Abstract
Objectives:
Schizophrenia is a serious and chronic mental illness known to have broad ranging impacts for individuals across the lifespan, yet research on the disease in older adults is sparse. This study provides a profile of the sociodemographic, environmental and diagnostic characteristics of older community residents with schizophrenia using a national database.
Methods:
A cross-sectional sample of individuals who underwent community needs assessment using the standardised Home Care International Residential Assessment Instrument between 1 September 2012 and 31 January 2016 was utilised. Sociodemographic, diagnostic, and social and environmental variables were measured for individuals with a diagnosis of schizophrenia and compared to those without a diagnosis of schizophrenia. Statistical investigations employed bivariable and multivariable logistic regression models.
Results:
A total sample of 71,859 was eligible and 517 (0.7%) had a diagnosis of schizophrenia. The majority of the sociodemographic variables were statistically associated with schizophrenia in the adjusted analysis, except for ethnicity (p = 0.35). Nearly all the measured social and environmental variables were adversely associated with having a diagnosis of schizophrenia, such as living in squalid conditions (adjusted odds ratio = 2.16; 95% confidence interval = [1.42, 3.28]). Participants with schizophrenia were significantly more likely to be diagnosed with all assessed psychiatric comorbidities (p < 0.001) and diabetes mellitus (p = 0.002), whereas coronary heart disease (p = 0.001) and other physical comorbidities (p = 0.001) were found at significantly lower rates.
Conclusion:
The profile of schizophrenia found here suggests some subtle differences in the demographic profile and distribution of medical comorbidities in the older population with schizophrenia. The results also suggest that this group continues to experience social disadvantage into old age. This requires the attention of policy-makers to ensure that services are tailored to the high social needs of these individuals.
Keywords
Introduction
Schizophrenia is a serious and often chronic mental illness with broad ranging impacts for individuals across the lifespan into old age. The prevalence of the disorder in the general adult population has been thoroughly investigated with a recent review indicating a lifetime prevalence estimate of 0.48% (Simeone et al., 2015). Although this rate is relatively small, the personal, social and clinical burden of the disease is significant, resulting in considerable research interest. However, much less attention has been paid to the prevalence and characteristics of schizophrenia in older adults.
The literature that is available suggests that older adults with schizophrenia have complex needs and remain impaired in old age when compared to their healthy counterparts (Folsom et al., 2009; Jeste et al., 2003, 2011). This includes factors such as the ongoing presence of psychotic symptoms (Jeste et al., 2003; Karim et al., 2005), high rates of comorbid depression (Diwan et al., 2007; Karim et al., 2005) and severe side-effects from long-term anti-psychotic use (Folsom et al., 2006; Jeste and Maglione, 2013; Karim et al., 2005). Moreover, it has been found that older adults with schizophrenia have a poorer quality of life (Cohen et al., 2008) and are less likely to achieve ‘successful aging’; a state of positive health and well-being (Ibrahim et al., 2010). This suggests that older adults with schizophrenia have high clinical and social needs, but further investigation is required to provide an adequate knowledge base to inform how these needs can be addressed.
The Home Care International Residential Assessment Instrument (interRAI-HC) is a comprehensive, evidence-based standardised assessment tool used by trained health professionals in New Zealand to determine individual need and allocate resource for older aged adults seeking publicly funded residential care or long-term community services. Since 2012, all people seeking these services in New Zealand are required to undergo assessment using the interRAI-HC (Schluter et al., 2016). Although the primary function of interRAI-HC is to establish needs and resources, permission to use the information for service planning and research purposes is explicitly obtained from participants (Schluter et al., 2016). The interRAI-HC elicits information pertaining to multiple domains such as biomedical, psychological, and social and environmental. The interRAI-HC assessed population is considerable in size as it encompasses over 10% of the New Zealand population aged ⩾ 65 years (Schluter et al., 2016). The sample consists of individuals that are seeking or receiving public health care support services, thus providing a sample that is representative of older adults that are more likely to access secondary services and who have greater physical, mental and social needs compared to the general population. This provides valuable information that is highly relevant to the planning and provision of health care services.
Using this national database, this study aims to first provide a sociodemographic, environmental and comorbid diagnostic profile of older adults with schizophrenia who access community health service support and, second, estimate the differences in these measures between those with a diagnosis of schizophrenia and those without. These objectives are a starting point in improving our understanding of the characteristics of older adults living with schizophrenia, which is fundamental knowledge that is currently lacking in New Zealand and internationally.
Methods
Study design
A cross-sectional study from a continuously recruited national cohort.
Participants
Participants were community-based people aged ⩾ 65 years with an interRAI-HC assessment undertaken between 1 September 2012 and 31 January 2016, inclusive of who provided consent to their data being used for planning and research purposes.
Instrument and primary measure
The interRAI-HC 9.1 instrument (©interRAI Corporation, Washington, DC, USA, 1994–2009), modified with permission for New Zealand, is used under licence to the Ministry of Health (www.interrai.co.nz). It is constructed of 236 questions divided over 20 distinct domains and produces internationally valid scales (Hirdes et al., 2008; Schluter et al., 2016). The domains used here consisted of (1) identification information, (2) psychosocial well-being, (3) disease diagnosis and (4) environmental assessment. The disease diagnosis domain captures schizophrenia, where a total of 21 distinct conditions are identified and labelled. The interviewer is prompted with the following preamble:
Disease/infection that doctor has indicated is present and affects client’s status, requires treatment, or symptom management. Also include if disease is monitored by a home care professional or is the reason for a hospitalization in last 90 days (or since last assessment if less than 90 days).
