symptomatic and functional recovery (Czobor et al., 2015), and relapse rates are high following cessation of oral antipsychotics or partial adherence (Alvarez-Jimenez et al., 2012; Novick et al., 2010). Non-adherence is, however, common in schizophrenia, with discontinuation rates ranging from 44% within 1 year after first episode (Kaplan et al., 2013) and as high as 74% over longer periods (Lieberman et al., 2005; Valenstein et al., 2006).
Long-acting injectable (LAI) antipsychotic formulations are effective in reducing the rate of antipsychotic discontinuation (Tiihonen et al., 2011) and significantly lower the risk of relapse and rehospitalisation (Lafeuille et al., 2013; Leucht et al., 2011). However, extrapyramidal side-effects and poor tolerability of conventional LAIs have limited their use and led to the development of second-generation LAI to improve adherence (Tiihonen et al., 2017). Since the introduction of LAI-risperidone in 2003, several other long-acting injectable atypical antipsychotic (LAI-A) formulations (including olanzapine, paliperidone and aripiprazole) have been developed with differences in dosing flexibility, costs, pharmacodynamics–kinetics and side-effect profile (Miyamoto and Fleischhacker, 2017). Several theories have been proposed to explain the potential advantages of broader and early use of LAIs for schizophrenia. First, LAI use facilitates close monitoring of adherence which is particularly important in the early phases of psychotic illness (Tiihonen et al., 2011). Second, patients may prefer the longer dosing interval, and third, single dosing prevents the risk of self-medication and harmful use that can occur with oral preparations (Maia-de-Oliveira et al., 2013; Patel and Davis, 2005). Finally, due to higher bioavailability and lower fluctuations in serum level (Ereshefsky and Mascarenas, 2003
Although psychosocial function as a term is widely used in literature, there has been a lack of uniformity in its construct. The patient’s well-being/quality of life (QoL) is not measured in some studies. More recently, a multidimensional construct that includes both measures of daily function and subjective well-being/QoL is gaining support as a more reliable, valid, comprehensive and holistic indicator (Dixon et al., 2010; Harvey, 2013; Leifker et al., 2010; Menezes et al., 2006; Olagunju et al., 2018; Ro and Clark, 2009). Thus, in this study, we used a multidimensional construct of psychosocial function that includes multiple domains of basic-daily function, socio-occupational function, interpersonal relationships, adaptive function, life satisfaction and subjective well-being/QoL.
The biological basis of psychosocial function is yet to be well understood; however, antipsychotics are thought to be neuroprotective against the cytotoxic effects of active psychosis (Kim and Na, 2017; Wyatt, 1991). Psychosocial function is known to worsen with frequent relapses or longer duration of untreated psychosis. In addition, performance-based skills decline with the disruption of social relationships, unemployment and institutionalisation following relapses or chronic illness (Emsley et al., 2013). LAI-A may therefore confer functional recovery through relapse prevention via better adherence (Kaplan et al., 2013). Furthermore, LAI-A have been linked to good subjective well-being and life satisfaction, which can in turn enhance social adaptation, autonomy and psychosocial function (Kane et al., 2015; Nasrallah et al., 2004). Atypical LAIs are now recommended in practice guidelines as first-line choice for maintenance treatment in non-adherent patients (Bai et al., 2006; Galletly et al., 2016; Lehman et al., 2004).
While the efficacy of LAI-A for symptom remission and relapse prevention is well established, the functional benefit of these treatments has not been interpreted across clinical trials. The majority of comparative effectiveness trials of LAI-A versus oral medications are modelled on proxy indices of functional outcome including relapse rates and rehospitalisation. These proxy endpoints are thought to be reliable to ascertain real-world functioning (Olivares et al., 2013) and have good face validity, however, hospitalisation as an outcome translates poorly between differing healthcare contexts, does not capture patient’s personalised experience and can be a negative therapeutic goal to end-users (Burns, 2007).
The results of randomised controlled trials (RCTs) of LAIs versus orals for relapse and rehospitalisation are mixed. Compared to RCTs, naturalistic studies are consistent in reporting LAI superiority, although they are not flaw-proof (Zhornitsky and Stip, 2012). The impact of these study biases on functional outcome measures need to be considered individually for all studies (RCT and observational) to appropriately interpret the benefits of LAI over oral treatment. In addition, experts have recommended effectiveness trials, in which post-randomisation involvement (manipulation of trial-related factors including adherence such that the performance of intervention reflects ideal or well-controlled circumstances) would be kept to a minimum to better reflect routine practice (Kane et al., 2013; Kishimoto et al., 2014). Such novel trials can be informed by an analysis of study quality in existing trials using psychosocial function measures.
To summarise and interpret the impact of LAI-A on function, we conducted a systematic review and meta-analysis using a multidimensional construct for function (‘psychosocial function’) that includes multiple domains of basic-daily function, socio-occupational function, interpersonal relationships, adaptive function, life satisfaction and subjective well-being/QoL (Menezes et al., 2006; Ro and Clark, 2009). Critical appraisal of all included reports was performed to assess the quality of the implementation of standardised measures of psychosocial function. We also systematically reviewed factors predictive of improvement in function with LAI-A treatment to identify characteristics that could be used to guide the decision to commence LAIs. Our specific aims were to
Investigate the longitudinal effects of LAI-A on measures of psychosocial function among people with schizophrenia or schizoaffective disorders;
Compare the effects of LAI-A on measures of psychosocial function with placebo or oral antipsychotic medications;
Describe the predictors of change in psychosocial function included in primary studies;
Appraise the quality of measures of psychosocial function.
Materials and methods
Inclusion criteria
This systematic review was conducted in accordance with Cochrane collaboration guideline and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline (Liberati et al., 2009; Moher et al., 2009) (checklist included as Supplementary S1). All literature till March 2018, with no language limits, was searched for RCTs that compared LAI-A with placebo or active controls in people with schizophrenia or schizoaffective disorders. Eligible studies included adults aged 18 years or older of either gender that provided information on measures of psychosocial function. We included reports on placebo-controlled trials and open-label active-controlled RCTs to improve study power and to allow a comprehensive appraisal of the quality of psychosocial function measurement. Studies with flexible or fixed doses of LAI-A within the recommended therapeutic range for clinical efficacy were shortlisted (Brissos et al., 2014). Non-randomised controlled studies and single-arm prospective experimental trials were excluded (Higgins and Green, 2011). Data concerning the predictors of functional outcomes were not consistent across studies and is described as a systematic review alone.
Outcome parameters
The main outcome was longitudinal change in psychosocial function associated with LAI-A treatment as defined in primary studies in comparison with placebo or oral antipsychotic medications. We indexed psychosocial function with study-defined multidimensional measures of domains of basic-daily function, socio-occupational function, interpersonal relationship, adaptive function, life satisfaction and subjective well-being/QoL. The composite scores of the psychosocial function scales in the included studies were used. LAI-A-controlled trials, designed to compare the efficacy of different LAI-A or of different doses of the same LAI-A, were included only in systematic review. Furthermore, we collected data on factors that predicted change in psychosocial function from the included studies.
