Abstract
Objective:
Interventions are required to redress the disproportionate tobacco-related health burden experienced by persons with a mental illness. This study aimed to assess the efficacy of a universal smoking cessation intervention initiated within an acute psychiatric inpatient setting and continued post-discharge in reducing smoking prevalence and increasing quitting behaviours.
Method:
A randomised controlled trial was undertaken across four psychiatric inpatient facilities in Australia. Participants (N = 754) were randomised to receive either usual care (n = 375) or an intervention comprising a brief motivational interview and self-help material while in hospital, followed by a 4-month pharmacological and psychosocial intervention (n = 379) upon discharge. Primary outcomes assessed at 6 and 12 months post-discharge were 7-day point prevalence and 1-month prolonged smoking abstinence. A number of secondary smoking-related outcomes were also assessed. Subgroup analyses were conducted based on psychiatric diagnosis, baseline readiness to quit and nicotine dependence.
Results:
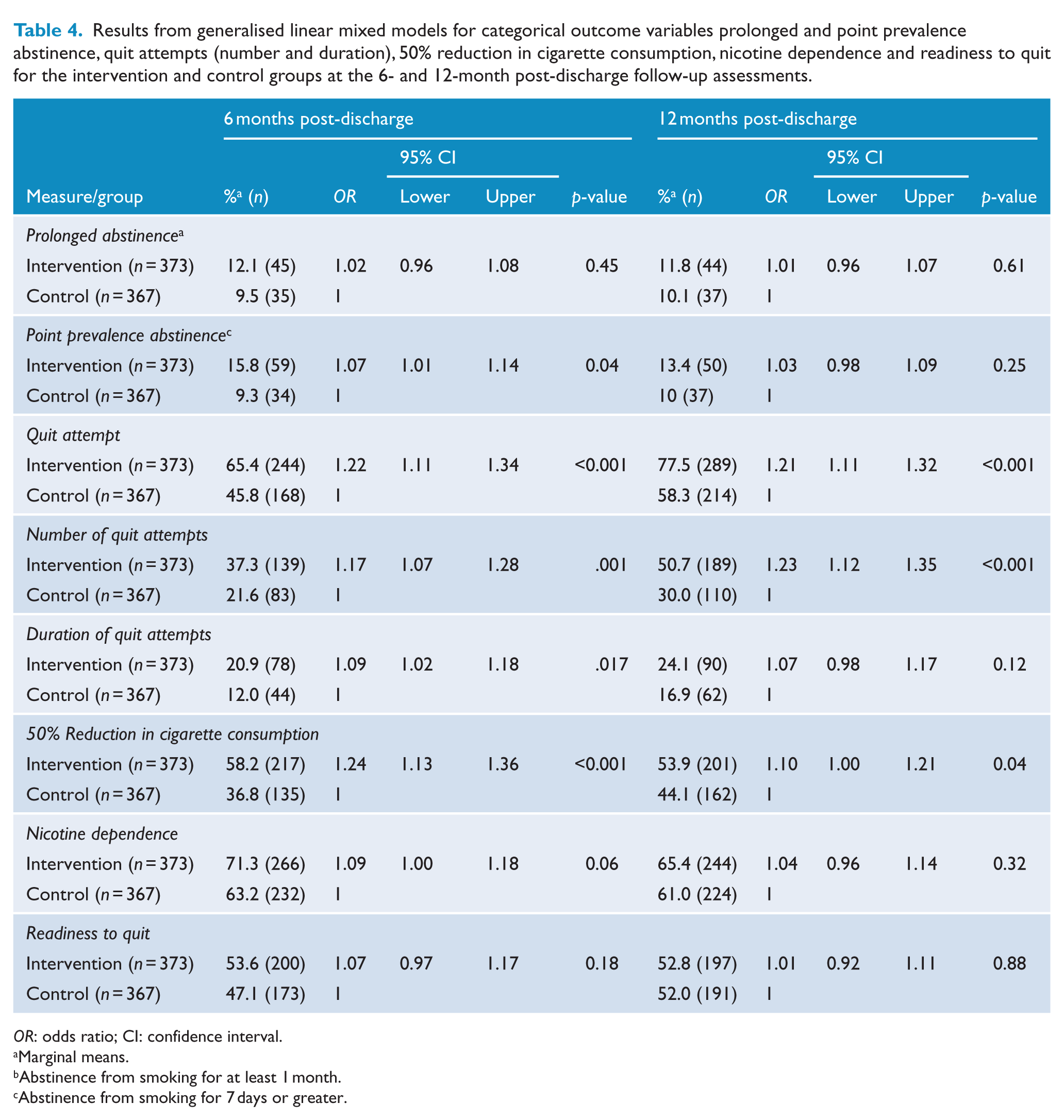
Seven-day point prevalence abstinence was higher for intervention participants (15.8%) than controls (9.3%) at 6 months post-discharge (odds ratio = 1.07, p = 0.04), but not at 12 months (13.4% and 10.0%, respectively; odds ratio = 1.03, p = 0.25). Significant intervention effects were not found on measures of prolonged abstinence at either 6 or 12 months post-discharge. Differential intervention effects for the primary outcomes were not detected for any subgroups. At both 6 and 12 months post-discharge, intervention group participants were significantly more likely to smoke fewer cigarettes per day, have reduced cigarette consumption by ⩾50% and to have made at least one quit attempt, relative to controls.
Conclusions:
Universal smoking cessation treatment initiated in inpatient psychiatry and continued post-discharge was efficacious in increasing 7-day point prevalence smoking cessation rates and related quitting behaviours at 6 months post-discharge, with sustained effects on quitting behaviour at 12 months. Further research is required to identify strategies for achieving longer term smoking cessation.
Background
The prevalence of tobacco smoking among persons with a mental illness has remained unchanged for the past 20 years (Steinberg et al., 2015; Szatkowski and McNeill, 2015) and is currently at least two to three times higher than that of the general population in a number of high-income countries, including Australia (Lawrence et al., 2009; Office for National Statistics, 2014). As a consequence, this group continues to experience disproportionate levels of preventable tobacco-related morbidity and mortality, and a reduced life expectancy of up to 25 years (Colton and Manderscheid, 2006). In addition, smoking prevalence is associated with mental illness severity and acuity, with up to 88% of those with psychotic disorders (De Leon and Diaz, 2005; McClave et al., 2010) and psychiatric inpatients (De Leon and Diaz, 2005; Stockings et al., 2013) reported to be smokers. Despite higher levels of nicotine dependence (Bowden et al., 2011; Lasser et al., 2000), smokers with a mental illness wish to quit smoking (Siru et al., 2009; Stockings et al., 2013), and attempt to do so at similar rates to smokers without a mental illness (McClave et al., 2010), using both pharmacological and psychosocial support strategies (Schuck et al., 2016; Stockings et al., 2014a). Despite this, a greater difficulty in successful quitting (Lasser et al., 2000; Lawrence et al., 2009) and a persistently elevated smoking prevalence (Steinberg et al., 2015; Szatkowski and McNeill, 2015) suggest that this group has not benefited as much from population-wide smoking cessation interventions relative to those without a mental illness (Cook et al., 2014; Williams et al., 2013). To address this inequity, the development of tailored smoking cessation intervention approaches have been recommended for this population group (Gelenberg et al., 2008; Royal College of Physicians and Royal College of Psychiatrists, 2013), and are a recognised clinical and public health priority (Royal College of Physicians and Royal College of Psychiatrists, 2013).
