Abstract
Objective:
Dementia with Lewy bodies is the second most common form of neurodegenerative dementia in older age yet is often under-recognised and misdiagnosed. This review aims to provide an overview of the clinical features of dementia with Lewy bodies, discussing the frequent challenges clinicians experience in diagnosing dementia with Lewy bodies, and outlines a practical approach to the clinical management, particularly in the Australian setting.
Methods:
This paper is a narrative review and a semi-structured database (PubMed and MEDLINE) search strategy was implemented. Articles were screened and clinically relevant studies were selected for inclusion.
Results:
Dementia with Lewy bodies is clinically characterised by complex visual hallucinations, spontaneous motor parkinsonism, prominent cognitive fluctuations and rapid eye movement sleep behaviour disorder. Neuropsychiatric features and autonomic dysfunction are also common. The new diagnostic criteria and specific diagnostic biomarkers help to improve detection rates and diagnostic accuracy, as well as guide appropriate management. Clinical management of dementia with Lewy bodies is challenging and requires an individualised multidisciplinary approach with specialist input.
Conclusion:
Dementia with Lewy bodies is a common form of dementia. It often presents as a diagnostic challenge to clinicians, particularly at early stages of disease, and in patients with mixed neuropathological changes, which occur in over 50% of people with dementia with Lewy bodies. Prompt diagnosis and comprehensive treatment strategies are important in improving patients’ care.
Introduction
Dementia is common in older people, with current estimates of over 47 million living with dementia worldwide (World Health Organization, 2015). The global prevalence estimates range between 5% and 7% for people over the age of 60, increasing to 20% of people over 85 years (Prince et al., 2013). In Australia, there are over 300,000 people living with dementia, as the leading cause of disability and the third leading cause of death; it is a National Health Priority (Australian Institute of Health and Welfare, 2012).
Dementia with Lewy bodies (DLB) is a common form of neurodegenerative dementia in older people, yet remains widely under-detected and often misdiagnosed. The histopathology of DLB is characterised by progressive accumulation and aggregation of the synaptic protein alpha-synuclein (α-syn) as Lewy bodies and Lewy neurites, in the brainstem, limbic and neocortical regions (Ferman et al., 2018; Vann Jones and O’Brien, 2014). Clinically, DLB manifests as progressive cognitive decline, typically in conjunction with cognitive fluctuations, parkinsonism and visual hallucinations (see Table 1). It shares many cognitive and non-cognitive symptoms with both Alzheimer’s disease (AD) and Parkinson’s disease (PD). An often devastating condition, DLB is associated with a poorer health-related quality of life, higher caregiver burden and mortality rates, compared to people living with AD (Bostrom et al., 2007a; Price et al., 2017). DLB patients also comparably utilise more resources with the costs of care twice those for AD patients (Bostrom et al., 2007b). Despite this, diagnostic accuracy remains a challenge with often significant delays in diagnosis (Galvin et al., 2010).
Common clinical features of dementia with Lewy bodies.

REM: rapid eye movement.
Core clinical features in 2017 consensus diagnostic criteria (McKeith et al., 2017).
This paper seeks to review the challenges in clinical diagnosis of DLB and to provide a framework for the management, particularly in the Australian setting. A semi-structured database (PubMed and MEDLINE) search strategy was implemented. Articles were screened and clinically relevant studies were selected for inclusion.
Clinical diagnosis of DLB
Cognitive symptoms
Patients with DLB often present with more prominent deficits in attention, executive function and visuospatial skill on cognitive assessment, as opposed to the more frequently occurring deficits in episodic memory seen in people with AD. Similar to other dementias, initial cognitive assessments may include the use of screening tools such as Mini-Mental State Examination (MMSE; Folstein et al., 1975), Montreal Cognitive Assessment (MoCA; Nasreddine et al., 2005) and Addenbrooke’s Cognitive Examination (ACE; Mathuranath et al., 2000). Low performance on these tests often represents global cognitive impairment and it is important to assess deficits in cognitive subdomains which may help in establishing a diagnosis. For instance, patients who have intact short-term recall but significantly impaired pentagon-copying skill on MMSE should raise the suspicion of possible DLB.
More recent studies have investigated the different cognitive profiles in the prodromal or mild cognitive impairment (MCI) stages of DLB (MCI-DLB) and AD (MCI-AD), and found that patients with MCI-DLB also showed greater impairment in visuospatial, visual attention as well as verbal fluency, compared to MCI-AD (Cagnin et al., 2015a). Interestingly, the pentagon-copying test, a subtest in MMSE, with a qualitative scoring method has been proposed in a number of studies as a valid predictive tool for patients with DLB as well as in MCI-DLB stage, i.e. patients with MMSE of 26 or more (Caffarra et al., 2013; Cagnin et al., 2015b). It is unlikely that clinicians are able to form a definitive diagnosis based on deficit in one single subtest, but it might help alert clinicians to assess for other features of DLB (Caffarra et al., 2013; Cagnin et al., 2015b).
Cognitive fluctuation is a core clinical feature of DLB and is one of the most difficult clinical features to confidently recognise. It can share features with the fluctuations seen in delirium, occurring as spontaneous alterations in cognition, attention and arousal (Morandi et al., 2017). Common examples of cognitive fluctuations in DLB include daytime drowsiness, blank stares and being disengaged in conversations. In some cases, patients may have undergone diagnostic workup for ‘persistent delirium’ or ‘transient ischaemic attacks’, further highlighting the diagnostic challenge. Assessment tools, including Mayo Fluctuations Scale and Clinical Assessment of Fluctuation, rely on proxy information from caregivers (Walker et al., 2000). The severity of cognitive fluctuation may also serve as a prognostic marker. DLB patients with more severe attentional impairment, but not global impairment, and more extensive thalamic atrophy on magnetic resonance imaging (MRI) were associated with a higher risk of mortality in one study (Watson et al., 2017). In AD, however, cognitive fluctuations typically occur as episodes of memory failure in response to higher cognitive demands and are often referred to as good and bad days, different to the experiences reported in DLB (Bradshaw et al., 2004).
