Abstract
Objective:
To examine the association between cord 25-hydroxyvitamin D2+3 (25(OH)D) and attention deficit hyperactivity disorder symptoms in toddlers, using Child Behaviour Checklist for ages 1.5–5.
Method:
In a population-based birth cohort, a Child Behaviour Checklist for ages 1.5–5 questionnaire was returned from parents of 1233 infants with mean age 2.7 (standard deviation 0.6) years. Adjusted associations between cord 25(OH)D and Child Behaviour Checklist–based attention deficit hyperactivity disorder problems were analysed by multiple regression.
Results
The median cord 25(OH)D was 44.1 (range: 1.5–127.1) nmol/L. Mean attention deficit hyperactivity disorder problem score was 2.7 (standard deviation 2.1). In adjusted analyses, cord 25(OH)D levels >25 nmol/L and >30 nmol/L were associated with lower attention deficit hyperactivity disorder scores compared to levels ⩽25 nmol/L (p = 0.035) and ⩽30 nmol/L (p = 0.043), respectively. The adjusted odds of scoring above the 90th percentile on the Child Behaviour Checklist–based attention deficit hyperactivity disorder problem scale decreased by 11% per 10 nmol/L increase in cord 25(OH)D.
Conclusion:
An inverse association between cord 25(OH)D and attention deficit hyperactivity disorder symptoms in toddlers was found, suggesting a protective effect of prenatal vitamin D.
Introduction
Attention deficit hyperactivity disorder (ADHD), defined as functional impairment due to persistent attention deficits and hyperactivity, is one of the most common psychiatric disorders in childhood, with a reported prevalence of 3.4–7.1% (Polanczyk et al., 2015; Thomas et al., 2015). Despite the high prevalence and its extensive socioeconomic impact (RFF, 2014), knowledge of the specific causes of the disorder is limited. A multifactorial aetiology is suggested with a wide variety of environmental factors moderating a primary genetic susceptibility (Thapar et al., 2013). The disorder is diagnosed based on Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) or International Classification of Diseases, Tenth Edition (ICD-10) criteria after a thorough assessment in a multidisciplinary setting (Atkinson and Hollis, 2010). ADHD is most frequently diagnosed in boys of school age (Polanczyk et al., 2014). However, early signs of psychopathology, including ADHD symptoms, can be assessed by using psychometric instruments in younger children, e.g. The Child Behaviour Checklist 1.5–5 (CBCL 1.5–5; ASEBA, n.d.).
A high prevalence of vitamin D insufficiency and deficiency in pregnancy and neonates has been reported worldwide (Bodnar et al., 2007; Cadario et al., 2015; Pratumvinit et al., 2015; Rodriguez-Dehli et al., 2015; Vinkhuyzen et al., 2015). While the impact on bone growth and mineralization has been known for more than a century, more recent research has focused on extra-skeletal manifestations of hypovitaminosis D in children (Christesen et al., 2012; Wacker and Holick, 2013). Recent findings include positive associations between levels of maternal pregnancy serum 25-hydroxyvitamin D (s-25(OH)D) and psychomotor function and language development in infants and children (Morales et al., 2012; Whitehouse et al., 2012).
Hypovitaminosis D has been linked to ADHD in children (Bener and Kamal, 2014; Goksugur et al., 2014; Kamal et al., 2014; Shang-Guan and Zhao, 2015). The amount of research concerning maternal or neonatal vitamin D status and ADHD symptoms is, however, limited and contradictory. A recent Spanish cohort study found an inverse association between maternal 25(OH)D levels in early pregnancy and ADHD-like symptoms in 4-year-old children (Morales et al., 2015). In contrast, a Danish cohort and a Swedish case-control study found no association in older children (Gustafsson et al., 2015; Strom et al., 2014).
Neonatal vitamin D associations to ADHD symptoms in toddlers have not been studied, but may be anticipated if the foetal environment is critical for the development of early signs of ADHD. We, therefore, examined the association between cord 25(OH)D concentrations and parent-rated ADHD symptoms in 2- to 4-year-old children, using CBCL 1.5–5 in the Odense Child Cohort (OCC) study.
Method
Study population
The study population was drawn from the OCC, a population-based observational mother-and-child cohort, including newly discovered pregnancies in the Danish Municipality of Odense between 1 January 2010 and 31 December 2012 (Kyhl et al., 2015). Serial questionnaires and blood samples were obtained. Of 6707 pregnant women eligible for participation, 2549 (38%) enrolled in OCC (Figure 1).
