Abstract
Background:
Eating disorders are serious mental disorders and are associated with substantial economic and social burden. The aim of this study is to undertake a systematic review of the cost-effectiveness studies of both preventive and treatment interventions for eating disorder.
Method:
Electronic databases (including the Cochrane Controlled Trial Register, MEDLINE, PsycINFO, Academic Search Complete, Global Health, CINAHL complete, Health Business Elite, Econlit, Health Policy Reference Center and ERIC) were searched for published cost-effectiveness studies of eating disorder prevention and treatment including papers published up to January 2017. The quality of studies was assessed using the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) checklist.
Results:
In all, 13 studies met the review inclusion criteria as full cost-effectiveness studies and 8 were published since 2011. The studies included three modelled and one trial-based study focused on prevention, two trial-based and one modelled study for anorexia nervosa treatment and three trial-based studies for bulimia nervosa treatment. The remaining studies targeted binge-eating disorder or non-specific eating disorder treatment. The average percent of CHEERS checklist items reported was 71% (standard deviation 21%). Eating disorder interventions were mainly cost-saving or more effective and more costly compared to comparators; however, some results did not reach statistical significance. In the two studies that achieved 100% CHEERS checklist, one study reported that a cognitive dissonance intervention might be cost-effective for prevention of anorexia nervosa and bulimia nervosa with a 90% participation rate and the second study supported lisdexamfetamine to be cost-effective in the treatment of binge-eating disorder. Insufficient evidence for long-term cost-effectiveness (e.g. over 2 years) was found.
Conclusion:
Cost-effectiveness studies in eating disorder appear to be increasing in number over the last 6 years. Findings were inconsistent and no firm conclusion can be drawn with regard to comparative value-for-money conclusions. However, some promising interventions were identified. Further research with improved methodology is required.
Introduction
Around 3 in every 100 Australians have a current major eating disorder (ED) with 3-month prevalence estimates of anorexia nervosa (AN) 0.4% (95% confidence interval (CI) = [0.2, 0.7]); bulimia nervosa (BN) 1.1% (95% CI = [0.7, 1.5]) and binge-eating disorder (BED) 1.5% (95% CI = [1.1, 2.0]; Hay et al., 2017). EDs are also associated with substantial economic and social burden (Ágh et al., 2016a; Deloitte Access Economics, 2012; Erskine et al., 2016; Stuhldreher et al., 2015) and there is evidence of an increase in the incidence of ED behaviours and in turn the incidence of the sub-threshold variants of AN, BN and BED that comprise the other specified feeding or eating disorder (OSFED) and unspecified feeding or eating disorder (UFED) over the last two decades (Hay et al., 2008, 2015; Mitchison et al., 2014). Furthermore, evidence shows that there is an increasing health burden of EDs in terms of prevalence and quality of life impairment (Hay et al., 2017; Mitchison et al., 2014). Together, the two main types of EDs, AN and BN, account for 1.9 million disability-adjusted life years (DALYs) and comprise the 12th major cause of burden of disease and injury in young females aged 15 to 24 (Haagsma et al., 2015). Additionally, the costs of EDs are substantial in terms of both direct health-care costs and broader societal costs including loss of productivity (Ágh et al., 2016a; Deloitte Access Economics, 2012; Samnaliev et al., 2015; Stuhldreher et al., 2015). Hospital care, composed of inpatient hospital stays, treatment at day clinics and rehabilitation, accounted for 90% of these costs in people with AN (Stuhldreher et al., 2015). Furthermore, the cost of disability payments for AN was reported in Canada as being between three-quarters and 30 times greater than the overall cost of tertiary care treatment (Su and Birmingham, 2003).
There are several preventive and treatment-based interventions which have been demonstrated to be effective for EDs. Effective preventive interventions include cognitive-behavioural therapy (CBT) or media literacy interventions (Le et al., 2017a). Effective treatment interventions include family-based treatment for young people with AN (Couturier et al., 2013) and CBT and antidepressant medications for adults with BN and BED (Hay et al., 2014). However, whether these interventions are cost-effective is uncertain.
