Abstract
Background:
Monitoring clinical response to treatment in depressed inpatients, particularly identifying early improvement, may be sub-optimal. This may impact adversely on patients through longer admissions and sub-optimal pharmacotherapy. Psychomotor speed is a prominent neuropsychological function which changes as recovery occurs. This study examines simple techniques used to quantify psychomotor change and their potential to contribute to monitoring recovery.
Methods:
Activity levels were continuously monitored in patients diagnosed with a major depressive episode from four acute psychiatric wards using two actigraphs (commercial and scientific) for 3 weeks and linear regression used to calculate a gradient to express rate of change. Psychomotor speed was assessed using the simple Coin Rotation Task. Mood and functioning were rated using the Quick Inventory of Depressive Symptoms, Clinical Global Impression Scale and Functioning Assessment Short Test. The assessments were completed at baseline and follow-up (3 weeks), and correlations were calculated for all change measures.
Results:
In all, 24 inpatients were recruited but not all completed baseline and follow-up measures. Change in activity count (N = 16) and psychomotor speed (N = 13) correlated significantly with improvement in clinical measures of depressive symptoms. Actigraphs were acceptable to hospital inpatients.
Limitations:
The limited size of this pilot study precludes the analysis of predictive power or the influence of other variables such as depression subtypes, age, gender or variations related to medications.
Conclusion:
Early change in simple activity and psychomotor speed warrant further investigation for utility in measuring treatment response in depressed inpatients.
Introduction
Patients suffering a major depressive episode make up a significant proportion of inpatients in any psychiatric hospital. In general, monitoring change in clinical symptoms relies on the observations of a changing, rostered group of nursing staff and other health professionals with various levels of skill (Mullen, 2002). Often there is no consistent objective measure of symptoms. The result is potentially inexact or anecdotal reporting; for example, ‘slept well’, ‘often stirred’, ‘isolative all day’, ‘out for several walks’ or ‘busy about the ward’. Initial signs of recovery from depressive episodes often occur early in treatment (Posternak and Zimmerman, 2005). Subtle changes in psychomotor characteristics can easily be missed in a busy ward which does not employ rigorous assessment tools. Any late recognition of change in symptom intensity may impact on patient progress by delaying the titration of treatment or change to alternatives. This may in turn delay discharge and return to normal life functioning, along with increased cost to health services. Monitoring symptoms consistently and objectively may prevent these adverse outcomes by detecting the earliest signs of improvement (or lack of it) (Matthews et al., 2014). Specific instruments to objectively assess psychomotor symptoms may assist accurate assessment of patient progress (Lemke et al., 1999).
Psychomotor functions have been postulated as likely predictors of clinical response (Bennabi et al., 2013; Sobin and Sackeim, 1997) and psychomotor speed may be one of the first functions to change with successful treatment of depression (Browning and Cowen, 1986). A review of longitudinal studies concerning neuropsychological function in depression suggests that psychomotor function most consistently changes in response to treatment of depression (Douglas and Porter, 2009), with further data suggesting that this is the case for inpatients with severe depression (Douglas et al., 2011). An extremely simple, validated measure of psychomotor speed (Hill et al., 2010; Mendoza et al., 2009), the Coin Rotation Task (CRT) is cheap and easy to administer suggesting possible utility by regular staff in the inpatient setting.
Activity levels may also provide an indication of response to treatment of depression. A review of the literature by Teicher (1995) identified a correlation between depression severity and daytime activity levels, and between change in depression severity and change in activity levels. Winkler et al. (2014) demonstrated the latter using actigraphic data to calculate percentage change in daytime activity levels following electroconvulsive therapy (ECT) and compared this with reduction in Hamilton Rating Scale for Depression (HRSD) scores where remission is <8. Although activity increased in the total sample (N = 15), the increase was more marked in remitters (49.8%) than non-remitters (3.1%). Other previous studies (Burton et al., 2013; De Crescenzo et al., 2016; Krane-Gartiser et al., 2014; McCall, 2015; Maxhuni et al., 2016; Raoux et al., 1994; Reichert et al., 2015; Teicher, 1995) have pointed to the usefulness of actigraphy as an objective clinical measure of psychomotor performance and suggested its utility in monitoring treatment progress in mood disorders.