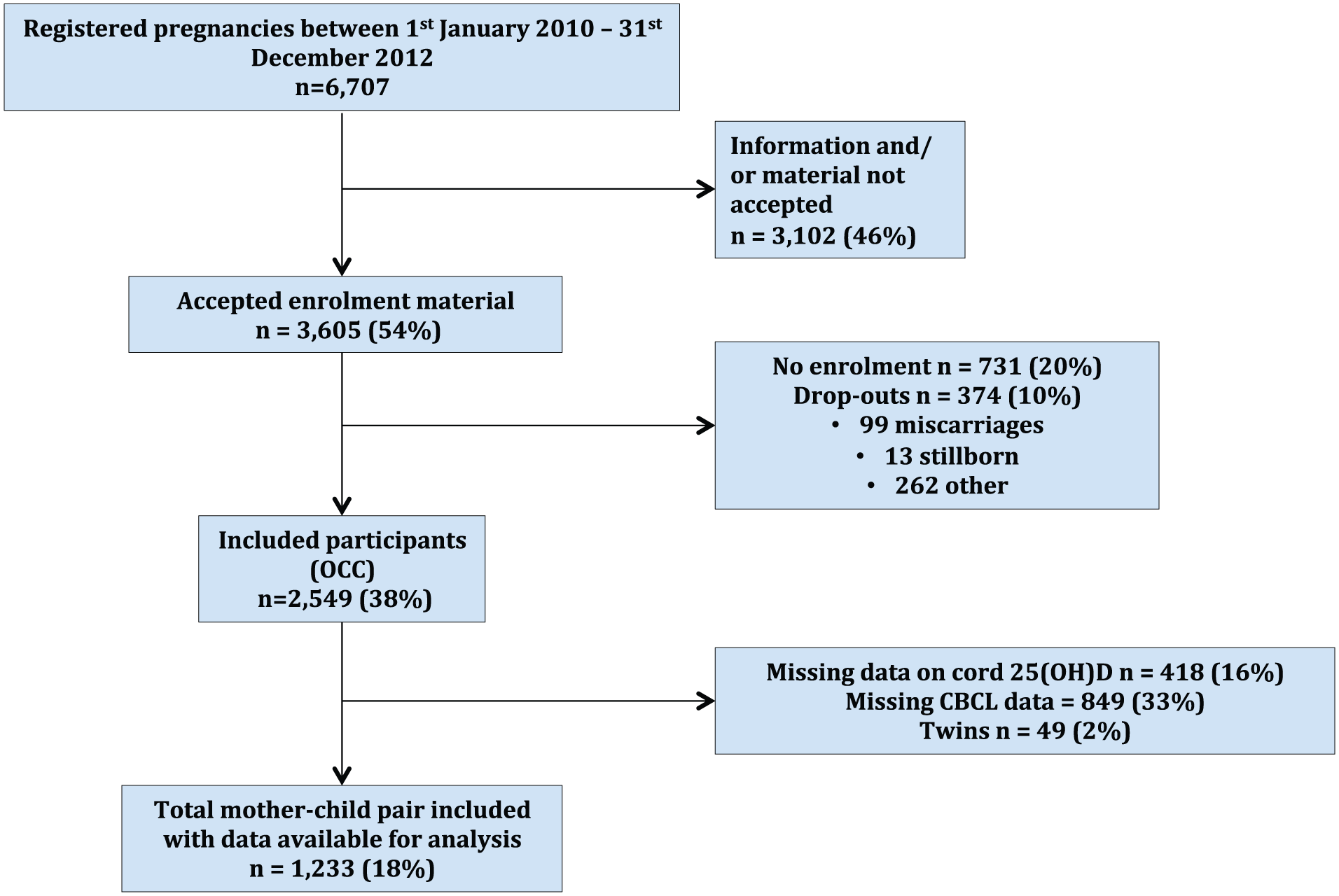
Flowchart.
Participants were included in this study if cord blood was donated with available s-25(OH)D measurements, and the parents had filled in a CBCL 1.5–5 questionnaire when the child was 2–4 years old (n = 1555). Exclusion criteria were multiple pregnancies, children with severe congenital malformations or syndromes and lack of information on covariates except alcohol consumption during pregnancy, maternal educational level and vitamin D supplementation. Parental psychiatric disorders were treated as a covariate.
Parameters studied
In short, following storage at −80°C, concentrations of s-25(OH)D2+3 were analysed using liquid chromatography-tandem mass spectrometry on a TurboFlow column (Thermo Scientific San Jose, CA, USA), as previously described (Andersen et al., 2013). The sum of D2 and D3 was reported as total 25(OH)D.