Hospitals have been identified as an opportune setting to initiate smoking cessation interventions (Dohnke et al., 2012), particularly following the introduction of smoke-free policies and the associated requirement to treat patient nicotine dependence (Fiore et al., 2008; New South Wales Department of Health, 2009). In general hospital settings, an inpatient stay has been associated with increased motivation to quit and likelihood of cessation (Duffy et al., 2010; Williams and Jones, 2012). The provision of smoking cessation support post-discharge has been shown to further increase the likelihood of successful quitting in general hospital patients by up to 37% (Rigotti et al., 2012). For persons with a mental illness, systematic review evidence suggests that a smoke-free psychiatric hospitalisation can have a positive impact on smoking behaviours including reduced daily cigarette consumption, motivation to quit and number of cessation attempts (Stockings et al., 2014b). However, without ongoing smoking cessation support, these effects are suggested to dissipate within 3 months of discharge (Stockings et al., 2014b).
Only three randomised controlled trials (RCTs) have assessed the efficacy of integrating inpatient smoking care with post-discharge cessation support for adult smokers with a mental illness (Hickman et al., 2015; Stockings et al., 2014a; Prochaska et al., 2014). Prochaska et al. (2014) demonstrated the efficacy of such an approach in a RCT involving predominantly Caucasian and medically insured smokers (n = 224) admitted to one acute psychiatric facility in the United States. All admitted patients were offered nicotine replacement therapy (NRT) during their hospitalisation. While inpatients, intervention group participants additionally received a computer-assisted, stage-tailored smoking cessation programme and cessation counselling. Post-discharge intervention was offered over 6 months and involved repetition of the stage-tailored computer programme at months 3 and 6. If during this time participants were assessed by the programme as ready to quit, a 10-week supply of nicotine patches was also offered. Patients randomised to the intervention condition were significantly more likely than usual care controls (brief cessation advice and quitline referral) to be abstinent for at least 7 days at 6 (14.4% vs 6.5%), 12 (19.4% vs 10.9%) and 18 (20.0% vs 7.7%) months post hospital discharge. A feasibility and replication controlled trial of the same intervention involving ethnically diverse, uninsured smokers with a mental illness (n = 100) admitted to both acute and non-acute units of an urban public hospital achieved similar effect sizes at 6 (17.5% vs 8.5%) and 12 (26.2% vs 16.7%) months post-discharge (Hickman et al., 2015).
A third study involving patients from one inpatient psychiatric facility in Australia assessed the efficacy, via RCT, of a universal smoking cessation intervention initiated during admission and continued immediately following discharge (Stockings et al., 2014a). Intervention components were proactively offered to all smokers, irrespective of clinical (e.g. psychiatric diagnosis) or smoking (e.g. readiness to quit) characteristics. Inpatient smokers (n = 205) were randomised to receive either a 4-month post-discharge smoking cessation intervention (comprising telephone counselling and combination NRT) or usual care (Stockings et al., 2014a). At 4 months post-discharge, those receiving the intervention had significantly higher rates of 7-day point prevalence abstinence compared to usual care controls (11.5% vs 2%), although this difference was not sustained at 6 months post-discharge. The study concluded that increased intervention intensity was required to achieve longer term effects.
To address the equivocal findings of the limited number of reported trials, an RCT was conducted of a smoking cessation intervention initiated for all smokers admitted to four Australian acute psychiatric inpatient facilities and continued for a period of 4 months post-discharge. The primary aim was to examine differences in rates of 7-day point prevalence and 1 month prolonged smoking abstinence between intervention and control groups at 6 and 12 months post-discharge. The secondary aims were (1) to assess differences in point prevalence and prolonged abstinence according to psychiatric diagnosis, baseline readiness to quit and nicotine dependence; and (2) to examine intervention effects in terms of cigarette reduction, quit attempts, nicotine dependence and readiness to quit.
Methods
Design and setting
A two-arm, parallel group RCT was undertaken with participants recruited from four public adult inpatient psychiatric facilities in one regional health district in New South Wales, Australia. Six clinical units (20–25 beds each) were included from the four facilities. A smoke-free policy was implemented in the facilities in 2006 (New South Wales Department of Health, 2005) with clinical guidelines directing staff to offer and provide cessation care to all hospitalised smokers (New South Wales Department of Health, 2009). The study methods have been previously reported (Metse et al., 2014).
The Human Research Ethics Committees of Hunter New England Health (reference no: 11/12/14/4.02) and the University of Newcastle (reference no: H-2012-0061) approved the research, which was registered on the Australian New Zealand Clinical Trials Registry (ANZTCN: ACTRN12612001042831).
Recruitment procedure, inclusion criteria and permissions
Research staff (trained in motivational interviewing), who were not employees of the hospitals, liaised with nurse unit managers daily to identify new patients sufficiently clinically stable to be approached for the study. Study eligibility criteria were having smoked any number of cigarettes in the month prior to hospital admission (Stockings et al., 2014a), 18 years of age or above, capable of providing informed consent and having a current telephone number. Eligible patients were invited to provide written consent to participate.
Randomisation
A statistician generated a patient random allocation sequence (using permuted block randomisation with a block size of 10), concealed from recruitment staff, prior to commencement of the study. Randomisation was carried out separately by site, stratified by diagnosis (psychotic/non-psychotic) using a 1:1 allocation ratio and implemented by provision of a sequentially numbered, opaque, sealed envelope to all consenting participants immediately following baseline data collection. Participants were informed by recruiting staff whether they would receive the multimodal intervention or treatment as usual control.
Treatment conditions
Intervention
While an inpatient, in addition to standard hospital smoking cessation care (New South Wales Department of Health, 2002), all smokers allocated to the intervention group received two additional evidence-based smoking cessation treatments from research staff: self-help material (Lancaster and Stead, 2005) tailored to smokers with a mental illness and a brief 10- to 15-minute motivational interview (Lai et al., 2010) designed to promote ambivalence about smoking, guide patients towards behaviour change and/or positively reinforce intentions to address smoking. At discharge, all such participants received a study-supplied sample pack of NRT (unless medically contraindicated) that included nicotine patches (7 × 21 mg), inhalator with cartridges (6 × 10 mg), gum (30 × 4 mg) and lozenges (20 × 4 mg), and instructions for NRT use. General practitioners and/or community mental health clinicians (where applicable) were notified of their client’s involvement in the study via inclusion of information in the standard hospital discharge summary, and asked to encourage use of the cessation supports provided.
Immediately following discharge, all participants were provided up to 4 months of tailored telephone behavioural smoking counselling support (minimum of 11 contacts) (Stead et al., 2006) and an additional 12 weeks of free NRT (weekly/fortnightly supplies provided following telephone support calls) (Stead et al., 2012). Participants were assigned to a telephone counsellor with the intention of receiving continuity of care from that counsellor. Telephone counselling support was provided (~15 minutes) every week during the initial 5 weeks post-discharge, weekly or fortnightly (contingent on participant preference) during the subsequent 7 weeks, and fortnightly during the last month of the intervention period. The counselling sessions were manualised, but tailored to the individual. A motivational interviewing framework was employed to increase motivation for smoking behaviour change and encourage use of the other cessation strategies (NRT and Quitline). Systematic monitoring of counselling calls was undertaken by a mental health clinician with experience in training in motivational interviewing.
NRT (patch and oral forms) was prescribed in accordance with an evidence-based, combination NRT algorithm for highly dependent smokers (Bittoun, 2006), offered by the telephone counsellors and delivered by mail. The counselling staff prompted participants to utilise the NSW Quitline, a free government-provided service providing telephone support and quit advice. If participant consent was provided, a referral was sent to the service on their behalf.