Three response options are available for diagnostic classification and include: (0) not present; (1) primary diagnosis/diagnosis for current stay; (2) diagnosis present, receiving active treatment; and (3) diagnosis present, monitored but no active treatment. For the purposes of this paper, schizophrenia was dichotomised into not diagnosed (code 0) and diagnosed (codes 1, 2 and 3) categories. The response options for all current and historical conditions, including schizophrenia, were only recorded if they were also listed in the participants’ medical records.
Sociodemographic characteristics and social and environmental measures
Age was derived from the difference between date of birth and date of interview. Sex response options included male, female, unknown and indeterminate; responses to the last two options were set to missing. A maximum of three ethnic groups could be self-identified by participants. Here, ethnicity was coded using a single-priority classification for those with multiple identifications (Allan, 2001), with Māori (indigenous people of New Zealand) having priority coding, followed by Pasifika, Asian and then NZ European/other. Residential living status at the time of assessment was classified as private (private home/apartment/rented room/board and care), assisted living (assisted living or semi-independent living/mental health residence/group home for person with physical disability), hospital (psychiatric hospital or unit/rehabilitation hospital or unit/acute care hospital) and other. It should be highlighted that, although some participants were in hospital for their interRAI-HC assessment, they usually (or until recently) resided in the community. As such, their assessment was conducted with the knowledge that they might require relocating to a residential service. Counting the indications of the 21 conditions (excluding schizophrenia) itemised in the disease diagnoses section established the number of comorbid conditions. These comorbidities were divided into psychiatric and non-psychiatric variables in a secondary analysis. Six questions pertaining to social relationships assessed the positive and negative social interactions the participants had with family and friends. The response options to these questions were: never, more than 30 days ago, 8–30 days ago, 4–7 days ago, in last 3 days and unable to determine. For environmental assessment questions, assessors coded (No or Yes) to five questions that made the home environment hazardous or uninhabitable. For the final environmental question, participants were asked to code (No or Yes) to the statement that ‘Because of limited funds, during the last 30 days made trade-offs among purchasing any of the following: adequate food, shelter, clothing; prescribed medications; sufficient home heat or cooling; necessary health care’.
Procedure
A comprehensive description of the interRAI-HC assessment instrument and procedure within New Zealand has been reported previously (Schluter et al., 2016). In summary, older people needing publicly funded long-term community services or age residential care undergo needs assessment using the interRAI-HC instrument. Individuals are referred for needs assessment by a health professional, and the assessments are conducted by trained assessors using the standardised interRAI-HC tool. The assessors, who are most commonly social workers or nurses, undertake a mandatory 2-day training programme and regular competency review (Schluter et al., 2016). Assessments take place in the older persons’ home environments and produce individualised care plans that inform the allocation of support services. All data are directly entered into the electronic interRAI-HC database, maintained by New Zealand’s Technical Advisory Services (TAS; http://centraltas.co.nz). With approval, consented data are released by TAS, through the Ministry of Health. The national interRAI-HC data set has been assessed for quality and has been shown to have good psychometric properties and high data reliability and validity (Schluter et al., 2016). Missing data were found to be rare, data matching with available duplicate assessments was adequate (2% error in fixed variables such as age, sex and ethnicity) and a high rate of continuity with the Ministry of Health’s mortality data was achieved (99.8%) (Schluter et al., 2016).
Statistical analysis
The STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) guidelines (www.strobe-statement.org) informed the analyses. Where an individual had more than one interRAI-HC assessment during the study period, only the first assessment was utilised here. Descriptive statistics of all variables of interest were obtained and reported. Fisher’s exact test was employed for categorical comparisons between those with and without a schizophrenia diagnosis. The associations between schizophrenia and the variables of interests were tested using bivariable and multivariable logistic regression models and estimated as odds ratios (ORs) and adjusted ORs (AORs), respectively, together with associated 95% confidence intervals (CIs). The Hosmer–Lemeshow goodness-of-fit test was used to check model assumptions. Only when goodness-of-fit was rejected would the Hosmer–Lemeshow test be explicitly reported. For the social and environmental variables, Pearson correlations were calculated. All analyses were executed using Stata SE version 14.1 (StataCorp, College Station, TX, USA), and α = 0.05 defined statistical significance.
Ethics
This study was approved by the Ministry of Health’s Health and Disability Ethics Committees (14/STH/140) and only includes de-identified data for those consenting to its use for planning and research purposes.