The parent-report questionnaire CBCL 1.5–5 consists of 100 questions. All items are rated on a 3-point Likert scale: 0 (not true), 1 (somewhat/sometimes true) and 2 (very true/often true). The purpose is to measure emotional and behavioural problems, including ADHD symptoms, in children between 1.5 and 5 years. A standardized version of CBCL 1.5–5 translated into Danish is available (Kristensen et al., 2010). In this study, a DSM-oriented ADHD problem scale (CBCL 1.5–5 extracted) consisting of 6 questions with a maximum score of 12 (Table 1) was used as outcome. Good reliability and validity have been reported for the CBCL 1.5–5 in both clinical and research settings (Ivanova et al., 2010; Kristensen et al., 2010).
CBCL 1.5–5 ADHD problem items used.
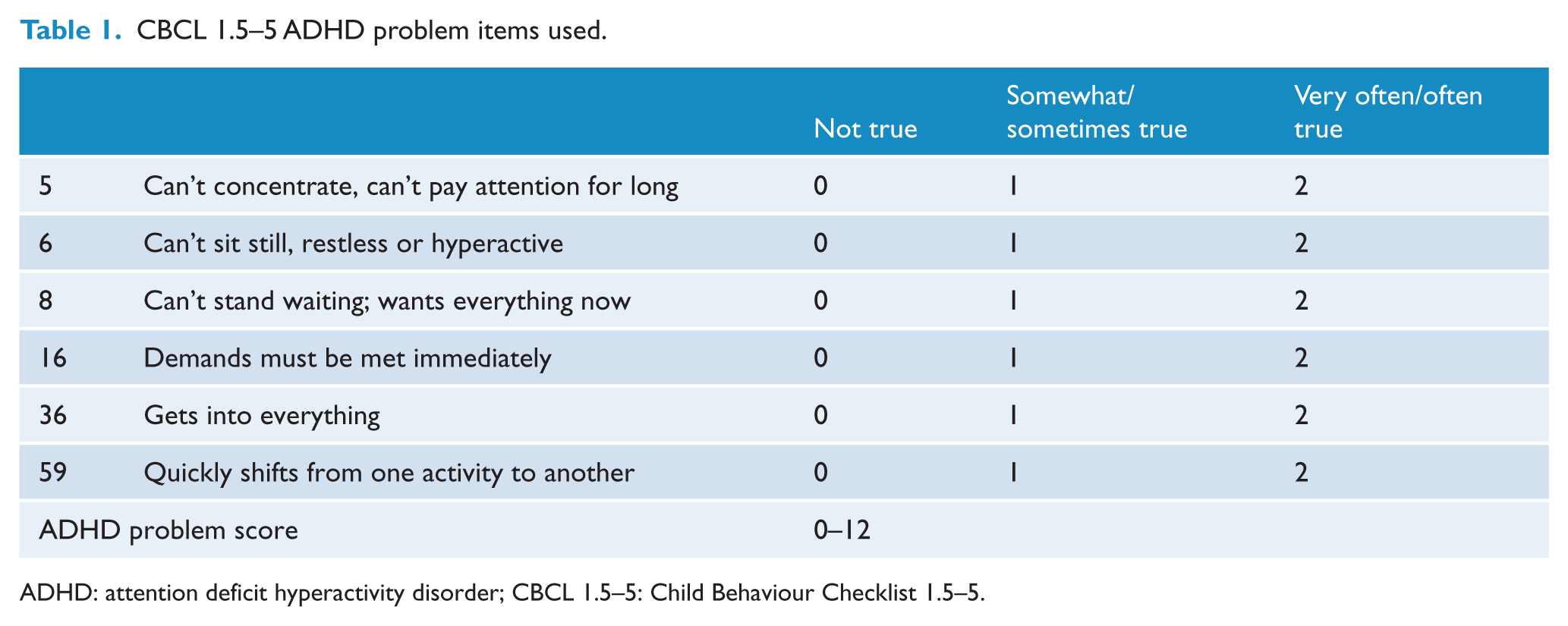
ADHD: attention deficit hyperactivity disorder; CBCL 1.5–5: Child Behaviour Checklist 1.5–5.
Covariates included a priori were maternal age, gestational age, sex of child, birth weight and season of year at birth. Parity, maternal smoking habits, alcohol consumption, educational level, vitamin D supplementation and pre-gestational height and weight for calculation of body mass index (BMI) were extracted from the OCC questionnaires as self-reported data. Parental psychiatric diagnoses were obtained from the Danish National Patient Register.
Statistical analyses
Descriptive data were presented as numbers (%), mean (standard deviation [SD]) or median (25–75% percentiles), and p-values were acquired with chi-square test or linear regression. Regression models were initially performed as linear, spline and cubic spline for categorical values.