The most recent systematic review of cost-effectiveness for ED interventions published in 2012 highlighted very few studies in this area (Stuhldreher et al., 2012). This review found that only two cost-effectiveness analyses alongside randomised controlled trials (RCT) included both intervention costs and costs beyond those of the specific intervention (i.e. cost offset). However, the review used a ‘strict’ classification of what constitutes a cost-effectiveness study, in which both intervention costs and costs beyond the intervention were considered (Stuhldreher et al., 2012). In the other words, this classification excludes cost-effectiveness studies in which costs beyond the intervention are not considered because such costs of health services are very context specific (Mihalopoulos and Chatterton, 2015). However, these studies may have important implications for decision making (Husereau et al., 2013). Furthermore, the search only included studies published up to January 2011.
The aim of this study was to conduct an up-to-date review of the evidence base for cost-effectiveness of both preventive and treatment interventions for EDs. This study also aimed to identify more recent (and therefore, most cost-relevant) higher quality cost-effectiveness studies and highlight the implication for policy makers in EDs.
Methods
The current review conformed to the evidence-based guidelines in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA; Moher et al., 2009).
Search strategy
Eligible cost-effectiveness studies were identified through searching in MEDLINE, PsycINFO, Academic Search Complete, Global Health, CINAHL complete, Health Business Elite, Econlit, Health Policy Reference Center and ERIC database for articles published until March 2017. The search used the following search terms (eating disorder* OR anorexia OR bulimia OR binge*) AND (cost* OR economic*). Hand searching was conducted in the table of contents for journals that commonly publish ED studies (e.g. International Journal of Eating Disorders, Journal of Eating Disorders, Eating Disorders and European Eating Disorders Review) over March 2017.
Inclusion and exclusion criteria
Studies were selected for inclusion if they were comparative cost-effectiveness studies of prevention or treatment of EDs and published in the English language. Studies were eligible for inclusion if they compared costs (including costs of interventions with or without costs beyond of the intervention) and outcomes of at least two interventions. Descriptive economic studies that reported only cost of the interventions without comparative outcomes or that reported cost and consequences of one intervention, or cost-of-illness studies were excluded.
Study selection and extraction
All citations were imported into an electronic database (Endnote® version X2) with duplicates subsequently removed. The first author (L.K.-D.L.) completed the literature search, screened the abstract of every record and reviewed the full text if appropriate for inclusion in the review. Decisions regarding inclusion of studies where it was not clear as to whether the inclusion criteria were met were resolved by consensus between the first and last authors. Included studies were classified as cost-effectiveness studies of prevention or treatment of EDs. Other information that was extracted included the type of cost-effectiveness study undertaken (including cost-effectiveness analysis [CEA, where outcomes are expressed in clinical units, such as clinical scales], cost-utility analysis [CUA, where outcomes are presented in generic units such as quality-adjusted life years (QALYs)] or cost-benefit analysis [CBA, where outcomes are expressed in monetary terms using techniques such as willingness to pay studies]); the perspective and time horizon; outcome measurement and incremental cost-effectiveness ratio (ICER) results. To compare the costs or ICERs across all the studies, all costs were converted into 2013 US dollars. We did this by first, using country-specific gross domestic product inflators to convert the currency of the study into the 2013 equivalent. Second, we used purchasing power parities (PPPs) to convert to US dollars for the studies which reported the costs in non-US dollar currencies. Finally, if the year was not reported, the assumption of 2 years prior to the publication date was made as the base year.
Quality assessment
The published Consolidated Health Economic Evaluation Reporting Standards (CHEERS) checklist (Husereau et al., 2013) was used to assess the quality of included studies. This checklist recently consolidated and updated previous health economic evaluation guidelines into a single standard (Husereau et al., 2013). The checklist includes 24 items which are subdivided into six main categories: title and abstract, introduction, methods, results, discussion and other (Husereau et al., 2013). Quality assessment was completed for each included study by the first author (L.K.-D.L.). A score of quality assessment was given based on the percentage of criteria met by each study that ranged from 0% to 100%. A study, of which percentage of met criteria ranked over 50th percentile, was classified as ‘adequate’ quality of reporting.
