Abstract
Objective:
To investigate the prevalence of occipital bending (an occipital lobe crossing or twisting across the midline) in subjects with schizophrenia and matched healthy controls.
Method:
Occipital bending prevalence was investigated in 37 patients with schizophrenia and 44 healthy controls.
Results:
Ratings showed that prevalence was nearly three times higher among schizophrenia patients (13/37 [35.1%]) than in control subjects (6/44 [13.6%]). Furthermore, those with schizophrenia had greater normalized gray matter volume but less white matter volume and had larger brain-to-cranial ratio.
Conclusion:
The results suggest that occipital bending is more prevalent among schizophrenia patients than healthy subjects and that schizophrenia patients have different gray matter–white matter proportions. Although the cause and clinical ramifications of occipital bending are unclear, the results infer that occipital bending may be a marker of psychiatric illness.
Introduction
Schizophrenia is a psychiatric disorder with approximately 1.0% lifetime prevalence in adults (Von dem Knesebeck et al., 2013). Various brain features have been implicated in schizophrenia, including reduced total brain volume (Honea et al., 2005), reduced gray matter (Lencz et al., 2001) and compromised white matter integrity (Di et al., 2009). Reduced head size has also been reported (Takagai et al., 2006), although this is not a consistent finding (Buckley et al., 2002).The earliest neuroimaging findings in subjects with schizophrenia were of enlarged lateral ventricles (Moriguchi, 1981; Schwarzkopf et al., 1990), particularly in the left hemisphere (Brown et al., 1986; Crow et al., 1989). More recent evidence of lateralized structural change of the ventricles are those of Degreef et al. (1992), Kawasaki et al. (2008) and Yotsutsuji et al. (2003).
We define occipital bending (OB; Figure 1) as indicating that one of the occipital lobes has crossed the antero-posterior axis (Corballis and Morgan, 1978). Rightward OB, for example, indicates that the left occipital lobe has crossed the midline pushing the right occipital lobe. We previously examined OB in patients with major depression (Maller et al., 2014b) and bipolar disorder (Maller et al., 2015) and found OB (primarily rightward) to be three to four times as prevalent among these patients when compared with matched controls.
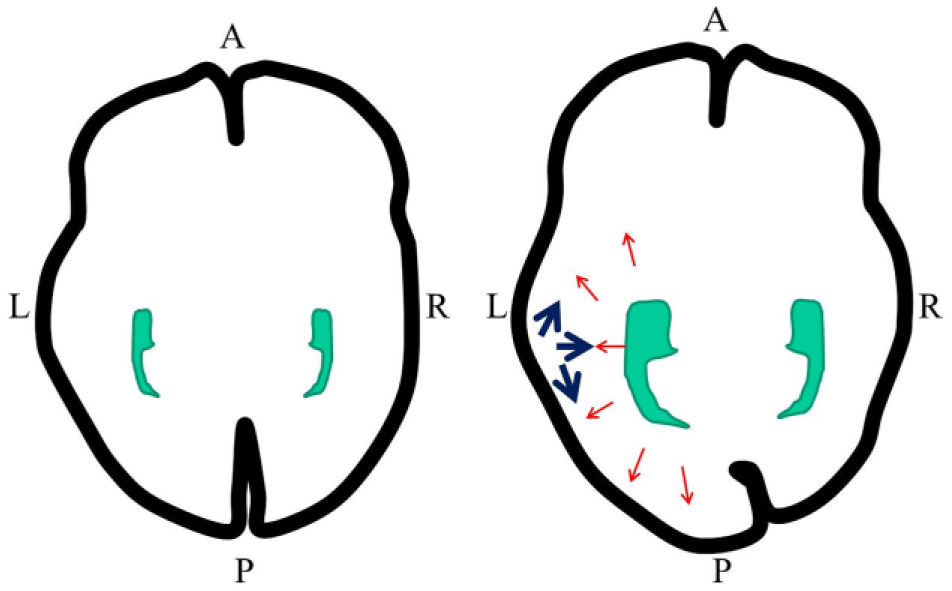
A model of occipital lobe displacement due to enlarged lateral ventricles (blue) leading to occipital bending. Red arrows denote the direction of occipital lobe displacement stemming from ventricular enlargement. Left: No ventricular enlargement or OB. Right: Ventricular enlargement with rightward OB. Images are in neurological convention.