Control
Participants allocated to the control group received routine hospital smoking cessation care only, as per the state clinical guideline (New South Wales Department of Health, 2009). Such care may have included assessment of smoking status and nicotine dependence on admission, brief advice to quit, provision of NRT while an inpatient, up to 3 days NRT supply upon discharge, referral to the NSW Quitline and/or a post-discharge smoking cessation care plan included on the discharge summary (New South Wales Department of Health, 2009). Limited and variable provision of such care has been reported previously in the facilities (Wye et al., 2010).
Data collection procedures
Baseline data pertaining to primary and secondary outcomes and smoking characteristics were collected between October 2012 and April 2014 by research staff via face-to-face interview during the period of admission. Patient clinical and demographic data were obtained from the facilities’ electronic medical record system and the baseline interview. For intervention participants only, measures of intervention uptake were collected by the telephone counsellor during each post-discharge telephone counselling call.
Follow-up outcome data were collected via computer-assisted telephone interview (CATI) at 6 and 12 months post hospital discharge (May 2013 to June 2015) – i.e. 4 and 8 months post the completion of the intervention, respectively. For those participants who self-reported 7-day point prevalence abstinence at 6 or 12 months post-discharge, carbon monoxide (CO) assessments were collected (using a Micro+ Smokerlyser), as soon as possible post CATI completion, by research staff either in participants’ homes or an enclosed public place. Research staff involved in outcome data collection, including CO assessment, were blind to participant allocation. The trial ceased at completion of follow-up data collection for all recruited participants.
Measures
Primary outcomes
The two primary outcomes were self-reported 7-day point prevalence and 1-month prolonged smoking abstinence. Patients were asked ‘when did you last smoke tobacco?’ For biochemical verification of self-reported 7-day point prevalence abstinence, expired breath CO assessments with readings <7 parts per million (ppm) were used to confirm abstinence (West et al., 2005). Participants with a reading ⩾7 ppm and those who refused/did not complete a CO assessment were classified as non-abstinent. If CO assessments were obtained >14 days after completion of the CATI interview (West et al., 2005), self-reported abstinence was used (Peckham et al., 2015).
Secondary outcomes
Secondary outcome measures were number of cigarettes smoked per day (Peckham et al., 2015; Stockings et al., 2014a), reduction in cigarettes smoked relative to baseline (proportion), quit attempts (a period of abstinence lasting at least 24 hours with the intention to quit; number and duration since hospital discharge), nicotine dependence (Fagerstrom Test for Nicotine Dependence, FTND) and readiness to quit (Readiness and Motivation to Quit Smoking Questionnaire).
Clinical and demographic information
Data collected for all admitted patients within the recruitment period from the electronic medical record system were age, gender, relationship status, Aboriginal and/or Torres Strait Islander status, primary mental health diagnosis at discharge, legal status on admission and total length of stay (days between admission and discharge) (Table 1).
Clinical and demographic characteristics of approached and not approached patients, and study participants and non-consenters.
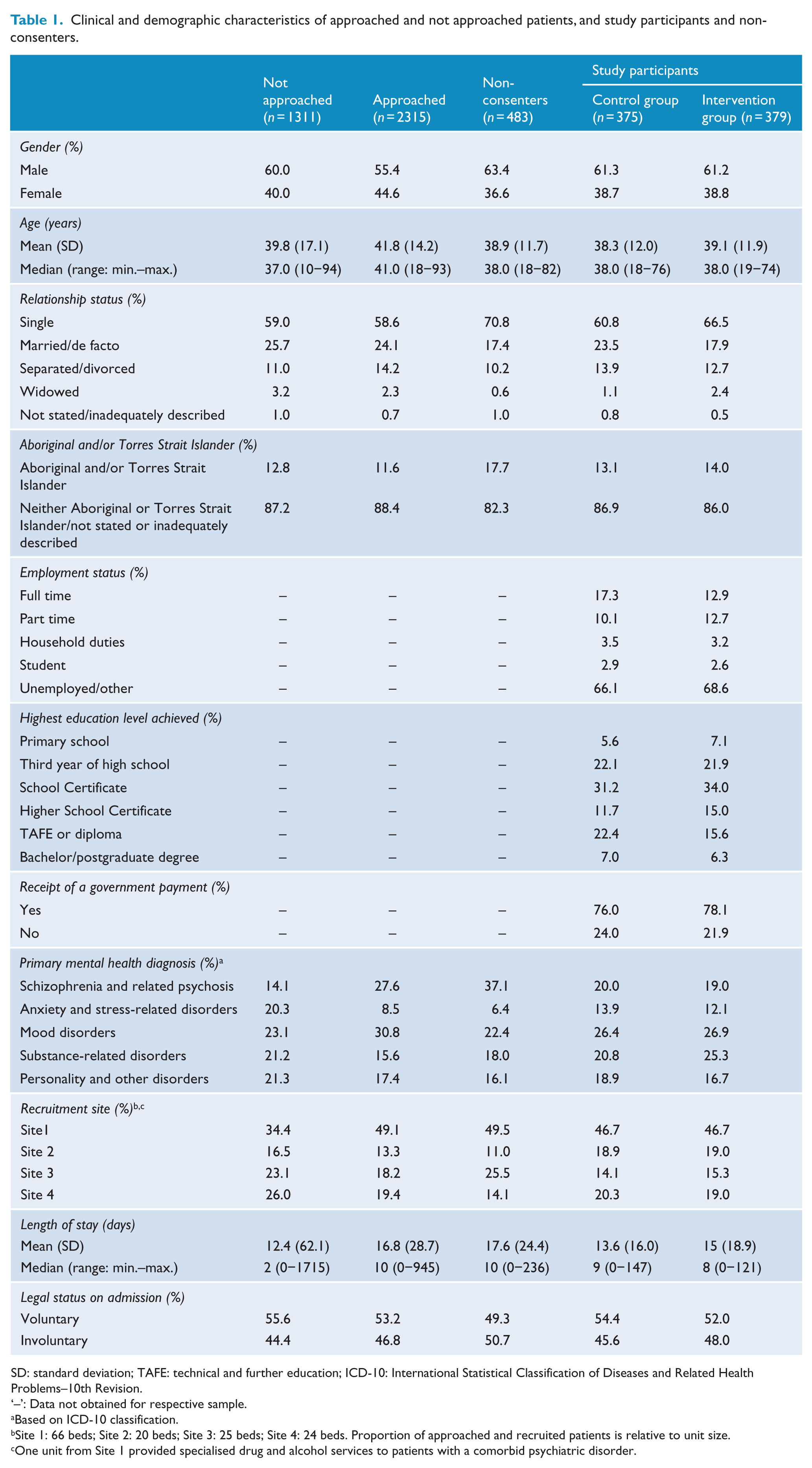
SD: standard deviation; TAFE: technical and further education; ICD-10: International Statistical Classification of Diseases and Related Health Problems–10th Revision.
‘–’: Data not obtained for respective sample.
Based on ICD-10 classification.
Site 1: 66 beds; Site 2: 20 beds; Site 3: 25 beds; Site 4: 24 beds. Proportion of approached and recruited patients is relative to unit size.
One unit from Site 1 provided specialised drug and alcohol services to patients with a comorbid psychiatric disorder.
Clinical and demographic information collected for participants only during the baseline interview were highest level of education attained, employment details, receipt of a government payment and alcohol use (Alcohol Use Disorders Identification Test–Consumption [AUDIT-C]) (Table 1).
Smoking characteristics
The following characteristics of smoking were collected for participants at baseline: smoking status, age of smoking initiation and number of years smoked (Table 2).
Participant baseline characteristics of smoking and alcohol use.