Results
The literature search identified 6017 articles. After removing duplicates and screening by abstracts, 235 articles were retained for further consideration. Of these, 13 studies were identified which met inclusion criteria (Figure 1). In the excluded studies, there were 10 studies, which have reported cost outcome of interventions but did not met the criteria of full cost-effectiveness study. In particular, two studies RCTs reported costs as an outcome but did not examine these in relation to study outcomes and provided insufficient methodological information to justify their inclusions as full cost-effectiveness studies (Agras et al., 2014; Schmidt et al., 2007). Two studies were excluded as they reported cost analysis or cost offset of the interventions in which, the outcomes were not defined (Bode et al., 2017; Kass et al., 2017). The remainder was excluded as they were cost-outcome descriptions of a single intervention. Further information of the excluded studies is available in Appendix S1.
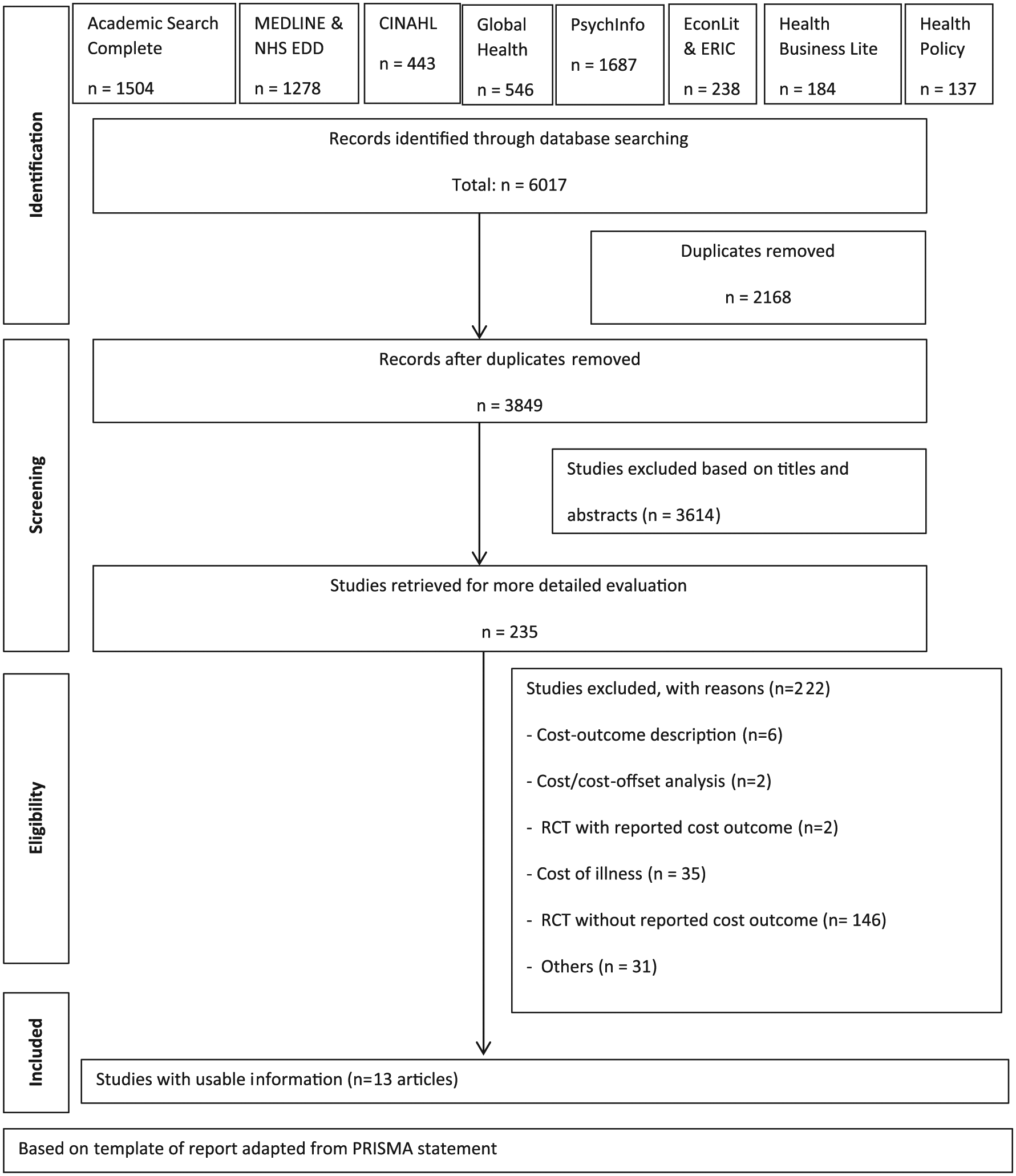
Flow chart of economic evaluation inclusion and exclusion.