We therefore aimed to determine whether simple techniques used to monitor change in psychomotor function would potentially be useful to monitor recovery in the acute inpatient setting. We investigated this using a scientifically validated actigraph, a commercially available ‘activity tracker’ and the CRT to determine change factors for psychomotor function which were then compared with the changes in measures of standard mood and functioning rating scales. Acceptability of actigraphy to participants was also assessed.
Methods
Participants
All patients admitted over an 8-month period to the general adult acute psychiatric wards of Hillmorton Hospital (Christchurch, New Zealand) with a primary diagnosis of major depressive episode (both unipolar and bipolar disorder—depressed phase) as assessed by experienced psychiatrists were considered for inclusion in an open label feasibility study of cognitive and behavioral activation therapies. Our investigation into changes in psychomotor speed involved the same patient group. The studies were approved by the University of Otago Human Ethics Committee (Health). As a pragmatic study, exclusion criteria were designed to be minimal. They included those with current severe drug or alcohol misuse disorders, those considered too severely depressed to give informed consent, patients who had already participated in the study on a previous admission and those patients currently receiving ECT. All participants received inpatient treatment as usual for depression and in addition attended up to three regular sessions per week with a therapist who administered the activation therapy programs for a maximum of 21 days as part of the wider pilot study. Inclusion in therapy and testing continued if the patient was placed on leave or discharged from hospital during the study period.
Measures
Coin rotation task
Participants held a New Zealand 20 cent coin (22 mm diameter) in their dominant hand and turned the coin through 180° as many times as possible in 20 seconds using their thumb and first two fingers. The time taken to retrieve a dropped coin and recommence the process was included in that period unless prolonged. The count was recorded at baseline and on completion of the wider study.
Actigraphic measures
Participants were asked to wear both a biaxial (recording acceleration in two planes) research actigraph (Actiwatch 2 using Philips Actiware 6.0.7) and a commercially available triaxial (three planes) ‘activity tracker’ (Fitbit Charge Heart Rate [HR]) for the entire study period. The Fitbit was removed while bathing (not waterproof), for recharging (every 5 days) and for ‘syncing’ to upload data (weekly) only, otherwise worn continuously. Fitbits were fitted with a face cover to preclude participant’s access to data which might have affected outcomes in the wider study by stimulating activity. Participants were asked to wear the Actiwatch continuously. Resulting data were retrieved on completion of the study period.
Participants were also asked to complete a questionnaire concerning the wearability of the devices on termination of the study period. The acceptability of the equipment to the participant has important implications for future clinical application.
Activity counts (or equivalent) were tabulated from both types of actigraphs. Clearly artefactual results, for example resulting from off wrist time, were estimated and the full day’s data deleted if a minimum of 16 hours of acceptable wear time data per day was not reached. While daily means are a relatively coarse measure, the use of activity measures spanning the nycthemeron dispenses with the need to allow for sleep, wake and napping periods.
Actiwatch activity count data (Actiware 6.0.7) used in this study is in the form of mean activity counts per minute per day. The Fitbit produces data in the form of minutes of ‘sedentary’, ‘light’, ‘fairly active’ and ‘very active’ levels of activity per day. An algorithm for quantitative breakdown of these levels into total activity counts per minute could not be obtained from the parent company (Fitbit); therefore, this study considered minutes of ‘light’ levels of activity (the largest component of total activity for our sample) and a combined ‘total’ of all levels of activity separately. Minutes of light, moderate and high activity were given the same ordinal value (regardless of intensity) when combined (noted as ‘total’) and treated as de facto activity counts.
Mood and functioning rating scales
Mood and functioning rating scales were carried out at baseline and on completion of the wider study. These included the Quick Inventory of Depressive Symptoms (QIDS-C16) (Rush et al., 2003), the Functioning Assessment Short Test (FAST) (Rosa et al., 2007) and the Clinical Global Impression: Severity scale modified (CGI-S) (Guy, 1976). Item 6—extremely ill and 7—severely ill of the CGI-S were combined as all participants were hospitalized.
Statistical analysis
As decided prior to the study, the linear trends in activity counts over time were summarized as the gradient of the trendline using all days with valid activity counts for each participant. This gradient has been adopted as the change factor for the Actiwatch, and Fitbit activity count data (mean activity count/minute/nycthemeron) recorded over the duration of the study period (⩽3 weeks). Actograms were inspected and clearly artefactual results, resulting for example from removal of the devices for periods of time, were totaled for each day. Days with less than a minimum number of 16 hours of acceptable data per day were deleted. The change between baseline and follow-up values for QIDS, CGI-S and FAST was examined using paired T tests. Correlation coefficients were calculated (Spearman’s rho was used as not all results were normally distributed) to test the strength of the association between changes in mood and functionality measures, and those of activity and psychomotor speed, SPSSv23.0 (IBM Corporation, Armonk, New York, USA) was used for all statistical analyses. Because of the likely inter-correlated nature of many of the measures, a correction for multiple tests was not made, preferring instead to examine the pattern of results. The practical aim of the current analysis is to provide simple results which might be of use in the development of future clinical assessment tools for severity of depression.