Results
Describing the sample
A total of 72,193 interRAI-HC assessments were available for individuals aged ⩾ 65 years. Of these, 318 (0.4%) had invalid encrypted National Health Index (NHI) numbers, a unique identifier that is assigned to every person who uses health and disability support services in New Zealand and were excluded. A further 16 repeat assessments were removed, leaving a final eligible sample size of 71,859 people. The average age of this eligible sample was 82.7 years (range: 65–106 years), with a larger proportion of female (61.0%), and the vast majority being NZ European (88.5%) – with 501 (0.7%) having their ethnicity classified as ‘Other’; see Table 1. The eligible sample was comparatively older, comprised a greater percentage of female, and had a reduced number of people with Asian ethnic identification, when compared with the New Zealand population aged ⩾ 65 years usually resident at the 2013 Census. The proportion of Māori in the inter-RAI sample (5.4%) was comparable to the New Zealand Māori population (5.6%).
Demographic distribution of eligible participants (N = 71,859) compared with the New Zealand population aged 65+ years usually resident at the 2013 Census (N = 607,035).
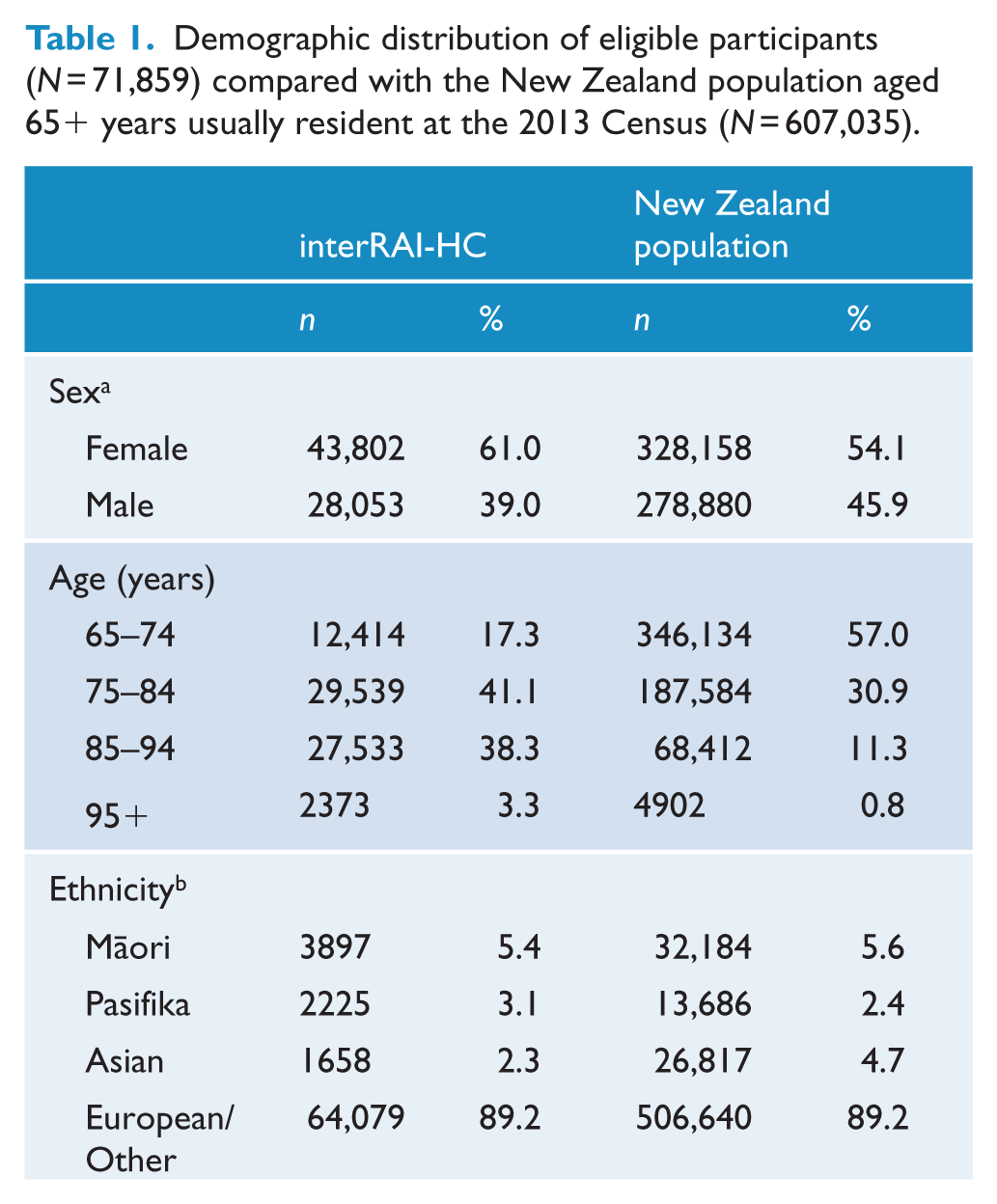
Four people had indeterminate sex values recorded in the eligible interRAI sample.
27,705 (4.6%) people aged 65+ years had unstated ethnicity in the 2013 Census.