Characteristics and main findings of included studies
Table 1 includes a comprehensive summary of included studies.
Included economic evaluations for preventive and treatment interventions for EDs.
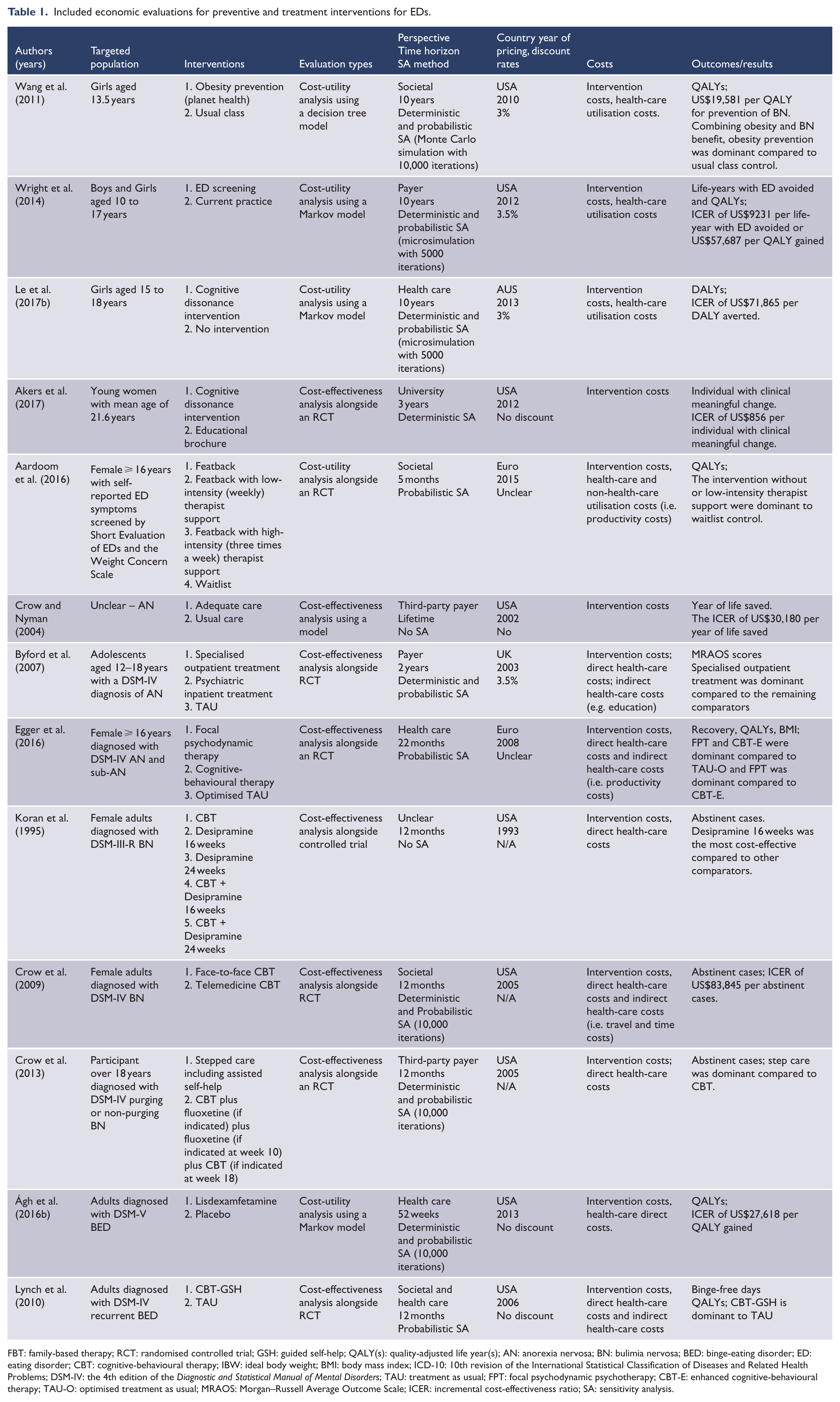
FBT: family-based therapy; RCT: randomised controlled trial; GSH: guided self-help; QALY(s): quality-adjusted life year(s); AN: anorexia nervosa; BN: bulimia nervosa; BED: binge-eating disorder; ED: eating disorder; CBT: cognitive-behavioural therapy; IBW: ideal body weight; BMI: body mass index; ICD-10: 10th revision of the International Statistical Classification of Diseases and Related Health Problems; DSM-IV: the 4th edition of the Diagnostic and Statistical Manual of Mental Disorders; TAU: treatment as usual; FPT: focal psychodynamic psychotherapy; CBT-E: enhanced cognitive-behavioural therapy; TAU-O: optimised treatment as usual; MRAOS: Morgan–Russell Average Outcome Scale; ICER: incremental cost-effectiveness ratio; SA: sensitivity analysis.
Preventive interventions for EDs
There were four studies that evaluated the cost-effectiveness of preventive interventions for EDs. Three studies were modelled evaluations focusing on adolescents aged 10–18 years, with 10-year time horizons, compared with a comparator of non-intervention or usual care (Le et al., 2017b; Wang et al., 2011; Wright et al., 2014). The remaining study was a 3-year trial-based CEA which focused on young females (mean age = 21.6 years; Akers et al., 2017). Most of these studies adopted narrow perspectives (i.e. from organisation perspectives to health-care perspectives). Wang et al. (2011) adopted a broader societal perspective. However, this study did not include productivity costs and therefore may be better characterised as a partial societal perspective. Wang et al. (2011) found that an obesity-prevention intervention for BN prevention (e.g. Planet Health Programme) was cost-effective with an ICER of US$18,291 per QALY gained when only benefits associated with ED were included. However, when benefits associated with both ED and obesity were considered the intervention became cost-saving. Results of cost-effectiveness of preventive interventions from other studies varied substantially as a result of differences in how outcomes were assessed. The study from Wright et al. (2014) assessed a screening intervention for preventing all types of Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) EDs and was the only preventive study that included both males and females. This study found that the screening intervention reported an ICER of US$57,687 per QALY gained or US$9,231 per life-year with ED avoided. The studies from Le et al. (2017b) and Akers et al. (2017) evaluated a cognitive dissonance intervention for preventing EDs in females with high body concerns. The results of these studies found that cognitive dissonance as a preventive intervention for ED was associated with an ICER of US$71,865 per DALY averted (Le et al., 2017b) and US$856 per additional at-risk person reducing ED symptom to a clinically meaningful degree (Akers et al., 2017).
