Abstract
Objective:
It is unclear whether blockade of the angiotensin system has effects on mental health. Our objective was to determine the impact of angiotensin converting enzyme inhibitors and angiotensin II type 1 receptor (AT1R) blockers on mental health domain of quality of life.
Study design:
Meta-analysis of published literature.
Data sources:
PubMed and clinicaltrials.gov databases. The last search was conducted in January 2017.
Study selection:
Randomized controlled trials comparing any angiotensin converting enzyme inhibitor or AT1R blocker versus placebo or non-angiotensin converting enzyme inhibitor or non-AT1R blocker were selected. Study participants were adults without any major physical symptoms. We adhered to meta-analysis reporting methods as per PRISMA and the Cochrane Collaboration.
Data synthesis:
Eleven studies were included in the analysis. When compared with placebo or other antihypertensive medications, AT1R blockers and angiotensin converting enzyme inhibitors were associated with improved overall quality of life (standard mean difference = 0.11, 95% confidence interval = [0.08, 0.14], p < 0.0001), positive wellbeing (standard mean difference = 0.11, 95% confidence interval = [0.05, 0.17], p < 0.0001), mental (standard mean difference = 0.15, 95% confidence interval = [0.06, 0.25], p < 0.0001), and anxiety (standard mean difference = 0.08, 95% confidence interval = [0.01, 0.16], p < 0.0001) domains of QoL. No significant difference was found for the depression domain (standard mean difference = 0.05, 95% confidence interval = [0.02, 0.12], p = 0.15).
Conclusions:
Use of angiotensin blockers and inhibitors for the treatment of hypertension in otherwise healthy adults is associated with improved mental health domains of quality of life. Mental health quality of life was a secondary outcome in the included studies. Research specifically designed to analyse the usefulness of drugs that block the angiotensin system is necessary to properly evaluate this novel psychiatric target.
Keywords
Introduction
Psychological illness has a considerable burden on global health and is often a challenge to treat. Remission of symptoms may present formidable difficulties, and full remission is only achieved by a small population of persons and usually after several courses of treatment (Global Burden and Disease Study, 2015; Kessler, 2012). This is aggravated by the lack of novel interventions capable of ameliorating depressive symptoms (Berk et al., 2011; Carvalho et al., 2014), and increasing attention and efforts are being paid to the repurposing of diverse and already developed drugs to psychiatric disorders.
Drugs that act on the angiotensin system, such as the antihypertensives angiotensin I converting enzyme (ACE) inhibitors and angiotensin II type 1 receptor (AT1R) blockers, have been suggested to possess antidepressive properties (Bortolato et al., 2016; Ping et al., 2014). Angiotensin II is a compound that is produced via the cleavage of angiotensin I by ACE and acts in the central nervous system via two receptor types, AT1R and AT2R. The AT2R plays a role in neurodevelopment while the AT1R is implicated in the regulation of the cellular stress response. Drugs that decrease angiotensin II activity have pleiotropic mechanisms of action that suggest potential efficacy as novel psychotropic agents for neuropsychiatric disorders (Benicky et al., 2011; Borrajo et al., 2014; De Souza et al., 2015; Kume et al., 2012; Li et al., 2010) and neuroprotection (Cernes et al., 2011). For instance, stimulation of AT1Rs expressed in the paraventricular nucleus of the hypothalamus contributes to the release of corticotrophin-releasing hormone, resulting in an over-activation of the hypothalamic-pituitary-adrenal (HPA) axis (Benicky et al., 2011). Blockade of AT1R in humans down-regulate HPA axis activation and decreases inflammation while also directly inhibiting microglial activation (Borrajo et al., 2014).
Evidence is emerging of the role of the angiotensin system in mood and related disorders. The angiotensin AT1R blocker valsartan induces antidepressant and anxiolytic-like effects in the chronic unpredictable model of depression in mice (Ping et al., 2014). The ATR blocker candesartan seems to display anti-manic efficacy in preclinical models (De Souza et al., 2015). Polymorphisms of angiotensin genes appear associated with mood and psychotic (Ancelin et al., 2013; Zhang et al., 2014) disorders. Such polymorphisms may be associated with depression and increased activity of ACE. Several large, high-quality clinical trials have also been conducted employing these drugs in several cardiologic and neurological conditions, measuring quality of life, including mental health related quality of life, as a secondary outcome.
Given the high burden of mental illness worldwide, it is imperative to continue to improve the quality and quantity of available treatments. To further investigate potential of ACE inhibitors and AT2R blockers in psychiatric disorders, we performed a systematic review and meta-analysis of all randomized, placebo or non-angiotensin-related agent controlled trials of ACE inhibitors and AT2R blockers with a mental health quality of life related outcome in asymptomatic populations. Our objective was to clarify the psychotropic potential of these drugs with the hypothesis that medications that decrease or block angiotensin II activity would improve quality of life related to psychological domains.
Methods
We adhered to the recommended guidelines for systematic reviews and meta-analyses of interventional studies statement (PRISMA) and to the Cochrane Collaboration (Jp, 2011). Two authors (D.J.B. and B.S.F.) performed the literature search, made decisions on inclusion and data extraction, and assessed quality control according to a pre-specified protocol (available under request).