Database search
We searched Medline/PubMed, PsychINFO, EMBASE, CINAHL, Scopus, Web of Science, Cochrane Central Register of Controlled Trials and Clinical Trial Registries (http://clinicaltrials.gov/) for all studies (published and unpublished) to avoid publication bias. In case of PubMed, the terms used as string in combination to search in titles, abstract and as MeSH terms included (1) ‘Antipsychotic Agents’ ‘Pharmacological Action’, ‘antipsychotic’ (2) ‘schizophren’ OR ‘schizo’ (3) ‘inject’ OR ‘depot’ OR ‘long acting’ OR ‘delayed-action preparations’ (4) ‘Quality of life’ OR ‘Function’. An additional search was done with the names and standard abbreviations of scales used to measure psychosocial function and QoL. The scales included the following: ‘Global Assessment of Function’, ‘Heinrichs–Carpenter QoL scale’, ‘Short Form-36 Health Survey’, ‘Personal and Social Performance scale’, ‘Strauss–Carpenter Levels of Functioning’ and ‘Social and Occupational Functioning Assessment’. The clinical trial registration number was used to identify all published reports on each of the clinical trials for comprehensive extraction of data. References of included studies and relevant reviews were snowball searched for additional studies. Authors of studies with missing data were contacted for further information.
A.T.O. screened titles and abstracts of 1104 articles to produce a shortlist of 98 potential articles. The full texts of the selected articles were reviewed by A.T.O. for eligibility as per the a priori criteria.
Data extraction and management
Data extraction was done by A.T.O. independently using piloted forms based on the PRISMA guidelines and included the PICOS (population, intervention, comparison, outcomes and setting) data items. Data extraction forms collected the following data: first author, funding source, study design, study country or sites, participants characteristics (age range, total number, gender, diagnosis and treatment setting), trial duration, antipsychotics (LAI-A vs controls), dosage, trial arms, measures of psychosocial function, clinical scales, findings on psychosocial function (baseline, endpoint and mean change) and identified predictors with statistical significance. During data collection, discrepancies were resolved through consultation with the senior author, B.T.B., where necessary.
Assessment of quality of studies with risk-of-bias tool
We assessed the risk of bias within individual studies using the Cochrane Collaboration methods for assessing bias in controlled clinical trials (Higgins and Green, 2011). Bias assessments were conducted at both the study and the outcome levels and included items such as random sequence generation, allocation concealment, blinding of personnel, blinding of outcome assessment, incomplete data reporting, selective reporting and other sources of bias. The presence of possible publication bias was visually assessed from funnel graphs and included Egger’s test (Higgins and Green, 2011; Leucht et al., 2012).
Synthesis of results and meta-analytic calculation
We performed both qualitative and quantitative analyses of psychosocial function but could only complete qualitative analyses for predictor variables. We compared LAI-A with placebo or oral antipsychotic medications on change in psychosocial function using standardised mean difference (SMD) as the primary effect size. Analysis was conducted considering all time points, with data for the last outcome time point in each study used to calculate change from baseline. All calculations were performed using Rev. Manager 5 software (Cochrane Collaboration, 2014) and meta-regression was done with STATA (StataCorp, 2017). A
Sub-group analyses and meta-regression
We conducted sub-group analyses for duration of trial; however, analyses were limited due to one short-term oral-controlled trial. To check the effects of trial duration, we categorised studies into short term for trials not beyond 3 months and those greater than 3 months were grouped as long term (Wahlbeck et al., 1999). We also performed sub-group analysis to assess any difference in functional outcomes for trials that used treatment as usual (TAU) with oral antipsychotics in comparison with LAI-A. We conducted meta-regression to check effects of study settings (outpatient vs inpatient), trial design, year of publication, industry sponsorship, baseline symptom severity and inclusion of psychosocial function as primary outcome in included trials (Borenstein et al., 2009).
Results
Study selection
A total of 1631 publications were retrieved from all sources and 527 duplicates were removed. Abstracts and titles of the remaining 1104 reports were screened, and 98 full texts were shortlisted for review. Of these 98 full texts, 25 (containing 26 studies) were included, while 73 were excluded due to non-RCT studies (
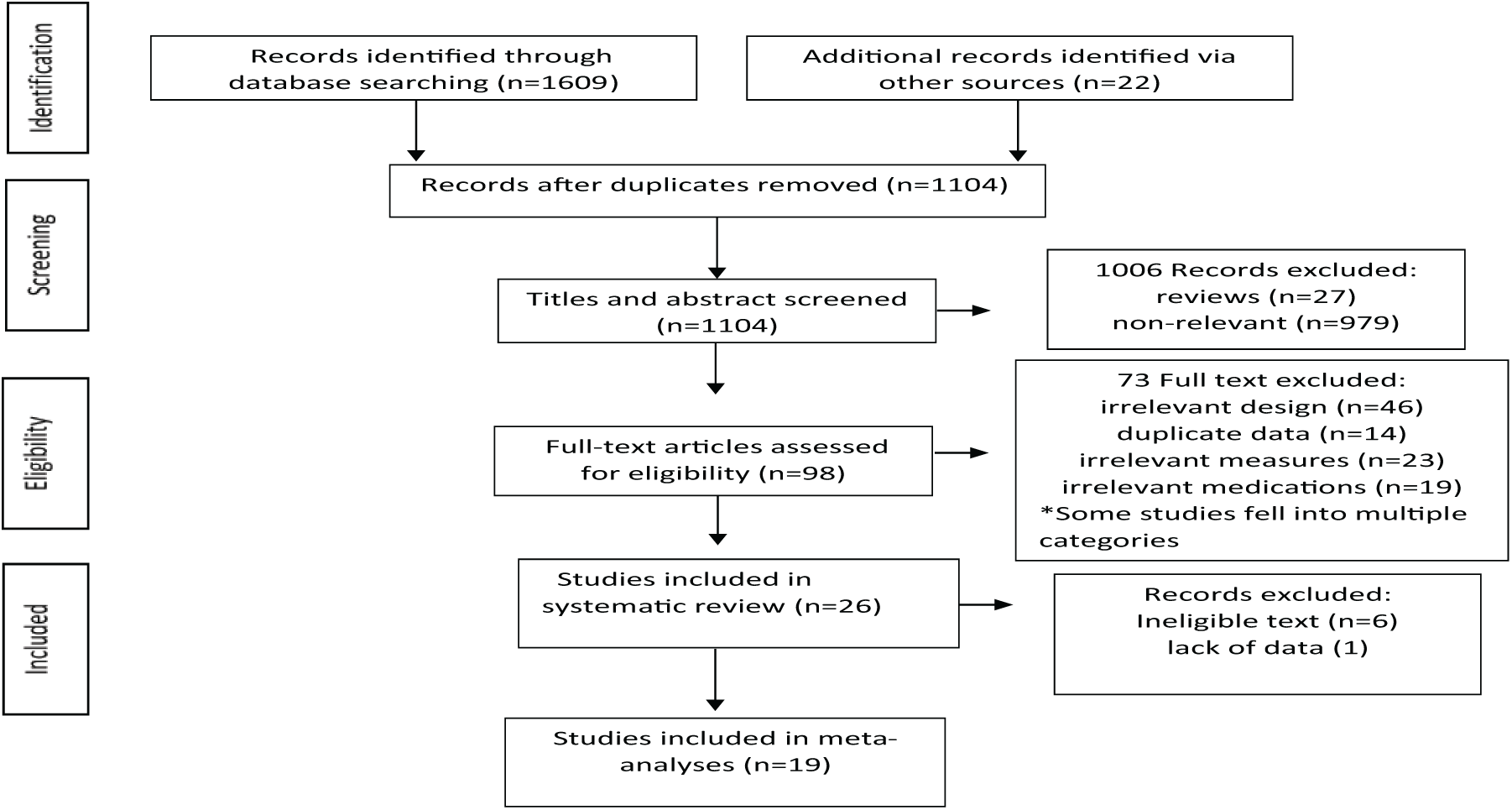
Flow diagram of study selection.
Study characteristics
The study characteristics are shown in Tables 1–3 and Supplementary Table S2. All 26 studies were conducted from 2003 to 2016 and included 11,097 participants with 67.1% males. Data on two trials were obtained solely from clinical trial registry (NCT00604279 and NCT00992407) because there were no publications indexed to them. Overall, 13 studies reported double-blind RCTs including all the 8 placebo-controlled studies, 3 LAI-A-controlled studies and 2 oral-controlled studies. Open-label studies were mostly oral-controlled (
Characteristics of studies on LAI-A vs placebo.