Neuropsychiatric features
Neuropsychiatric symptoms in dementias are common and often distressing for patients and caregivers. They can indistinctly occur in all forms of dementias, but a higher prevalence has been reported at early stages of DLB, compared to early AD (Simard et al., 2000). These non-cognitive behavioural symptoms, encompassing behavioural, mood and psychotic symptoms, are often associated with higher caregiver burden leading to subsequent admission into residential care facilities (Bostrom et al., 2007a; Rongve et al., 2014).
Recurrent visual hallucinations are a core clinical feature in DLB and are a frequent signpost to diagnosis for clinicians (McKeith et al., 2017). They are present in up to 80% of patients with DLB (Simard et al., 2000; Urwyler et al., 2016). However, these symptoms also commonly occur in other conditions such as delirium, psychiatric disorder and eye disease. Visual hallucinations in DLB are typically complex and well formed or panoramic, featuring people, children and animals. Patients and/or caregivers are often able to report these experiences with good insight (Urwyler et al., 2016). In clinical practice, visual hallucinations are commonly assessed based on patients’ and/or caregivers’ history and specific questions to determine the phenomenology of visual hallucinations help in identifying the underlying causation (Urwyler et al., 2016). Visual hallucinations in DLB often occur with cognitive fluctuations and are correlated with deficits in visual attention and executive functions on cognitive testing (Cagnin et al., 2013).
Apart from visual hallucinations, other modalities of hallucination, although less common, can also occur in DLB. For instance, auditory hallucinations have been reported in around 30% of pathologically confirmed DLB patients in one study (Klatka et al., 1996).
Delusions frequently occur in people with DLB and examples include misidentification delusion, paranoid delusion and phantom boarder. A previous review of 47 articles (mostly case reports and retrospective studies) reported that misidentification delusion was more common in DLB, while theft delusion (e.g. money being stolen) was more commonly experienced by AD patients (Simard et al., 2000). Capgras syndrome, a form of misidentification delusion, has been well described in DLB. It is characterised by the fixed false belief that a familiar person, usually a family member or caregiver, has been replaced by an imposter and is more likely to occur in the moderate stages of disease (Thaipisuttikul et al., 2013).
Depression and dementia frequently coexist, with higher rates of depression reported in DLB compared with AD (Fritze et al., 2011; Klatka et al., 1996; Simard et al., 2000). Approximately 20% to 60% of DLB patients suffer from co-morbid depression and it can be associated with poorer mental health in caregivers (Borroni et al., 2008; Chiu et al., 2017; Fritze et al., 2011; Simard et al., 2000). Similar to depression, anxiety also occurs frequently, affecting up to 20% to 65% across all stages of DLB (Borroni et al., 2008; Breitve et al., 2016). Apathy, defined as the lack of motivation or interest not due to mood disorder, has been observed in approximately 55% of people with DLB (Borroni et al., 2008). Apathy can be present without depression and it is clinically important to differentiate between these two conditions as the use of selective serotonin reuptake inhibitors to treat depression can potentially exacerbate apathy in DLB patients (Barnhart et al., 2004).
Motor symptoms
Spontaneous symptoms of motor parkinsonism occur in around 50% of people with DLB in the early stages and affect up to 85% as the disease progresses (Ferman et al., 2011, 2014). It is important to note that the presence of motor parkinsonism is not needed to diagnose DLB. Furthermore, the presence of only one of the three cardinal features of parkinsonism (i.e. tremor, bradykinesia and rigidity) is required to meet criteria for parkinsonism according to the revised consensus diagnostic criteria (McKeith et al., 2017). When motor parkinsonism is present in DLB, patients more commonly experience motor features mediated by the non-dopaminergic systems, namely, the postural-instability-gait-disorder (PIGD) subtype, as opposed to tremor-dominant (TD) subtype in PD patients (Burn et al., 2003). This PIGD motor subtype tends to be less responsive to levodopa therapy (Lucetti et al., 2010). Clinically, it is also important to consider the potential effect of medications such as antipsychotics and vascular contribution, which are common in older people.
Relationship to PD
Lewy body diseases (LBDs) are the result of abnormal accumulation of the synaptic protein alpha-synuclein and include PD, Parkinson’s disease dementia (PDD) and DLB. Cognitive impairment is common among PD patients and the prevalence of dementia in PD is approximately 30% to 40%, increasing to 80% after 20 years (Goetz et al., 2008; Hely et al., 2008). DLB and PDD have considerable overlaps clinically and pathologically and are both Lewy body dementias. DLB is diagnosed when the onset of dementia precedes or occurs within a year of the onset of motor parkinsonism, while PDD is diagnosed when cognitive decline occurs in the context of well-established PD (at least 1 year after the onset of motor symptoms). The distinction between DLB and PDD has been an area of research and debate for years among experts. The ‘1-year rule’ is useful for research purposes to assist with cross-study comparisons and data pooling. However, in the clinical setting, these definitions can, on occasion, be unhelpful, and general terms such as Lewy body disease or Lewy body dementia may be more appropriate.