Results
Of the 39 patients invited to participate, 24 consented (22 unipolar, 1 Bipolar 1, 1 Bipolar 2). Age of participants ranged from 18 to 56, with mean age being 38.08 (standard deviation [SD] 13.625). The cohort consisted of 15 males (one transitioning from female) and 9 females. In all, 19 identified as NZ European and 5 as Maori. From the initial 24 patients, usable actigraphy results were obtained from 17 Actiwatch wearers and 19 Fitbit wearers. Analysis showed that in general participants showed linear changes (increase or decrease) in their activity levels over time as demonstrated by mean correlations for Actiwatch of 0.32 and for Fitbit-light 0.30 and Fitbit-total 0.27.
Wear periods ranged from 10 to 22 days (M = 18.4, SD = 3.9) for Fitbit and 13–22 days (M = 16.9, SD = 4.0) for Actiwatch. Usable recording periods ranged from 5 to 19 days (M = 14.5, SD = 4.4) for Fitbit and 9–21 days (M = 16.7, SD = 3.8) for Actiwatch. Six participants were discharged prior to the completion of the study. Most of the participants had leave away from the hospital of various lengths during the study period.
Scores on mood and function scales are shown in Table 1.
Mean change in mood and functioning rating scales.
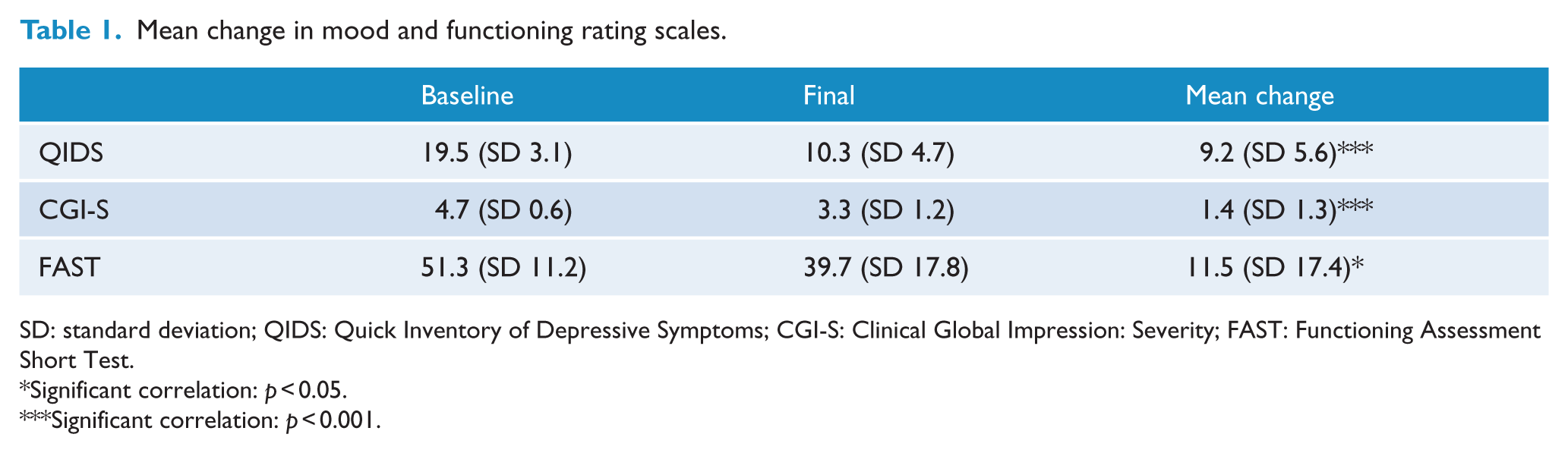
SD: standard deviation; QIDS: Quick Inventory of Depressive Symptoms; CGI-S: Clinical Global Impression: Severity; FAST: Functioning Assessment Short Test.
Significant correlation: p < 0.05.