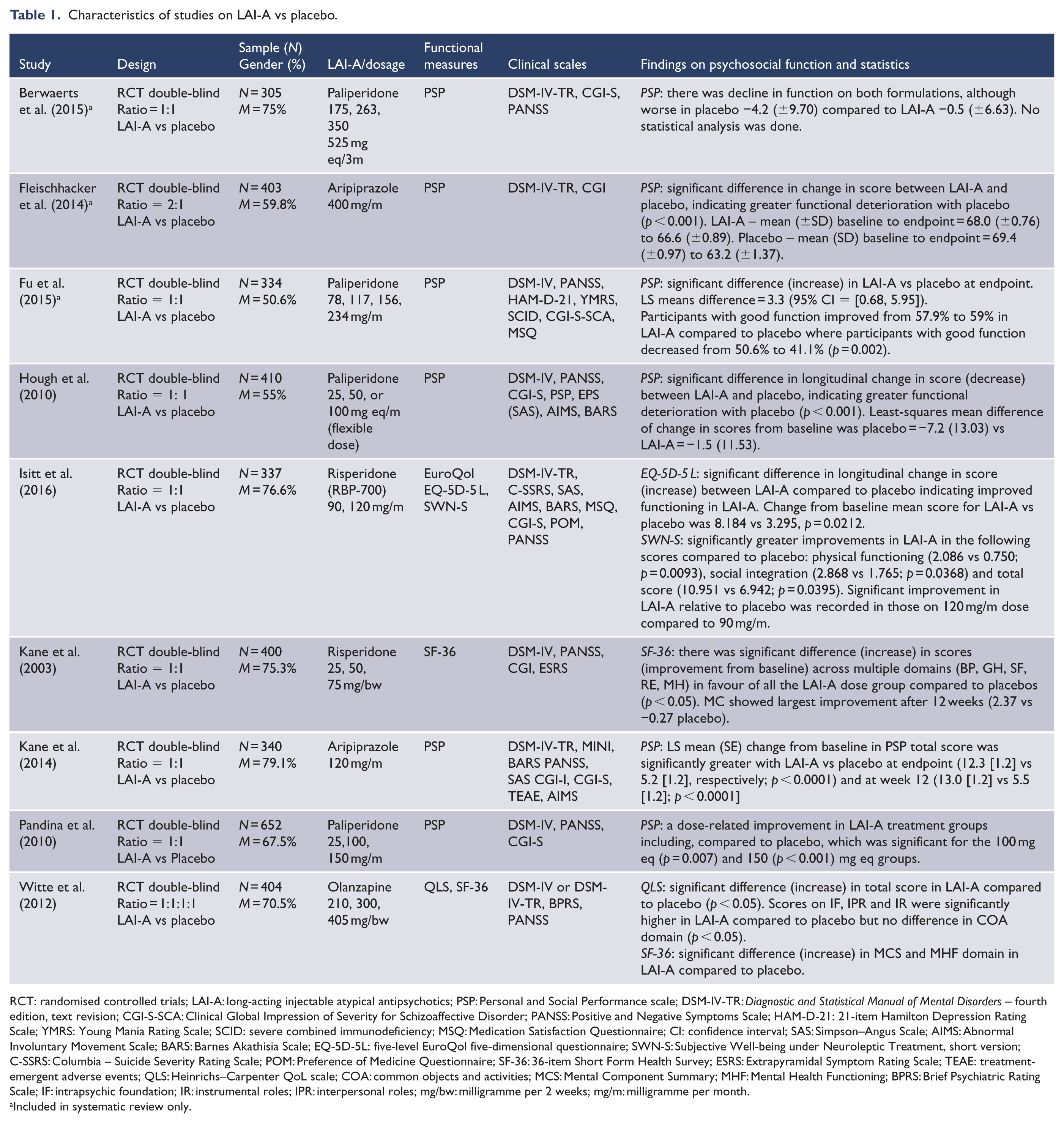
RCT: randomised controlled trials; LAI-A: long-acting injectable atypical antipsychotics; PSP: Personal and Social Performance scale; DSM-IV-TR:
Included in systematic review only.
Characteristics of studies on LAI-A vs oral antipsychotic medications.
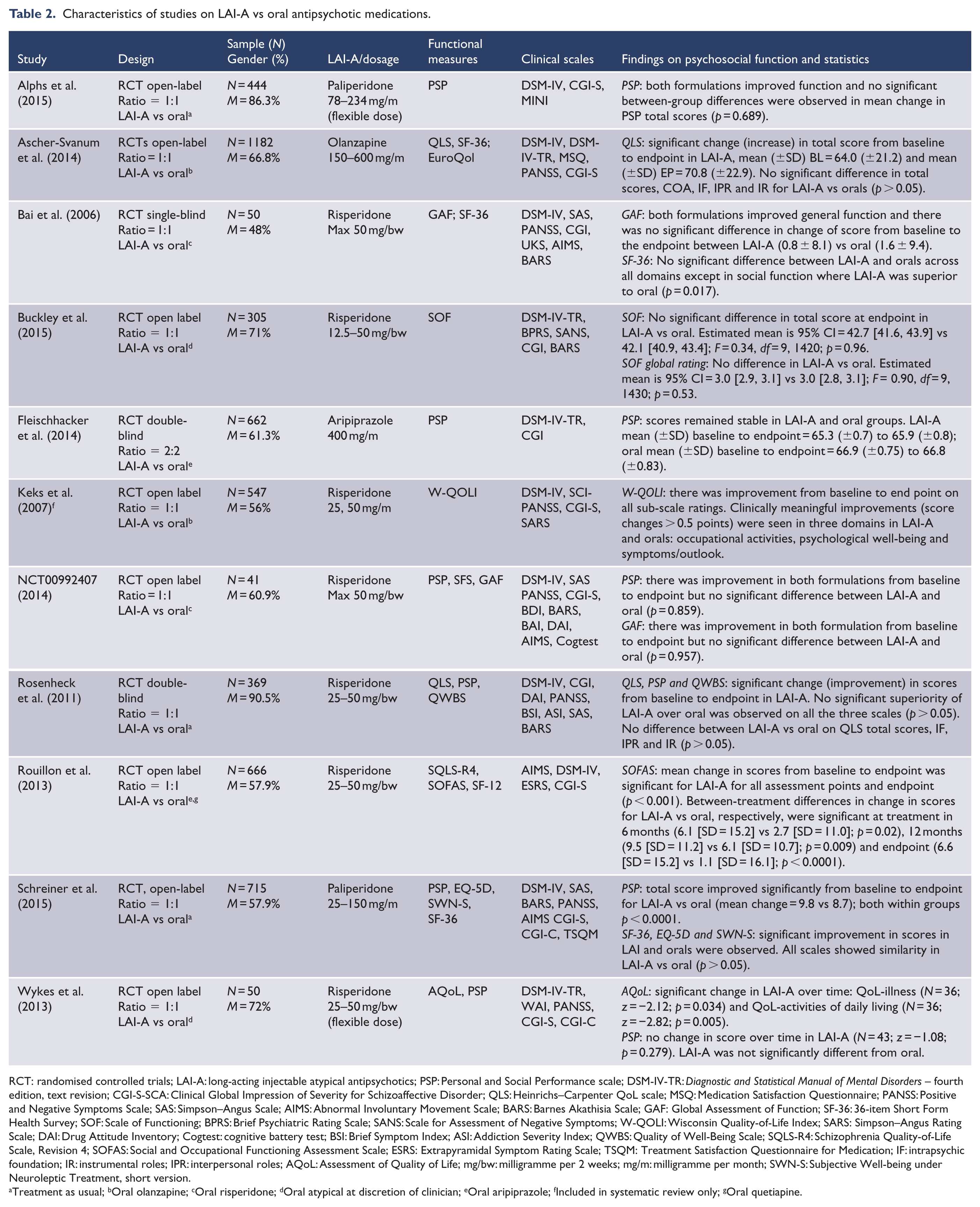
RCT: randomised controlled trials; LAI-A: long-acting injectable atypical antipsychotics; PSP: Personal and Social Performance scale; DSM-IV-TR:
Treatment as usual; bOral olanzapine; cOral risperidone; dOral atypical at discretion of clinician; eOral aripiprazole; fIncluded in systematic review only; gOral quetiapine.