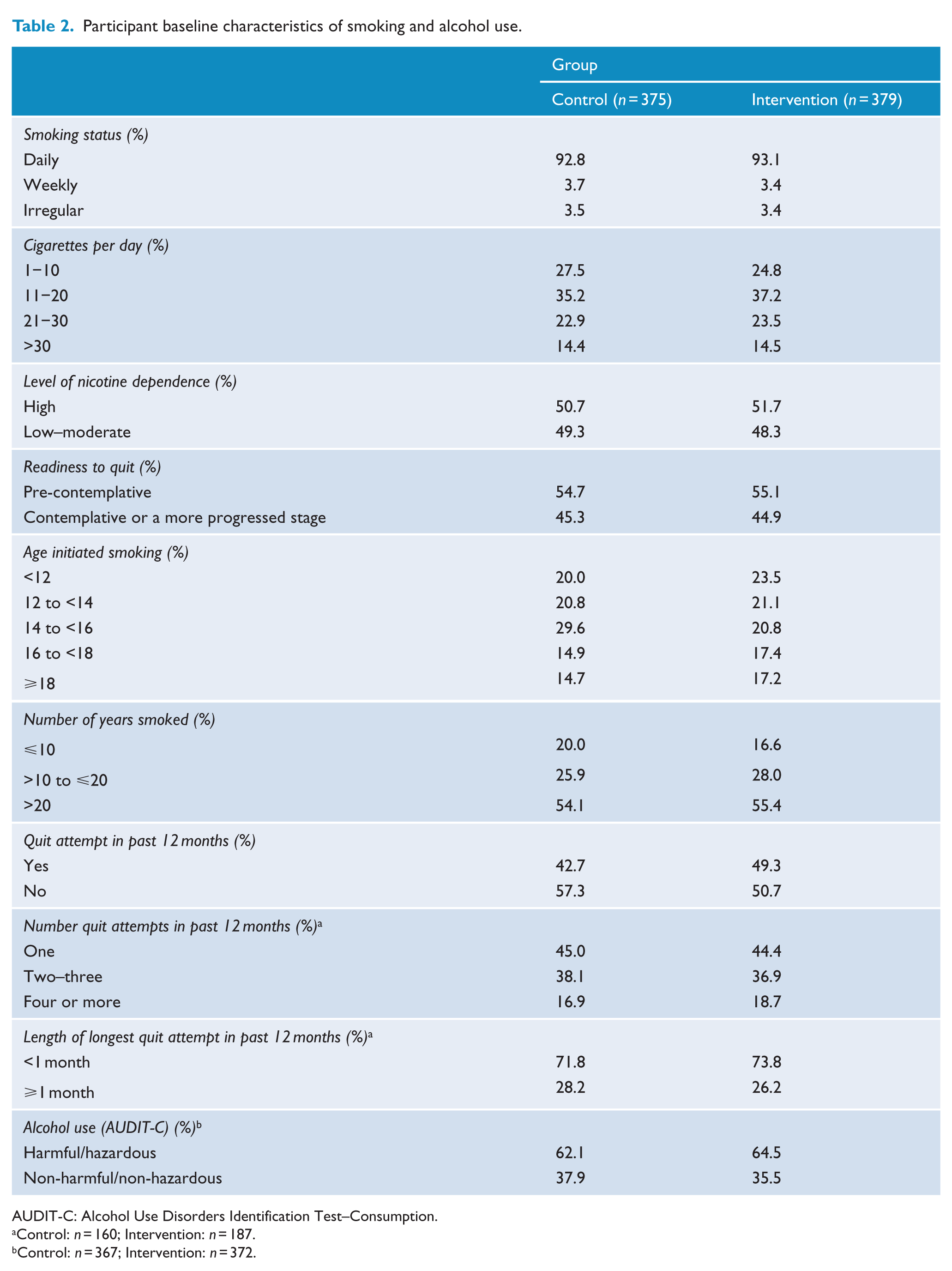
AUDIT-C: Alcohol Use Disorders Identification Test–Consumption.
Control: n = 160; Intervention: n = 187.
Control: n = 367; Intervention: n = 372.
Uptake of offered intervention
For participants allocated to the intervention group, data pertaining to the uptake of the offered post-discharge intervention components (telephone behavioural smoking counselling provided by the study, NRT and Quitline referral) were collected and have been the focus of a previous paper (Metse et al., 2016).
Variable transformation
The following secondary outcome variables were transformed from continuous to categorical format for the purpose of conducting analyses: number of quit attempts (two measures: 0 vs ⩾1; ⩽1 vs ⩾2), duration of longest quit attempt (<1 month, ⩾1 month) (Borland et al., 2012; Okoli and Khara, 2014) and nicotine dependence (low–moderate [FTND score ⩽ 5], high [FTND score ⩾ 6]). Categorical variables reduced to two levels were diagnosis (psychotic, non-psychotic), employment status (paid, unpaid), Aboriginal and/or Torres Strait Islander status (Aboriginal and/or Torres Strait Islander, neither Aboriginal or Torres Strait Islander/not stated or inadequately described), smoking status (daily smoker, weekly/irregular smoker) and readiness to quit (pre-contemplative, contemplative or a more progressed stage). Highest level of education attained was reduced to three levels (up to school certificate, beyond school certificate and up to Higher School Certificate [HSC], tertiary). To improve normality of the distribution, a log transformation was applied to the secondary outcome variable, cigarettes per day.
Analyses
Data were analysed using SPSS Statistics version 22. Descriptive statistics were used to summarise clinical, demographic and smoking (where applicable) characteristics of admitted, approached and non-consenting patients, and study participants.
Primary and secondary outcomes analyses
Outcome analyses were conducted using intention-to-treat (ITT) principles, with all participants retained in originally assigned groups. Generalised linear mixed models (GLMMs) were used to assess for differences in primary and secondary outcomes by allocation group over time. Intervention effects were determined via allocation group (intervention and control) by time (baseline, 6 and 12 months post-discharge) interactions. A compound symmetry residual covariance structure was used to model correlation associated with repeated time measurements. For outcomes where no baseline measure was applicable (i.e. 7-day point prevalence and 1-month prolonged abstinence, and proportion of cigarettes cut down), an additional set of models with main effects only were fitted. Marginal means, odds ratios (ORs) and 95% confidence intervals (CIs) were determined from the fitted models. Allocation group differences at 6 and 12 months post-discharge were assessed using simple effects.
Clinical and demographic factors significantly associated with attrition (age, Aboriginal and/or Torres Strait Islander status, education level; Table 3), determined via chi square analyses with a p-value ⩽ 0.05 were entered into all models to control for the missing at random (MAR) assumption inherent in GLMMs. Recruitment site was also entered and, where the main effect was significant, differential effects were tested using interaction terms (all two and three-way interactions with allocation group and time). Sensitivity analyses were conducted for primary outcomes, using a conservative approach where all missing data were coded as ‘not abstinent’ (National Research Council, 2010). The threshold for statistical significance for all models was set at α = 0.05.
Baseline characteristics of participants lost to follow-up at 12 months.