There is research suggesting reduced cerebral asymmetry in schizophrenia (Crow, 2008; Venkatasubramanian and Debnath, 2014), particularly of the superior temporal gyrus (Shenton et al., 2001). However, the literature is inconsistent as it has been reported that those with schizophrenia have an enlarged or reduced left planum temporale and Heschl’s gyrus, which are regions central to receptive language (Guo et al., 2014). This asymmetry forms during development (Venkatasubramanian and Debnath, 2014), and it has been suggested that OB ‘twist’ creates a prominent shape difference between the left and right posterior temporal regions and a larger left planum temporale (Glasel et al., 2011). This therefore supports a developmental underpinning to the development of schizophrenia with auditory–verbal hallucinations. With the addition of enlarged lateral ventricles, pressure may be applied to surrounding structures, effectively displacing them in medial, anterior and posterior directions. Consistent with this hypothesis, Zhao et al. (2009) reported ‘interhemispheric fissure bending’ to be more frequent in schizophrenia than in controls and the amount of OB to be more pronounced. Those findings suggested that torque occurs in various regions along the midline, not only in the occipital lobe. Furthermore, they recently reported occipital asymmetry in a group of medication-naive schizophrenia subjects (Pepe et al., 2013).
We hypothesized that OB prevalence would be greater in schizophrenia patients than in controls and that OB would be more rightward than leftward. We also hypothesized that lateral ventricular volume and the volume in receptive language regions would be greater among those with schizophrenia and greatest in those with OB, especially on the side of OB.
Method
Participants
The sample (Table 1) included 37 subjects (males = 22) with a diagnosis of schizophrenia or schizoaffective disorder confirmed by the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition (DSM-IV) (SCID; First et al., 2001), aged 20–62 years (mean age = 39.24 years, standard deviation [SD] = 11.02 years; male mean value = 36.86, SD = 10.29; female mean value = 42.73, SD = 11.46), and 44 controls (male = 23; mean age = 39.66 years, SD = 12.90 years; male mean age = 38.35 years, SD = 10.91 years; female mean age = 41.10 years, SD = 14.93 years). Severity of positive and negative symptoms was measured with the Positive and Negative Symptom Scale (PANSS; Kay et al., 1987). Participants were recruited either by public notice or word of mouth