Search strategy
We conducted a systematic search for all possibly eligible peer-reviewed articles using PubMed and clinicaltrials.gov on 5 January 2017. Studies were limited to those published in English. No year or country restrictions were used (Higgins and Green, 2011). The Boolean terms used for the electronic database search were [(angiotensin converting enzyme OR angiotensin receptor blocker OR angiotensin converting enzyme inhibitors OR captopril OR enalapril OR losartan OR ace inhibitors OR angiotensin converting enzyme inhibitors OR AT1 receptor antagonist OR candesartan OR benazepril OR cilazapril OR fosinopril OR imidapril OR lisinopril OR moexipril OR perindopril OR quinalapril OR ramipril OR espirapril OR trandolapril OR zofenopril OR candesartan OR eprosartan OR irbesartan OR olmesartan OR telmisartan OR valsartan) AND (trial OR randomised OR randomized) AND (quality of life)]. Study selection eligibility and exclusion criteria were pre-specified.
Eligibility criteria
Studies included in this meta-analysis met the following inclusion criteria: (1) adult subjects without any major physical symptoms such as heart failure or cardiovascular disorders, which could lead to an improvement of mental health related quality of life with drugs that block the angiotensin system due to an improvement of physical symptoms and not due to a primary effect in psychiatric or psychological symptoms, (2) randomized controlled trials (RCTs) of any ACE inhibitor or AT1R blocker versus placebo or a non-ACE inhibitor or a non AT1R blocker, (3) studies with a parallel design, and (4) studies with analysis intention-to-treat or on treatment analyses. Exclusion criteria were as follows: (1) duplicate reports, (2) studies regarding subjects with major physical symptoms, (3) studies that measured QoL with a non-validated scale, or (4) studies that measured QoL with a scale that only addressed physical domains.
General outcomes
Our aim was to verify if medications that decrease angiotensin II activity improve quality of life related to psychological domains. The scales assessed were the Psychological General Wellbeing Index, Short Form 36 Questionnaire and the Symptom Rating Test. Although well validated, the Goteborg Quality of Life Instrument (GQL) (Tibblin et al., 1990) was not included because it does not thoroughly assess mental domains, instead focussing primarily on physical well-being, and a total score for overall QoL is not provided. The scales included are summarized as follows.
Psychological General Wellbeing Index
The Psychological General Wellbeing (PGWB) index, used in eight studies (Applegate et al., 1991; Croog et al., 1990; Dahlof et al., 1997, 2005; Omvik et al., 1993; Rake et al., 2001; Rasmussen et al., 2006; Steiner et al., 1990), is designed to assess six domains to determine quality of life: anxiety, depressed mood, positive well-being, self-control, general health and vitality (Wiklund and Karlberg, 1991). The index consists of 22 questions, with each question being scored from 1 to 6, giving a total minimum of 22 and maximum of 132, with a higher score indicating better outcomes. The validity and reliability of this scale has been well documented (Wiklund and Karlberg, 1991). We used only the scores for anxiety, depressed mood, positive well-being and total quality of life.
Short Form 36 Questionnaire
The Short Form 36 Questionnaire (SF-36), constructed for the Medical Outcomes Study, assesses 8 domains: physical functioning, social functioning, bodily pain, general physical health, general mental health, emotional functioning and general health perceptions (Ware and Sherbourne, 1992). Only the score for general mental health and total quality of life were included in this meta-analysis. Domains vary in the number of items and maximum scores, however, higher scores describe better quality of life. The SF-36 has been documented as both valid and reliable (Mchorney et al., 1994; Russo et al., 1998), and was used in two studies (Kusek et al., 1996; Williams et al., 2008).
Symptom Rating Test
One study (Fletcher et al., 1990) used the Symptom Rating Test (SRT) as a means to assess QoL. The SRT consists of a checklist of 38 items, covering the subscales of anxiety, depression, inadequacy, and somatic and has been determined to be valid and reliable (Kellner and Sheffield, 1973). Conversely to other scales, a lower score in the SRT indicates better QoL. To ensure data harmonization, the effect size was multiplied by (–1).
Data extraction
Data were extracted using a predesigned form. Where there was missing data and email addresses provided, authors of original studies were contacted and the necessary information was requested. We extracted the following data: sample size, age, sex, medication in use, study population, design of the study and blood pressure at both baseline and follow-up. Overall QoL scores and suitable domains were assessed by scales at both at baseline and at follow-up. For the analyses, we extracted data regarding sample size, mean and standard deviation (SD). When applicable, standard errors and quartile ranges were manually converted to SD using appropriate statistical formulae.
Risk of bias assessment
We assessed the risk of bias in all included studies as suggested by the Cochrane Collaboration (Higgins and Green, 2011). The evidence was assessed and ranked according to the grading of recommendations assessment, development and evaluation (GRADE) for high risk of bias, imprecision, indirectness, heterogeneity and publication bias (Andrews et al., 2013).
Publication bias
Studies with negative results are less likely to be published than studies with positive results (Carvalho et al., 2016a; 2016b; Higgins and Green, 2011). We analysed the funnel plot graph for verifying publication bias, which is a scatter plot of the SMD against a measure of study size, calculated the Egger test (Egger et al., 1997; Soeken and Sripusanapan, 2003) and employed the Orwin’s fail-safe N test to quantify the number of possible negative omitted studies that would be required to make our results non-significant (p ⩾ 0.05). Furthermore, we implemented Duval and Tweedie’s trim and fill test as a final method to adjust for any potential publication bias.