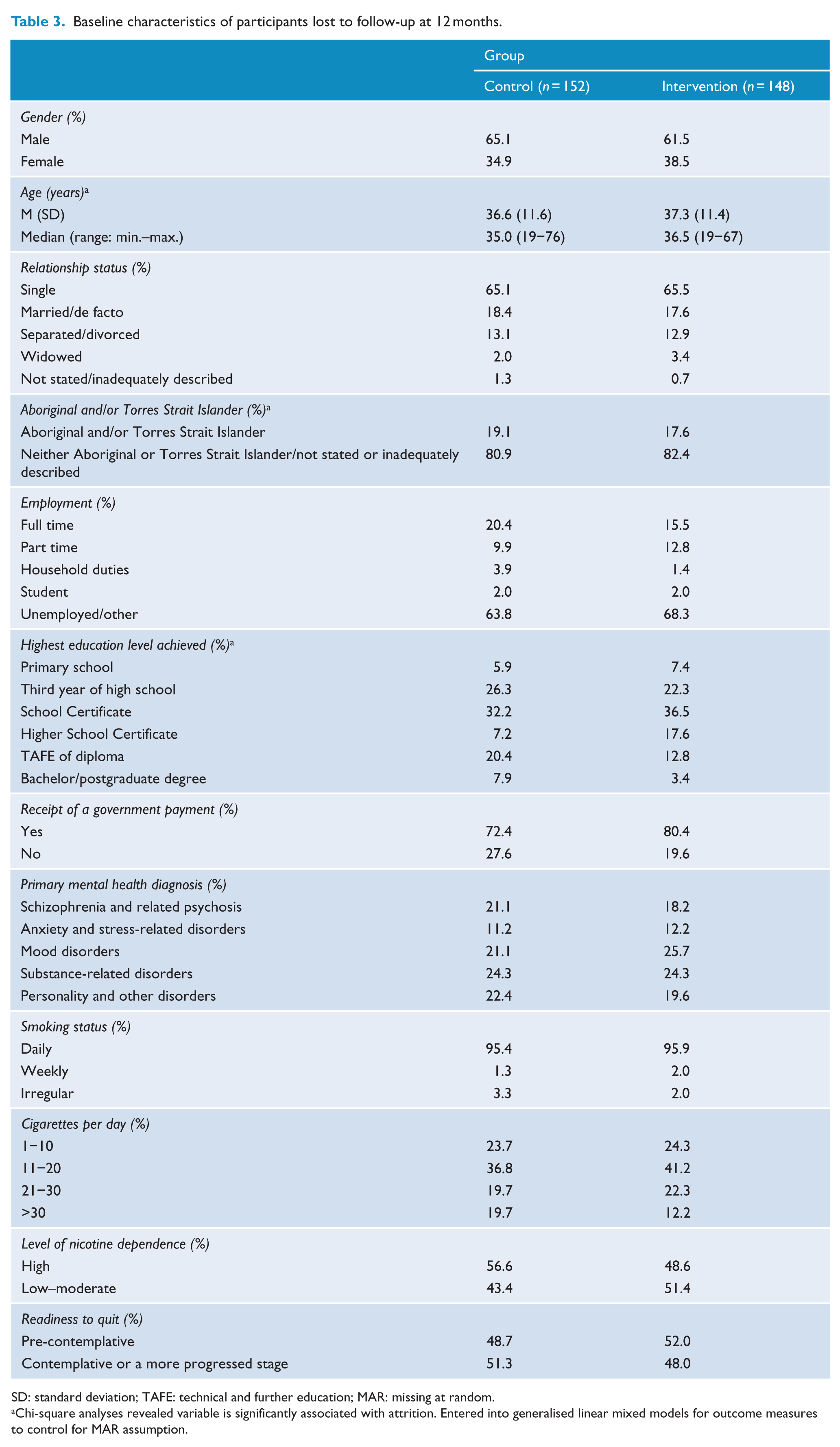
SD: standard deviation; TAFE: technical and further education; MAR: missing at random.
Chi-square analyses revealed variable is significantly associated with attrition. Entered into generalised linear mixed models for outcome measures to control for MAR assumption.
Subgroup analyses
As specified prior to trial inception, to determine whether intervention effects differed according to psychiatric diagnosis and baseline nicotine dependence and readiness to quit, subgroup analyses were undertaken for both primary outcomes at 6 and 12 months post-discharge. Differential effects were assessed via entry of interaction terms (subgroup × allocation group, subgroup × allocation group × time) into the model.
Per protocol analyses
Adopting the same statistical method described for the primary analytical approach, per protocol analyses were undertaken for the two primary outcome measures: 7-day point prevalence and 1-month prolonged abstinence, at both 6 and 12 months post-discharge. Such analyses were explorative and assessed for differences in the primary outcomes between the control group and intervention subgroups, which were defined in terms of the amount of the intervention received and calculated as a percentage using the formula: (number of calls completed + number of instances using NRT + use of Quitline)/(number of calls offered [n = 15] + resupplies of NRT offered [n = 13] + offer of proactive Quitline referral [n = 1]) × 100. Seven such per protocol models were created, with percent intervention received increasing by 5% intervals from 55% to 85%.
Results
Participants
Of 3626 patients admitted within the recruitment period, 64% (n = 2315) were approached by research staff, with patients not being approached primarily due to either a short length of stay (⩽one night; 38%), psychiatric instability for the duration of time spent as an inpatient (35%) or discharge prior to staff availability to undertake the assessment interview (27%). Of the 2315 patients approached, 2078 (90%) agreed to be assessed for study eligibility, of which 841 (40%) were ineligible, predominantly due to non-smoking status (n = 797, 95%; overall smoking prevalence: 62%). In all, 61% (n = 754) of eligible smokers consented and were randomised to the intervention (n = 379) or control group (n = 375). Follow-up rates did not differ by allocation and were as follows: for the intervention group, 6 month post-discharge = 55.9%, 12 month post-discharge = 60.9%; and for the control group, 6 month post-discharge = 58.4%, 12 month post-discharge = 59.5%. Figure 1 describes the flow of participants through the trial.
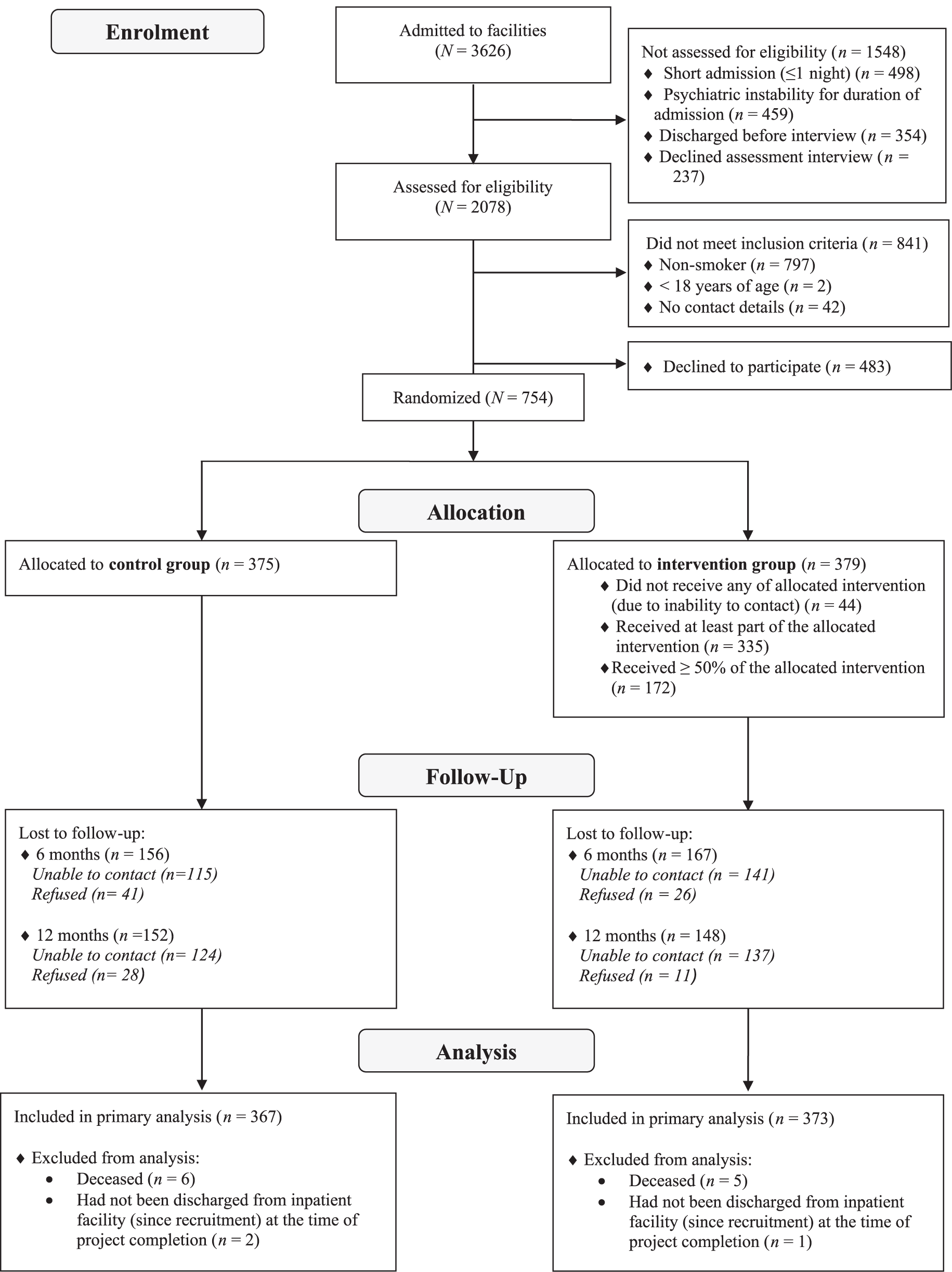
Participant flow diagram.