Sleep
Sleep disturbances are common in people with dementia. Rapid eye movement (REM) sleep behaviour disorder (RBD), present in up to 80% of DLB patients, is a parasomnia manifested by recurrent dream enactment behaviours associated with an absence of normal REM sleep atonia (Ferman et al., 2014; Pao et al., 2013). The behaviours can range from vocalisation, jerks to more complex motor movements such as punching or kicking out, which can potentially result in injuries to self and/or bedpartner. RBD is now regarded as a core clinical feature of DLB in the revised diagnostic criteria (McKeith et al., 2017) due to its high specificity and sensitivity for DLB compared to other forms of dementia, as assessed in post-mortem studies (Ferman et al., 2011; Iranzo et al., 2013). Isolated RBD has also been increasingly recognised as a prodromal phase (Iranzo et al., 2013). Longitudinal cohort studies have consistently demonstrated high risk of progression of isolated RBD to a synucleinopathy with time and may precede the onset of dementia by 6 to 10 years (Boeve et al., 2013; Ferman et al., 2011; Iranzo et al., 2013). It is important to directly enquire as to these symptoms, from the patients and more importantly their partners. As a symptom, it may have been present for many years prior to the onset of cognitive symptoms and therefore may not be considered relevant by patients and/or families at the time of assessment. Questionnaires have been developed to assist with screening for RBD, for instance, the Mayo Sleep Questionnaire to be completed by bedpartner or informant (Boeve et al., 2011). Previous validation study of Mayo Sleep Questionnaire has demonstrated a high sensitivity of 98% and specificity of 74% for the diagnosis of RBD, compared to polysomnography (PSG; Boeve et al., 2011). However, in some cases, a sleep study may still be necessary to accurately diagnose RBD and to exclude possible obstructive sleep apnoea.
The absence of normal muscle atonia during REM sleep on PSG is the electrophysiological hallmark of RBD and is now an indicative biomarker of DLB (McKeith et al., 2017). A clinical history of RBD accompanied by confirmation of REM sleep without atonia on PSG produced a 98% likelihood of a synucleinopathy, as confirmed on a neuropathologic study (Boeve et al., 2013). This can be helpful clinically, particularly where there is doubt about the clinical diagnosis of RBD and also to address possible obstructive sleep apnoea to guide appropriate treatment.
Excessive daytime somnolence, or hypersomnia, has also been found to be more common in DLB compared to AD (Ferman et al., 2014). Its presence was shown to be independent of night-time sleep efficiency, RBD and dementia severity in DLB (Ferman et al., 2014).
Autonomic features
Autonomic dysfunction is frequently under-recognised despite its common occurrence in approximately 30% to 50% of people with DLB (Allan et al., 2006, 2007). The pathophysiology is thought to be related to the accumulation of Lewy pathology in the autonomic nervous system as well as the result of cholinergic dysfunction (Allan et al., 2007). Clinical manifestations of autonomic dysfunction include orthostatic hypotension (without compensatory tachycardia), constipation, bladder incontinence, excessive sweating and erectile dysfunction, which can all have significant implications on quality of life (Allan et al., 2006). They can potentially result in repeated falls and syncope, which are also supportive clinical features on the revised diagnostic criteria (McKeith et al., 2017).
123Iodine-metaiodobenzylguanidine (123I-MIBG) myocardial scintigraphy is a nuclear medicine study using analogue of noradrenaline to evaluate cardiac sympathetic denervation which frequently occurs as autonomic dysfunction in LBDs, including DLB. A prospective multicenter study using 123I-MIBG myocardial scintigraphy has demonstrated sensitivity and specificity for DLB of 69% and 87%, respectively, compared to non-DLB dementias (Yoshita et al., 2015). It may also be used as a predictor of conversion of possible to probable DLB at 1 year (Oda et al., 2013). However, some of the disadvantages of this scan were that the results can be affected by common cardiac conditions such as myocardial infarction, cardiac failure and dilated cardiomyopathy, as well as medications such as calcium channel blockers and labetalol, somewhat limiting its use.
Other clinical features
Presence of Lewy bodies in the olfactory bulb, clinically presenting as hyposmia or reduced ability to smell, has been hypothesised as one of the earliest sites of involvement in DLB and may predate the onset of cognitive impairment by several years (Chiba et al., 2012; Funabe et al., 2013). Clinically, hyposmia is rarely the primary presenting complaint in patients, but its presence supports the diagnosis of DLB.
Diagnostic criteria
The clinical diagnostic criteria for DLB was first developed in 1996 by consensus (McKeith et al., 1996), which required one or two of the three core clinical features of recurrent visual hallucinations, spontaneous features of motor parkinsonism and cognitive fluctuation to make a diagnosis of possible or probable DLB, respectively. These criteria were found to be highly specific up to 90%, but the poor sensitivity indicated that many DLB cases were being missed during life (Luis et al., 1999; Rizzo et al., 2018). To address this, the consensus criteria were revised in 2005 (McKeith et al., 2005), which added suggestive features of RBD, severe neuroleptic sensitivity and reduced dopamine transporter uptake in basal ganglia on single photo emission computed tomography (SPECT) or positron emission tomography (PET) imaging. Both population-based and clinical prevalence studies conducted using the revised 2005 diagnostic criteria for DLB have reported significantly higher prevalence rates than those using the original 1996 criteria, with an increase from 3.7% to 8.2% for population studies and 6.7% to 11.4% for clinical studies (Vann Jones and O’Brien, 2014).