Significant correlation: p < 0.001.
The correlation among measures is shown in Table 2. As shown, the Actiwatch activity count gradients correlated strongly with both QIDS and FAST changes and also significantly with CGI-S change values. The Fitbit activity count gradients (for both ‘light’ and ‘total’ values) did not correlate significantly with change values for QIDS, CGI-S or FAST. They also did not correlate with the Actiwatch gradients (with Fitbit ‘light’ 0.418 p = 0.095 and with Fitbit ‘total’ 0.336 p = 0.188). The change in CRT scores showed correlations with change measures of the QIDS, CGI-S and FAST and was also correlated with the Actiwatch gradients. There was a significant correlation (Spearman’s rho) between changes in QIDS and changes in FAST (n = 19, r = 0.848, p < 0.001).
Correlation of activity and psychomotor speed changes with mood and functionality changes.
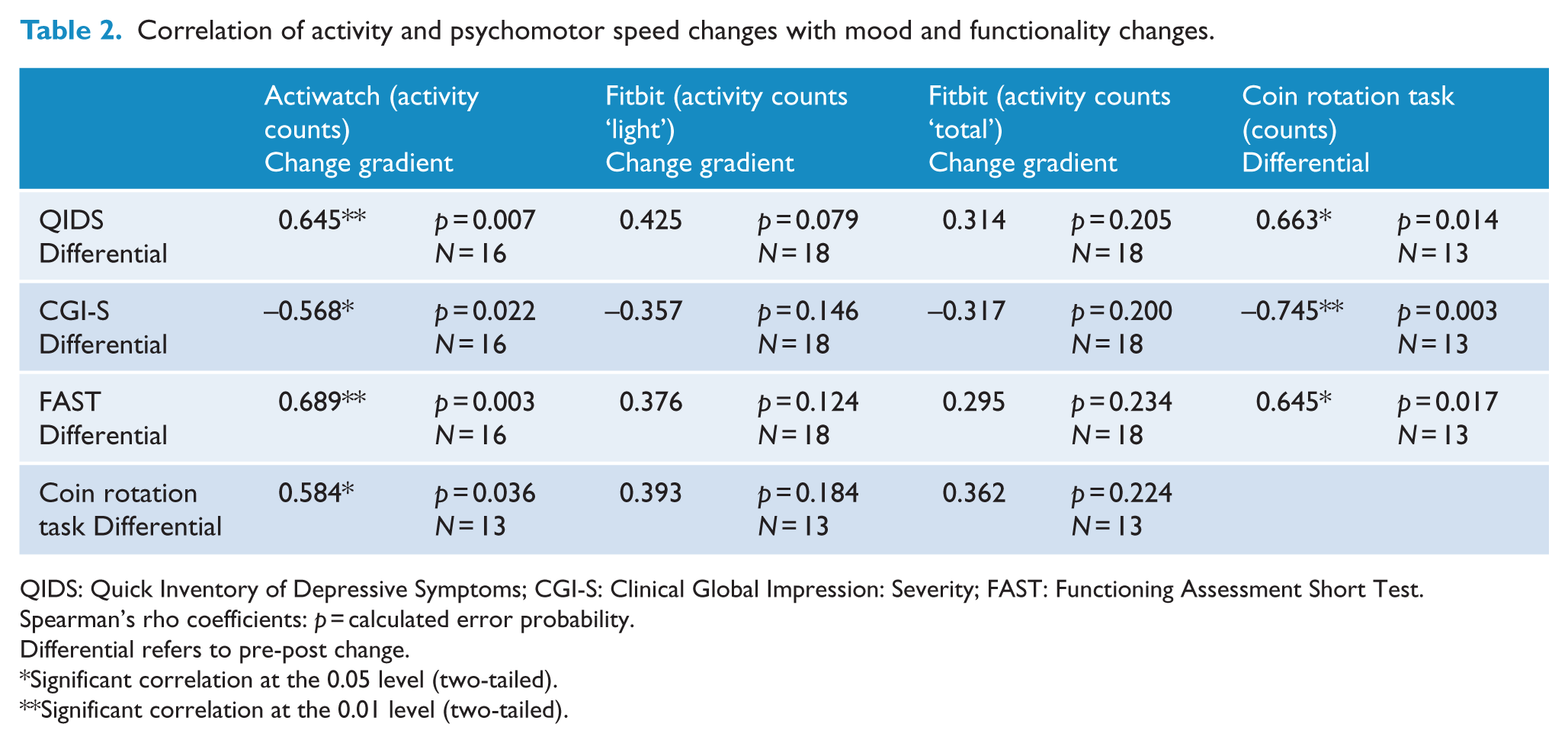
QIDS: Quick Inventory of Depressive Symptoms; CGI-S: Clinical Global Impression: Severity; FAST: Functioning Assessment Short Test.