from the clinical services of The Alfred Hospital, Victoria, Australia, and the Centre for Addiction and Mental Health, University of Toronto, Canada. Exclusion criteria included axis I comorbidity (except for the presence of generalized anxiety disorder or social phobia), current active medical problem, a known neurological disease or a contraindication to magnetic resonance (MR) scanning. Healthy control subjects were required to have no history of psychiatric illness. All subjects provided written informed consent on a form approved by the Alfred Human Subjects Research and Ethics Committee and the Centre for Addiction and Mental Health Research Ethics Board.
Descriptive statistics for SCZ and control subjects.
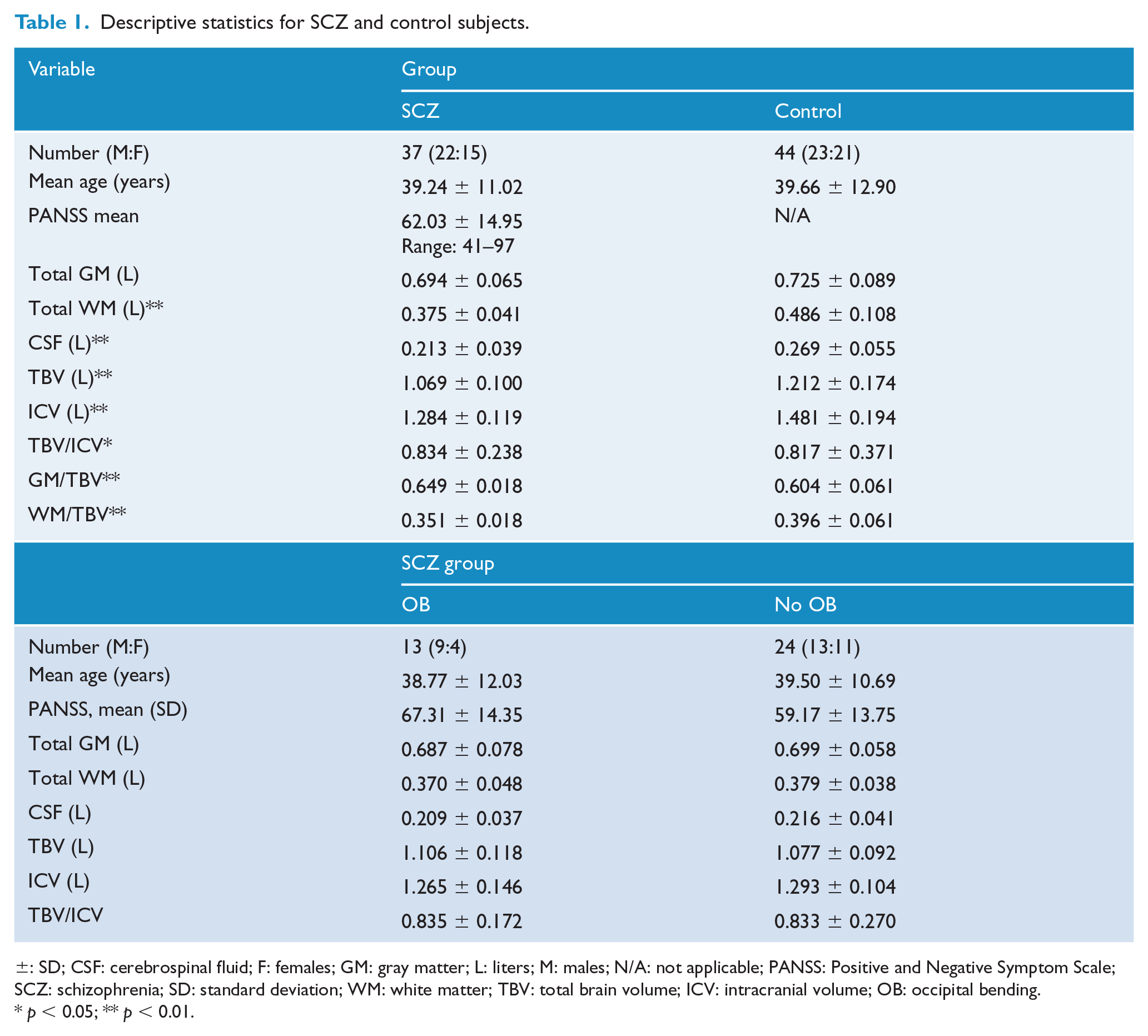
±: SD; CSF: cerebrospinal fluid; F: females; GM: gray matter; L: liters; M: males; N/A: not applicable; PANSS: Positive and Negative Symptom Scale; SCZ: schizophrenia; SD: standard deviation; WM: white matter; TBV: total brain volume; ICV: intracranial volume; OB: occipital bending.
p < 0.05; ** p < 0.01.
Magnetic resonance imaging
A 1.5-T GE Signa Imaging System (General Electric Medical Systems, Milwaukee, WI) was used to acquire a contiguous sagittal inversion recovery (IR)-prepared SPoiled Gradient Recalled (SPGR) T1-weighted sequence for volumetric estimations (TR = 8.628; TE = 1.924; IT = 300; matrix size = 256 × 256, 0.94 mm × 0.94 mm; number of excitations = 1; slice thickness = 1.5 mm) for each subject.
Structural T1-weighted scans were viewed in MRIcro (www.mccauslandcenter.sc.edu/mricro/mricro/) from axial perspective and visually inspected for OB, whereby one occipital pole extended across the midline, that is, interhemispheric fissure; this fissure refers to the narrow groove separating the left and right cerebral hemispheres and is identified by looking for a narrow space separating the two cerebral hemispheres which contains a dural septum called the falx cerebri. This is verified from coronal orientation which demonstrates a warped large narrow space in those with OB. Hence, one occipital pole may cross the interhemispheric fissure, but the fissure must appear warped to be classified as OB, as demonstrated with three-dimensional cortex rendering (Figure 2). Rating of OB, and the degree of OB (measured using Analyze 9.0, Brain Imaging Resource; Mayo Clinic, Rochester, MN), was carried out as previously described (Maller et al., 2015). Two raters (J.J.M. and R.J.A.) independently rated each scan blinded to group membership. Kappa across all scans was 1.0.