Treatment interventions for EDs
There were nine studies that evaluated the economic credentials of ED treatments. For AN treatment, both studies from Egger et al. (2016) and Byford et al. (2007) were trial-based cost-effectiveness studies with time horizons of 2 years. The study by Crow and Nyman (2004) was a modelled evaluation with a lifetime horizon. Target populations varied between adolescents aged 11–18 years (Byford et al., 2007) and female adults (Egger et al., 2016) and were unclear in the Crow and Nyman (2004) study. While the studies from Byford et al. (2007) and Crow and Nyman (2004) were conducted from a government as payer perspective, the study from Egger et al. (2016) used a health-care perspective in the base case and a broader societal perspective (i.e. included productivity costs) in their sensitivity analysis. Studies examine different outcomes: recovery (defined as having a body mass index (BMI) > 17.5 kg/m2 and a score on the psychiatric status rating (PSR) scale of 1 or 2); QALY gains (based on the EuroQol – five dimensions (EQ-5D)); BMI changes (Egger et al., 2016); changes on the Morgan–Russell Average Outcome Scale scores (MRAOS; Byford et al., 2007) and years of life saved (Crow and Nyman, 2004). Results from the Egger et al. (2016) study indicated that both focal psychodynamic therapy and CBT were more effective at lower cost compared to optimised treatment as usual (TAU). Similarly, Byford et al. (2007) found that specialised outpatient treatment was more effective and less costly compared to psychiatric inpatient treatment or TAU. However, the results from both papers did not reach statistical significance. The remaining study found that implementing the ‘‘adequate care’’ approach (defined as inpatient weight restoration to close to 100% of ideal body weight followed by extensive care) led to an ICER of US$30,180 per year of life saved when compared to usual care (Crow and Nyman, 2004). However, the study did not include any uncertainty or sensitivity analyses, so it is unknown how robust the results were.
For BN treatment, all three studies were trial-based CEAs with 12-month follow-up and were undertaken to evaluate the cost-effectiveness of CBT on females aged 16 years or over who were diagnosed with Diagnostic and Statistical Manual of Mental Disorders (3rd ed.; revised; DSM-III-R) BN within the US context (Koran et al., 1995) or DSM-IV BN (Crow et al., 2009, 2013). All of these studies used abstinence as the primary outcome measure. In terms of the economic perspective, while Crow et al. (2009, 2013) adopted a third-party payer and societal perspective, Koran et al. (1995) did not state which perspective was used (although from the results it appears that a health-care perspective was adopted). Results indicated that face-to-face CBT was not considered to be cost-effective in comparison with 24-week desipramine (⩽300 mg/day) or telemedicine CBT or a stepped care intervention. Both studies from Crow et al. (2009, 2013) included insufficient details of how the resource uses were costed while the remaining study used ‘projected costs’ of a treatment plan instead of actual costs (Koran et al., 1995).
The current review identified two studies for BED treatment from Ágh et al. (2016b) and Lynch et al. (2010). The first study developed a population model to estimate the costs and benefits of lisdexamfetamine relative to placebo, while the second study evaluated the cost-effectiveness of CBT with guided self-help added to TAU. Both studies were conducted with a time horizon of 1 year, were within the US context, targeted adults with BED and clearly presented the way that resources were identified and costed. Lynch et al. (2010) found that adding CBT-guided self-help to TAU was more effective and less costly (or dominant) compared to TAU alone for recurrent BED treatment under both the societal perspective and health-care perspectives. Their conclusion did not change following a sensitivity analysis that assessed only cases with complete data (Lynch et al., 2010). Ágh et al. (2016b) reported an ICER of US$27,618 per QALY gained for lisdexamfetamine compared to the placebo with an 82% probability that the intervention was cost-effective at a willingness to pay threshold of US$50,000/QALY gained. The result of this study was most sensitive to the utility of the ‘non-symptomatic BED’ health state in the sensitivity analysis.
One remaining study evaluated the cost-effectiveness of an Internet-based intervention with or without clinician support relative to a waitlist control group. This study did not specify the type of ED but included adults with self-reported ED symptoms measured by the Short Evaluation of EDs and the Weight Concern Scale. The study analysed costs from a societal perspective within a 5-month time horizon and measured outcomes using QALY gains based on the EQ-5D-3L. This study found that the active intervention was more cost-effective compared to the waitlist control, although both cost and QALYs were not statistically significantly different across treatment arms.