Clinical and demographic characteristics of approached and not approached patients, as well as non-consenters and study participants (by allocation group) are provided in Table 1. Given the large sample sizes, statistical tests were not conducted (Moher et al., 2010); however, differences in the median length of stay and diagnosis of patients were suggested for those approached and not approached to participate in the trial, and in the diagnosis between study participants and non-consenters. Participants allocated to the intervention and control groups did not differ in terms of clinical, demographic (Table 1) or smoking (Table 2) characteristics at baseline or among those lost to follow-up (Table 3).
Primary outcomes
Seven-day point prevalence abstinence
Biochemical verification tests were conducted for 68% of participants self-reporting 7-day point prevalence abstinence at 6 and 12 month post-discharge follow-up (6 months, 67% [34/51]; 12 months, 70% [40/58]). For the remaining 32%, tests were not completed due to issues contacting and reaching participants, compounded by a large geographical study area (130,000 km2). Of the completed tests, 84% were undertaken within 14 days (6 months, 71% [27/34]; 12 months, 88% [35/40]). Of the tests conducted in 14 days, 81% confirmed the participant as being abstinent. Agreement between self-reported abstinence and biochemical verification did not differ between allocation groups, χ2(1, N = 62) = 0.016, p = 0.90.
The proportions of participants abstinent for 7 days or greater were 15.8% and 9.3% for intervention and control groups at 6 month post-discharge, and 13.4% and 10.0% at 12 months, with no significant group-by-time interaction, F(1, 841) = 1.03, p = 0.31, and a borderline significant main effect of group, F(1, 841) = 3.56, p = 0.06; OR = 0.72, 95% CI = [0.42, 1.25]. Simple effects models showed that at 6 months post-discharge, those in the intervention group were significantly more likely than control participants to be abstinent (OR = 1.07, 95% CI = [1.01, 1.14]; p = 0.04) (Table 4), although this effect was not sustained at 12 months (OR = 1.03, 95% CI = [0.98, 1.09]; p = 0.25). Sensitivity analyses showed no intervention effect via the group-by-time interaction, F(2, 1467)=0.001, p = 1.00; main effect of group, F(1, 1467) = 1.76, p = 0.19; or simple effects models (6 month: OR = 1.02, 95% CI = [0.98, 1.05]; 12 month: OR = 1.02, 95% CI = [0.98, 1.05]).
Results from generalised linear mixed models for categorical outcome variables prolonged and point prevalence abstinence, quit attempts (number and duration), 50% reduction in cigarette consumption, nicotine dependence and readiness to quit for the intervention and control groups at the 6- and 12-month post-discharge follow-up assessments.
OR: odds ratio; CI: confidence interval.
Marginal means.
Abstinence from smoking for at least 1 month.
Abstinence from smoking for 7 days or greater.
Subgroup analyses
Subgroup analyses revealed that for participants allocated to the intervention group, the odds of 7-day point prevalence abstinence did not differ on the basis of psychiatric diagnosis (6 month: OR = 1.01, 95% CI = [0.91, 1.13]; 12 month: OR = 1.00, 95% CI = [0.91, 1.11]), readiness to quit (6 month: OR = 1.05, 95% CI = [0.95, 1.16]; 12 month: OR = 1.09, 95% CI = [0.98, 1.18]) or nicotine dependence (6 month: OR = 0.92, 95% CI = [0.84, 1.02]; 12 month: OR = 0.95, 95% CI = [0.99, 1.19]).
Per protocol analyses
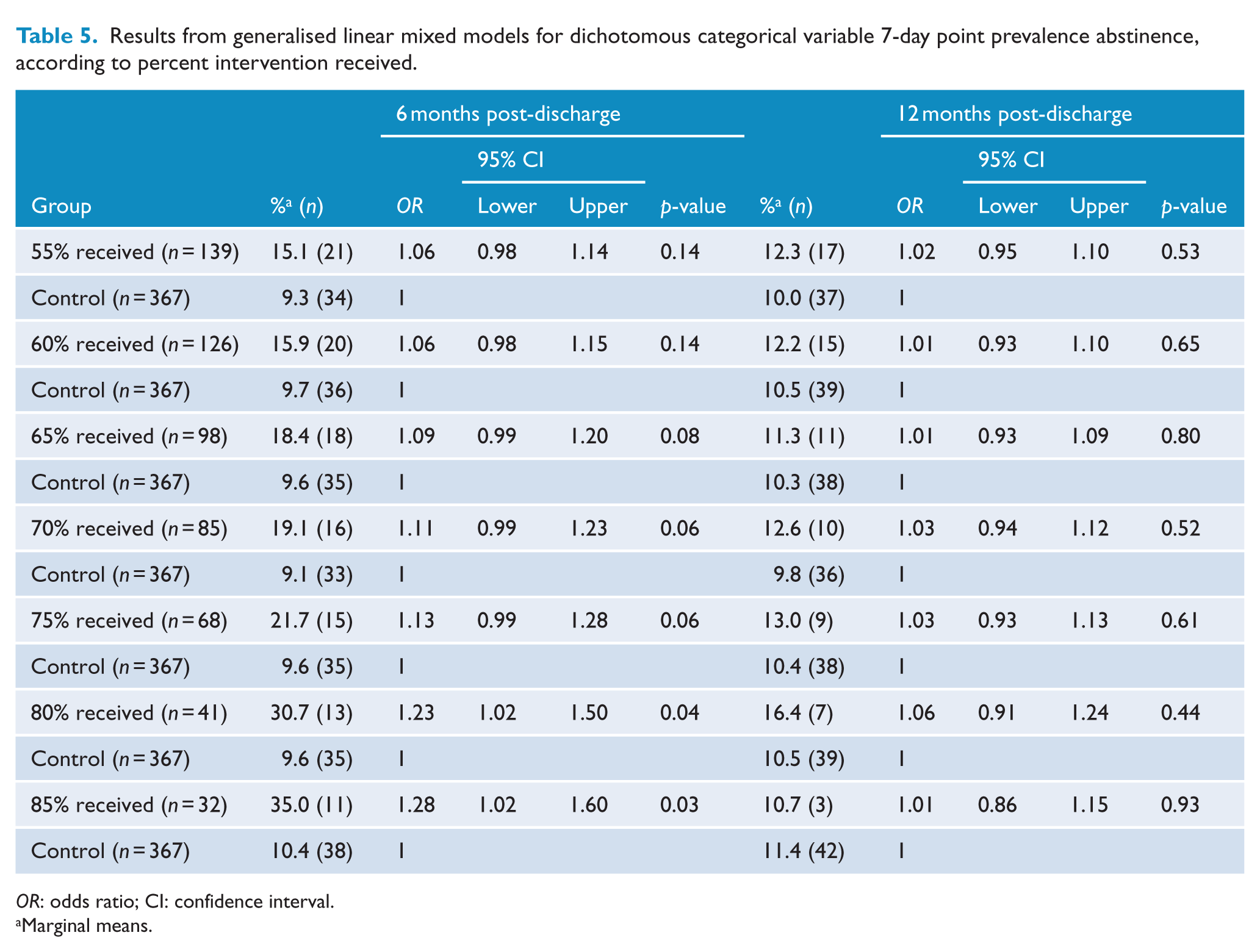
The data used to calculate percent intervention receipt groups are reported in Supplementary Table 1 (see Metse et al. [2016] for a more detailed description). For 7-day point prevalence abstinence, the group-by-time interaction was significant for the 85% intervention receipt group, F(1, 462) = 7.41, p = 0.017, and borderline significant for the 80% intervention receipt group, F(1, 475) = 3.66, p = 0.06. Simple effects models revealed at 6 months post-discharge that those receiving 80% (OR = 1.23, 95% CI = [1.02, 1.50]) and 85% (OR = 1.28, 95% CI = [1.02, 1.60]) of the offered intervention were significantly more likely than control participants to be abstinent for at least 7 days, although these effects were not sustained at 12 months post-discharge (both ps > 0.44; Table 5). Group-by-time interactions and simple effects models were non-significant for the 55–75% intervention receipt models (all ps ⩾ 0.06).