Characteristics of studies on LAI-A vs LAI-A.
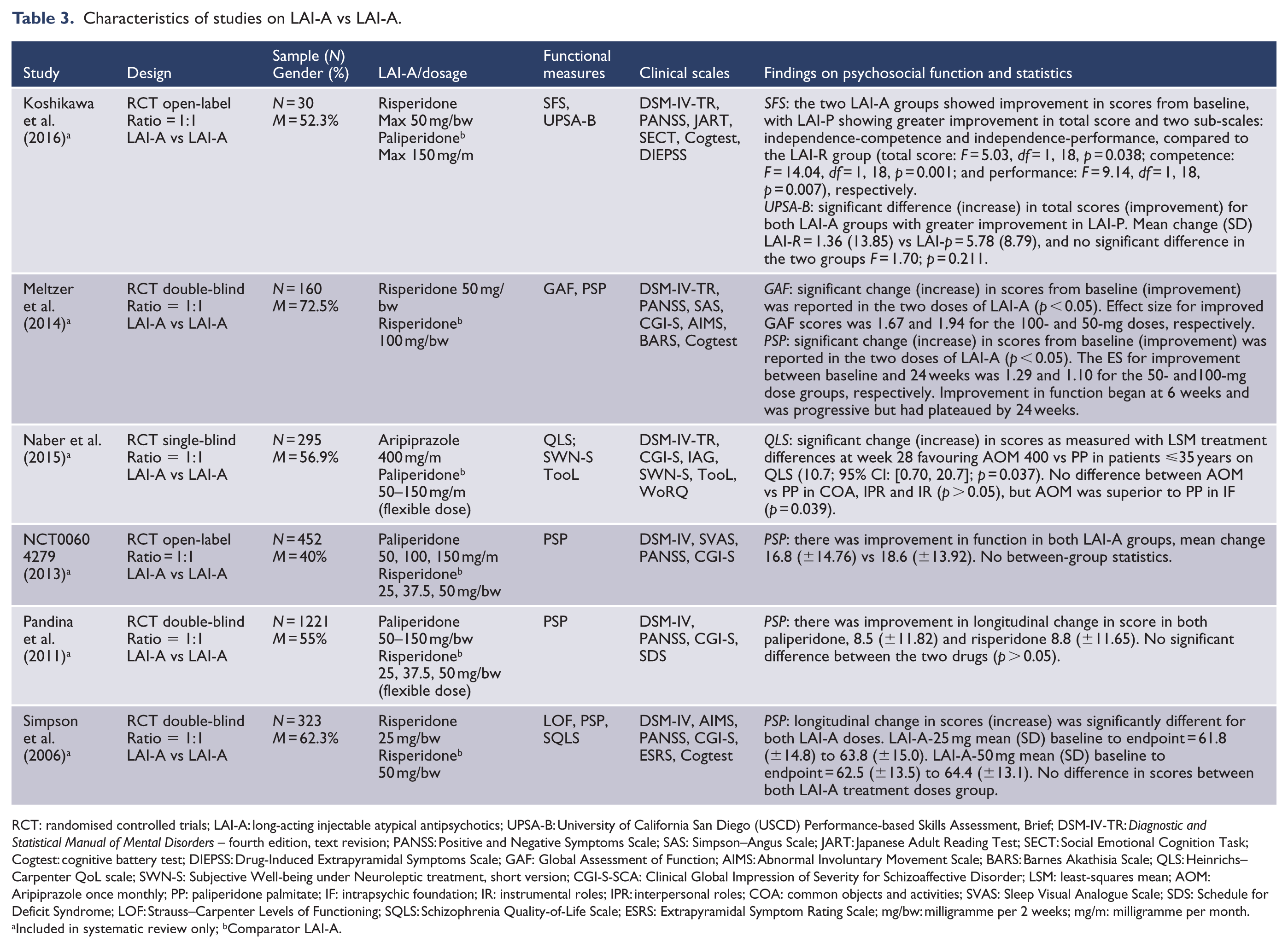
RCT: randomised controlled trials; LAI-A: long-acting injectable atypical antipsychotics; UPSA-B: University of California San Diego (USCD) Performance-based Skills Assessment, Brief; DSM-IV-TR:
Included in systematic review only; bComparator LAI-A.
Diagnoses of schizophrenia and schizoaffective disorder were based on the
In 12 studies, LAI-risperidone was compared with placebo (
A total of 17 different measures of psychosocial function were used across studies. And 13 studies each used single and multiple scales. Personal and Social performance scale (PSP) was used in 17 studies, Short Form Health Survey in 6 studies, Heinrichs–Carpenter QoL scale (QLS) in 5 studies, EuroQol in 2 studies, Global Assessment of Function (GAF) in 3 studies, University of California San Diego (USCD) Performance-based Skills Assessment, Brief (UPSA-B) and Social and Occupational Functioning Assessment Scale (SOFAS) in 1 study each. Only three studies assessed psychosocial function as primary study objective a priori. All the included studies were industry sponsored except two.
Assessment of quality of studies with risk-of-bias tool
The findings on study quality using the risk-of-bias tool are presented in Figures 2 and Supplementary S3. We included 13 double-blind RCTs studies, 11 open-labelled and 2 rater-blinder studies. All 26 studies were randomised but only 11 studies described the process of sequence generation for randomisation including 7 that used interactive web software, 3 that allocated by randomly permuted blocks and 1 study by stratified lot. All participants were aware of treatment allocation in two studies. There was no confirmation of effectiveness of blinding in any of the 26 studies. While selective reporting was low in 24 studies, it was high and unclear in 1 study each. Incomplete outcome data were high in 6 studies due to early withdrawal, drop out from relapse and lack of efficacy, and low in the remaining 20 trials. Other bias was high in three studies, low in one study and unclear in the remaining trials.