Spearman’s rho coefficients: p = calculated error probability.
Differential refers to pre-post change.
Significant correlation at the 0.05 level (two-tailed).
Significant correlation at the 0.01 level (two-tailed).
The relationship between activity (Actiwatch) gradients, psychomotor speed (CRT) change and change in QIDS for each participant is shown in Figure 1. Data have been normalized for each measure so they are on a common scale for visual comparison. The process of normalization re-expresses each individual change as a proportion of the range of changes across all individuals for each measure (where available).
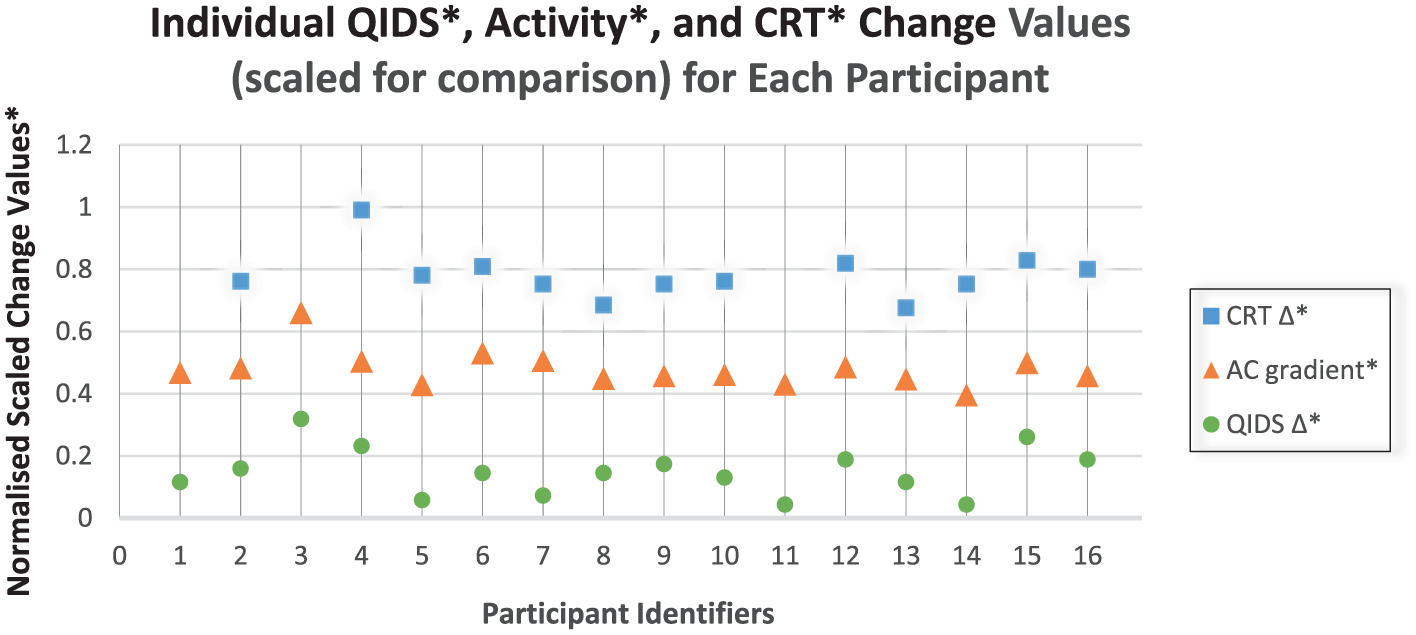
Change in Actiwatch activity levels, psychomotor speed and depression rating for individual participants.
Acceptability of actigraphs to participants and researchers
In total, 18 participants completed questionnaires upon completion of the study period. Overall, the units were well tolerated. In all, 16 wore both the Fitbit and Actiwatch with few issues of discomfort or inconvenience reported even though only 5 participants reported usually wearing a watch. In all, 11 participants did not notice the actigraphs or readily became accustomed to wearing them. Three felt self-conscious when wearing the actigraphs, especially when on leave outside the hospital. In total, 15 reported no effects on their sleep as a result of wearing them, but 3 (referring to the Fitbit) reported redness, itchiness or a rash and of those one was discontinued when staff were notified. General comments received included the inconvenience of removing the unit for washing (Actiwatches are waterproof, Fitbits are not) and being annoyed when the unit needed to be recharged (Actiwatches are pre-charged only, Fitbits required recharging regularly during the study). There was at least one instance of a Fitbit face cover removal and data accessed.