With the aim to further improve diagnostic accuracy and detection rates, the diagnostic criteria were subsequently revised again in 2017 with increased weighting given to RBD as a core clinical feature (see Table 2; McKeith et al., 2017). Furthermore, a new classification of ‘indicative biomarkers’ was added, which included reduced dopamine transporter uptake in basal ganglia on SPECT or PET, low uptake on 123I-MIBG myocardial scintigraphy and polysomnographic confirmation of REM sleep without atonia. The presence of at least two core clinical features or one core clinical feature with one or more indicative biomarkers is required for the diagnosis of probable DLB.
Revised criteria for the clinical diagnosis of probable and possible DLB (McKeith et al., 2017).
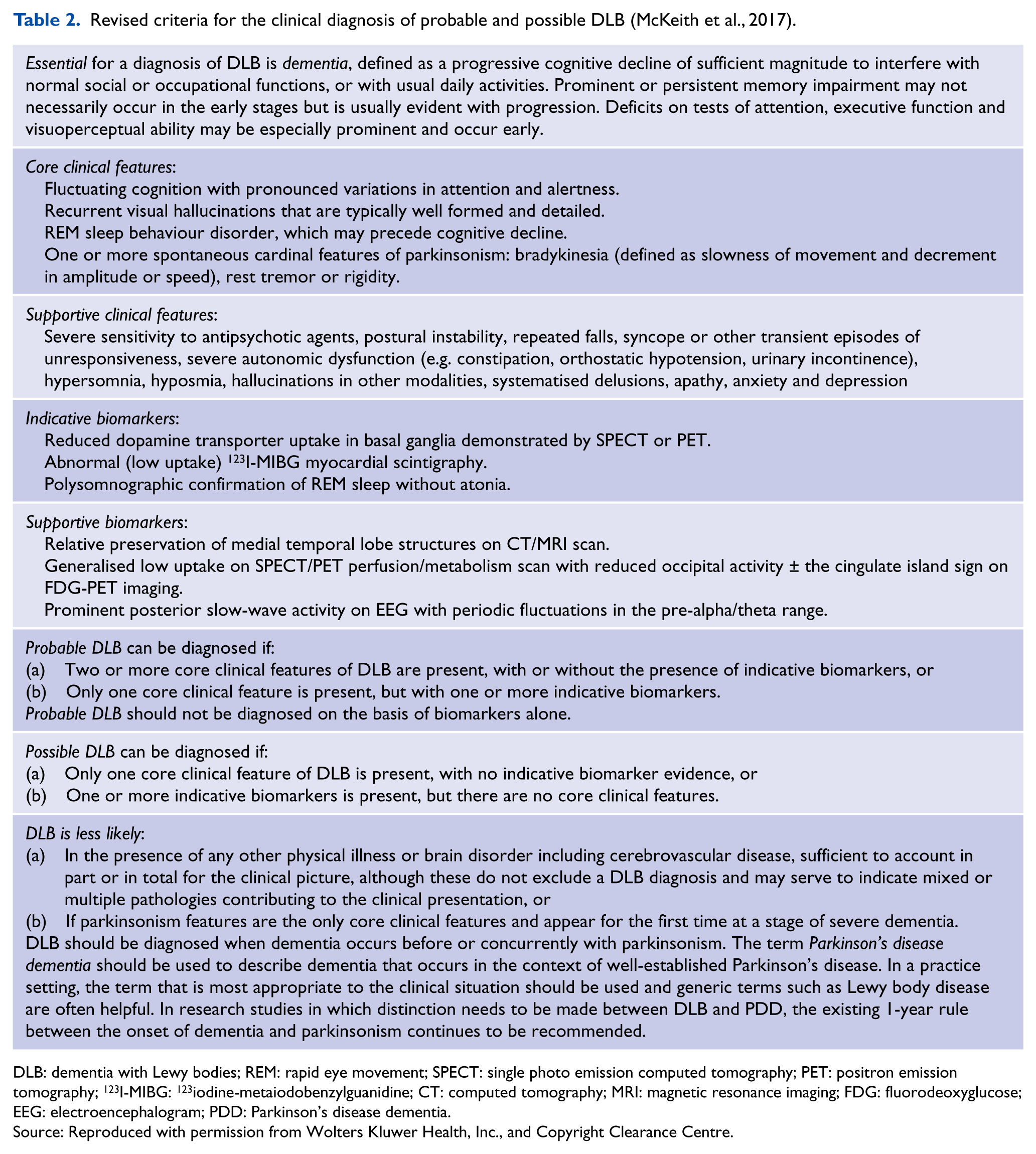
DLB: dementia with Lewy bodies; REM: rapid eye movement; SPECT: single photo emission computed tomography; PET: positron emission tomography; 123I-MIBG: 123iodine-metaiodobenzylguanidine; CT: computed tomography; MRI: magnetic resonance imaging; FDG: fluorodeoxyglucose; EEG: electroencephalogram; PDD: Parkinson’s disease dementia.
Source: Reproduced with permission from Wolters Kluwer Health, Inc., and Copyright Clearance Centre.
Specific assessment toolkits (https://doi.org/10.1002/gps.4948), aligned with the new consensus diagnostic criteria, have also been developed to facilitate the diagnostic process for DLB, especially for less experienced clinicians. These toolkits incorporate structured and validated questions and aim to assist clinicians in more efficiently detecting the core features of DLB in routine clinical practice (Thomas et al., 2018).
However, despite the availability of specific diagnostic criteria, the diagnosis of DLB remains a challenge for clinicians, particularly at early stages of disease, for patients with mixed AD and DLB pathologies as well as in non-expert centres. Reasons for difficulties making the diagnosis include underuse or lack of availability of biomarkers, underuse of possible DLB diagnosis, insufficient neuropsychological evaluation and the fact that atypical presentations are common.