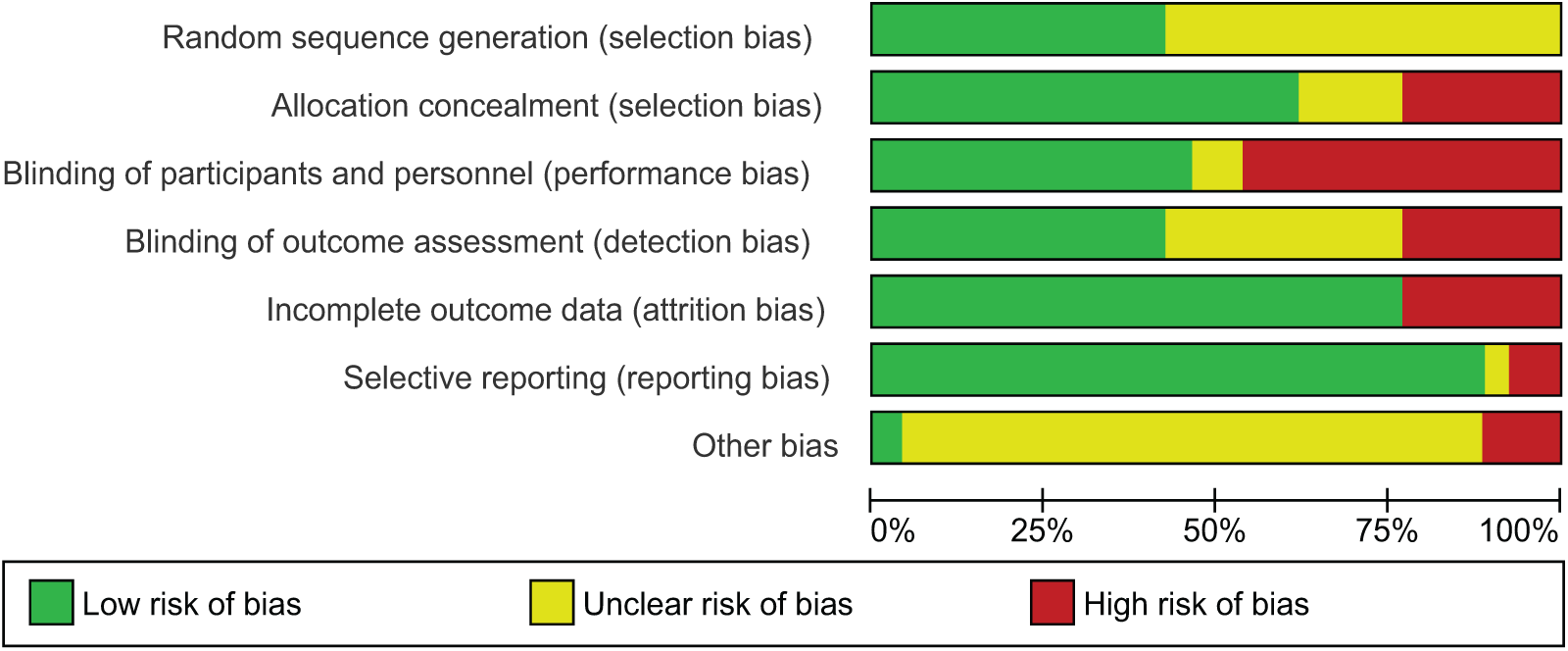
Quality assessment of studies with risk-of-bias tool.
Findings on outcome
Qualitative description of effects of LAI-A across studies on psychosocial function
Qualitative findings on the effects of LAI-A on psychosocial function are presented in Tables 1–3. All the 26 studies reported positive effects of LAI-A on longitudinal change in psychosocial function except three (Berwaerts et al., 2015; Fleischhacker et al., 2014; Hough et al., 2010). LAI-A superiority was reported in three oral-controlled and eight placebo-controlled trials. Head-to-head comparisons of LAI-A were reported in six trials and showed mixed results (Koshikawa et al., 2016; Meltzer et al., 2014; Naber et al., 2015; Pandina et al., 2011; Simpson et al., 2006). Data showing the clinical significance of LAI-A benefits on psychosocial function were reported in few placebo and oral-controlled trials. Findings varied across studies and between LAI-A and comparators. For example, in one study, patients treated with LAI-paliperidone with good functioning (PSP score > 70) increased from 57.9% at baseline to 59.0% at endpoint, whereas there was reduction in the proportion of placebo-treated subjects with good functioning from 50.6% at baseline to 41.1% at trial endpoint. The difference between LAI-A versus placebo was significant (Fu et al., 2015). In one oral-controlled trial, the proportion of patients treated with LAI-paliperidone with good functioning (PSP score > 70) increased from 6.4% at baseline to 36.7% at endpoint, while those on oral antipsychotics with good functioning increased from 6.5% to 34.6% at endpoint (Schreiner et al., 2015). Similarly, 19.2% of LAI-olanzapine-treated patients had a good level of functioning (QLS score ⩾ 84.5) at baseline, which increased to 27.5% (
Effects of LAI-A on psychosocial function compared to placebo
Figures 3–5 show the overall effects of LAI-A compared to placebo on psychosocial function. When the nine included studies were pooled, LAI-A treatment showed significant superiority in functional measures over placebo (SMD = 0.39; 95% confidence interval [CI] = [0.32, 0.47];
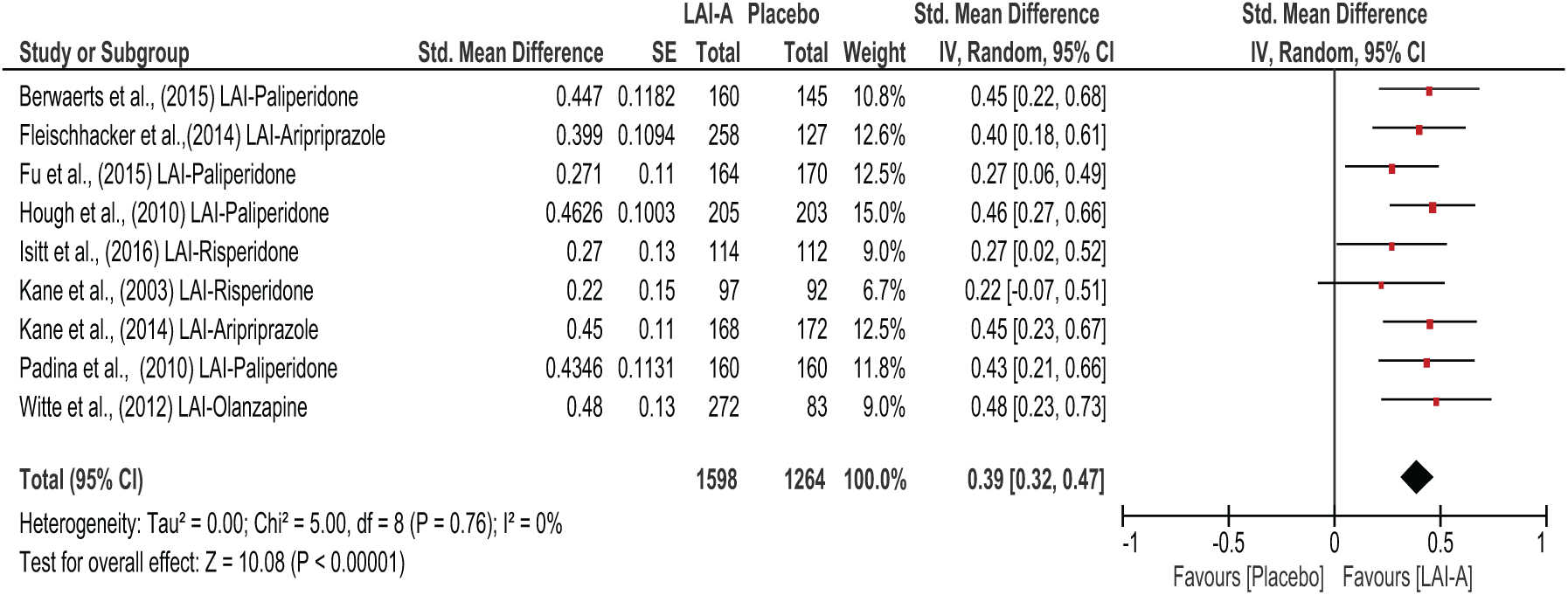
All LAI-A vs placebo studies.