Statistical analysis
We employed Comprehensive Meta-analysis software (CMA Version 2) to calculate the standard mean difference (SMD) for continuous outcome measures and the 95% CI. The SMD was calculated as the standardized difference in means between both arms in the follow-up. The standardized mean difference expresses the size of the intervention effect in each study relative to the variability observed in that study. The direction of the SMD values was positive if subjects showed an improvement in QoL with ACE inhibitor or AT1R blocker, and negative if they showed improvement with placebo or other non-ACE inhibitor or AT1R blocker drugs. For trials that did not report SD or other dispersion measure, the SD of similar trials was imputed to ensure standardizing constants were comparable across trials as per the method of Furukawa (Furukawa et al., 2006).
Heterogeneity across studies was assessed using the Cochran Q test, a weighted sum of the squares of the deviations of individual study SMD estimates from the overall estimate, with a p value of <0.10 considered significant (i.e. showing heterogeneity)(Higgins and Green, 2011; Lau et al., 1997). Inconsistency across studies was evaluated using the I2 metric, which describes the percentage of the variability in effect estimates that is due to heterogeneity rather than sampling error(Higgins and Green, 2011) and is considered substantial when greater than 50% (Bowden et al., 2011).
Results
We initially identified 693 studies, all of which had an English abstract. Of those, 635 were excluded through title and abstract screening due to irrelevance, not meeting inclusion criteria or being a duplicate study, leaving 61 studies for a full text review. We additionally excluded 18 studies (Boissel et al., 1995; Breed et al., 1992; Bursztyn et al., 1993; Chan et al., 1997; Croog et al., 1994; Degl’Innocenti et al., 2004; Frimodt-Moeller et al., 1991; Glik et al., 1990; Herrick et al., 1989; Hill et al., 1985; Kundu et al., 1990; Levine et al., 1987, 1995; Malmqvist et al., 2000; Os et al., 1991; Van Bortel et al., 2005; Weir et al., 1996b, 2005) due to insufficient data required for this analysis. To supplement data, attempts to contact the authors of three studies who provided email addresses were made but none replied (Malmqvist et al., 2000; Van Bortel et al., 2005; Weir et al., 2005). Twenty studies did not use a validated scale for measuring QoL and were hence excluded (Bracchetti et al., 1990; Conlin et al., 1998; Croog et al., 1986; Ebert and Kirch, 1999; Fagher et al., 1991; Fletcher et al., 1992; Freytag et al., 2001; Hu and Zhu, 1999; Lacourciere, 1988; Lewis et al., 1996; McMahon et al., 1990; Neaton et al., 1993; Oparil et al., 1996; Paran et al., 1996; Perry et al., 1994; Tanser et al., 2000; Tedesco et al., 1999; Testa et al., 1993; Weir et al., 1996a; Yodfat et al., 1985). One study was excluded as it employed a scale that assessed a purely physical domain of QoL (Ostergren et al., 1996). Three studies analysed drugs that decrease angiotensin activity in both arms (Breeze et al., 2001; Chien et al., 2002; Marques et al., 2015), four studies were crossover randomized control trials (Dahlof and Dimenas, 1992; Handa et al., 1991; McCorvey et al., 1993; Palmer et al., 1992), and five studies were not available in English (Andreeva et al., 2004; Carre et al., 1988; Gralec et al., 1997; Metelitsa et al., 1996; Siegrist et al., 1991). In total, 11 studies fulfilled our inclusion criteria (Figure 1).
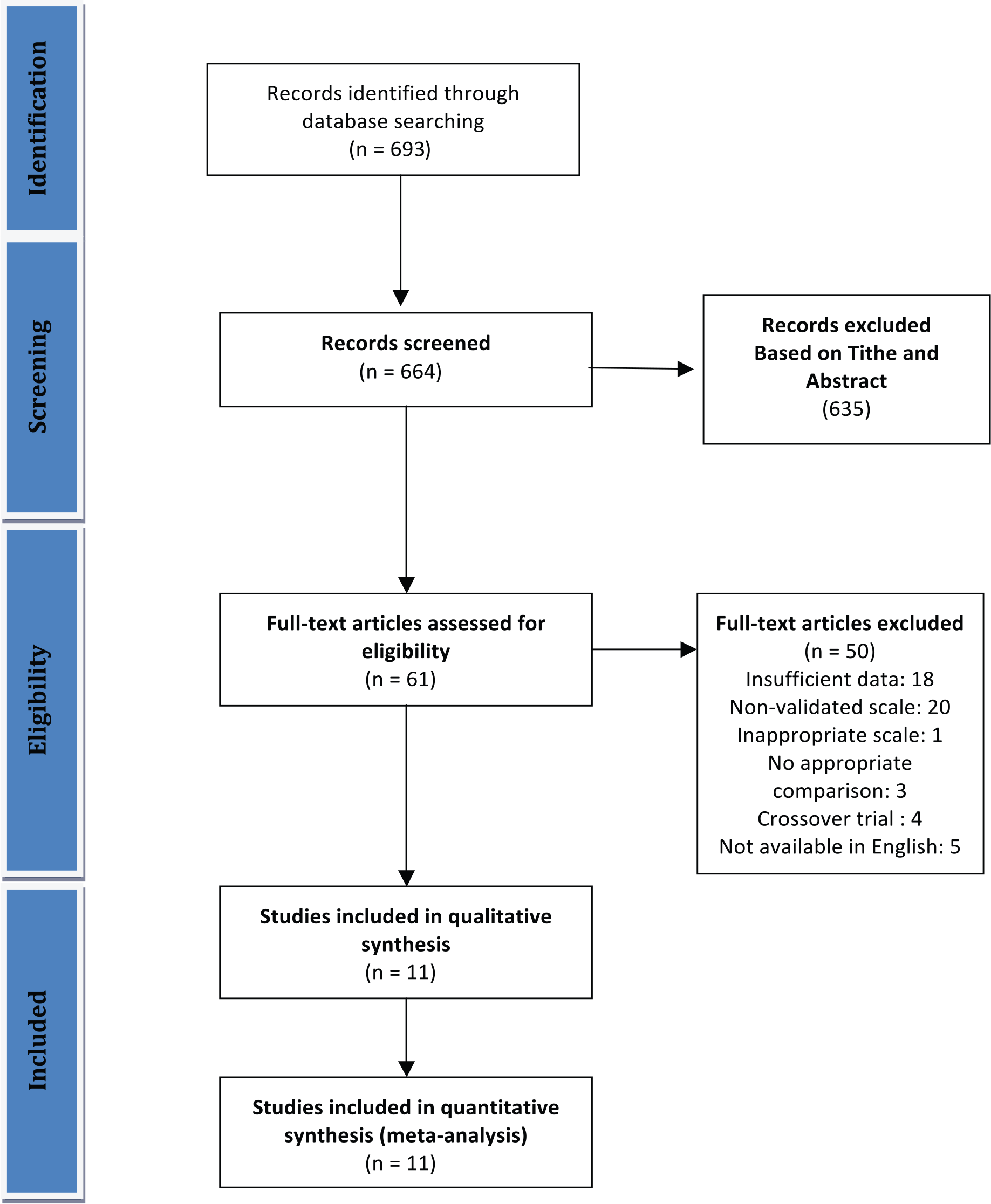
Flow diagram of the systematic review and meta-analysis.