Overall schizophrenia prevalence and associations with sociodemographic variables
A total of 517 (0.7%) participants had a schizophrenia diagnosis; 163 (31.5%) with primary diagnosis/diagnosis for current stay; 296 (57.3%) with diagnosis present, receiving active treatment; and 58 (11.2%) participants with diagnosis present, monitored, but receiving no active treatment. Table 2 includes the distributions and logistic regression estimates for sociodemographic variables and the number of comorbidities. All variables were statistically associated with schizophrenia in crude analyses, except for sex (p = 0.87) and number of comorbid diagnoses (p = 0.85), and in adjusted analyses, except for ethnicity (p = 0.35) and number of comorbid diagnoses (p = 0.74). Never married participants, those living alone or with non-relatives, and those living in assisted living places of residence had a comparatively higher rate of schizophrenia than those without a schizophrenia diagnosis. Pearson’s estimated correlation between these variables ranged from a maximum of r = 0.63 (between ‘marital status’ and ‘living arrangements’) and a minimum of r = –0.16 (between ‘ethnicity’ and ‘age group’).
Distribution, prevalence, odds ratios (ORs) and adjusted ORs (AORs) together with associated 95% CIs of schizophrenia within the sample (N = 71,859) by sociodemographic characteristics.
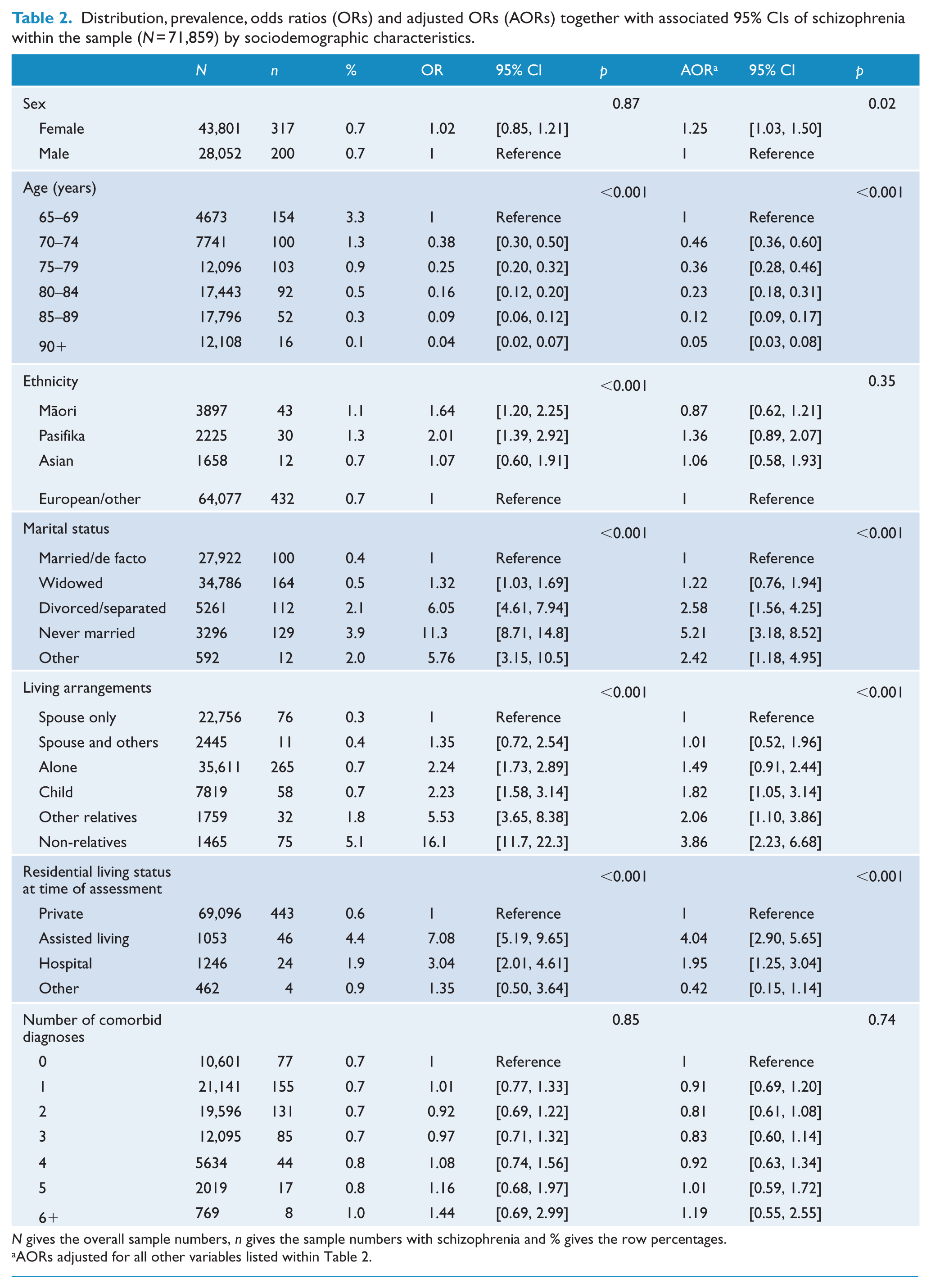
N gives the overall sample numbers, n gives the sample numbers with schizophrenia and % gives the row percentages.
AORs adjusted for all other variables listed within Table 2.