Quality assessment
All the studies included in this review met over 50% of the CHEERs criteria checklist. The average percent of items reported in the studies was 71% (standard deviation: 20.8%). The studies that were published more recently (time cut-off point of 2011) achieved higher scores than the earlier studies (average scores of 77% vs 60%). Only two studies from Ágh et al. (2016b) and Le et al. (2017b) met 100% of the CHEERs criteria. Half of the studies were classified as ‘adequate’ with respect to the quality of their reporting, ranking them in 50th percentile. A common reason why studies did not achieve 100% of the criteria was that the research question did not include key elements such as the perspective, study population or time horizon. None of the studies undertaken alongside RCTs met the criterion of characterising heterogeneity. However, it is important to note that these trials were not necessarily powered to detect subgroup differences (Mihalopoulos and Chatterton, 2015). Only 3 out of 13 studies reported any conflict of interest. Approximately half of studies reported costs and outcomes for every treatment alternative but did not report the incremental costs and outcomes between the interventions. Further details of the quality appraisal may be found in APPENDIX S2.
Discussion
Summary of the main findings
The current review found that the number of cost-effectiveness studies in EDs has more than doubled in the last 6 years. The methodological quality of studies has also improved in more recent studies (since 2011). Studies from Le et al. (2017b) and Ágh et al. (2016b) were the highest quality cost-effectiveness studies which met 100% CHEERs checklist criteria. The former study highlighted that increasing the participation rate of the cognitive dissonance intervention might render the intervention to be cost-effective for AN and BN prevention, while the latter study supported the cost-effectiveness of lisdexamfetamine in BED treatment. Due to the variability in the types of interventions and alternatives evaluated and heterogeneity of targeted population, no conclusive recommendations can be drawn regarding the most cost-effective interventions for prevention and treatment in EDs. Further research of cost-effectiveness with improved methodological rigour is needed.
Methodological limitations of included studies
While there have been considerable improvements in both quantity and quality of cost-effectiveness studies in comparison to previous review by Stuhldreher et al. (2012), there are still some persistent methodological limitations in the current studies reviewed. Many studies provided insufficient information on how resources were measured and costed. Furthermore, the analytical methods were often not clearly reported and justified. Information regarding the methods for dealing with skewed or missing data and/or how to address population heterogeneity was often missing. Approximately 40% of included studies did not report currency, reference year or conversion methodologies. Although three studies stated that they adopted societal perspectives, productivity costs were not included in the analyses (Byford et al., 2007; Lynch et al., 2010; Wang et al., 2011); instead, included societal costs appeared as education sector costs (Byford et al., 2007) or time and travel costs (Lynch et al., 2010). This is possibly because the target populations were adolescents who were not yet in the workforce (Byford et al., 2007; Wang et al., 2011). Having said that, the cost-effectiveness of interventions might be underestimated given that the societal costs associated with EDs are substantial (Ling et al., 2016, Stuhldreher et al., 2012, 2015). Another limitation is that conclusions regarding the long-term cost-effectiveness of the interventions, especially in ED treatment, cannot be made since only 4 out of 13 studies included time frames beyond two years.
Of the 13 studies, eight evaluated were cost-effectiveness analyses alongside clinical trials. While some trials in EDs provided useful cost results, they did not include a full CFA (Agras et al., 2014; Schmidt et al., 2007). Importantly, the findings from these trial-based cost-effectiveness studies should be interpreted with caution. The differences between costs and/or outcomes across treatment arms in several studies did not reach statistical significance (Aardoom et al., 2016; Byford et al., 2007; Crow et al., 2009, 2013; Egger et al., 2016; Koran et al., 1995; Lynch et al., 2010). One reason for this may be that the sample sizes were too small to detect statistical significance in use of healthcare resources and cost measures (Briggs, 2000). Having said that, cost-effectiveness studies with low power should ideally report the CIs along with the probability of being cost-effective at different willingness to pay threshold.