There was a wide variation between participants in medication used. Antidepressants—at baseline: six patients were taking selective serotonin reuptake inhibitors (SSRIs) of whom two continued and four were switched to venlafaxine; five were on venlafaxine of whom four continued and one switched to bupropion; four were on no antidepressant of whom three were discharged on none and one was started on an SSRI; one was on nortriptyline and switched to venlafaxine. Mood stabilizers—at baseline 1 was on lithium carbonate and continued on it; one was commenced on lithium carbonate and one was commenced on sodium valproate (neither had previously been on mood stabilizers). Antipsychotics—four were on olanzapine of whom three continued the same dose and one was increased; two were on risperidone of whom one continued (increased dose) and one switched to olanzapine; one was commenced on risperidone; one started on quetiapine and continued with an increased dose.
Discussion
In depressed inpatients, there was a significant correlation between change in QIDs, FAST and CGI-S and change in movement measured by actigraphy (activity levels [Actiwatch 2]) and CRT (psychomotor speed). There was no significant correlation when activity was measured using the Fitbit Charge HR.
Correlation between measures of patient activity at a certain point, with clinical depression ratings at the same point, have been shown in many previous studies (Aronen et al., 1996; Attu et al., 2012; Iverson, 2004; O’Brien et al., 2017; Song et al., 2012) and activity has been proposed as a real-world biomarker for depression. Change in activity levels in depressed inpatients following a period of treatment has been demonstrated (see meta-analysis by Burton et al. (2013)).
The current study differs from previous similar studies by virtue of having examined the correlation of clinical rating scale changes and changes in activity level measured (by actigraphy) continuously over the course of treatment. Previous studies have tended to examine activity before and then after treatment with the exception of an early study (Godfrey and Knight, 1984) showing partial correlations between repeated measures of (daytime) activity using an actometer (a modified self-winding wristwatch) and change in Depression Adjective Check List (DACL) scores over an average treatment period of 15.2 days (five inpatients). In only one patient was this correlation significant. The inconsistent results may result from the use of the actometer as similar instruments have been shown to provide poor intersubject reliability (Teicher, 1995). Benoit et al. (1985) monitored 10 depressed female inpatients over the period of their admission, using actigraphy to record activity levels (averaged over 3 days) on admission and at discharge. They identified increased (daytime) activity, decreased immobility and decreases in HRSD and Montgomery Asberg Depression Rating Scale (MADRS; Montgomery and Asberg, 1979) scores but demonstrated no correlations between the changes in these measures. In contrast to our study, measurements were taken only at the beginning and end (a variable time interval) of the admission and not in between. Winkler et al. (2014) demonstrated increased activity (wrist worn Actiwatch Plus) in depressed inpatients (N = 15) who showed remission after ECT (none was found in non-remitters), which correlated with the change in HRSD scores. However, this study monitored activity before treatment with ECT and after treatment, but not during the course of treatment. A recent actigraphic study (10 inpatients and 2 outpatients) found that motor activity was associated with a short-term increase in mood and suggested that the increase in motor activity was driving an increase in mood (Bewernick et al., 2017).
A contrasting inpatient study measuring activity intensity and quantity compared data for a 7-day period in weeks 1 and 4 of treatment (27 inpatients and 27 healthy matched controls). Todder et al. (2009) found that although daytime activity intensity increased and was correlated with change in HRSD ratings, the quantity of activity did not. This may be due to methodological issues which included all patients receiving 50–100 mg of quetiapine per day as well as antidepressant medication. The impact of medication on activity is clearly demonstrated in a study of outpatients (N = 14) using continuous wrist actigraphy for the first 10 days of a medication trial (Stanley et al., 1999). It was demonstrated that patients taking dothiepin showed less overall daytime activity than those taking fluoxetine by comparing composite graphs of mean activity counts per hour for each group.