Schizophrenia associations and other comorbidities
While the overall number of comorbid diagnoses were not different between those with and without a schizophrenia diagnosis (p = 0.74), the distribution of individual comorbidities is given in Figure 1. The most common comorbid conditions found in the overall sample were coronary heart disease (31.8%) and diabetes mellitus (20.6%). Alzheimer’s disease and other dementias were diagnosed for 7.6% and 14.3%, respectively. Participants with a schizophrenia diagnosis were significantly more likely to be diagnosed with all psychiatric comorbidities (anxiety, bipolar disorder and depression) than their reference category counterparts (all Fisher’s exact tests p < 0.001). A higher rate of comorbid diabetes mellitus (Fisher’s exact test p = 0.002) was also found among those with schizophrenia compared to those without schizophrenia, whereas comorbid diagnoses of Alzheimer’s disease (Fisher’s exact test p < 0.001), coronary heart disease (Fisher’s exact test p < 0.001), congestive heart failure (Fisher’s exact test p < 0.001), cancer (Fisher’s exact test p = 0.002), other fractures (not hip) during the last 30 days (Fisher’s exact test p = 0.03) and stroke/cerebrovascular accident (CVA) (Fisher’s exact test p = 0.048) were found at a significantly lower rate for those with schizophrenia.
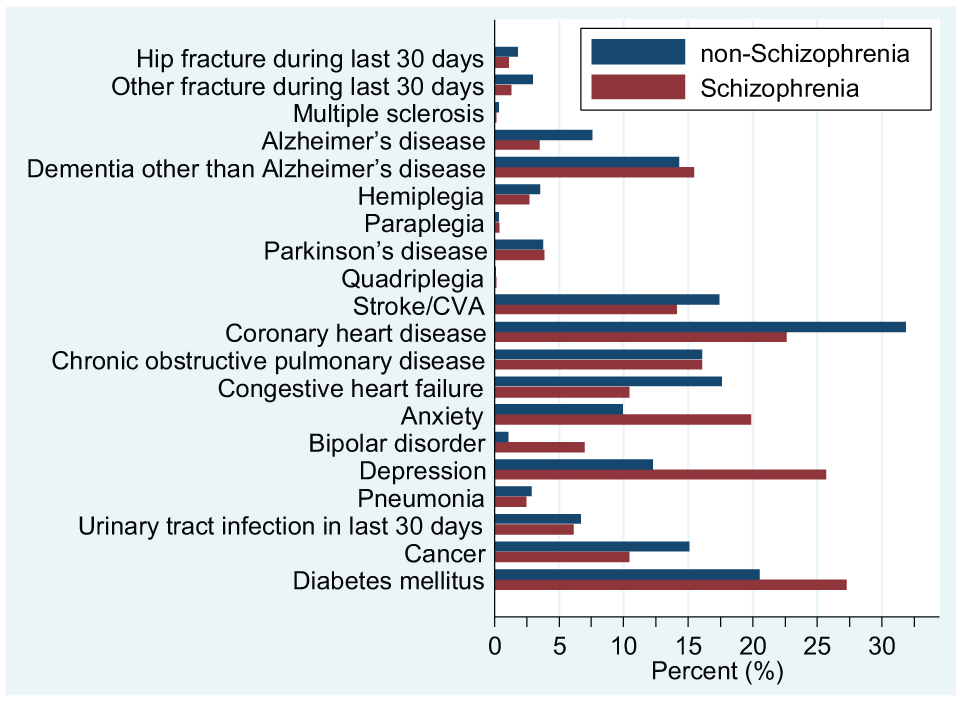
Distribution of individual comorbidities between participants with and without a diagnosis of schizophrenia.
In the secondary analysis, partitioning comorbidities into psychiatric and non-psychiatric conditions, 13,247 (18.4%) and 58,911 (82.0%) participants were indicated, respectively, of whom 10,900 (15.2%) were indicated for both. A higher percentage of psychiatric comorbidity was established for participants with schizophrenia (38.7%) compared to those without the diagnosis (18.3%), alongside a ratio that was significantly higher (AOR = 2.13; 95% CI = [1.75, 2.60]; p < 0.001). Conversely, non-psychiatric comorbidities were identified for 398 (77.0%) participants with schizophrenia compared to 58,511 (82.0%) without schizophrenia (AOR = 0.69; 95% CI = [0.56, 0.85]; p = 0.001), the ratio indicated a significantly lower rate for participants with schizophrenia than that of participants without schizophrenia.
Schizophrenia associations with social and environmental conditions
Social relationship and environmental associations with schizophrenia are presented in Table 3. All considered variables were significantly related to schizophrenia in crude analyses (except for the environmental assessment variable ‘limited access to home or rooms in home’ p = 0.62) and adjusted analyses (except for environmental assessment variables ‘disrepair of the home’ p = 0.10, and ‘limited access to home or rooms in home’ p = 0.29). A moderate, yet significant number of participants reported being neglected, abused or mistreated – 988 (1.4%), with an additional 2232 (3.1%) responses unable to be determined. Those with schizophrenia were more likely to report such mistreatment or not respond to the question. Having a diagnosis of schizophrenia was also associated with other negative social interactions such as conflict or anger with family and friends, or being fearful of a family member or close acquaintance. In terms of the environmental assessment variables, a range of negative characteristics were observed, from access limitations reported by 5850 (8.1%) participants to living in squalid conditions reported by 924 (1.3%). People with schizophrenia were more likely to be living in these squalid conditions (AOR = 2.16; 95% CI = [1.42, 3.28]) and have inadequate heating or cooling (AOR = 2.02; 95% CI = [1.43, 2.87]).