Results from generalised linear mixed models for dichotomous categorical variable 7-day point prevalence abstinence, according to percent intervention received.
OR: odds ratio; CI: confidence interval.
Marginal means.
One-month prolonged abstinence
The proportions of participants abstinent for 1 month or longer were 12.1% and 9.5% for intervention and control groups at 6 month post-discharge, and 11.8% and 10.1% at 12 months post-discharge, respectively, with no significant group-by-time interaction, F(2, 841) = 0.02, p = 0.98 (Table 4), or main effect of group, F(1, 841) = 0.80, p = 0.37; OR = 0.86, 95% CI = [0.48, 1.54]. Sensitivity analyses similarly showed no intervention effect for the group-by-time interaction, F(2, 1467) = 0.05, p = 0.95, or main effect of group, F(1, 1467) = 0.47, p = 0.49.
Subgroup analyses
Subgroup analyses revealed the odds of 1-month prolonged abstinence did not differ among intervention group participants contingent on psychiatric diagnosis (6 month: OR = 1.04, 95% CI = [0.93, 1.15]; 12 month: OR = 1.04, 95% CI = [0.95, 1.14]), readiness to quit (6 month: OR = 1.08, 95% CI = [0.98, 1.19]; 12 month: OR = 1.09, 95% CI = [0.996, 1.19]) and level of nicotine dependence (6 month: OR = 0.94, 95% CI = [0.86, 1.02]; 12 month: OR = 0.99, 95% CI = [0.91, 1.07]).
Per protocol analyses
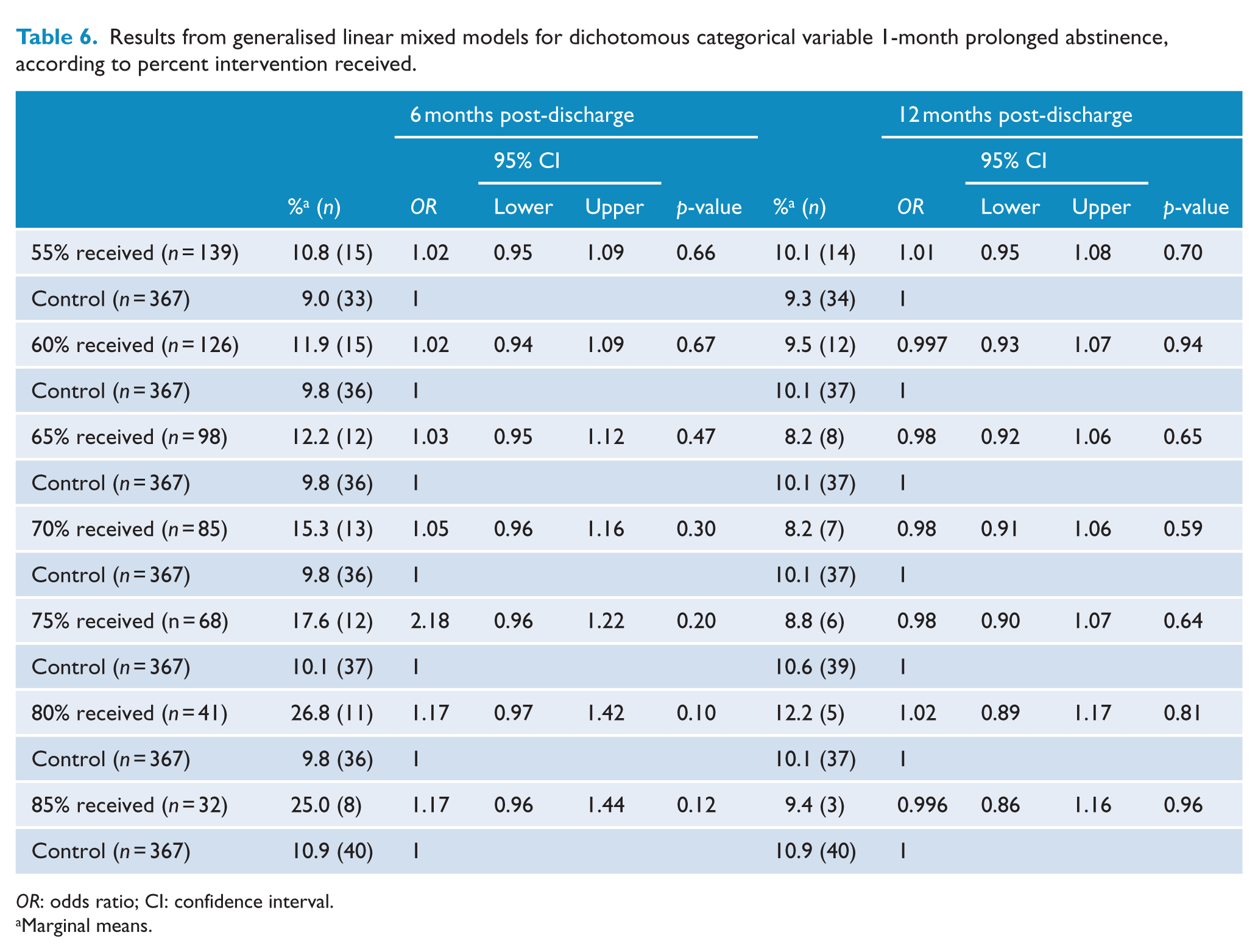
The likelihood of 1-month prolonged abstinence at 6 and 12 months post discharge did not vary between the control group and any of the intervention receipt groups, with all group-by-time interactions and main effects of group being non-significant (all ps > 0.13; Table 6).