For clinical use in early identification of treatment response, the continuous measurement of psychomotor change during treatment may be more useful and accurate. Wielopolski et al. (2015), using a multi-sensor actigraphic armband (SenseWear Pro3 Armband for 24-hour [at days 1 and 7] and 48-hour [day 21] periods) in unipolar depressed inpatients (N = 19), found that physical activity levels and total and active energy expenditures increased significantly in parallel with clinical improvement over the period of study. They note that the size of the armband makes continuous use over longer periods problematic. Finazzi et al. (2009) used actigraphy (Actiwatch Score) to study the correlation between depression severity and activity measures (mean values from weekly samples) in six adolescent outpatients over 9 weeks. They found correlations between weekly assessed depression measures (Children’s Depression Rating Scale–Revised) and mean motor activity (significant only in the 1200–1800 hour period), mean waking activity and 10 most active hours per week. A more marked degree of change in mean motor activity was demonstrated at the conclusion of week 1 than the following weeks.
This study is therefore the only one to monitor activity continuously over the first 2–3 weeks of treatment (mean 16.3 days) and to examine the correlation of change with the change in clinical measures. Actigraphic gradients did in fact correlate with treatment response over the study period. Further research is required to explore whether this method may be used successfully at earlier time points (say baseline to 7 and/or 14 days). This may allow an earlier examination of whether there is a change in activity which is likely to reflect clinical response.
Of the two actigraphs tested, the Actiwatch proved to be simplest to administer and most convenient for participants, but accessing and interpreting data was more difficult. Actiwatch advantages were waterproofness, no recharging or data uploads during the 21-day period of the study (Fitbit needed to be recharged/5 days and data uploaded/7 days), and Actiwatch data were inaccessible to the wearer (the face cover fitted to Fitbits was not secure and accessing data may have affected motivation and activity levels). Advantages to researchers of Fitbits included being considerably less expensive and providing easy access to much of the collected data by simple download from a website on any computer. However, some data are not available by download and have to be transcribed manually. Also, the Fitbit data stream is a less continuous activity record as a result of being removed regularly. Actiwatch software was uploaded to dedicated computers only but provided full access to all data in spreadsheet format. Data retrieval and interpretation required more expertise, but this interpretation resulted in richer and more accurate data. Regardless of these practicalities, gradient of change in activity measured with the Actiwatch correlated with change in all clinical measures while change in Fitbit activity correlated with no clinical variables. Other comparative studies have suggested poor correspondence between consumer-ready devices and validated clinical actigraphs (Ancoli-Israel et al., 2015).
In this study, psychomotor speed as measured using the CRT correlated with change in clinical measures. This is in keeping with the review by Douglas and Porter (2009) examining longitudinal studies in depression which suggests that the neuropsychological domain most sensitive to clinical state is PM speed. In a group of depressed inpatients similar to those in this study, psychomotor function (assessed using computer-based reaction time tasks) was the only cognitive function to differ significantly between responders and non-responders after 6 weeks of treatment (Douglas et al., 2011). Early clinical change (reduced psychomotor retardation) was identified in some patients receiving antidepressant treatment (desipramine, N = 70) using HRSD self-report. This differentiated them from others who had not responded at 6 weeks (Katz et al., 2004).
As a measure of PM speed, the CRT was found to be simple and easy to administer. Previously, it has been used to demonstrate change in PM function in the assessment of various neurodegenerative disorders including Parkinson’s disease, hemispheric damage and multiple sclerosis; conditions such as apraxia, schizophrenia and aging; and changes caused by dopaminergic medications. In a study comparing the CRT (N = 204) to a psychomotor subscale of the Modified HIV Dementia Scale, Minor and others concluded that this simple test is not only a valid test of PM performance in HIV sufferers, but may be the more accurate, particularly if lower education level was a factor (Minor et al., 2010). In depression, it has been used to assess change in PM speed (Thornton, 2014) in patients undergoing ECT (treatment-resistant depression = 10, bipolar disorder mania = 3). Effect sizes (baseline to follow-up) were small, perhaps due in part to the mixed group and the treatment-resistant nature of the patients. Change in PM speed in this study correlated significantly with the Actiwatch activity gradients and change in all three mood and functionality measures (particularly the QIDS). This indicates that PM speed testing in some form may also have practical clinical applications in monitoring inpatient treatment for depression although practice effects have previously been reported with the CRT (Thornton, 2014) which may limit its use for multiple measures.