DLB vs delirium
The challenge of differentiating between delirium and DLB is especially problematic and the interface merits special attention. This is because DLB and delirium share many similar features including severity of fluctuations, and persistent delirium lasting over 6 months in increasingly recognised (Meagher et al., 2012; Morandi et al., 2017). However, the pharmacological treatments are markedly different and therefore correctly detecting the diagnosis is essential for safe prescribing. Cholinesterase inhibitors have been shown to be harmful in patients with delirium but conversely are treatment for cognitive and non-cognitive symptoms of DLB (Stinton et al., 2015; Van Eijk et al., 2010). Whereas antipsychotics are avoided in patients with DLB, it can be used to treat distressing symptoms associated with delirium where risks are present (Meagher et al., 2017). To help differentiate DLB from persistent delirium, a collateral history in search of a clear insult is essential. Typically, persistent delirium, which may be misdiagnosed as DLB, is preceded by a severe insult, such as severe brain injury following a cardiac event, whereas DLB comes on insidiously. Equally when considering a diagnosis of DLB, it is prudent to always ask about the presence of early autonomic dysfunction or RBD in the history, these features being more suggestive of DLB than delirium.
The importance of making the correct diagnosis is further emphasised by the fact that patients with DLB are more ‘delirium prone’ than other dementias. Detection of delirium superimposed on dementia is therefore key, however limited by an absence of bedside tests for delirium superimposed on dementia and the impracticality of using biomarkers such as 123I-FP-CIT SPECT (DaTscan), if available (Morandi et al., 2017; Richardson et al., 2016).
Diagnostic biomarkers
Given the challenges in clinical diagnosis of DLB, the development of specific biomarkers is essential to help improve diagnostic accuracy, particularly in the earlier stages of disease. The consensus criteria have now described ‘indicative’ and ‘supportive’ biomarkers (McKeith et al., 2017).
Structural neuroimaging
CT (computed tomography) or MRI is usually the first choice of neuroimaging in evaluation of patients with cognitive concerns due to their wider availability. It can be useful in helping to establish the pattern of cerebral atrophy as well as to rule out any intracranial pathologies, e.g. brain tumour (Australian Clinical Practice Guidelines, 2016). Relative preservation of medial temporal lobe structures on CT or MRI is a relatively robust finding across imaging studies of DLB compared to AD and is a supportive biomarker of DLB (Watson et al., 2010). However, a proportion of people with DLB will have medial temporal lobe atrophy, which may reflect the contribution of AD-related pathology (Nedelska et al., 2015). One study with autopsy validation found that using MRI visual rating scale, medial temporal lobe atrophy differentiated AD from DLB with a sensitivity of 64% and specificity of 68% (Harper et al., 2016). In addition, there is generally less cortical grey matter atrophy in DLB than AD with greater subcortical involvement in DLB on structural imaging (Watson et al., 2016). The subcortical atrophy observed, however, does not account for the substantial functional changes seen using SPECT or PET imaging (Watson et al., 2010).
Functional imaging
Functional imaging using perfusion SPECT and fluorodeoxyglucose (FDG) PET highlight the characteristic patterns of cerebral blood flow and glucose metabolism seen in different types of neurodegenerative dementias. In DLB, more profound deficits are observed in the parieto-occipital lobes, while in AD, temporoparietal areas, in particular the medial temporal and posterior cingulate areas, are more commonly affected (O’Brien et al., 2014). Perfusion deficits and glucose hypometabolism in the occipital lobes on SPECT and FDG-PET imaging correlate with visual hallucinations and visuospatial deficit clinically observed in DLB patients (Hanyu et al., 2006). Occipital hypoperfusion on SPECT and occipital hypometabolism on PET were reported to have a sensitivity of 65% to 85% and 75% to 99%, and specificity of 72% to 87% and 71% to 93%, respectively, for distinguishing DLB from AD (Davison and O’Brien, 2014; Inui et al., 2014). When comparing SPECT to PET, previous systematic review and a subsequent head-to-head comparison study have both reported superiority of FDG-PET over perfusion SPECT, when differentiating DLB from AD (Davison and O’Brien, 2014; O’Brien et al., 2014). Relative preservation of posterior or mid-cingulate metabolism, compared with the parieto-occipital hypometabolism on FDG-PET, known as the cingulate island sign (CIS; see Figure 1), has been described with high specificity in DLB (Graff-Radford et al., 2014; Lim et al., 2009). Interestingly, CIS on PET has also been reported to correlate with lower tau burden, but not amyloid, in DLB at autopsy (Graff-Radford et al., 2014). However, the use of CIS as a marker of AD-related pathology and the influence of tau pathology in DLB are not well understood and require further investigation.