Cord s-25(OH)D and ADHD scores were treated as both categorized and continuous variables. Cord s-25(OH)D was categorized into quartiles and routine cut-offs according to Institute of Medicine (IOM) (Ross et al., 2011) and Danish recommendations, i.e. s-25(OH)D <50 nmol/L, defined as insufficiency and s-25(OH)D <25 nmol/L defined as deficiency. ADHD score was split at the 90th percentile. Categorizing ADHD score in other percentiles, e.g. at the 97.5th percentile, led to too small numbers of children allowed for statistical analysis (possible type II error).
In the adjusted models, the initially included covariates were associated with either the ADHD score or cord s-25(OH)D. All self-reported data as well as parity, season of birth and parental psychiatric diagnoses were analysed as categorical variables. Maternal age and pre-pregnancy BMI were analysed as continuous variables. BMI was also categorized as <18.5, 18.5–24.9, 25–30 and >30 mg/m2. Results were expressed as beta coefficients and odds ratios (OR) with 95% confidence intervals (CI) using linear and logistic regression, respectively. Missing data on covariates (maternal educational level, alcohol and vitamin D supplementation) were included in the statistical analysis as a missing categorical variable. A p-value < 0.05 was considered statistically significant, and p-values between 0.05 and 0.10 were considered trends.
A power analysis showed that our study would detect a true change of 0.02 ADHD scores per 10 nmol/L change in cord s-25(OH)D (α: 0.05, β = 0.8, 25(OH)D SD 22 nmol/L, ADHD score SD 0.6, n = 1200).