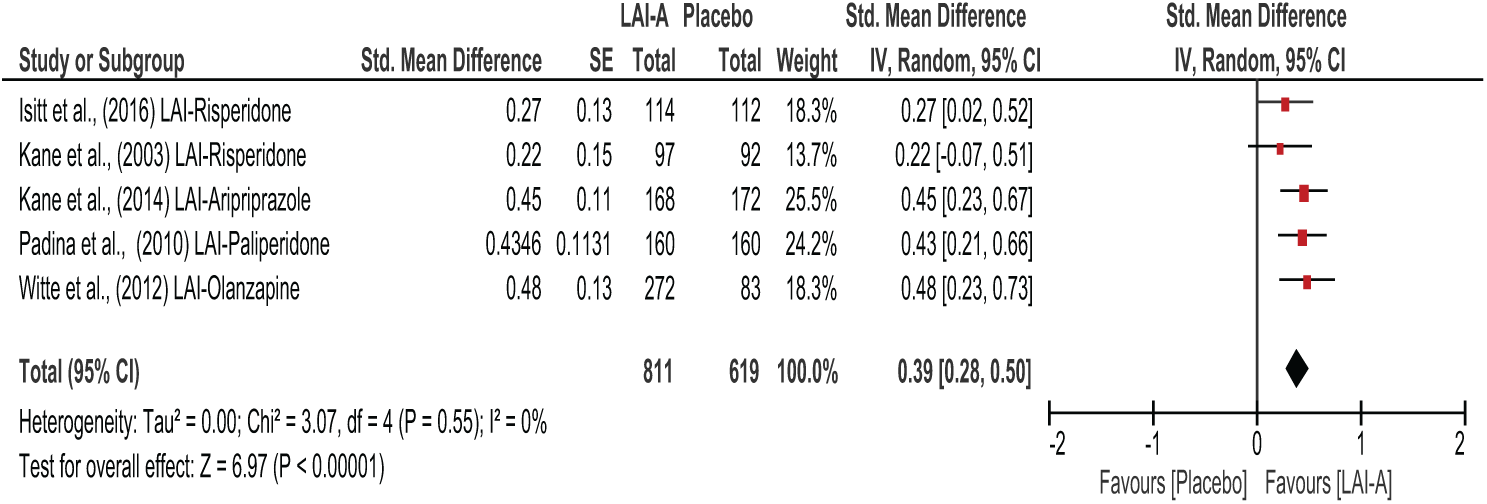
Short-term trials of LAI-A vs placebo.
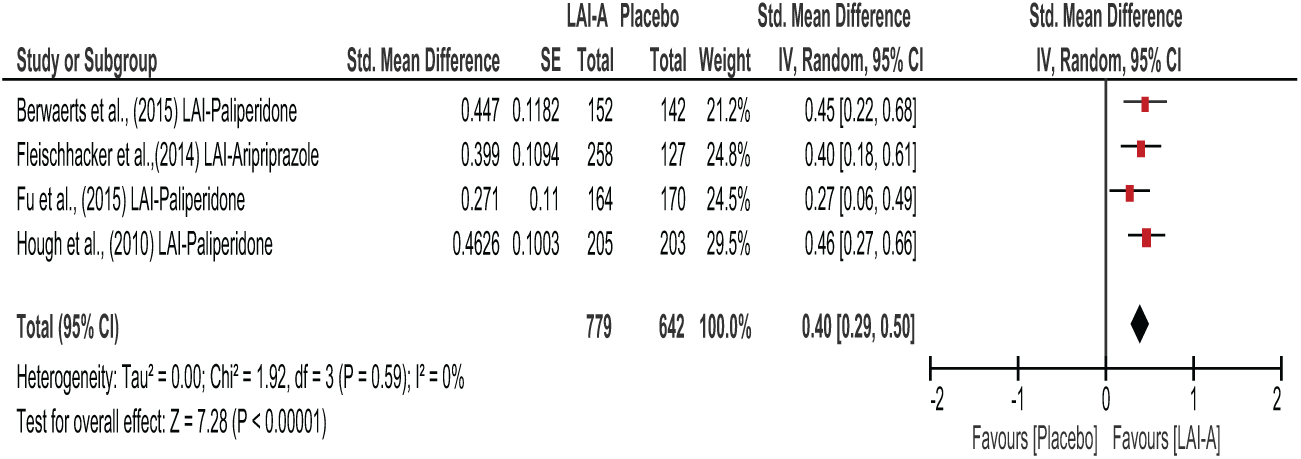
Long-term trials of LAI-A vs placebo.
Effects of LAI-A on psychosocial function compared to oral antipsychotic medications
LAI-A superiority in total score on functional outcomes was reported in three oral-controlled trials (Alphs et al., 2016; Buckley et al., 2015; Rouillon et al., 2013). Seven trials, Ascher-Svanum et al. (2014), Bai et al. (2016), Fleischhacker et al. (2014), NCT00992407, Rosenheck et al. (2011), Schreiner et al. (2015) and Wykes et al. (2013), reported no statistically significant difference in total functional improvement between LAI-A and orals. However, pooled effects of all the 10 oral-controlled studies showed that LAI-A-treated patients displayed a small but significantly greater improvement in functional outcomes compared to oral antipsychotic medications (SMD = 0.16; 95% CI = [0.01, 0.31];
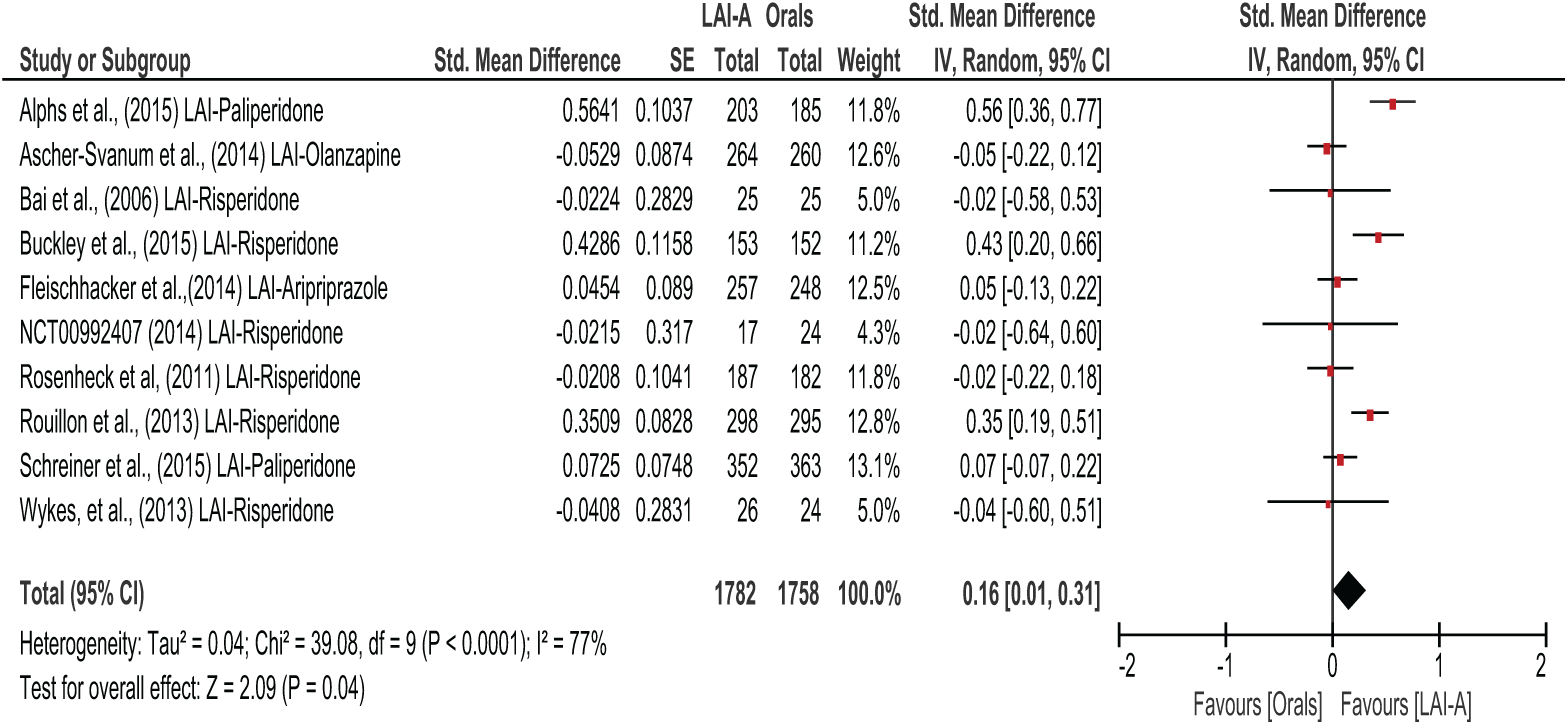
All LAI-A vs orals studies.