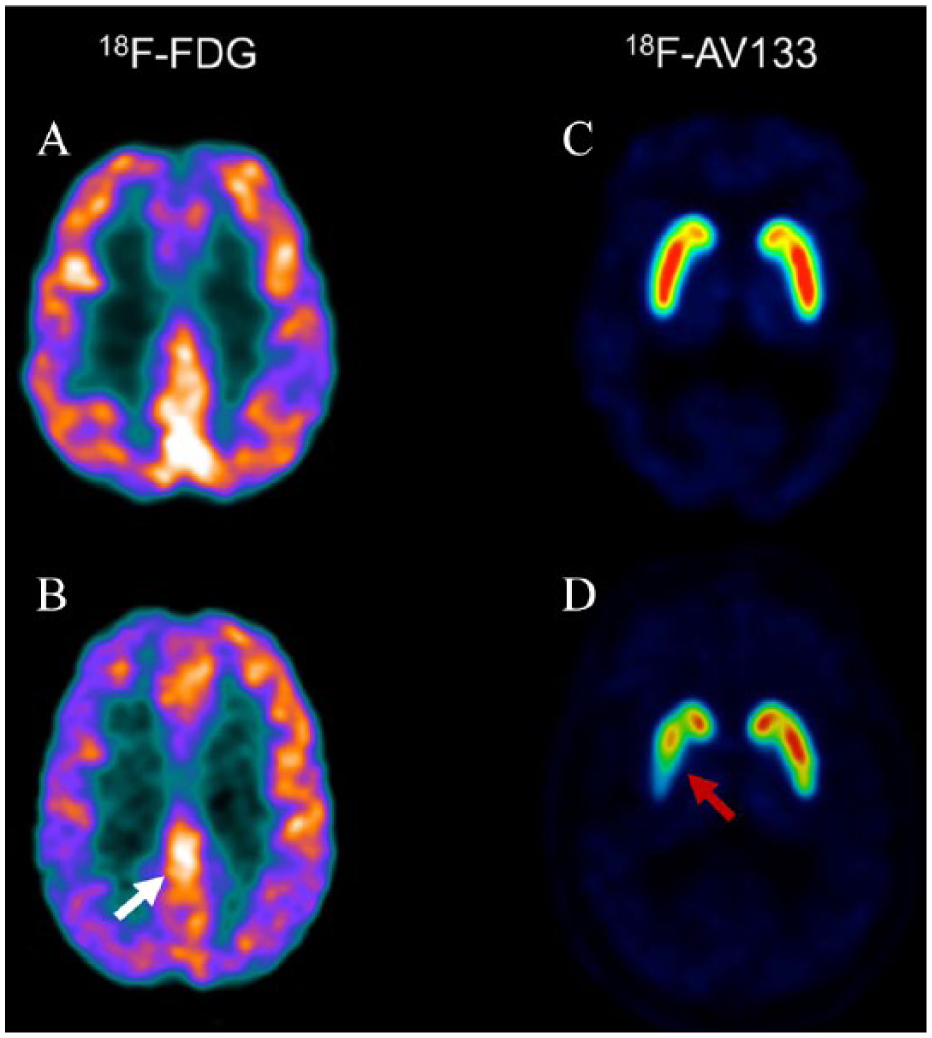
FDG-PET and VMAT2 ([18F]AV-133) PET scans of healthy control vs DLB. (A) FDG-PET of a healthy control; (B) FDG-PET for DLB showing cingulate island sign (CIS) (white arrow) and parieto-occipital hypometabolism, worse on the right than left; (C) VMAT2 PET of a healthy control; and (D) VMAT2 for DLB showing reduced uptake in the right caudate and putamen (red arrow) as well as moderate decrease in the tail of the left putamen.
Imaging the Dopamine Transporter
Neuroimaging studies using the tracer 123I-N-fluoropropyl-2b-carbomethoxy-3b-(4-iodophenyl) nortropane (123I-FP-CIT) to image the dopamine transporter (DaTscan) have demonstrated reduced uptake in the caudate and anterior and posterior putamen in people with DLB, compared to people with AD and healthy older controls (McKeith et al., 2007; O’Brien et al., 2004). It has also been subsequently validated in autopsy study which showed a sensitivity of 80% and specificity of 92% in diagnosis of DLB, more accurate than a clinical diagnosis (Thomas et al., 2017). However, it is important to note that a normal 123I-FP-CIT SPECT does not rule out DLB as patients may have Lewy pathology predominantly in the limbic and neocortical regions, highlighting the variable presentations common to this condition. Approximately 10% to 20% of people with DLB have normal DaTscan and often clinically have none-to-milder motor symptoms (Shimizu et al., 2017; Thomas et al., 2017). Despite DaTscan being a highly specific and relatively sensitive biomarker, it is not currently licensed for use in Australia. Even in countries where DaTscan is available, it is typically only used when there is diagnostic uncertainty and is limited to specialist centres. Similar to DaTscan, the evaluation of dopaminergic activity in the basal ganglia can also be achieved with PET scans targeting vesicular monoamine transporter type 2 (VMAT2). PET study using tracer such as 18F-labelled tetrabenazine derivative, 9-fluoropropyl-(+)-dihydrotetrabenazine ([18F]AV-133) has demonstrated reduced VMAT2 binding in the caudate, anterior and posterior putamen in DLB patients compared with AD, with a diagnostic accuracy of 95% (Villemagne et al., 2011; see Figure 1). VMAT2 PET scan is currently only available for research use in Australia.
Electroencephalogram
There is building evidence to support specific EEG (electroencephalogram) patterns as a DLB biomarker. Prominent posterior slow-wave EEG activity with periodic fluctuations in the pre-alpha/theta range is supportive of the diagnosis of DLB (McKeith et al., 2017). However, the current national dementia guideline in Australia does not recommend the routine use of EEG for investigation of dementia, and EEG should only be considered if delirium or Creutzfeldt-Jakob disease is suspected clinically (Australian Clinical Practice Guidelines, 2016).
Cerebrospinal fluid biomarkers
Many studies have investigated specific cerebrospinal fluid (CSF) biomarkers to distinguish LBD from other neurodegenerative dementias. Considering heterogeneity in study designs, a systematic review has reported a trend of lower CSF α-syn level in DLB compared to AD, without neuropathological confirmation (Lim et al., 2013). The utility of CSF α-syn as a potential diagnostic biomarker for LBDs is attractive but remains inconclusive and requires ongoing investigation.