Results from generalised linear mixed models for dichotomous categorical variable 1-month prolonged abstinence, according to percent intervention received.
OR: odds ratio; CI: confidence interval.
Marginal means.
Secondary outcomes
Group-by-time interactions revealed a significant intervention effect for four of seven secondary outcomes including cigarettes per day, F(2, 1440) = 5.41, p = 0.005 (Table 7); proportion of cigarettes cut down, F(1, 5.16) = 5.410, p = 0.02; quit attempt, F(2, 1578) = 7.11, p = 0.001; and number of quit attempts, F(2, 1525) = 6.2, p = 0.002 (Table 4), with intervention group participants smoking significantly fewer cigarettes per day and being more likely to reduce cigarette consumption and attempt to quit one or more times at both the 6- and 12-month post-discharge follow-ups, relative to controls.
Results from generalised linear mixed models for continuous outcome variable cigarettes per day for the intervention and control groups at the 6- and 12-month post-discharge follow-up assessments.

SE: standard error; CI: confidence interval.
There was no significant group-by-time interaction for nicotine dependence, F(2, 1579) = 1.25, p = 0.29; readiness to quit, F(2, 1580) = 0.99, p = 0.37; or duration of longest quit attempt, F(2, 1415) = 1.03, p = 0.36.
Discussion
The universal smoking cessation intervention initiated during admission to a psychiatric inpatient facility and continued for a period of 4 months immediately post-discharge was efficacious in reducing the 7-day point prevalence of smoking at 6 months post-discharge but not at 12 months. The intervention did not impact rates of 1-month prolonged abstinence at either 6 or 12 month post-discharge. Among those receiving the intervention, per protocol analyses showed that the degree of intervention received was positively associated with a greater likelihood of quitting for 7 days, while diagnosis, readiness to quit and nicotine dependence were not. The intervention resulted in reduced daily cigarette consumption and increased quit attempts, behaviours that may precede future successful cessation (Caponnetto and Polosa, 2008; Hymowitz et al., 1997). The findings suggest a short-term benefit of linking all smokers in inpatient mental health facilities with community-based post-discharge smoking cessation support. Further research is required to identify strategies for achieving sustained smoking cessation, the importance of doing so supported by the high smoking prevalence typical of psychiatric inpatients (62% in the present study).
The finding of a significant 7% absolute difference in 7-day point prevalence of abstinence at 6 months post discharge in this trial is similar to that of two US trials (8% and 9%) (Hickman et al., 2015; Prochaska et al., 2014) and greater than that reported in an earlier Australian trial (2%) (Stockings et al., 2014a). The absence of a significant intervention effect in 7-day point prevalence abstinence at 12 months however contrasts with the outcomes in the US trials (Hickman et al., 2015; Prochaska et al., 2014), and may be related to participant, methodological or jurisdictional differences between the studies. In the present study, the universal intervention approach reflected clinical guidelines recommending that clinicians systematically assess smoking status and offer cessation care to all smokers, regardless of assessed readiness to quit (Fiore et al., 2008; National Institute for Health Care Excellence, 2013). This approach is in keeping with research that suggests quitting can occur spontaneously and that the availability of cessation assistance can promote a quit attempt (Caponnetto and Polosa, 2008; Ferguson et al., 2009). In contrast, the cessation support in the US studies entailed an offer of one intervention component, NRT contingent on patient ‘readiness’ to quit, with such offer being made at any point over a period of up to 6 months post-discharge. The US researchers suggest that their sustained outcome, with an apparent increasing intervention effect over time for point prevalence abstinence is consistent with such a stage-based approach (Prochaska et al., 2014).
Results from subgroup analyses suggest the intervention component was acceptable and efficacious for all smokers – with neither measure of abstinence being associated with psychiatric diagnosis, readiness to quit or level of dependence. Neither US trial (Hickman et al., 2015; Prochaska et al., 2014) assessed for differences in intervention effects according to the specified subgroups. However, across both intervention and control conditions, Hickman et al. (2015) found those with psychotic disorders to be more likely to be abstinent relative to those with unipolar depression; and Prochaska et al. (2014) found that participants who were ready to quit and those with lower nicotine dependence were more likely to be abstinent. The lack of parallel approach to analyses precludes direct comparisons, nevertheless the absence of differential intervention effects in the current study may have been related to equal provision of intervention components, including NRT, to all participants regardless of readiness to quit and NRT prescription occurring in accordance with degree of nicotine dependence.
In line with the findings of previous research among both smokers generally (Fiore et al., 2008; Stead and Lancaster, 2012) and those with a mental illness (Baker et al., 2006; Okoli and Khara, 2014), results from the per protocol analyses suggest a dose–response relationship between the degree of intervention receipt and likelihood of abstinence: smokers in receipt of greater amounts of the intervention (80+%) were 23–28% more likely to achieve 7-day point prevalence abstinence at 6 months post-discharge, compared to controls. Such a finding suggests the importance of strategies for optimising access to and utilisation of cessation aids (Hollands et al., 2015). For smokers without a mental illness, systematic review evidence suggests further participant education regarding the benefits of tobacco cessation aids and provision of additional problem-solving-based support to overcome potential barriers to aid utilisation and to enhance adherence to cessation interventions (relative risk [RR] 1.14, 95% CI = [1.02, 1.28], n = 1630), which in turn leads to a higher likelihood of cessation (RR = 1.16, 95% CI = [1.01, 1.34], n = 3049) (Hollands et al., 2015). As no such research exists for persons with a mental illness, identification of strategies to improve access and/or utilisation of cessation supports for this group of smokers is needed.
The positive effects of the intervention on secondary outcomes including reduced cigarette consumption and greater number of quit attempts are congruent with those of the previous Australian trial (Stockings et al., 2014a). Given such changes in smoking behaviour have been shown to increase the subsequent likelihood of successful cessation among smokers generally (Caponnetto and Polosa, 2008; Hymowitz et al., 1997), these findings suggest the intervention is likely to have been efficacious in progressing smokers with a mental illness towards cessation. Only one paper was identified that has previously investigated the possible relationship of such changes in smoking behaviours on cessation for smokers with a mental illness. Okoli and Khara (2014) reported a positive relationship between the length of a previous quit attempt and 7-day point prevalence abstinence.
Strengths of this study included a large and heterogeneous study population, promoting external validity and allowing for investigation of intervention effect that may have been contingent on psychiatric diagnosis, readiness to quit, degree of nicotine dependence and dose of intervention received. Limitations included high participant attrition and incomplete biochemical verification of point prevalence abstinence, both of which may have resulted in biases likely to result in overestimation of intervention effect. Other smoking trials with persons with a mental illness have encountered such issues, some similarly adopting a biochemically verified self-report hybrid measure of abstinence (Peckham et al., 2015). A further limitation may have been sub-optimal intervention exposure, which in contrast was likely to have resulted in an underestimate of intervention effect.
Footnotes
Acknowledgements
The authors would like to thank the participants, clinicians and research team that enabled this study to be undertaken.
Availability of supporting data
The datasets generated and analysed during the current study are not publicly available to preserve the privacy of participants; however, they are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Prochaska has consulted to Pfizer, which makes smoking cessation medications, and has been an expert witness for plaintiffs’ counsel in lawsuits against the tobacco companies. All other authors declare no conflicting interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Health and Medical Research Council (NHMRC) (grant number: G1100130). Dr Stockings is supported by an Australian NHMRC early career fellowship (grant number: 1104600). Dr Prochaska is supported by the National Institutes of Health (grant number: HL117736-01) and the State of California Tobacco-Related Disease Research Program (grant number: 24RT-0035).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