Effects of LAI-A across sub-domains of the psychosocial scales
The majority of the studies did not report on sub-domains of the functional scales used. Four studies reported positive change in QLS total scores among LAI-A and also published sub-domain data (Ascher-Svanum et al., 2014; Naber et al., 2015; Rosenheck et al., 2011; Witte et al., 2012). Compared to placebo, LAI-A showed significant improvement in three domains of QLS including intrapsychic functioning (adaptive function and life satisfaction), instrumental roles (socio-occupational function) and interpersonal roles (interpersonal relationship) but no difference in common objects and activities (basic-daily function) (Witte et al., 2012). LAI-risperidone and oral olanzapine treatment showed similar improvements in the sub-domains of occupational activities, psychological well-being and symptoms/outlook based on the Wisconsin Quality-of-Life Index (W-QOLI; Keks et al., 2007). Individual studies suggested improvements for LAI-A over oral antipsychotics in the social function scale of the 36-item Short Form Health Survey (SF-36; Bai et al., 2006) and illness and activities of daily living scales of the Assessment of Quality of Life (AQoL; Wykes et al., 2013). Details are included in Tables 1–3.
Investigation of heterogeneity and meta-regression
Heterogeneity was present in the oral-controlled trials (χ2 = 39.08;
Meta-regression did not show trial effects of study settings (outpatient vs inpatient), trial design, year of publication, industry sponsorship, baseline symptom severity and inclusion of psychosocial function as primary outcome. Egger’s test was not significant for publication bias considering all studies (intercept = 0. 22; 95% CI = [−0.122, 0.563];
Predictors of change in psychosocial function
Predictors of psychosocial function are presented in Table 4. Considering the seven studies that addressed predictors, poor psychosocial function outcome was predicted by baseline factors including high general psychopathology in one study (Witte et al., 2012), high negative symptoms in four studies, (Gharabawi et al., 2007; Koshikawa et al., 2016; Pandina et al., 2013; Witte et al., 2012) and severe illness based on poor CGI-S (Peuskens et al., 2012). Furthermore, positive change in insight, longer duration of treatment, lesser disorganised thoughts and higher cognitive performance predicted better psychosocial function (Gharabawi et al., 2007; Pandina et al., 2013). Witte et al. (2012) reported that high baseline general psychopathology predicted poorer instrumental role, intrapsychic foundations, common objects and activities, interpersonal relations and overall function. More severe negative symptoms predicted poorer recreational and prosocial functioning with medium effect size (Koshikawa et al., 2016). In the study of Gharabawi et al. (2007), a combination of predictive factors explained larger variability (27%) in psychosocial function compared to the 2.1% explained by insight, 10.3% by negative symptoms and 1.6% explained by duration of treatment alone.
Predictors of change in functioning in LAI-A trials.
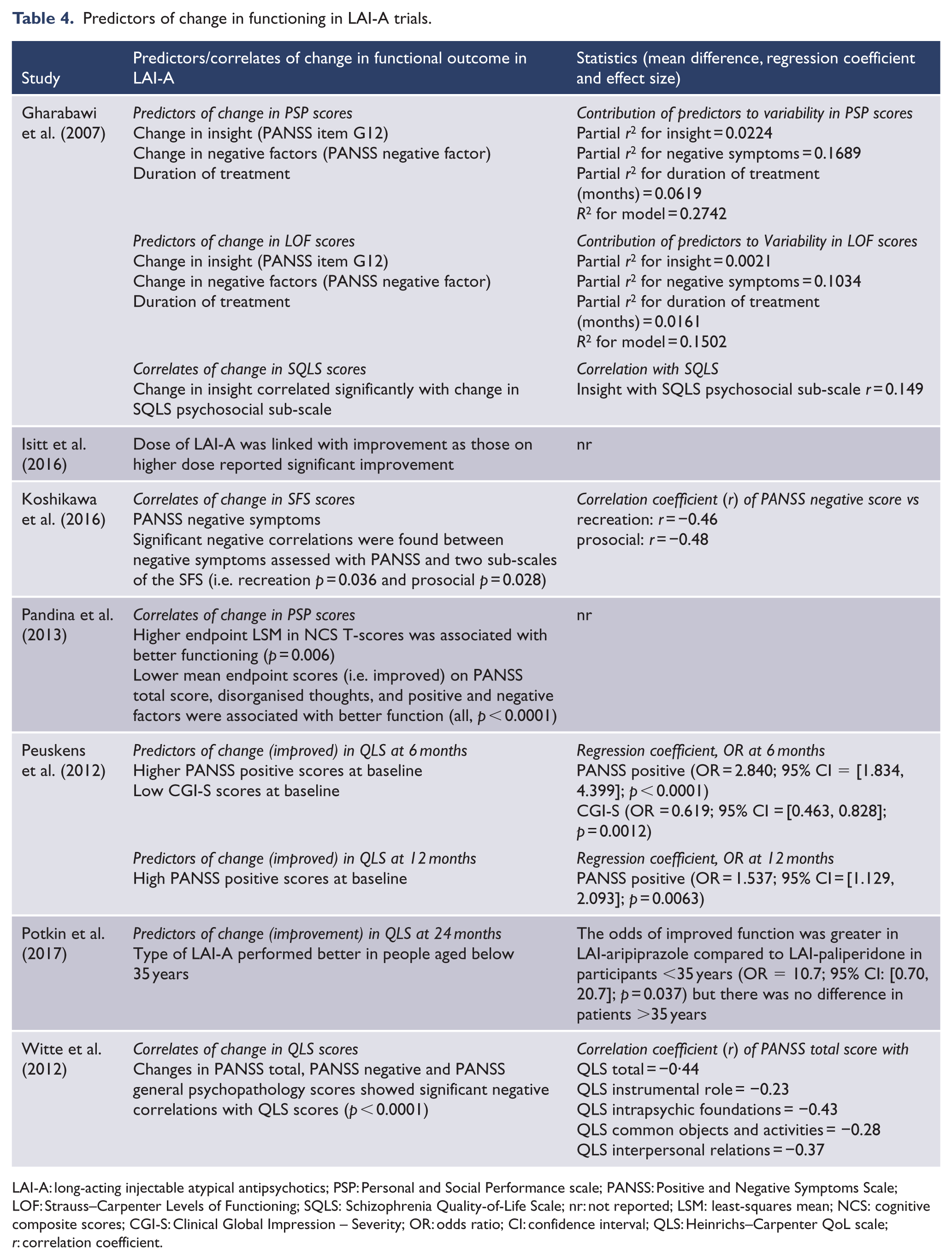
LAI-A: long-acting injectable atypical antipsychotics; PSP: Personal and Social Performance scale; PANSS: Positive and Negative Symptoms Scale; LOF: Strauss–Carpenter Levels of Functioning; SQLS: Schizophrenia Quality-of-Life Scale; nr: not reported; LSM: least-squares mean; NCS: cognitive composite scores; CGI-S: Clinical Global Impression – Severity; OR: odds ratio; CI: confidence interval; QLS: Heinrichs–Carpenter QoL scale;
Discussion
To our knowledge, this systematic review and meta-analyses is the first to analyse the effects and predictors of LAI-A on psychosocial function compared to placebo or oral antipsychotics. We were able to review evidence from 26 studies, including 11,097 participants treated with second-generation LAIs where psychosocial function was indexed by a wide range of measures including QoL (Ro and Clark, 2009).