The influence of Alzheimer’s pathology on DLB
Amyloid plaques
The burden of amyloid plaques, one of the pathological hallmarks of AD, has been studied in people with DLB. Around 50% of DLB cases have significant amyloid-beta (Aβ) deposition present on amyloid PET imaging, with the prevalence increasing with age (Donaghy et al., 2015; Petrou et al., 2015). The degree and distribution of amyloid deposition in DLB were also similar to that seen in AD, with increased binding in frontal, posterior cingulate and precuneus, temporoparietal region and the striatum (Donaghy et al., 2015). There is increasing evidence in neuroimaging and neuropathological studies suggesting that high amyloid load correlated with worse cognition and a more aggressive decline in DLB (Donaghy et al., 2015; Howlett et al., 2015). This warrants further investigation given the recent advances in anti-amyloid therapy trials which might benefit a specific subgroup of DLB patients with high amyloid burden.
Neurofibrillary tangles
Neurofibrillary tangles, formed by hyperphosphorylation and aggregation of tau proteins, another pathological hallmark of AD, are also a common co-pathology in DLB. Pathological total tau and phosphorylated tau levels have been detected in the CSF of approximately 28% and 25% of DLB patients, respectively (Skillback et al., 2015). Previous PET study using radioligand fluorine 18-labelled AV-1451 ([18F]AV-1451) showed greater cortical tau uptake in DLB compared to healthy controls, again signifying the co-occurrence of Alzheimer’s pathology in DLB (Kantarci et al., 2017). DLB patients with significant tau burden were found to be less likely to manifest the core clinical features of DLB, making clinical diagnoses even more challenging (Merdes et al., 2003).
Understanding the relationship between Lewy body and Alzheimer’s pathologies is crucial as it might potentially affect clinical diagnostic accuracy, has impact on disease progression and outcome and influences treatment options. Longitudinal studies, investigating environmental and genetic risk factors, and utilising novel biomarkers, would be helpful in providing insight into the role of co-morbid neuropathologies and in developing effective treatments.
Clinical management
The management of DLB is challenging and complex and a patient-centred multidisciplinary approach is needed given the disease heterogeneity (see Table 3). There are relatively few randomised-controlled trials that have looked at specific treatment strategies for DLB. Non-pharmacologic interventions such as psychoeducation of caregiver and patient, physical exercise and cognitive training are recommended in all types of dementia, including DLB. Physical exercise, in particular, has been reported to help in improving cognition, motor symptoms and mood (Morrin et al., 2018). Medication rationalisation should be actively considered in DLB patients, particularly reducing medications with known anticholinergic effects, such as oxybutynin. Fall risk minimisation and advance care planning should also form part of the comprehensive treatment in DLB.
Recommendations on clinical management of DLB.

DLB: dementia with Lewy bodies.
Caregiver education is especially important given the relatively higher levels of caregiver stress in DLB compared to other dementias. Earlier entry to residential care facilities in people with DLB is closely linked to the level of caregiver stress and the severity of neuropsychiatric symptoms (Rongve et al., 2014). Typically, education involves explaining the diagnosis, emphasising the aim being on symptomatic control and establishing priorities to guide treatment decisions. In people with DLB, the treatment of one symptom can potentially result in worsening of the other symptoms. For example, the use of levodopa to manage motor symptoms can exacerbate psychotic symptoms or hypotension, or the use of clonazepam for RBD can worsen confusion and increase fall risks. Therefore, ongoing management from a dementia specialist (aged psychiatrist, geriatrician or neurologist) can be beneficial to support these discussions and help guide management. Furthermore, linking patients and their families to important resources, such as the Lewy Body Society (https://www.lewybody.org/) and Dementia Australia (https://www.dementia.org.au/), may be beneficial.
Cognitive symptoms
Most of the pharmacologic treatment options available for DLB target symptoms and improve quality of life, rather than prevent or slow disease progression. Cholinesterase inhibitors, both donepezil and rivastigmine, in DLB have been studied in a number of randomised-controlled trials which showed improvements in cognition (approximately two-point increase in MMSE score), neuropsychiatric symptoms and activities of daily living, benefits which might be maintained for up to 52 weeks (Aarsland et al., 2012; Ikeda et al., 2013; Matsunaga et al., 2015; Mori et al., 2012; Stinton et al., 2015). The benefit of cholinesterase inhibitor on cognitive function in DLB was reported to be greater than in AD (Aarsland et al., 2012; Ikeda et al., 2013). This is likely to be attributed to the more extensive cholinergic deficits observed in DLB compared to AD (Shimada et al., 2015). While cholinesterase inhibitors have been found to be effective in DLB, they are not licensed under the Pharmaceutical Benefits Scheme (PBS) for DLB in Australia. Currently, cholinesterase inhibitors are accessible by DLB patients on private prescription, i.e. at higher costs, which might potentially impact medication compliance. National guidelines for dementia in Australia and the United Kingdom both recommend that cholinesterase inhibitors should be considered routinely in DLB patients (Australian Clinical Practice Guidelines, 2016; The National Institute for Health and Care Excellence, 2018). Furthermore, extra caution is recommended when discontinuing cholinesterase inhibitor in DLB as sudden withdrawal can be detrimental resulting in marked deterioration in cognition and behavioural symptoms (Minett et al., 2003). For memantine trials, studies have generally reported an improvement in clinical global impression of change, but no significant changes in cognitive measures, compared to placebo (Aarsland et al., 2009; Emre et al., 2010). The efficacy of memantine in DLB is less clear, though increasingly memantine is used in addition to a cholinesterase inhibitor in clinical practice (Aarsland et al., 2012; Stinton et al., 2015).