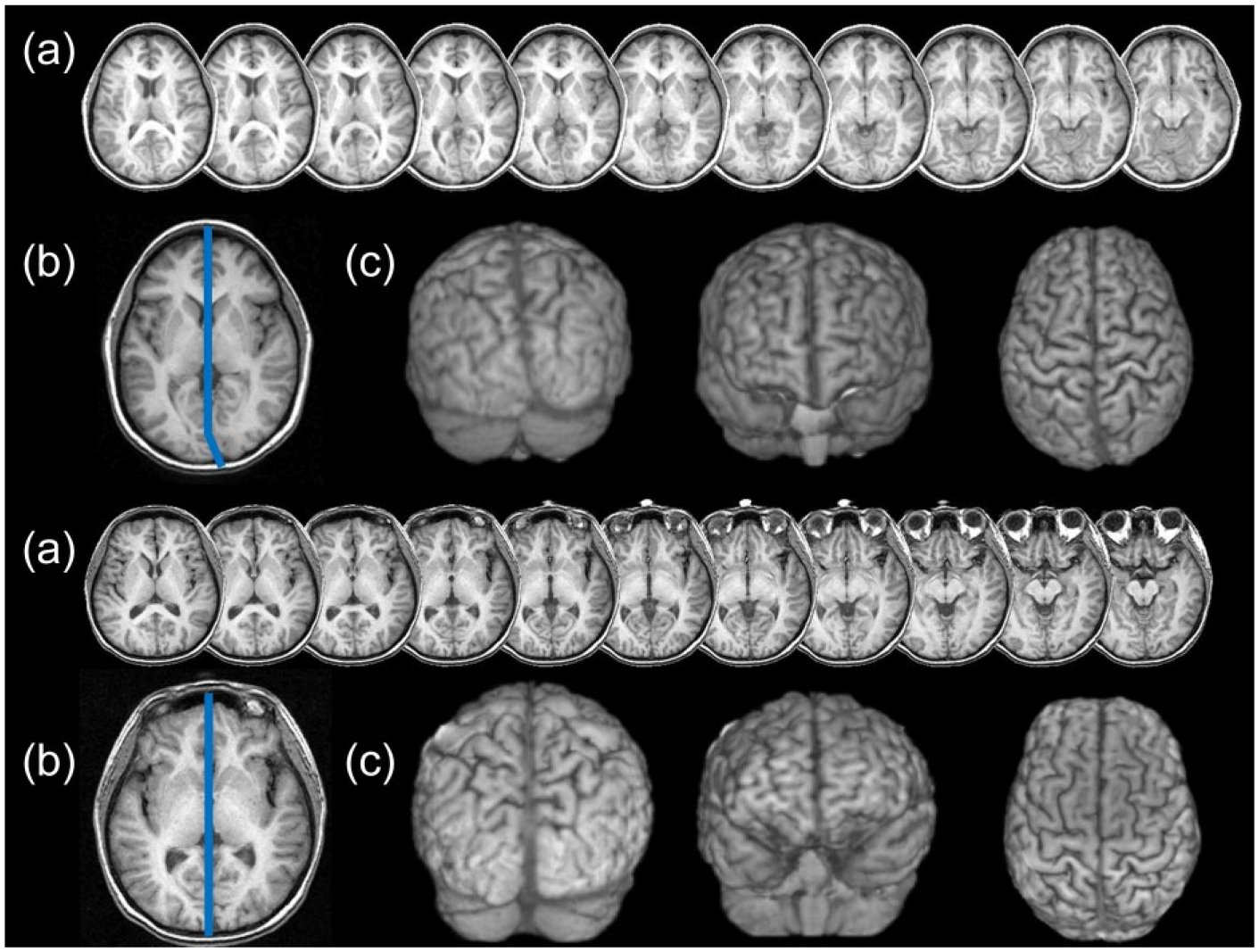
(a) Axial slices 2 mm apart at the level of the occipital lobes. (b) Measurement of OB angle (blue line). (c) Three-dimensional rendering from posterior, superior, anterior perspectives. Upper: Schizophrenia patient with OB. Lower: Control subject with no OB. Images are in neurological convention.
All scans were also processed through the FreeSurfer image analysis suite (http://surfer.nmr.mgh.harvard.edu/) to calculate regional brain volumetrics, which then established occipital and lateral ventricular volumes so that they could be statistically analyzed for relationships with OB. The subsequent parameters within Qdec (the analysis module within FreeSurfer, which displays regional cortical morphometrics including volume, thickness and gyrification) were used for all of the analyses: Measure = Volume (and then Thickness), Smoothing = 10, Monte Carlo simulation was used to correct for multiple comparisons using a threshold of 2.0 (p < 0.01) and the appropriate two-tailed test was applied.
Subcortical volumetric data were exported from FreeSurfer into SPSS for Windows, version 22.0 (SPSS Inc., Chicago, IL) for statistical analysis as Qdec does not analyze these data. Furthermore, FreeSurfer does not calculate extra-ventricular cerebrospinal fluid (CSF) volume; hence, we used SPM8 which includes extra-ventricular CSF volume (Valverde et al., 2015) to calculate gross volumetrics (total gray matter, white matter, CSF volume and intracranial volume [ICV], which is the sum of gray matter, white matter and CSF volumes).