Distribution, prevalence, odds ratios (ORs) and adjusted ORs (AORs) together with associated 95% CIs of schizophrenia within the sample (N = 71,859) for social relationship and environmental association variables.
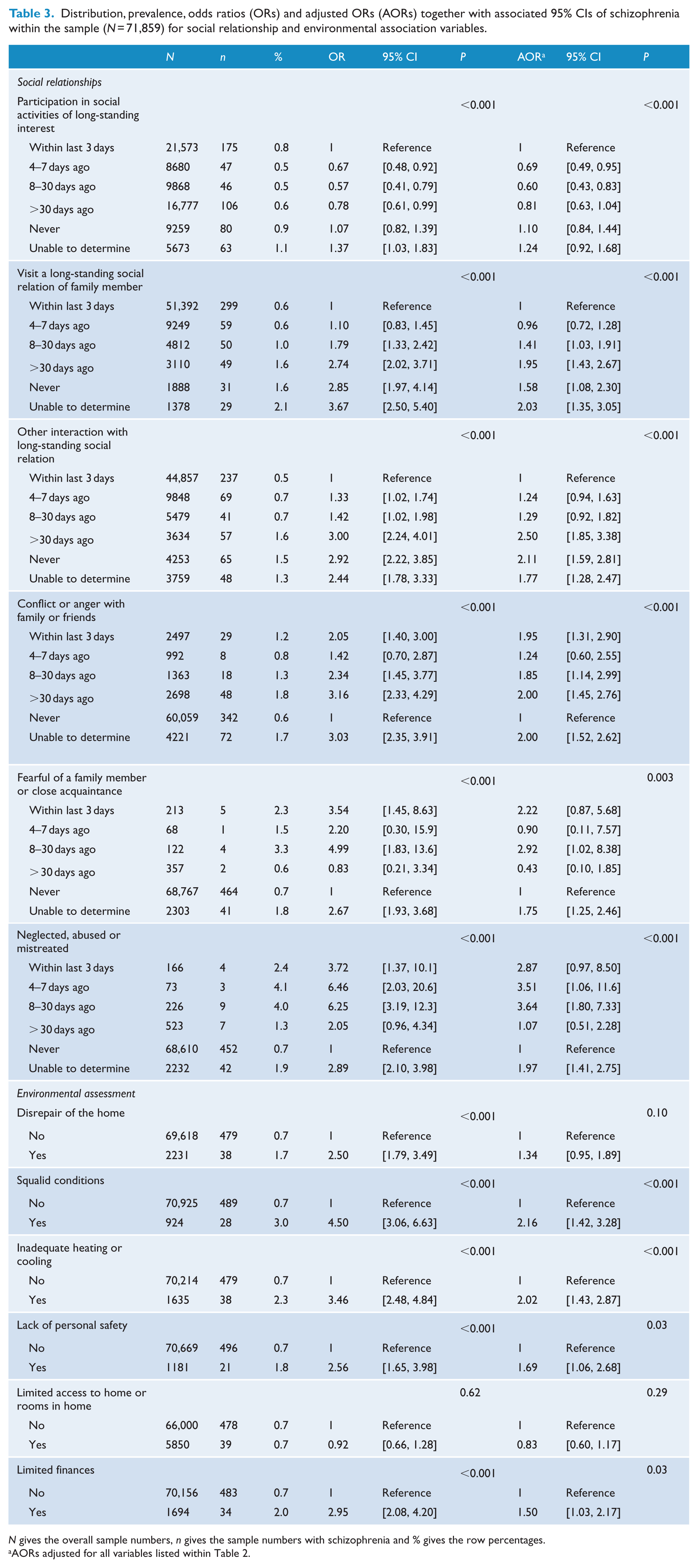
N gives the overall sample numbers, n gives the sample numbers with schizophrenia and % gives the row percentages.
AORs adjusted for all variables listed within Table 2.
Pearson correlation tests indicated a degree of inter-correlation between the social and environmental assessment variables presented in Table 3. For the social relationship variables, a strong correlation was established between the variables ‘fearful of a family member or close acquaintance’ and ‘neglected, abused, or mistreated’ (r = 0.75), alongside a moderate association between ‘conflict or anger with family or friends’ and ‘fearful of a family member or close acquaintance’ (r = 0.54). The correlations concerning ‘conflict or anger with family or friends’ and ‘neglected, abused, or mistreated’ (r = 0.48), and ‘participation in social activities of long-standing interest’ and ‘visit a long-standing social relation of family member’ (r = 0.23) were weak. All other social variable correlations were between 0.08 and 0.11. For the environmental assessment variables, the maximum estimated Pearson correlation occurred between ‘disrepair of the home’ and ‘squalid conditions’ (r = 0.40), followed by ‘disrepair of the home’ and ‘inadequate heating or cooling’ (r = 0.31) and to a minor extent between ‘squalid conditions’ and ‘inadequate heating or cooling’ (r = 0.25). All other correlations for the environmental variables were between 0.05 and 0.19.