Neuropsychiatric symptoms
The first-line treatment of neuropsychiatric symptoms in dementia should be non-pharmacologic interventions, such as caregiver training, physical activities, social stimulation and environmental modification (Soril et al., 2014). Generally, a safe, constant and familiar environment with orientation cues and appropriate level of stimulation is recommended. Identification and treatment of potentially reversible triggers (e.g. pain, hunger, sensory impairment) are also crucial but often overlooked. Many non-randomised trials evaluating non-pharmacologic therapies have included patients with all types of dementia, including DLB, and found positive effects on neuropsychiatric symptoms (Morrin et al., 2018). In Australia, services such as Dementia Behaviour Management Advisory Service (DBMAS) and Severe Behaviour Response Teams (SBRTs) (https://www.dementia.com.au/) are available to provide individualised assessment, support and advice focusing on non-pharmacologic interventions, to people and caregivers living with dementia.
Antipsychotic medications should be avoided in DLB patients due to the potential for severe neuroleptic sensitivity reactions. This does not merely refer to worsening of parkinsonism, but profound changes in conscious state which can be fatal and occur in around 50% of people with DLB (Aarsland et al., 2005; Ballard et al., 1998). These reactions can occur with both typical and atypical antipsychotics, and are not dose-related (Ballard et al., 1998). However, the absence of neuroleptic sensitivity does not exclude DLB. When indicated clinically, antipsychotics should only be prescribed under the supervision of a dementia specialist, utilising atypical agents at the lowest dose possible. Regardless, any use of antipsychotic medications warrants regular reviews with a careful plan for cessation as it is unlikely to yield any long-term benefit and is associated with an increased risk of cerebrovascular events (Mittal et al., 2011). Furthermore, the more disruptive behaviours such as constant pacing, wandering and vocalisation seem to be least responsive to antipsychotics. Despite limited efficacy, quetiapine, an atypical antipsychotic, seems to be the generally preferred option by clinicians in severe neuropsychiatric disturbances due to the perceived less risk of parkinsonism (Aarsland et al., 2012; Kurlan et al., 2007; Stinton et al., 2015). Currently, risperidone is the only antipsychotic agents licensed under PBS in Australia for treatment of moderate-to-severe behavioural symptoms of dementia.
Cholinesterase inhibitors, on the other hand, appear to be an effective and more favoured option in treatment of neuropsychiatric symptoms in DLB (Aarsland et al., 2012; Matsunaga et al., 2015; Mori et al., 2012). Donepezil, in particular, may be beneficial for patients experiencing hallucinations or delusions (Stinton et al., 2015). One longitudinal study also found that the use of cholinesterase inhibitors in DLB potentially delays admission to residential care facilities (Rongve et al., 2014).
Depression and anxiety are frequently associated with DLB, but there are very few studies assessing the effectiveness of antidepressant medications in DLB (Chiu et al., 2017). One uncontrolled trial evaluated the use of citalopram and found no effect on mood or neuropsychiatric symptoms, complicated with poor tolerability of the medication (Culo et al., 2010). Electroconvulsive therapy (ECT) was reported to improve depression in people with LBD in a systematic review, but the long-term cognitive effect of ECT is not clear (Morrin et al., 2018). It might be considered in cases of severe depression (not responding to antidepressants) in consultation with aged psychiatrists.
Motor symptoms
Physical therapy, despite the lack of robust evidence, has been reported to improve motor symptoms and balance in people with LBD (Morrin et al., 2018). Fall prevention strategies should be considered in all patients with motor parkinsonism. Levodopa is the mainstay of PD treatment. Levodopa has been shown to benefit around 30% of DLB patients with motor symptoms but the response is typically less pronounced compared to PD, with an increased risk of psychosis, including hallucinations (Aarsland et al., 2012; Goldman et al., 2008; Lucetti et al., 2010). Therefore, the use of levodopa in DLB patients involves careful titration and monitoring. Other agents such as dopamine agonists can substantially worsen psychotic symptoms and exacerbate confusion, and should be avoided (Goldman et al., 2008).
Sleep
Treatment of RBD is not always needed. However, when indicated due to excessive daytime sleepiness or risk of injury to patient and/or bedpartner, melatonin is an effective and generally well-tolerated option (Aarsland et al., 2012; Jung and St Louis, 2016; Medeiros et al., 2007). Clonazepam and pramipexole are second-line treatment options for RBD, but the potential side effects of drowsiness and confusion limit their use in DLB (Aarsland et al., 2012). Cholinesterase inhibitors, particularly rivastigmine, as well as memantine may also help (Jung and St Louis, 2016; Larsson et al., 2010).
Conclusion
DLB is a common form of dementia with rates as high as 1 in 7. Despite being a debilitating condition with many negative consequences, it is often under-detected and poorly managed in clinical practice. The new diagnostic criteria promote the importance of diagnostic biomarkers and REM sleep behaviour disorder as a core clinical feature, which can support clinical practice and improve case detection. Clinical management of DLB is complex and requires an individualised multidisciplinary approach including support and education for patients and their caregivers.
Footnotes
Acknowledgements
We wish to thank Associate Professor Victor Villemagne for providing the image.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
K.S.C. is supported by Yulgilbar Alzheimer’s Research Program and Research Training Program Scholarship by the Australian Commonwealth Government. R.W. is supported by Yulgilbar Alzheimer’s Research Program.
