Abstract

To the Editor
Body weight and body mass index (BMI) have a crucial impact on serum concentrations of psychotropic drugs affecting drug disposition (Paulzen et al., 2016), thereby altering treatment efficacy. However, the impact of body weight or BMI acute changes following bariatric surgery or breast reduction surgery (BRS) remains unclear. Pharmacokinetic changes following BRS might be associated with changes in hormonal homoeostasis and diminished distribution volume for lipophilic agents while the gastrointestinal tract remains intact. There are no data in the literature regarding pharmacokinetic changes following BRS hitherto. We therefore report a clinically intriguing case with a female patient on stable antipsychotic treatment with clozapine with increased serum concentrations after she underwent BRS.
Mrs A. is a 57-year-old outpatient with residual schizophrenia (International Classification of Diseases, 10th Revision [ICD-10]:F20.5). Her body weight was stable over the last years with a BMI of 38.75 kg/m2. She was in good condition under a stable treatment regimen with clozapine 325 mg, aripiprazole 15 mg, paroxetine 40 mg and pirenzepine 100 mg per day without adverse drug reactions (ADRs) except hypersalivation. Due to bilateral gigantomasty (ICD 10:N62.0) with orthopedic complaints, she underwent BRS in August 2015. A total of 6.450 g (14.22 lb) of breast tissue was removed, leading to a BMI reduction of 2.231 kg/m2 (postoperative BMI: 36.33 kg/m2). In the outpatient setting, she complained of tiredness and reduced concentration. Blood tests revealed slight changes in alanine transaminase (43 U/L, ref. range <35 U/L) and gamma-glutamyl transpeptidase levels (42 U/L, ref. range <40 U/L). We also detected enhanced clozapine and norclozapine serum concentrations (926.0 and 374.0 ng/mL) (Table 1). Accordingly, the dosage was reduced to 300 mg and later on to 250 mg. Nevertheless, enhanced clozapine and norclozapine concentrations remained (1192.0 and 530.0 ng/mL), while tiredness persisted.
Serum concentrations and dose-adjusted serum concentrations before and after breast reduction surgery.
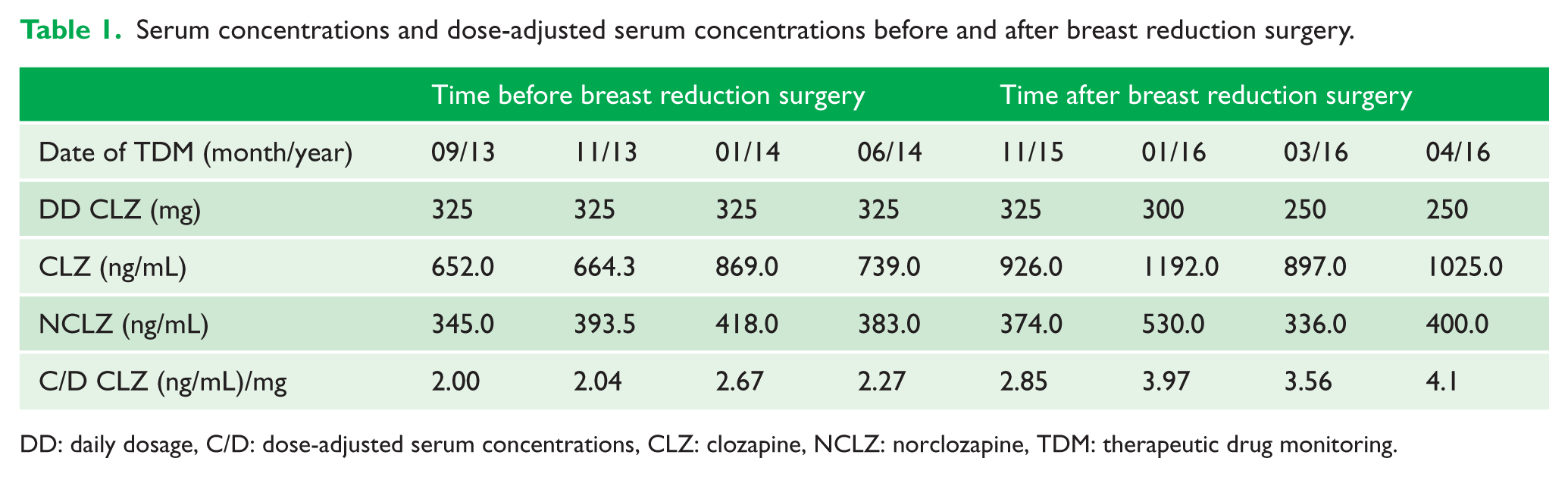
DD: daily dosage, C/D: dose-adjusted serum concentrations, CLZ: clozapine, NCLZ: norclozapine, TDM: therapeutic drug monitoring.
In times of increasing BMIs and excess fat accumulation, the number of weight loss operations has massively increased. The postoperative care may encounter beyond the surgical complications additional challenges, including pharmacokinetic alterations of ongoing pharmacotherapies. Alterations have been described for bariatric surgeries (Padwal et al., 2010). However, data for BRS are lacking. In our patient, a massive BMI reduction led to highly increased serum concentrations of clozapine associated with tiredness and reduced concentration. The ADRs subsided after clozapine dose adjustment. We suggest that the reduction in the distribution volume of clozapine, driven by the fat tissue reduction, underlies this pharmacokinetic finding. Associations of high clozapine drug concentrations with an increased occurrence of ADRs have been previously reported. Clinicians should be aware of pharmacokinetic alterations following weight loss operations.
Footnotes
Declaration of Conflicting Interests
The research study did not receive funds or support from any source. Gerhard Gründer has served as a consultant for Boehringer Ingelheim (Ingelheim, Germany), Cheplapharm (Greifswald, Germany), Eli Lilly (Indianapolis, Ind, USA), Lundbeck (Copenhagen, Denmark), Ono Pharmaceuticals (Osaka, Japan), Roche (Basel, Switzerland), Servier (Paris, France) and Takeda (Osaka, Japan). He has served on the speakers’ bureau of Eli Lilly, Gedeon Richter (Budapest, Hungary), Janssen Cilag (Neuss, Germany), Lundbeck, Roche, Servier and Trommsdorf (Aachen, Germany). He has received grant support from Boehringer Ingelheim and Roche. He is co-founder of Pharma Image GmbH (Düsseldorf, Germany) and Brainfoods UG (Selfkant, Germany). He reports no conflict of interest with this publication. Georgios Schoretsanitis received grant from the bequest ‘in memory of Maria Zaoussi’, State Scholarships Foundation, Greece, for clinical research in psychiatry for the academic year 2015–2016. Michael Paulzen and André Kirner-Veselinovic declare no conflicts of interest.
Ethical Approval
Ethical approval for this work was provided by the local regulatory authority (Registration number 244/16).
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