Discussion
Our findings suggest the ongoing presence of social and environmental deficits in older age schizophrenia, alongside some subtle differences to participants without schizophrenia in the demographic profile and distribution of medical comorbidities. The individuals with schizophrenia were also more likely to report negative social interactions and undesirable living conditions. The distribution of ethnicity was not found to be significantly different for individuals with schizophrenia in the adjusted analysis, which was an unexpected finding given the high rate of schizophrenia known to effect the Māori population in New Zealand (Kake et al., 2008). An increased rate of psychiatric comorbidity and diabetes mellitus was also found, while a lower rate of all other physical comorbidities including chronic heart disease and congestive heart failure was established in the group with schizophrenia.
Although there was a small degree of correlation, a number of the social and environmental variables were detrimentally associated with schizophrenia, such as higher levels of conflict and anger with friends and relatives, and having inadequate funds for the necessities of living. The 2010 Australian Survey of High Impact Psychosis (SHIP) identified similar social disadvantage in the adult population (18–64 years) of people with schizophrenia (Morgan et al., 2017). This nationally representative data identified a number of significant social challenges for people with psychotic illnesses including high rates of unemployment, poverty, inadequate and unstable housing, increased loneliness and poor social functioning. Our findings suggest that these disparities persist into old age. Loneliness has received particular attention in the schizophrenia literature given it has wide ranging consequences for individuals experiencing the illness including a detrimental relationship with metabolic syndrome (Badcock et al., 2018; Trémeau et al., 2016), drug abuse/dependence (Trémeau et al., 2016), cognitive functioning (Badcock et al., 2015) and, in older adults, quality of life (Cohen et al., 2008). The participants with schizophrenia also had a higher likelihood of living alone, in squalid conditions, or in assisted living. Housing difficulties are known to disproportionately affect people with schizophrenia including issues with homelessness and inappropriate supported accommodation options (Harvey et al., 2012; Morgan et al., 2017). The latter is further compounded in old age by the limited availability of suitable housing options and additional concerns regarding the quality of care received by individuals with severe mental illness in nursing homes (Fullerton et al., 2009).
Theoretical and empirical explanations for social and environmental disadvantages in schizophrenia have taken many courses. Individuals with schizophrenia are known to have impairments in social cognition such as emotional processing, social perception and theory of mind, which has been linked to the functional status of those affected (Sergi et al., 2006). A further consideration has been the influence of the social environment on the development of the disorder (Allardyce and Boydell, 2006; Muntaner et al., 2004), as well as the impact the disorder has on individuals’ social status and living conditions. This includes circumstances such as individuals with schizophrenia aggregating in adverse social environments (Allardyce and Boydell, 2006), a decreased participation in the socio-economic arena limiting asset gains throughout the lifespan (Muntaner et al., 2004), and poverty and social disability resulting in a lack of suitable and stable housing (Galletly et al., 2016).
From this study, we are unable to determine if diminished supports and adverse environmental conditions were a consequence of the illness, but what these findings do reinforce is that older persons with schizophrenia require a high level of psychosocial support as do younger people with schizophrenia. Current treatment recommendations suggest that in addition to cautious anti-psychotic use, older persons with schizophrenia require access to quality psychological and psychosocial interventions (Galletly et al., 2016; Jeste and Maglione, 2013; McKay et al., 2015). It is likely that the majority of this sample had a long disease history, yet the social and environmental deficits remained. This suggests that current services may not be sufficiently catering to the psychosocial needs of these patients earlier in treatment.
When adjusted for sociodemographic profiles, no significant ethnic differences were found, despite the overall percentage of Māori in our sample being consistent with the percentage of Māori in the New Zealand population for a similar age group. The lack of ethnic variation in the adjusted analysis was unexpected given other research findings that indicate a significantly higher prevalence of schizophrenia among Māori compared to non-Māori in the adult population (Ellis and Collings, 1997; Kake et al., 2008), and an over-representation of Māori in youth psychosis and schizotypal cohorts (Linscott et al., 2006; Turner et al., 2006). It has not previously been investigated whether these differences persist into older age. It may be that our results are related to the InterRAI sample characteristics, namely, the older distribution and the potential for ethnic differences in accessing publicly funded support. Survivor bias could also be a factor given the high rate of mortality known to affect Māori (Phillips et al., 2017).
Participants with schizophrenia were less likely to be diagnosed with the majority of physical comorbidities with a clear exception for diabetes mellitus. There is a paucity of research that has assessed medical comorbidity in the elderly population with schizophrenia, and what is available is heterogeneous due to the variability of the study designs and geographic contexts. For example, a recent Danish study found equal physical comorbidity among older adults with schizophrenia compared to their peers in the general population (Brink et al., 2017), yet an American investigation found higher rates of congestive heart failure, chronic obstructive pulmonary disease and hypothyroidism in older adults with schizophrenia (Hendrie et al., 2014). The reduced rate of physical comorbidities found in our schizophrenia group could be due to sample bias, whereby those with schizophrenia were disproportionately more likely to require assessment for complex needs despite having fewer physical comorbidities.