All data analyses were performed using STATA software, versions 13.1 and 14.0 (StataCorp, College Station, TX, USA).
Ethics
All women gave written informed consent to participate. The study complied with the Helsinki Declaration and was approved by the Regional Scientific Ethical Committee of Southern Denmark, no. S-20090130, and the Danish Data Protection Agency.
Results
The study included 1233 mothers and their children, of whom 654 (53.0%) were boys. The mean (SD) maternal age was 30.4 (4.4) years, and the mean child age when parents returned the CBCL questionnaires was 2.7 (0.6) years. The median cord s-25(OH)D concentration was 44.1 (25–75%: 30.4–59.3, range: 1.5–127.1) nmol/L. Mean (SD) CBCL-based ADHD problem score was 2.7 (2.1).
Table 2 presents the maternal, offspring and seasonal characteristics in relation to ADHD score, split by the 90th percentile. In univariate analysis, a score at or above the 90th percentile was significantly associated with lower cord s-25(OH)D concentrations, lower maternal age, primiparity, BMI <18.5 or ⩾30 kg/m2, maternal smoking and alcohol consumption during pregnancy and lower maternal educational level. Trends were seen for parental psychiatric diagnoses, male sex and vitamin D supplementation.
Distribution of maternal, offspring and environmental factors in CBCL 1.5–5 scores.
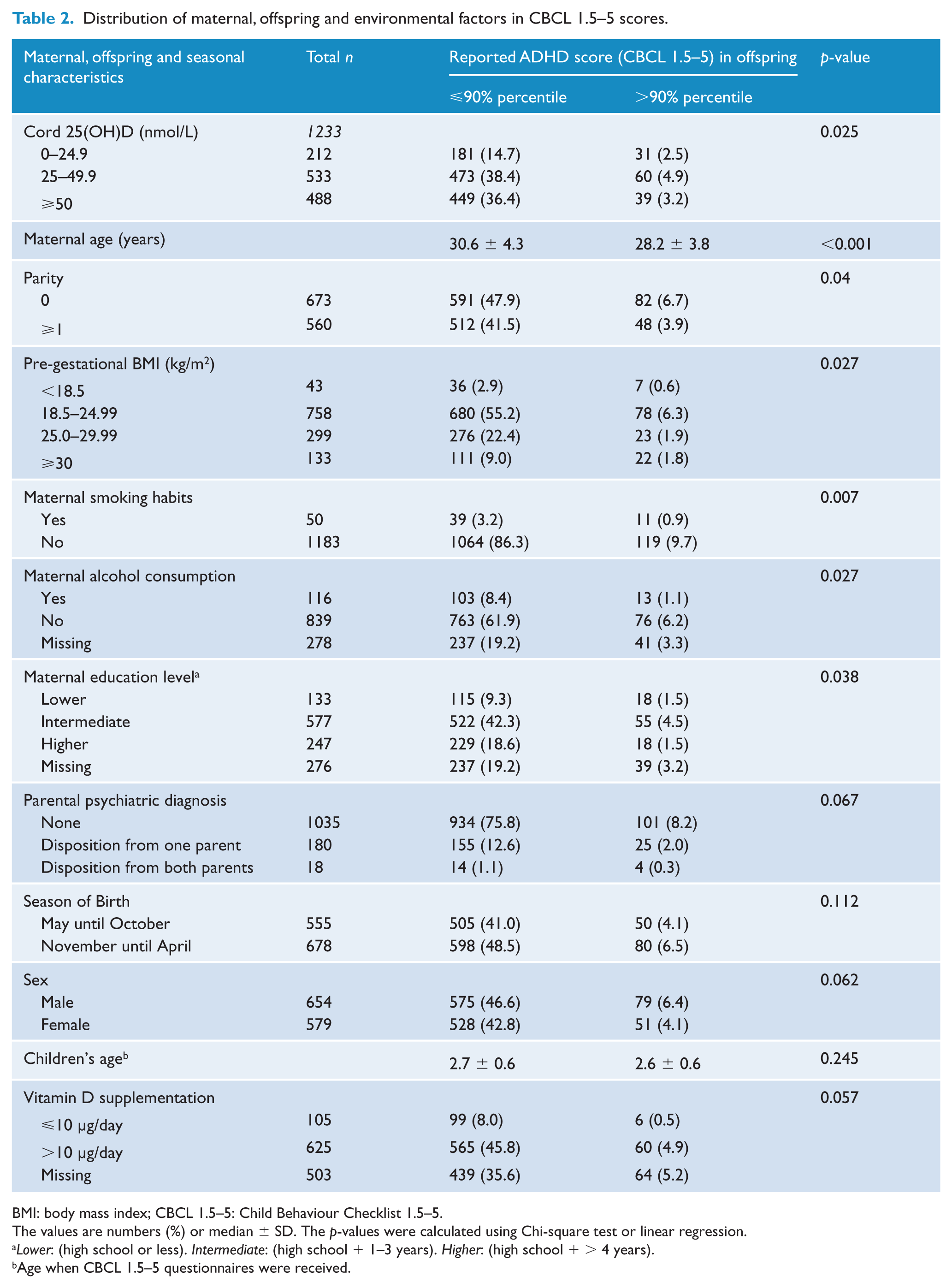
BMI: body mass index; CBCL 1.5–5: Child Behaviour Checklist 1.5–5.
The values are numbers (%) or median ± SD. The p-values were calculated using Chi-square test or linear regression.
Lower: (high school or less). Intermediate: (high school + 1–3 years). Higher: (high school + > 4 years).
Age when CBCL 1.5–5 questionnaires were received.