Statistical analysis
All data were statistically analyzed using SPSS for Windows, version 22.0 (SPSS Inc., Chicago, IL). Analyses were two-tailed and evaluated for significance at the 0.05 alpha level. Simple chi-square analysis and analysis of variance (ANOVA) were employed to compare demographics, gross volumetrics and prevalence of OB between groups. Ratios of left to right (left/right × 100) occipital lobe volumes were also calculated as were ratios of left to right lateral ventricular volumes. Age and sex were analyzed as covariates, as suggested by Whitwell et al. (2001) and Chen et al. (2014).
Results
Demographics
There were no significant differences between the proportion of males and females overall nor between their mean ages, between or within groups (p > 0.05; Table 1).
Gross volumetrics
Schizophrenia and controls
Mean total brain volume was statistically reduced in the schizophrenia group (Table 1; F(1, 80) = 19.297, p < 0.001) as was ICV (F(1, 80) = 28.827, p < 0.001), and the ratio of total brain volume to ICV was larger in the schizophrenia group (F(1, 80) = 5.369, p = 0.023). All volumetrics were therefore normalized by total brain volume (in addition to sex and age). Normalized total gray matter was significantly greater among the schizophrenia group, but normalized total white matter volume was significantly less (F(1, 80) = 19.605, p < 0.001). The ratios of left and right white matter, gray matter and total hemispheric volume were not significantly different between schizophrenia and control groups.
Within the schizophrenia group
There was no significant difference between mean total brain volume or ICV of those who had and did not have OB within the schizophrenia group. There was also no significant age difference between those with and without OB, either as whole groups or when divided by sex.
Prevalence of OB within and between groups
Schizophrenia and controls
The prevalence of OB within the schizophrenia group was 35.1%, which was statistically greater than in the control group (13.6%; χ2(1) = 5.174, p = 0.022; Table 2).The direction of OB occurred more often in the left-to-right (rightward) direction in both groups (only four subjects, three schizophrenia and one control, had leftward OB). One scan demonstrated frontal right-to-left (leftward) torquing.
Prevalence of occipital bending.
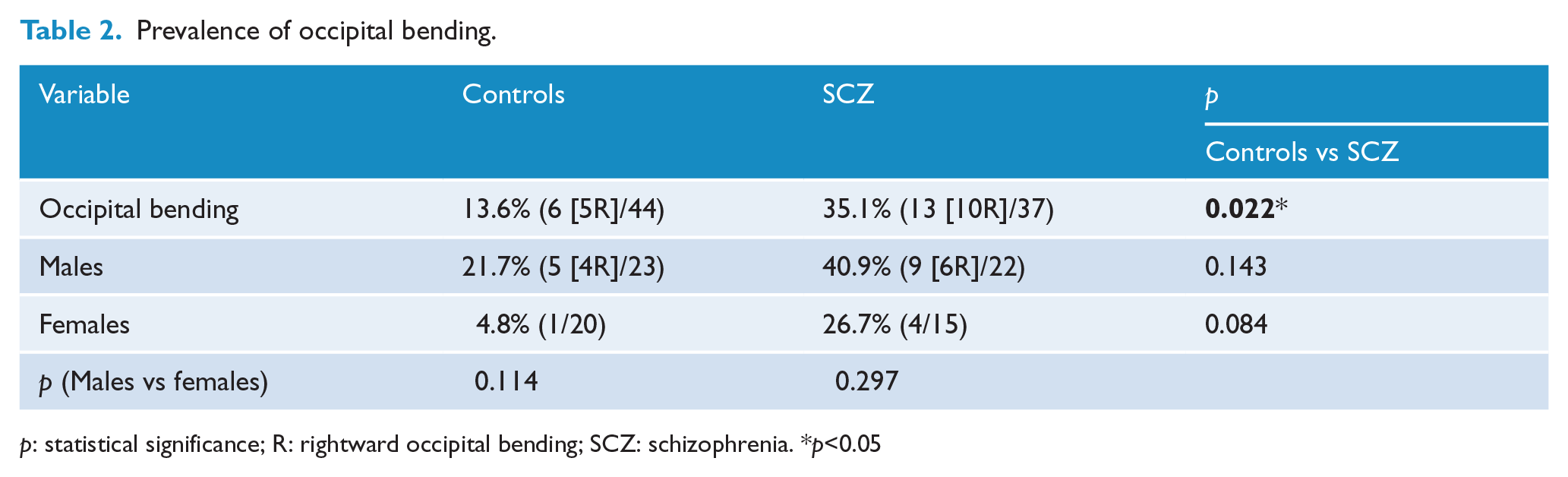
p: statistical significance; R: rightward occipital bending; SCZ: schizophrenia. *p<0.05
There was also no statistical difference in OB prevalence between males and females within the control or schizophrenia group. There was no significant statistical difference in OB prevalence between the males of the groups (p > 0.05). The angle of OB statistically differed between the groups (control mean = 1.35°, SD = 3.56°, range = 0.00°–12.15°, schizophrenia mean = 3.46°, SD = 5.20°, range = 0.00°–17.74°, F(1, 79) = 4.687,p = 0.033; when controlling for age and sex, t(1) = 4.351, p = 0.040), but did not statistically differ between sexes within or between groups (p > 0.05).