Tables 1 and 2 summarize the characteristics and demographics of the included studies. The studies were published from 1990 to 2005 and varied in sample size from 84 to 947. The specific type and dose of ACE inhibitors and AT1R blockers used varied between studies, as did the class of anti-hypertensive used as the non-angiotensin agent comparison. All included studies were RCTs. Ten were double-blind, and one was single-blind. A placebo washout period (between 4 and 8 weeks) was used in 9 of the studies (Applegate et al., 1991; Croog et al., 1990; Dahlof et al., 1997, 2005; Omvik et al., 1993; Rake et al., 2001; Rasmussen et al., 2006; Steiner et al., 1990). The follow-up time varied from 6 weeks to 7 years from the start of active treatment to the completion of the trial. The mean participant age varied from 48.6 to 70.3 years. All medications were comparable in terms of blood pressure control, and attrition percentages were within reasonable limits and were even between treatment groups (range 1.6–19.7%).
Key characteristics of included studies.
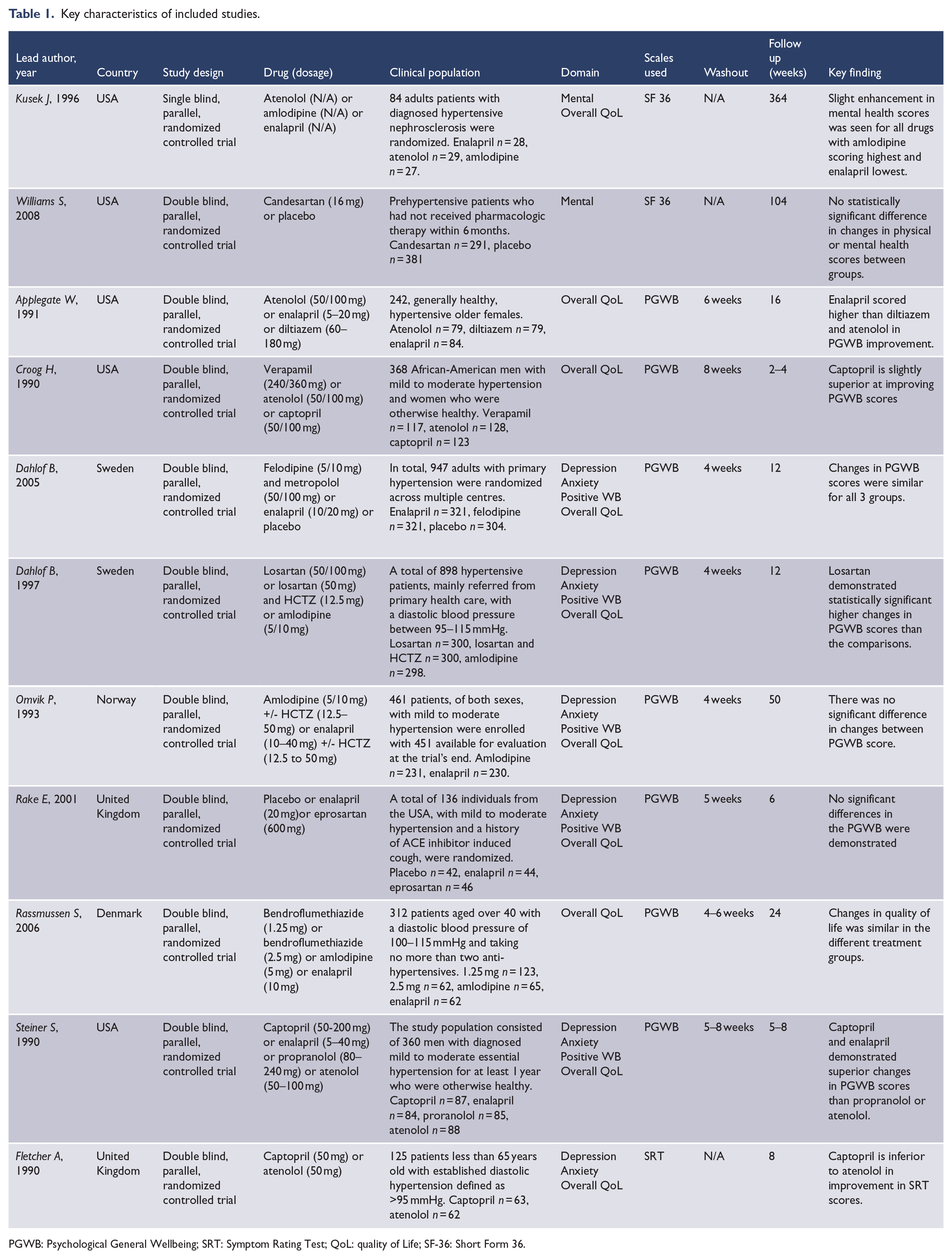
PGWB: Psychological General Wellbeing; SRT: Symptom Rating Test; QoL: quality of Life; SF-36: Short Form 36.