We identified no continuous association between cord s-25(OH)D concentrations and ADHD score, unadjusted (p = 0.180), adjusted (p = 0.320), Table 3.
Associations between cord 25(OH)D and CBCL-based ADHD scores (n = 1233).
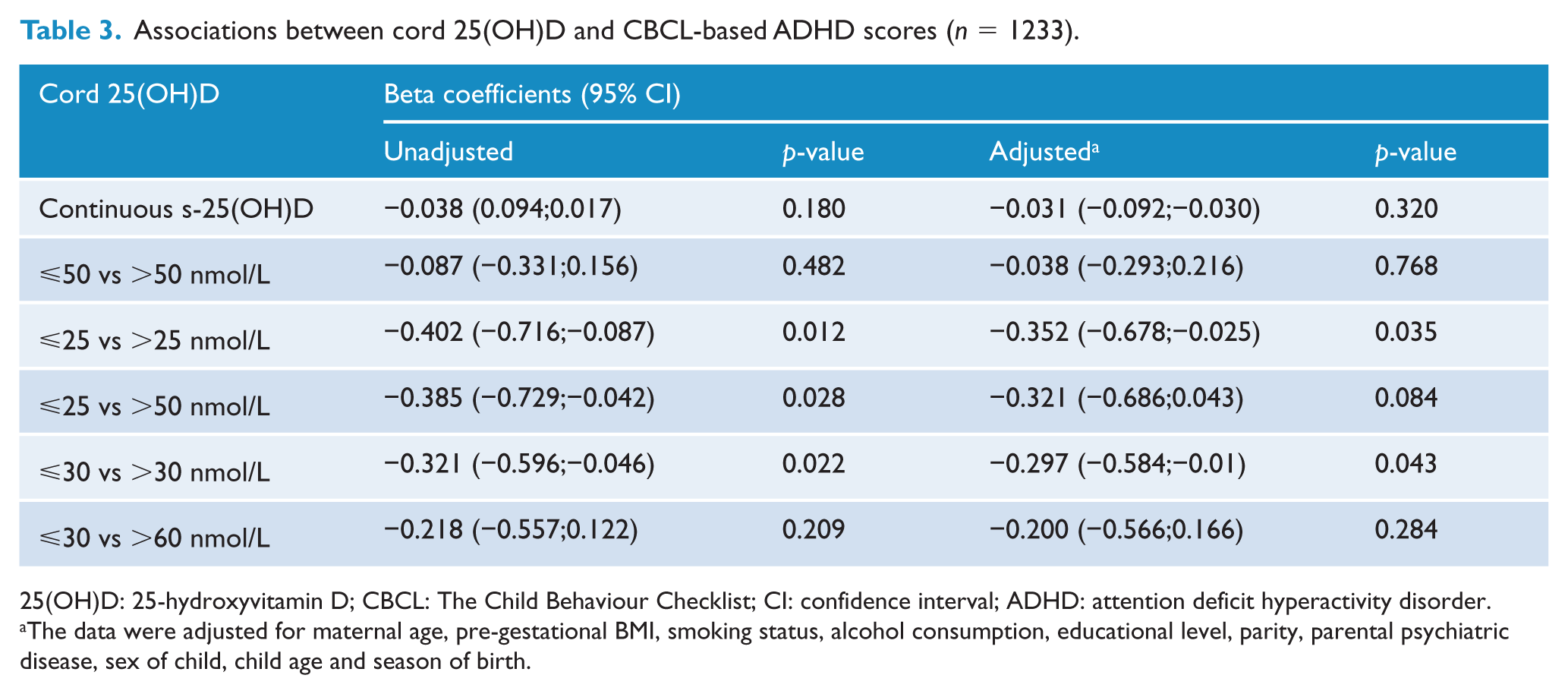
25(OH)D: 25-hydroxyvitamin D; CBCL: The Child Behaviour Checklist; CI: confidence interval; ADHD: attention deficit hyperactivity disorder.
The data were adjusted for maternal age, pre-gestational BMI, smoking status, alcohol consumption, educational level, parity, parental psychiatric disease, sex of child, child age and season of birth.
The crude association was not strengthened by spline (p = 0.277) or cubic spline (p = 0.184) regression. Split by routine cut-offs, cord s-25(OH)D >25 nmol/L had significantly lower ADHD scores compared to levels ⩽25 nmol/L, p = 0.035. Split by quartiles, cord s-25(OH)D >30 nmol/L had significantly lower ADHD scores compared to levels ⩽30 nmol/L, p = 0.043.
The adjusted odds of being in ⩾90th percentile decreased by 11% per 10 nmol/L increase in cord s-25(OH)D(p = 0.039), see Table 4. Split by routine cut-offs, the adjusted odds of an ADHD score ⩾90th percentile were decreased for cord s-25(OH)D >50 nmol/L compared to ⩽50 nmol/L (trend, p = 0.071) and >50 nmol/L compared to ⩽25 nmol/L (trend, p = 0.053). Split by quartiles, the adjusted odds of an ADHD score ⩾90th percentile were decreased for cord s-25(OH)D >30 nmol/L compared to ⩽30 nmol/L (trend, p = 0.065) and >60 nmol/L compared to ⩽30 nmol/L (trend, p = 0.062).
Associations between cord 25(OH)D and CBCL-based ADHD scores split by the 90th percentile (n = 1233).