Within the schizophrenia group
Although the prevalence of OB was greater among males diagnosed with schizophrenia than females diagnosed with schizophrenia, this difference was not statistically significant.
Morphometrics
Schizophrenia and controls
FreeSurfer volumetrics and cortical thickness
The results from the FreeSurfer analyses demonstrated four significant clusters of greater right hemisphere cortical thickness among the schizophrenia group (Table 3 and Figure 3) and two significant clusters of thinner cortical thickness. Results were similar within the left hemisphere.
Significant FreeSurfer clusters detected in the right and left hemispheres between controls and schizophrenia subjects after multiple corrections controlling for total brain volume, sex and age.
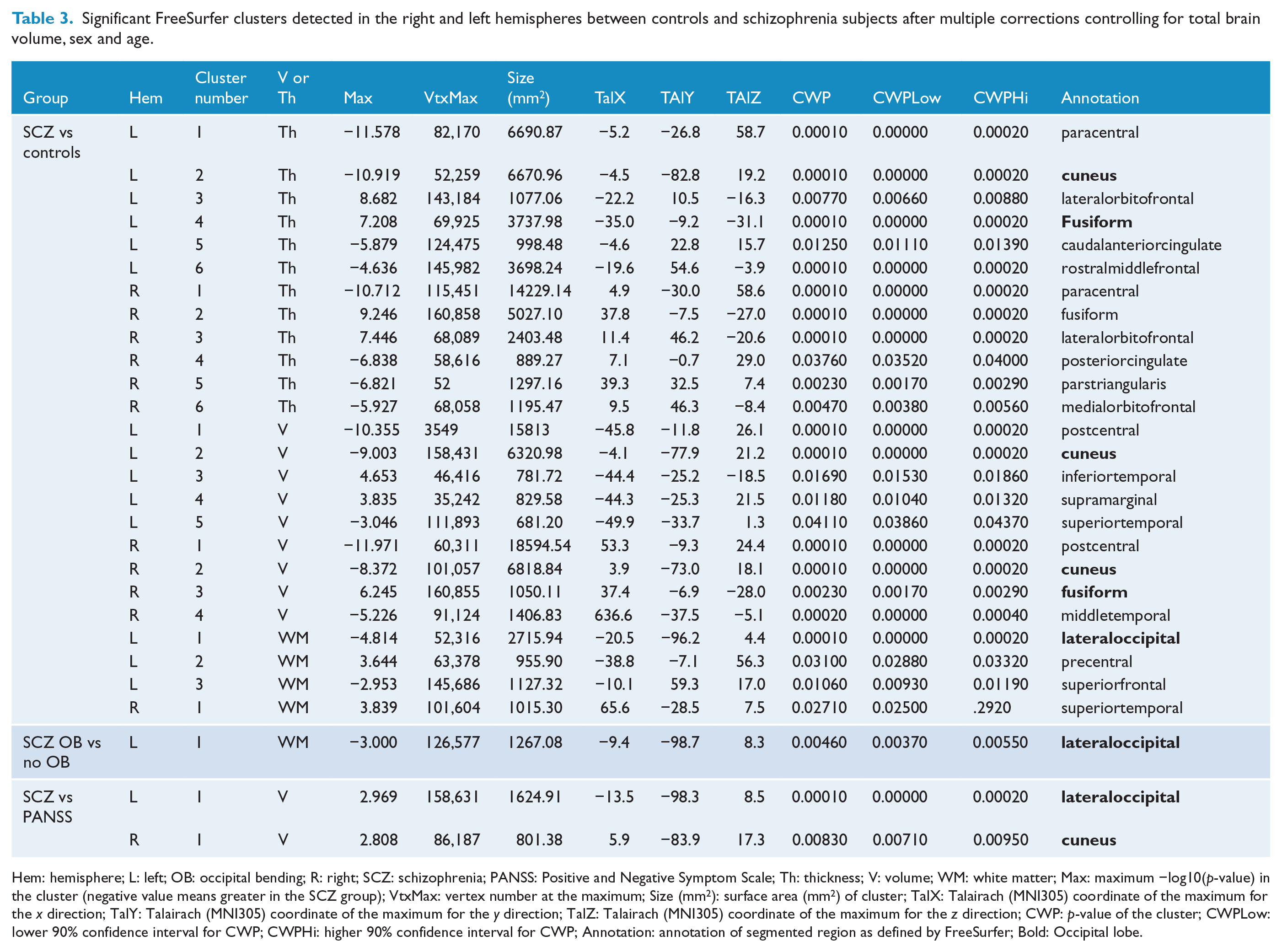
Hem: hemisphere; L: left; OB: occipital bending; R: right; SCZ: schizophrenia; PANSS: Positive and Negative Symptom Scale; Th: thickness; V: volume; WM: white matter; Max: maximum −log10(p-value) in the cluster (negative value means greater in the SCZ group); VtxMax: vertex number at the maximum; Size (mm2): surface area (mm2) of cluster; TalX: Talairach (MNI305) coordinate of the maximum for the x direction; TalY: Talairach (MNI305) coordinate of the maximum for the y direction; TalZ: Talairach (MNI305) coordinate of the maximum for the z direction; CWP: p-value of the cluster; CWPLow: lower 90% confidence interval for CWP; CWPHi: higher 90% confidence interval for CWP; Annotation: annotation of segmented region as defined by FreeSurfer; Bold: Occipital lobe.