Demographics of included studies.
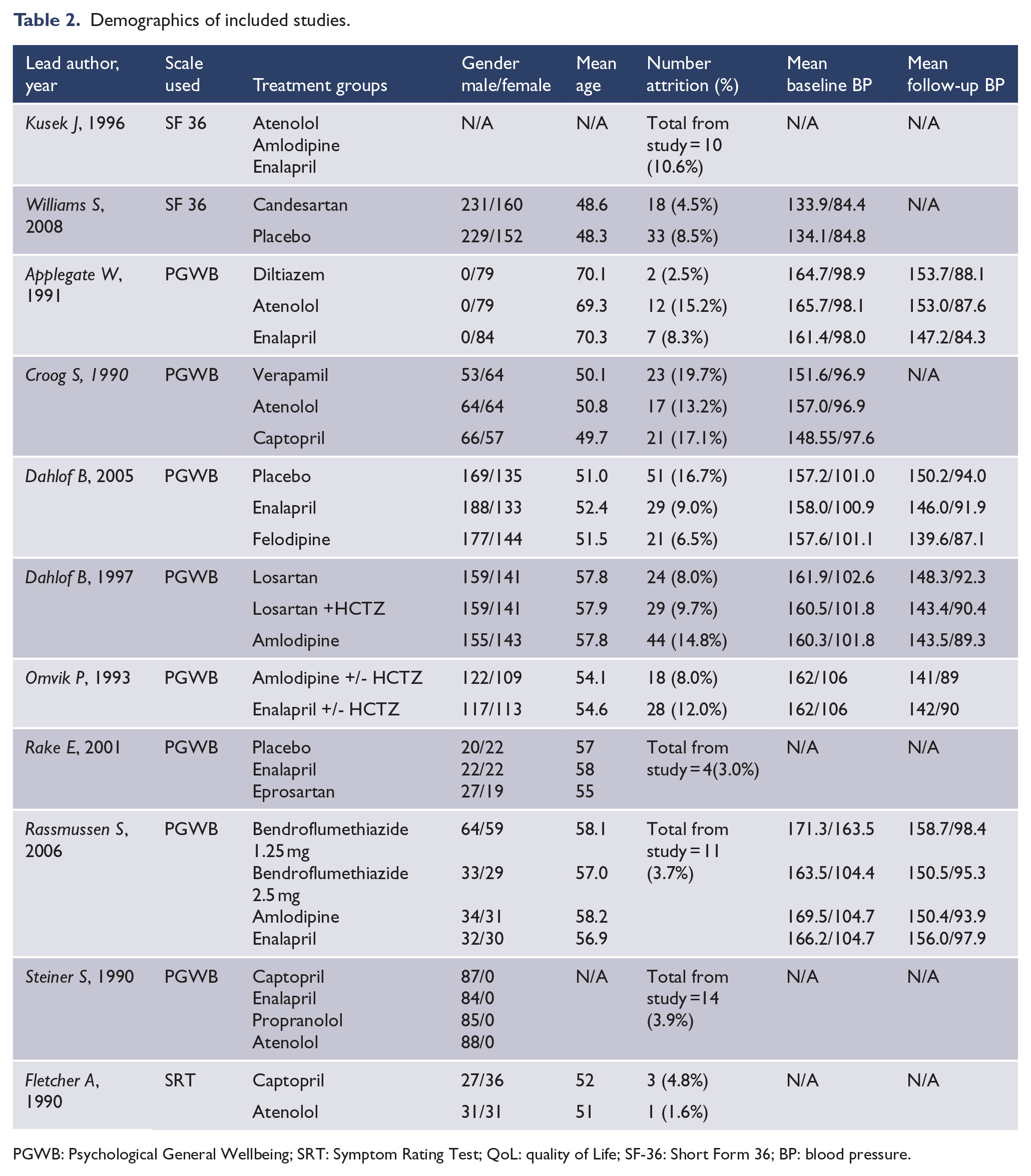
PGWB: Psychological General Wellbeing; SRT: Symptom Rating Test; QoL: quality of Life; SF-36: Short Form 36; BP: blood pressure.
ACE inhibitors and AT1R blockers improve mental-related quality of life
Overall, random-effects meta-analysis showed that, when compared to placebo or other anti-hypertensive medications, blockade of the angiotensin system improved overall quality of life (SMD = 0.11, 95% CI = [0.08, 0.14], p < 0.001). Subjects receiving angiotensin system blockade or inhibitors consistently presented better scores in the positive wellbeing (SMD 0.11, 95% CI = [0.05, 0.17], p < 0.001), mental (SMD 0.15, 95% CI = [0.06, 0.25], p < 0.001) and anxiety (SMD 0.08, 95% CI = [0.01, 0.16], p = 0.03) domains of QoL. Results for the depression domain indicated no significant differences between blockers of the angiotensin system and the comparison (SMD 0.05, 95% CI = [–0.02, 0.12], p = 0.15) (Figure 2).