Schizophrenia is also associated with a significantly reduced life expectancy with a recent systematic review establishing a weighted average of 14.5 years of potential life lost and an average life expectancy of 64.7 years (Hjorthøj et al., 2017). The confounding effects of psychiatric treatment including the metabolic side effects of second-generation anti-psychotics, in addition to lifestyle factors, are thought to play a significant role (Hjorthøj et al., 2017; Jeste et al., 2011). Surprisingly, a lower rate of coronary heart disease (CHD) was found in the schizophrenia group, which is in contrast to the high rate of cardiovascular disease associated with schizophrenia (Hennekens et al., 2005). CHD-related mortality is higher in individuals with schizophrenia than the general population, with estimates as high as two-thirds of all people with schizophrenia dying from CHD (Hennekens et al., 2005). Brink et al. (2017) suggest that individuals with schizophrenia that actually survive into old age may in fact represent a healthier cohort of individuals due to the high morbidity and mortality associated with the disease. This is potentially relevant to the low rate of CHD found here, in that those with concurrent schizophrenia and CHD may be less likely to survive into old age. This is in addition to the possibility of the sample bias described above, whereby individuals with schizophrenia are more likely to require InterRAI assessment for complex needs regardless of fewer physical comorbidities such as CHD.
The higher rate of diagnosed diabetes mellitus in the schizophrenia group is consistent with what is already known regarding the comorbidity of the two illnesses. Individuals with schizophrenia are 2–5 times more likely to be diagnosed with diabetes mellitus, and this risk increases with age (Suvisaari et al., 2016). Factors that have been linked with diabetes, such as obesity, sedentary lifestyle, smoking and poor diet, are also common among individuals with schizophrenia (Gough, 2005; Suvisaari et al., 2016). Furthermore, the impact of anti-psychotic treatments on the development of diabetes is twofold; there is the known effect that these medications have on insulin sensitivity, alongside the compounding effects of weight gain that is a common side-effect of all anti-psychotics (Suvisaari et al., 2016). Second-generation anti-psychotics, in particular, are associated with an increased relative risk for diabetes compared to first-generation anti-psychotics (Smith et al., 2008). Interestingly, loneliness in psychotic illness has been associated with an increased risk of cardiometabolic disorders (Badcock et al., 2018), which further emphasises the potential benefits of psychosocial interventions in schizophrenia. There is also some evidence to suggest schizophrenia is directly associated with an underlying genetic vulnerability to metabolic abnormalities even in the absence of neuroleptic treatment (Mitchell et al., 2013).
Unsurprisingly, high rates of all the assessed comorbid psychiatric disorders in the schizophrenia group were established. One of the key characteristics of schizophrenia in older persons is elevated rates of comorbid depression (Cohen et al., 2008; Diwan et al., 2007; Karim et al., 2005), and our findings further contribute to this understanding. The excess of bipolar disorder most probably illustrates the diagnostic overlap between the two disorders, and the high rate of comorbid anxiety is consistent with other research findings (Achim et al., 2011).
While this study has many strengths, including the large sample size, use of an internationally recognised standardised instrument, self-identified ethnicity and the adjustment for several confounding factors with few missing values, there are some limitations that require discussion. Significantly, the study was not carried out in a random community sample, and therefore, the results cannot be generalised to the wider New Zealand population. However, the sample covers over 10% of the national population aged ⩾ 65 years and reflects persons who are accessing and requiring healthcare, which is a substantial sample relevant to health service provision and planning. The elevated prevalence of schizophrenia in this sample (0.7% compared to a recently established lifetime prevalence of 0.48% in the adult population (Simeone et al., 2015)) is most likely related to these sample characteristics. What this increased prevalence rate does establish is that there is a clear presence of older adults with schizophrenia accessing health services. Furthermore, the higher ratio of female in the schizophrenia group needs to be interpreted with caution given the higher proportion of female in the sample. An additional limitation that requires highlighting is that the age of onset of schizophrenia was not determined. Schizophrenia in the older age population consists of two groups: those that developed the disorder early in life and have grown old, and those that are diagnosed at age 45 years or older who are categorised as having late-onset schizophrenia (Folsom et al., 2006). Late-onset schizophrenia is thought to have a better prognosis and generally requires less aggressive treatment with anti-psychotics (Jeste and Maglione, 2013). Differential age of onset, including the distinction between early- and late-onset schizophrenia groups, could not be explored in relation to the findings. This information could be collected longitudinally and compared to the data set, but such information is not consistently or routinely collected. Finally, multiple statistical comparisons were made within this study, thus 5% of significant tests presented may be spurious due to chance alone. However, all comparisons were planned a priori, care was taken to present both crude and adjusted estimated effect sizes and associated confidence intervals and so no Bonferroni or Bonferroni-type adjustments were implemented (Perneger, 1998).
It has been shown that older adults with schizophrenia remain significantly impaired when they are evaluated next to their peers. Yet, there is a dearth of research that addresses the topic, limiting our understanding of how services can cater to the needs of this highly complex group. This research starts to contribute to this gap in knowledge by providing insights into the distinct characteristics of older adults with schizophrenia and supports the assertion made by Jeste and Maglione (2013) that ‘just as children are not small adults, older people are not merely chronologically older adults’ (p. 966). The higher prevalence rate demonstrates a clear presence of older adults with schizophrenia accessing health services with persistent social and environmental challenges. This information offers an evidential basis for understanding the extent and nature of the problem, upon which the development and provision of health care services can be built.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