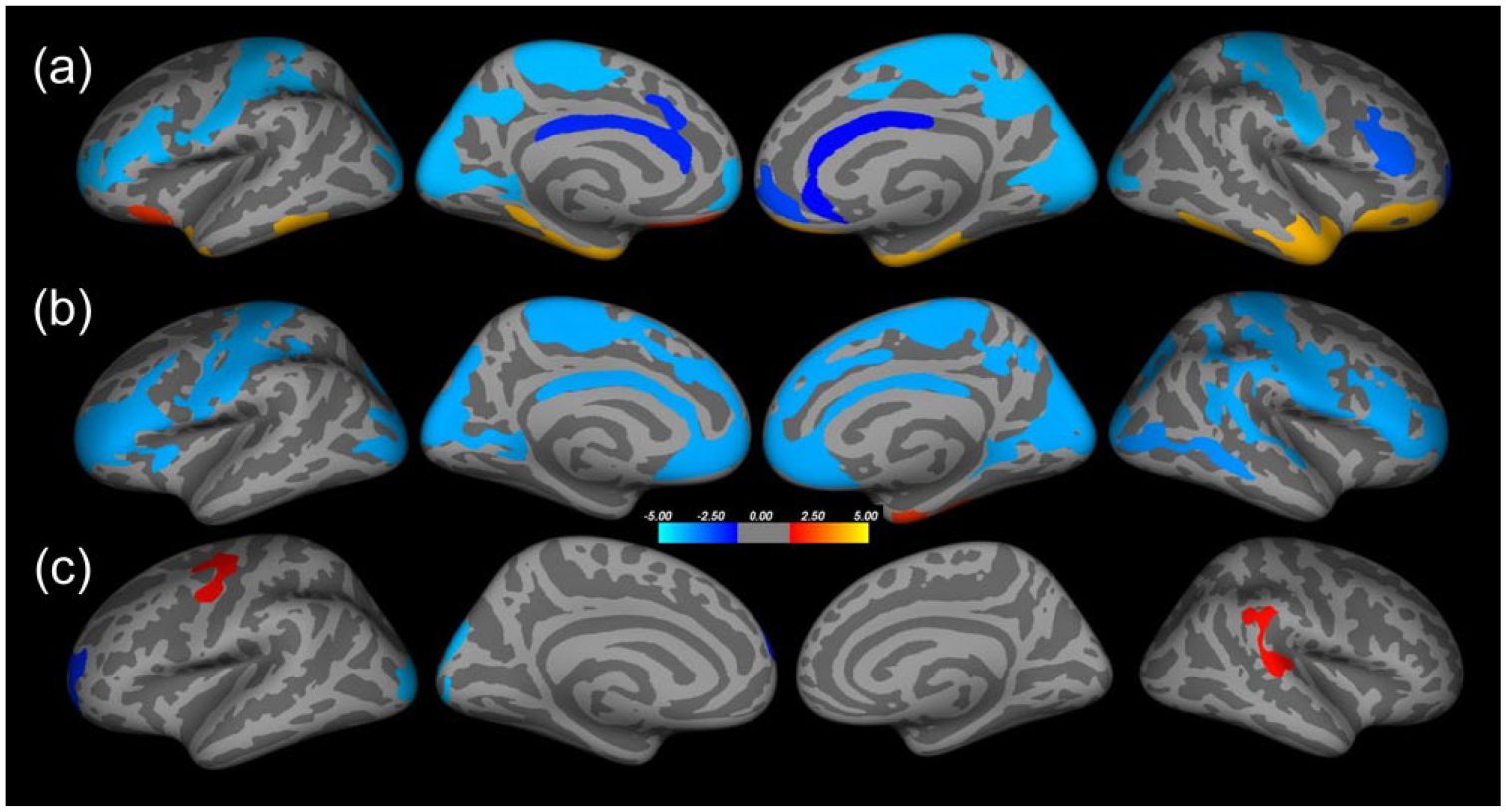
Significant FreeSurfer clusters between groups, corrected for multiple comparisons and controlling for total brain volume and age. (a) Schizophrenia vs controls: cortical thickness. (b) Schizophrenia vs controls: cortical volume. (c) Schizophrenia vs controls: white matter. Negative (blue) values represent increased values in schizophrenia.
There were three clusters representing larger gray matter volume in the schizophrenia group in the left hemisphere, while two clusters were of reduced volume. On the right, there were two clusters of significantly greater gray matter volume in the schizophrenia group and two clusters of reduced volume.
There were two significant clusters of greater white matter volume among the schizophrenia group in the left hemisphere and one significant cluster of lower white matter volume. In the right hemisphere, there was one cluster of smaller white matter volume within the schizophrenia group.
When Heschl’s gyrus (transverse temporal gyrus) was analyzed in SPSS using a general linear model (GLM; which is an analysis procedure commonly used in neuroimaging research to test for differences between groups), white matter volume was significantly larger on the left and right in the schizophrenia group (left: t(1) = 10.684, p = 0.002; right: t(1) = 15.224, p < 0.001). There were no significant differences in the gray matter volumes in this region.
SPSS analysis of subcortical volumetrics