We found that LAI-A were superior to placebo in improving psychosocial function with a medium effect size of SMD = 0.39, consistent over short- and long-term trials. LAI-A also demonstrated a small but significant benefit (SMD = 0.16) compared to oral antipsychotic medications for functional outcome in long-term clinical trials. While there was clear evidence of clinically meaningful benefit for LAI-A treatment over placebo, where reported absolute differences between oral and LAI-A treatment in terms of percentage of patients achieving good function were small, this benefit is smaller than that reported in mirror-image and some large cohort studies, potentially due to higher levels of oral adherence and under representation of non-compliant patients in selective and closely monitored clinical trials (Kane et al., 2013; Tiihonen et al., 2011; Zhornitsky and Stip, 2012). Thus, there is a need for true effectiveness trials, in which post-randomisation involvement would be kept to a minimum to better reflect routine practice (Kane et al., 2013; Kishimoto et al., 2014). Non-adherence with oral antipsychotic medication is associated with high rates of relapse and poor functional outcomes (Leucht et al., 2011). Adherence is affected by many factors including insight, side-effects, patient treatment goals, attitude to treatment and the effectiveness of the treatment itself (Kikkert et al., 2006). Depot treatments can reduce day-to-day fluctuations in compliance that occur with oral dosing and allow clear monitoring of adherence, leading to early psychoeducation and intervention.
Systematic review of predictors in seven studies showed that patients with more severe symptoms (Ascher-Svanum et al., 2014; Koshikawa et al., 2016; Simpson et al., 2006; Witte et al., 2012), cognitive impairment (Pandina et al., 2013) and poor insight (Simpson et al., 2006) at decision to trial LAI-A were less likely to improve in psychosocial function. Our findings are consistent with the existing evidence of a clear association between cognitive impairment, severity of psychopathology and poor insight with poor functional recovery (Bowie and Harvey, 2006). This association is complex but modifiable, such that adherence, less severe psychopathology and functional recovery are associated with better insight, while insight is cognition-dependent, particularly on executive performance and working memory (Aleman et al., 2006; Donohoe et al., 2005; Rajji et al., 2014; Rossell et al., 2003). Evidence suggests that insight (Pijnenborg et al., 2013) and cognition can be improved by a wide range of interventions including cognitive behavioural therapy, assertive community treatment and cognitive remediation. These interventions are recommended for the promotion of functional recovery and in combination with pharmacotherapy lead to the best psychosocial outcomes (Galletly et al., 2016; Mueser et al., 2013; Pfammatter et al., 2006). Further research is required to predict poor functional outcomes and fully personalise the selection of medications and interventions (Jaaskelainen et al., 2013). More fundamentally, there is a need to develop better understanding of the pathophysiology of functional decline and studies of the biological basis of function are ongoing (Clark et al., 2018; Wojtalik et al., 2017).
On a different note, our findings underscore the impact of antipsychotics on overall outcome, and by extension support the benefits of maintenance antipsychotics in patients with schizophrenia. Several studies have reported decline in functional recovery and increased relapse rates with cessation of maintenance antipsychotic treatment (Thompson et al., 2018). Notwithstanding the small advantage of LAI-A over oral antipsychotic medications on psychosocial function, careful interpretation of this finding is required. The choice of antipsychotic formulation for individual patient should entail a balanced consideration of clinical factors, patient attributes and evidence from clinical guidelines through a shared decision process (Correll et al., 2016).
There are several limitations to this study. First, in all but two included studies, psychosocial function was not the primary outcome measure. While some trials reported statistical corrections to address potential effects of this limitation on findings, the quality of evidence would be much improved if cofounding effects and study power were considered in relation to psychosocial function a priori (Freemantle, 2001). Given the increasing burden of mental illness-related disability and the emerging understanding that symptomatic improvement is not prerequisite nor necessarily sufficient for improved function, there is a need to consider functional measures as primary outcomes in studies of antipsychotic efficacy (GBD 2016 DALYs and HALE Collaborators, 2016; Whiteford, 2017). In keeping with a broad composite concept of psychosocial function, 50% of the studies reviewed considered both QoL and general functional measures. Standardisation of these approaches will make future trials of depot efficacy more comparable.
Similar to previous systematic reviews of the literature, we found poor reporting of the randomisation process, concealment and sequence generation was common (Leucht et al., 2011). We used a random-effects model to address heterogeneity in psychosocial function scales and grouped trials into clinical, meaningful placebo-controlled versus oral-controlled groups during analyses and sensitivity analyses. The oral-controlled data showed heterogeneity of 77%, accounted for by three studies that showed superiority of LAI-paliperidone and LAI-risperidone over orals (Alphs et al., 2016; Buckley et al., 2015; Rouillon et al., 2013). The oral arms of these trials were not well standardised or monitored. For example, LAI-A were not necessarily compared to the same atypical oral; however, recommended dosing was generally implemented. The outcome of meta-analysis was not changed when studies using a TAU oral comparator were excluded.
Only two non-industry trials could be included suggesting risk of industry-sponsored bias (Buckley et al., 2015; Koshikawa et al., 2016). Studies varied in active comparators and drug dosage; however, all trials used doses within the recommended ranges. Trial length did not appear to influence functional outcome in depot over placebo treatment, suggesting a ceiling effect as optimal function was met within the first 3 months (Carter, 2006; Swartz et al., 2007). Most of the trials did not look at predictors of psychosocial function and useable data were poorly reported.
Conclusion
We conclude that LAI-A improve psychosocial function, but in clinical trials, the magnitude of effect is small when compared to oral antipsychotic medications. Patients with more severe symptoms and cognitive impairment at decision to trial were less likely to improve in psychosocial function and may benefit from more intensive psychosocial therapies in adjunct to pharmacological treatment. Consideration of baseline attributes at diagnosis could help to stratify patients who may benefit from early adjunct interventions. Further analysis of predictors of long-term function is needed, potentially leading to trials of indicated psychosocial intervention. Future clinical trials should improve on the quality of reporting and design, and include composite psychosocial function as a measure of efficacy a priori.
Supplemental Material
Revised_supplmentary_material_for_ANZJPF31012019 – Supplemental material for Long-acting atypical antipsychotics in schizophrenia: A systematic review and meta-analyses of effects on functional outcome
Supplemental material, Revised_supplmentary_material_for_ANZJPF31012019 for Long-acting atypical antipsychotics in schizophrenia: A systematic review and meta-analyses of effects on functional outcome by Andrew T Olagunju, Scott R Clark and Bernhard T Baune in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to thank Suzanne Edwards for providing statistical advice and support. This research was supported by an Australian Government Research Training Programme (RTP) Scholarship. The abstract was presented in the New Investigator Award Programme at the 2018 Annual Meeting of the American Society of Clinical Psychopharmacology, Miami, Florida.
Author contribution
All authors were involved in the study design and interpretation of results. Article selections were independently performed by A.T.O. Data extraction was completed by A.T.O. independently using a piloted form. Clarification was resolved by consultation with the senior author (B.T.B.). A.T.O. and S.R.C. drafted the manuscript with supervision from B.T.B. All authors participated in critical revision of manuscript drafts and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