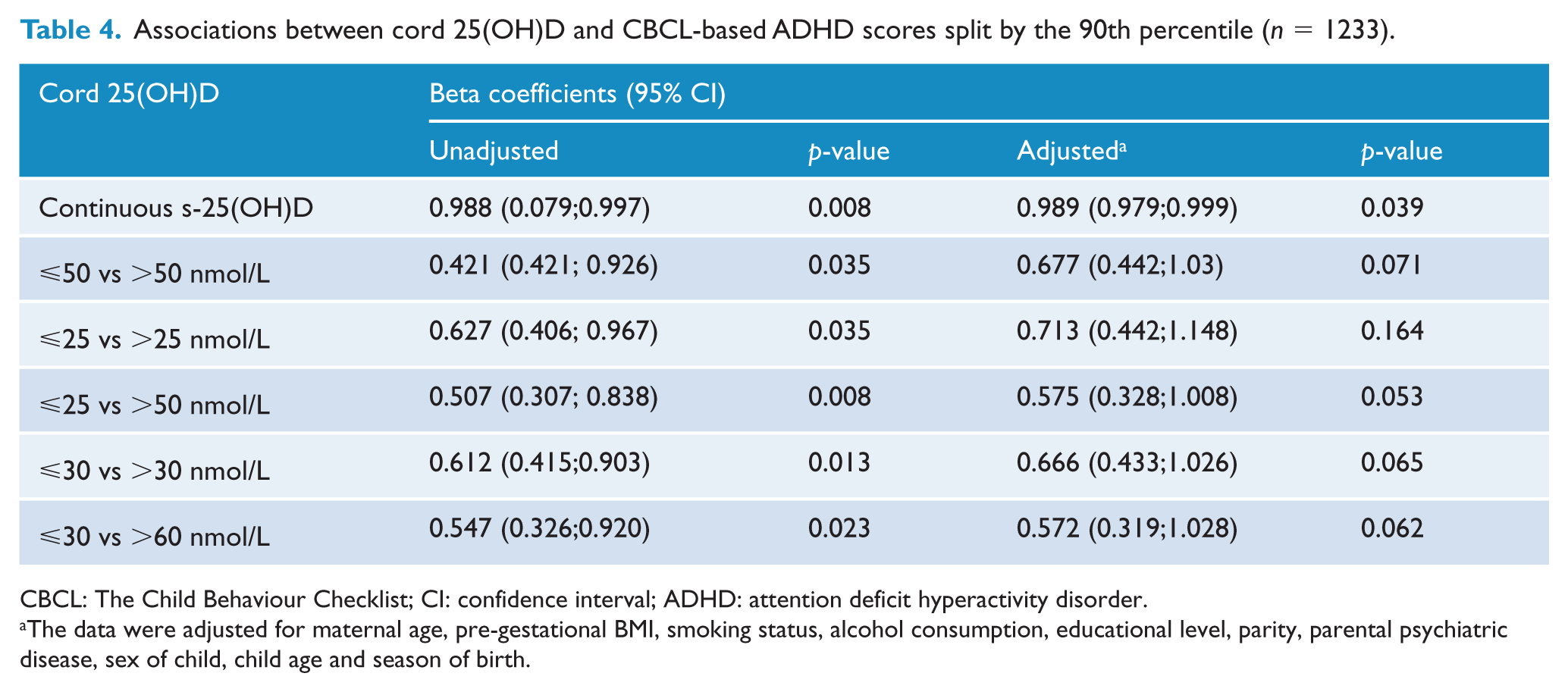
CBCL: The Child Behaviour Checklist; CI: confidence interval; ADHD: attention deficit hyperactivity disorder.
The data were adjusted for maternal age, pre-gestational BMI, smoking status, alcohol consumption, educational level, parity, parental psychiatric disease, sex of child, child age and season of birth.
In addition, lower maternal age (p = 0.004), psychiatric disease in parents (p = 0.014), maternal smoking during pregnancy (p = 0.047) and male sex (p = 0.006) were independently associated with increasing ADHD score.
The adjusted odds of being in the top 10 percentile of the ADHD scale were associated with lower maternal age (p = 0.002), maternal smoking during pregnancy (trend, p = 0.061), male sex (trend, p = 0.072) and missing alcohol data (trend p = 0.064).
We performed a post hoc analysis without adjustment for ‘season of birth’ in the model as it has been suggested that season of birth may not have impact on an eventual brain development during pregnancy. However, this did not change the results in the linear models, i.e. no significant association between continuous s-25(OH)D and ADHD score (β-coefficient [95% CI]: −0.047 [−0.10;0.010], p = 0.109) but strengthened the association found in the logistic models, the odds of being in the ⩾90th percentile decreased 14% per 10 nmol/L increase in cord s-25(OH)D (p = 0.004).
Discussion
This prospective cohort study of a population-based Danish birth cohort showed novel inverse associations between cord s-25(OH)D concentrations and lower parent-rated ADHD problems in toddlers. Other variables showed significant associations with higher ADHD problem scores, including lower maternal age, male sex, smoking during pregnancy and parental psychiatric disorders. These variables have previously been associated with clinically assessed ADHD (Gustafsson et al., 2015; Thapar and Cooper, 2015), supporting the hypothesis that toddlers with high CBCL 1.5–5-based ADHD scores actually represent an ADHD high-risk group.
The association between early s-25(OH)D and ADHD symptoms has only been studied sparsely, and the association between cord s-25(OH)D and CBCL-based ADHD problem scores has, to our knowledge, not been studied previously. Discrepancies in previous studies may, in addition to methodological issues and age of the children, be a matter of timing of blood sampling in relation to a possible time window of the impact of the exposure on the developing brain and differences in cut-offs for s-25(OH)D or ADHD evaluation scores.