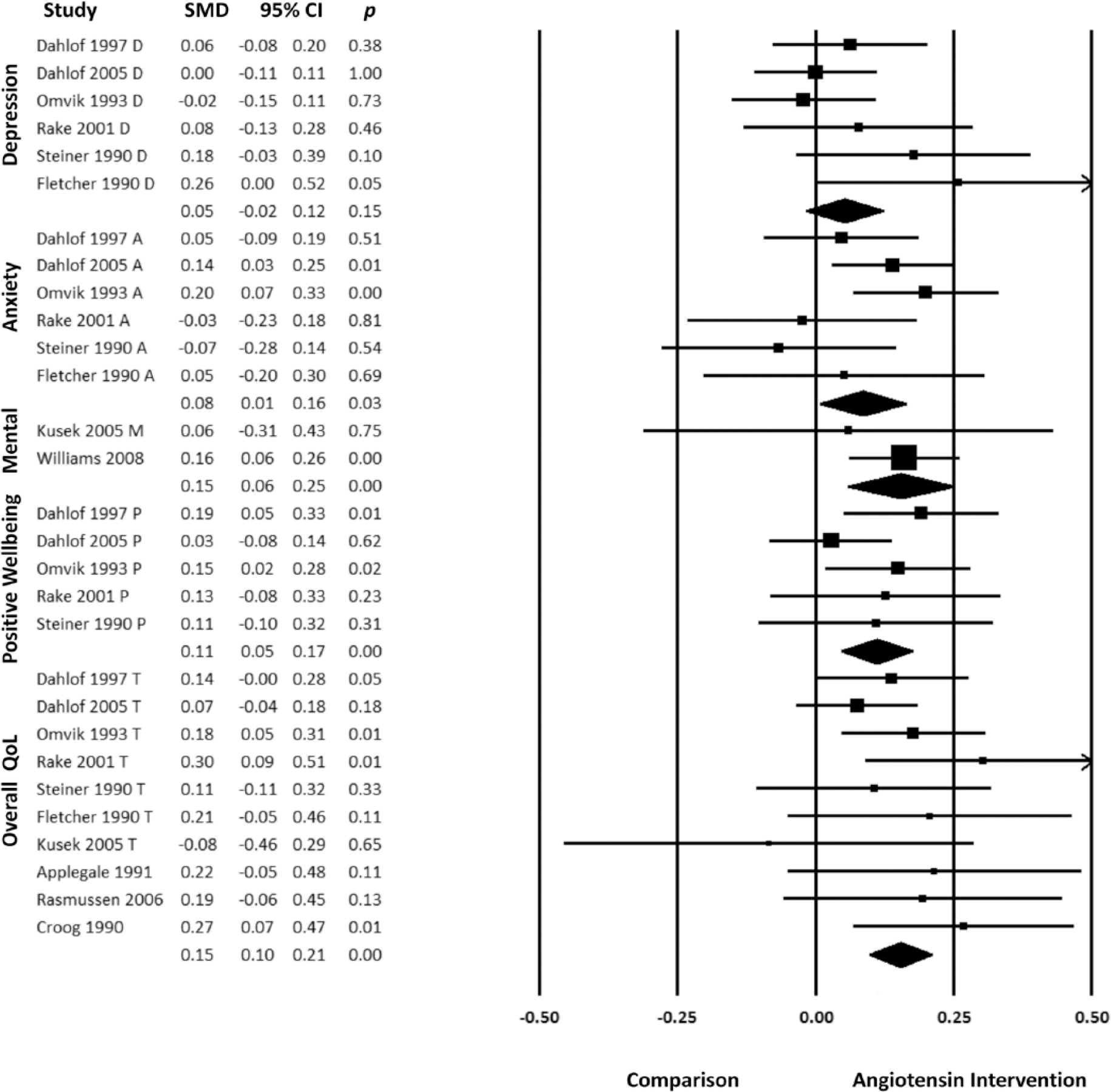
Forest plot for random effects meta-analysis.
Publication bias
The visual inspection of the funnel plot showed it to be symmetrical and without evidence of publication bias, and the Egger’s test was not significant (p = 0.50). Although the Duval and Tweedie’s trim and fill method suggested two possible negative missing studies, the overall SMD remained almost identical when adjusted for the two missing studies (from 0.109 to 0.101). Furthermore, The Orwin’s fail-safe N test yielded 353 studies as the number of negative studies necessary to turn our positive results into a null result. These analyses suggest that our positive results are unlikely to be due to publication bias.
Sensitivity analyses
The heterogeneity between studies was low (I2 = 13.267%, p = 0.263). We conducted a sensitivity analysis in all meta-analyses excluding studies one at a time to determine the robustness of the analyses and to verify if one study was responsible for the results. No single study explained the results, and the results remained unaltered in all cases.