However, model-based cost-effectiveness studies are useful accompaniments to trial-based studies for both prevention and treatment of EDs. Such research then provides important information to decision-makers as to whether interventions represent good value for money (Sculpher et al., 2006). However, there is an important gap for modelled cost-effectiveness studies especially for ED treatment, with only one out of nine studies using this type of analysis. A very important consideration, particularly for modelled evaluations in this field, is that the synthesis of clinical effectiveness data should be presented adequately.
Directions for future research
While the current review has demonstrated that the evidence base of economic evaluations for interventions targeting EDs has doubled from 2011 to 2017 (albeit with a small total number of studies), several evidence-based interventions for prevention and treatment of EDs have not been examined for cost-effectiveness. In particular, CBT has strong evidence as a treatment intervention, which has been found to be superior to non-intervention control in BN treatment (Hay et al., 2009). Family-based treatment is also an evidence-based intervention for the treatment of young people with AN and is commonly provided in many contexts including Australia (Couturier et al., 2013; Hughes et al., 2014; Watson and Bulik, 2013). Importantly, only two cost-effectiveness studies were identified of treatment for BED and there have been no cost-effectiveness studies of treatment for other types of Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) ED including OSFED or UFED. Given that the prevalence of these disorders is far higher than those of AN and BN, in both community and clinical samples, this would appear to be a major shortcoming of the existing evidence base. Further research is needed to investigate the cost-effectiveness of these interventions.
Another gap in the existing evidence base is that cost-effectiveness studies of existing interventions, both preventive and treatment, have been almost exclusively confined to studies involving females. Findings from recent epidemiological studies demonstrated that there are substantial increases in the population health burden of ED behaviour in males and BED is relatively common in males (Mitchison et al., 2014; Mitchison and Mond, 2015). Furthermore, there is increasing concern associated with the prevalence and adverse effects of ‘muscularity-oriented disordered eating’ (Murray et al., 2016). Given that, the cost-effectiveness of intervention involving males is an important direction for future research.
Finally, cost-effectiveness studies of interventions for ED appear to be confined, thus far, to clinical preventive and treatment approaches. Given that there is increasing awareness of the role that health promotion and early intervention programmes play on the health burden of ED (Mond, 2016), it is important that further research is needed to establish the effectiveness and cost-effectiveness of these types of programmes.
Conclusion
In conclusion, this review found growth in terms of number of cost-effectiveness studies for prevention and treatment of EDs published in the past 6 years. This is an encouraging finding given that there were very few studies identified in the previous review (Stuhldreher et al., 2012). However, methodological differences between the studies have limited their comparability. It is, therefore, difficult to formulate general conclusions on which interventions offer the best value for money. There is an urgent need for further research evaluating the cost-effectiveness credentials of evidence-based ED interventions. Moreover, future research of cost-effectiveness in EDs should adhere to established reporting standard, such as CHEERs checklist, to ensure high-quality reporting and conduct of cost-effectiveness studies.
Footnotes
Acknowledgements
The authors acknowledge Miss Jenifer David for proof reading of the manuscript.
Declaration of Conflicting Interests
L.K.-D.L., P.H. and C.M. were authors of one study that was included in the review. In the past 5 years, P.H. has received sessional fees and lecture fees from the Australian Medical Council, Therapeutic Guidelines publication and New South Wales Institute of Psychiatry and royalties/honoraria from Hogrefe and Huber, McGraw Hill Education, BioMed Central, PLOS Medicine and Blackwell Scientific Publications.
Funding
The study was funded by a National Health and Medical Research Council (NHMRC) Centre for Research Excellence Grant (APP1041131). The views expressed in this study are solely those of the authors and do not reflect the views of the NHMRC. L.K.-D.L. is a recipient of CREMSI (APP1041131) and Deakin University Post-Graduate Scholarship. During the conduct of this work, CM was funded by a NHMRC Early Career Fellowship Grant (APP1035887).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