Our data did not support a linear correlation throughout the whole range of exposure, and outcome parallels Strom et al. (2014), who found no association between maternal pregnancy s-25(OH)D and prescription of ADHD medication in late adolescents (n = 850, ADHD, n = 24), and, Gustafsson et al. (2015), who also found no association between low cord s-25(OH)D and ADHD in children between 5 and 17 years of age (n = 404). Whitehouse et al. (2012) found no association between maternal s-25(OH)D in pregnancy and behaviour in 2-year-old children when using CBCL 2–3 (n = 743 children).
In contrast, one large study has supported an association between low s-25(OH)D and ADHD-like symptoms. Morales et al. (2015) found that the adjusted risk of ADHD symptoms in children aged 4.8 years (n = 1650) decreased by 11% per 25 nmol/L increase in maternal pregnancy s-25(OH)D. However, Keim et al. (2014) found no association between maternal or cord s-25(OH)D and behaviour in children aged between 8 months and 7 years, including hyperactive behaviours in 4-year-old children (n = 3896).
The study by Morales et al. (2015) is the study most comparable to our study regarding age of assessment and sample size and it supports our findings. For other studies, methodical differences, most importantly ADHD assessment tools and age of evaluation, hamper direct comparison with our results.
Existing research suggests that vitamin D is crucial to the foetal neuroanatomical development through a variety of pathways, including neuronal differentiation and regulation of reactive oxidative species (Eyles et al., 2011). However, the reason why higher vitamin D concentrations potentially protect against ADHD symptoms remains uncertain. The preventing component could be that vitamin D, through its anti-inflammatory abilities, inhibits harmful environmental triggers related to potential neurodevelopmental anomalies, as discussed by Morales et al. (Morales et al., 2015). Moreover, vitamin D deficiency in pregnant mice has been linked to increased glucocorticoid exposure in the foetus (Tesic et al., 2015), which may in turn, through increased foetal brain exposure, lead to neurodevelopmental anomalies. Sufficient vitamin D concentrations may protect the foetal brain against excessive glucocorticoid exposure, thus protecting against neurodevelopmental disorders like ADHD.
The major strengths of this study include the relatively large population-based and prospective study design and use of the gold standard method for analysis of s-25(OH)D (Farrell et al., 2012). Furthermore, the cord vitamin D analysis and the CBCL assessment process were blinded from each other, and the statistical analyses performed were thorough, including search for optimal cut-offs and inclusion of missing data.
The limitations include the observational nature of the study, allowing no causative conclusions; minor discrepancies between cohort participants and the background population and the use of only parents as informers of ADHD symptomatology. We have no information on mother–child interactions, which could have led to confounding due to possible mediation from, e.g. daily activities and sun exposure.
However, most Danish children in this age group are in kindergartens with planned daily out- and indoor activities.
The Danish population is rather homogenous and so is this cohort, with only 3.5% being non-Caucasians. This group has lower vitamin D levels, but due to its small size, it did not alter the outcomes significantly (results not shown). Furthermore, the young age of assessment in this study may lead to higher CBCL-scores due to possible natural over-activity or restless children. Last, only 18% of the eligible participants were included which could have an impact on the generalizability.
Conclusion
In conclusion, associations were detected between higher levels of cord s-25(OH)D and lower ADHD problem score in children aged on average 2.7 years, suggesting that higher cord s-25(OH)D levels may protect against early ADHD symptoms in toddlers. The key findings in this study were summarized in Figure 2. The study further underlines the multifactorial aetiology of the disorder, identifying other independently associated factors such as low maternal age and parental psychiatric diagnoses. A follow-up at school age will be necessary to further validate the possible psychopathologic impact of vitamin D deficiency in late pregnancy and the development of ADHD.

Key findings.
Clinical implications
Based on our results, we suggest that women use vitamin D supplementation during pregnancy, as it seems likely that it may reduce the risk of developing ADHD symptoms in toddlers.
Footnotes
Acknowledgements
The authors thank the participants in the cohort study, the staff at Odense Child Cohort and our collaborators at the Department of Clinical Biochemistry and Pharmacology at Odense University Hospital.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was funded by The Region of Southern Denmark, the University of Southern Denmark, Aase & Ejner Danielsen’s Foundation, the King Christian IX and Queen Louise’s Jubilee Grant, the A.P. Møller Foundation for the Advancement of Medical Science, Torben and Alice Frimodt’s Foundation, Odense University Hospital, The National Board of Social Services and Mental Health Research Fund of Southern Denmark.