There has been considerable interest in the relationship between cognitive function and general functioning in mood disorders. Specifically, in inpatients with depression, two studies suggest a correlation between measures of cognitive function and general functioning. While Withall et al. (2009) showed only a relationship with executive function measures, Jaeger et al. (2006) showed a close correlation between general functioning and cognitive function on a range of cognitive measures including those of psychomotor speed, measured using another very simple task—the Grooved pegboard task. We did not examine single time point correlation but did find a correlation between activity gradients and psychomotor function, and change in a measure of general function—the FAST. Larger studies are needed to assess further the contribution of impaired activity and psychomotor function to general function in people with depression.
There are some potential limitations which relate to the interpretation of the findings. First, the data were collected in the process of conducting an open-label trial of activation therapy in addition to treatment as usual. This then differs from usual clinical practice and the focus on activating patients may have led to greater increases in psychomotor function and activity, thereby inducing correlations with response which would not occur in usual treatment. Second, discharge or extended leave during the study period may also have confounded results somewhat as opportunities and/or motivations for activity may be greater in these circumstances. However, this is the reality of treatment of depression in most inpatients units and these methods of monitoring will only be of use if they are robust in the face of this. In future research, groups of patients should be large enough to examine the impact of periods of home leave. Third, research assistants supervising the clinical rating scales were not necessarily blinded to baseline outcomes, which introduces the possibility of rater bias. However, they were blinded to actigraphic data and CRT results are not subject to this error. Fourth, the use of multiple measures and therefore multiple statistical tests could have resulted in chance findings. However, the positive correlations all occur in a block of tasks and lack of statistical significance all occurs in tests using one method of measurement. It is unlikely that this is a chance finding. Fifth, the study was small and certainly requires replication in a larger sample. Finally, findings may apply disproportionately to inpatients with melancholia and less to other depression subtypes. Psychomotor changes noted may well be driven by a subgroup of patients who have melancholic depression, which is characterized by psychomotor disturbance (Parker et al., 2010). This is particularly relevant to this study as evidence suggests that many inpatients with major depression meet the criteria for the melancholic subtype (Stage et al., 1998).
As expected, patients were on a variety of medications at baseline and a variety of changes were made. While medication may certainly affect psychomotor speed, it is difficult to see how these changes in medication could systematically have biased the results.
Strengths include the conduct of the study in a busy inpatient unit with minimal exclusion criteria, making the study findings applicable to, we believe, many inpatient units in public health services.
Conclusion
This study monitored activity levels (using 0000–2400 hours continuous actigraphy) and psychomotor speed (CRT measures) over a 3-week period and compared the results with change in measures of mood and functioning (QIDS, FAST and CGI-S) for a small group of depressed inpatients. Significant correlation between simple measures of psychomotor activity and clinical response provide encouraging evidence for the possibility of early, objective identification of response to treatment in depression. It also provides preliminary evidence regarding methods of activity monitoring, with simple Fitbit monitoring unlikely to be useful.
Of the two actigraphs tested, the Actiwatch proved to be the simplest to administer and most convenient for the participants. Further work is required to develop a user-friendly actigraphy interface with a focus on how data can be accessed more readily in real time in the inpatient setting in a form which could be interpreted by clinical staff. Future studies should be larger to allow analysis of predictive power and the influence of various other variables such as unipolar versus bipolar depression, melancholic depression or not, age and variations related to medications. The results of this study suggest that further research using actigraphic activity measures as well as other measures of psychomotor function may provide data which is a useful addition to clinical assessment of response and may predict clinical response at an early stage in the clinical course.
Footnotes
Acknowledgements
The authors would like to acknowledge the actigraphy support given by Mr Steve Edwards (Product Support Technical Specialist, Philips Respironics, Philips Home Healthcare Solutions).
Declaration of Conflicting Interests
The authors have received no financial support from the makers of the Actiwatches used (Philips Respironics, Philips Home Healthcare Solutions) and declare no other conflicts of interest. R.J.P. uses software for research at no cost from Scientific Brain Training Pro.
Funding
This work was supported by the Canterbury District Health Board, Christchurch, New Zealand; the University of Otago (Department of Psychological Medicine), Christchurch, New Zealand; Te Pou o te Whakaaro Nui, PO Box 108-244, Symonds Street, Auckland 1150, New Zealand (education funding); and the Canterbury Medical Research Foundation, 1/230 Antigua Street, Christchurch, New Zealand (research funding reference number 2016 NFS). None of these have contributed to the study design; the collection, analysis or interpretation of data; in the writing of the report; or in the decision to submit the article for publication.