Quality of evidence
The included studies were published between 1990 and 2005, before the practice of registering a clinical trial, employment of guidelines for reporting clinical trials and the exercise of publishing the research protocol. Therefore, the quality of the evidence was ‘unclear’ in most situations despite the large sample size of many of these randomized trials (Ghanizadeh and Hedayati, 2013). Attrition bias was the best described type of bias, with most studies rated as low risk of bias in this category as there were either no drop-outs or the missing data was balanced across groups, and intention-to-treat analyses were used (Ghanizadeh and Hedayati, 2013; Haghighi et al., 2014) (Table 3). In accordance to the low heterogeneity and most of the studies not providing enough detail to ascertain the risk of bias, the GRADE assessment was ‘unclear’ for all considered outcomes.
Risk of bias assessment of the included studies.
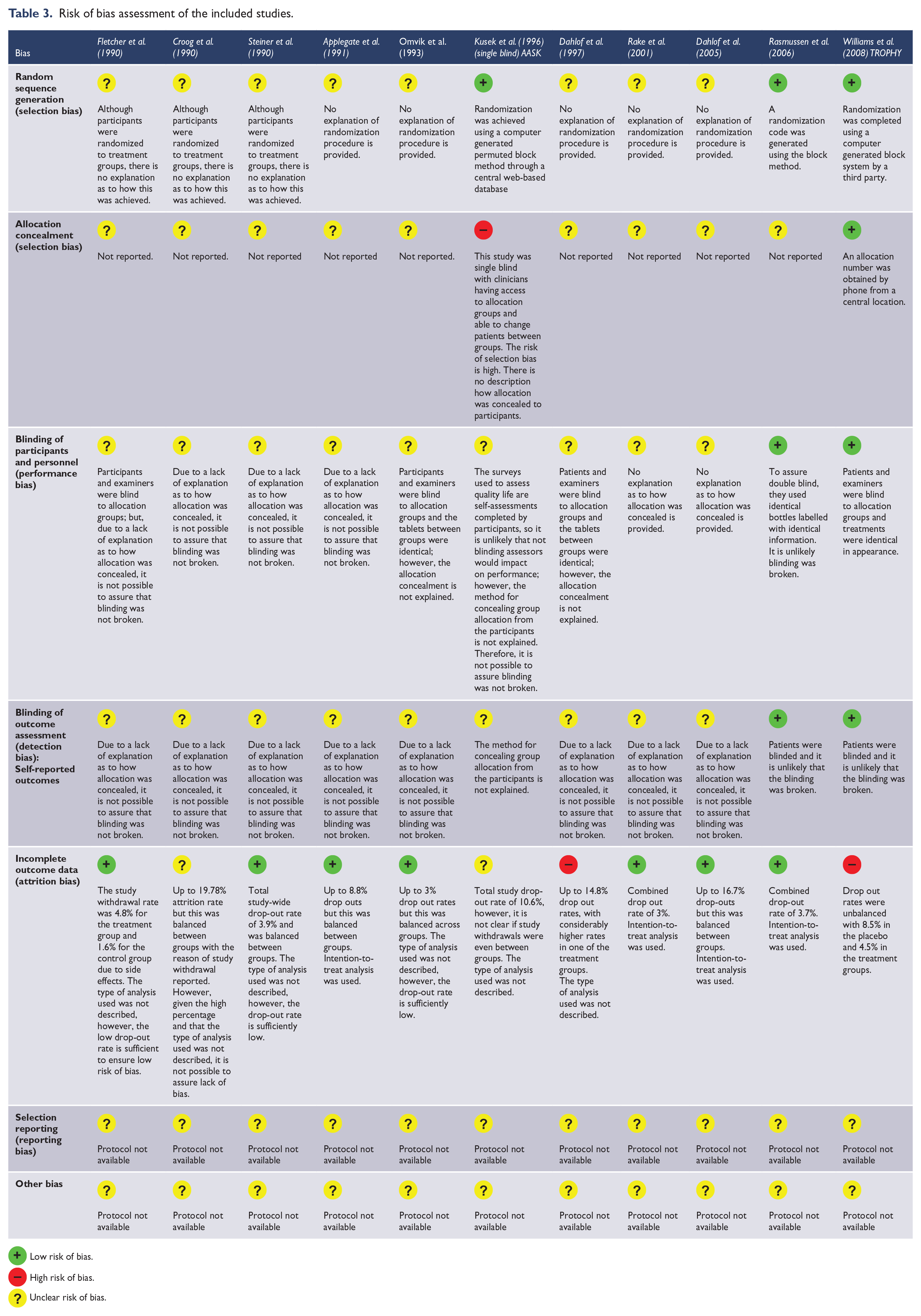
 Low risk of bias.
Low risk of bias.
 High risk of bias.
High risk of bias.
 Unclear risk of bias.
Unclear risk of bias.
Discussion
This meta-analysis shows that blockade of the angiotensin system is associated with improved mental-related QoL, with subjects receiving AT1R blockers or ACE inhibitors consistently presenting better scores in the positive wellbeing, mental and anxiety domains of QoL. Results for the depression domain indicated no effect of AT1R blockers and ACE inhibitors. This may be affected by the much lower rates of depression in the general population compared to other domains such as anxiety, and, in this context, the changes in quality of life of the domains included reflect mostly prevention and not treatment. Results showed that the magnitude of the improvement in mental-related QoL is small, however, since the studied populations were mostly asymptomatic and without psychiatric disorders, it would be unrealistic to expect larger effects in prevention. The inclusion of only randomized clinical trials with placebo or a non-angiotensin antihypertensive drug as a comparison treatment in an asymptomatic population speaks in favour of a true effect of drugs that target the renin-angiotensin system on mental-related QoL and significantly decreases the odds of our results being due to confounders.
Despite considerable effort and investment from industry, few novel compounds for treating depression have become available over the last decade (Fernandes et al., 2017). As such, drugs already developed for the management of organic illness are increasingly being investigated and repurposed to treat psychiatric disorders. Depression is characterized by a systemic low-grade inflammatory state. Drugs that decrease inflammation, such as statins (Salagre et al., 2016), celecoxib (Na et al., 2014) and N-acetyl-cysteine (NAC) (Fernandes et al., 2016) have shown beneficial effects on depression at a meta-analytic level of evidence. Both ACE inhibitors and AT1R blockers can decrease neuroinflammation by decreasing angiotensin II activity and have therefore been proposed as novel therapeutic targets for inflammatory brain diseases as well as for cognitive disorders (Benicky et al., 2011; Borrajo et al., 2014; De Souza et al., 2015, Kume et al., 2012; Li et al., 2010).
The idea that ACE inhibitors could improve mood is not novel. In the early 1980s several case reports revealed that captopril promoted possible mood elevation in patients with unipolar depression (Germain and Chouinard, 1988) and mood benefits also reported with lisinopril (Hertzman et al., 2005). Their antidepressant effects can be further inferred by mood elation in healthy volunteers taking enalapril (Cohen et al., 1984).
The evidence for potential benefits of decreasing angiotensin activity also exists in epidemiological studies. The Nord-Trøndelag Health Study (HUNT) (Johansen et al., 2012), a large population study of 55,472 participants conducted in Norway, showed an important trend in favour of reduction of depressive symptoms as assessed by the Hospital Anxiety and Depression Rating Scale in patients with systemic hypertension taking an ACE inhibitor or AT1R blockers, when compared to people with untreated systemic hypertension. Interestingly, those on beta-blockers or on calcium-channels antagonists did not show a reduction of depressive symptoms compared to the untreated systemic hypertension group. These results were replicated in another study by Boal et al. (2016) which examined mood related hospital admissions of 144,660 patients treated with antihypertensive monotherapy for a five-year follow-up. Those on no antihypertensive medication and thiazide diuretics showed no significant difference. This again suggests that ACE inhibitor and AT1R blockers pharmacological benefits in depression are independent of its anti-hypertensive effects. More recently, in a 5-year follow-up from the Geelong Osteoporosis Study (GOS), the incidence of a first episode of depression was 5.3% in men with no use of ACE inhibitors, and zero on those on ACE inhibitors (Williams et al., 2016).
Our article also has some limitations. First, meta-analysis is retrospective research in nature, affected by the methodological rigor of the studies included, comprehensiveness of search strategies, and possibility of publication bias. We tried to minimize the probability of bias by doing a thorough search for data, however, 18 potentially eligible studies were not included because necessary information was lacking. Second, individual studies varied greatly with respect to the specific ACE inhibitor and AT1R blockers employed. We cannot know if all ACE inhibitors and AT1R blockers drugs possess the same class effects. Importantly, we cannot know if ACE inhibitors and AT1R blockers are equally beneficial, since no direct comparison of ACE inhibitors to AT1R blockers were done. Based on pharmacodynamics, it is likely that AT1R blockers would be potentially more beneficial than ACE inhibitors, since they provide the best antagonism to angiotensin II. Moreover, this meta-analysis cannot provide evidence of efficacy of ACE inhibitor and AT1R blockers drugs for treatment of depression, since the population studied was largely asymptomatic. However, since our study would mimic effects on mental-related QoL in what is mostly a preventive fashion, we can hypothesize that subjects on ACE inhibitor or AT1R blockers drugs presented better mental-related QoL on the follow-up due to a decrease in the incidence of psychiatric symptoms. Finally, since all subjects on the included studies have primary hypertension, we cannot extrapolate our results to those without hypertension.
In conclusion, asymptomatic persons taking ACE inhibitor or AT1R blockers drugs presented better mental-related quality of life outcomes. Our meta-analytic findings provide further support for the angiotensin system as a novel therapy target, and that drugs that decrease activity of angiotensin, specifically ACE inhibitor and AT1R blockers drugs, can lead to benefits from a psychiatric perspective. If ACE inhibitor and AT1R blocker drugs are effective in the treatment or prevention of anxiety or depression remains to be determined by randomized, placebo-controlled clinical trials specifically designed for these outcomes.
Footnotes
Acknowledgements
We thank all the authors of the included papers and study participants.
Declaration of Conflicting Interests
M.B. has received grant/research support from the NIH, Cooperative Research Centre, Simons Autism Foundation, Cancer Council of Victoria, Stanley Medical Research Foundation, MBF, NHMRC, Beyond Blue, Rotary Health, Geelong Medical Research Foundation, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Meat and Livestock Board, Organon, Novartis, Mayne Pharma, Servier and Woolworths; has been a speaker for Astra Zeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck, Merck, Pfizer, Sanofi Synthelabo, Servier, Solvay and Wyeth; and served as a consultant to Allergan, Astra Zeneca, Bioadvantex, Bionomics, Collaborative Medicinal Development, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck Merck, Pfizer and Servier. The remaining authors declare no conflicts of interest regarding this manuscript. None of these agencies had any role in this manuscript.
Funding
B.S.F. is supported by a postdoctoral fellowship from Deakin University, Australia. C.A.K. is supported by a postdoctoral scholarship from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES; Brazil). A.F.C. is supported by a research fellowship award from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; Brazil). J.Q. is a 1A CNPq Research Fellow. M.B. is supported by a NHMRC Senior Principal Research Fellowship 1059660.