There were a number of subcortical volumes that were significantly reduced in the schizophrenia sample, including the left and right thalamus (left: t(1) = 27.654, p < 0.001; right: t(1) = 55.981, p < 0.001); pallidum (left: t(1) = 12.869, p = 0.001; right: t(1_ = 20.398, p < 0.001); accumbens (left: t(1) = 8.174, p = 0.005; right: t(1) = 6.210, p = 0.015); left anterior cingulate (left: t(1) = 6.200, p = 0.015); posterior (t(1) = 5.361, p = 0.023), midanterior (t(1) = 3.982, p = 0.050) and anterior (t(1) = 6.045, p = 0.016) corpus callosum; and white matter of the parahippocampus (left: t(1) = 23.972, p < 0.001; right: t(1) = 19.990, p < 0.001). The right and left mean amygdalae were significantly larger in the schizophrenia group (left: t(1) = 23.890, p < 0.001; right: t(1) = 10.427, p = 0.002).
SPSS analysis occipital lobe volumes and lateral ventricular volumes
There was no significant difference in the ratio of L:R occipital lobe volumes between the groups. When controlling for age and sex in a GLM, the schizophrenia group had larger right, brain volume – normalized, occipital lobe volumes (t(1) = 7.771, p = 0.007) and larger right and left, total brain volume – normalized, lateral ventricular volumes (left: t(1) = 6.057, p = 0.016, right: t(1) = 6.151, p = 0.015). There was no significant difference in the L:R ratio of the lateral ventricular volumes between these groups.
Within the Schizophrenia group with and without occipital bending
FreeSurfer volumetrics and cortical thickness
There was one significant cluster showing larger mean white matter volume among those with schizophrenia and OB in the left pericalcarine region (Figure 4). There were no significant clusters for cortical volume or thickness in either hemisphere.
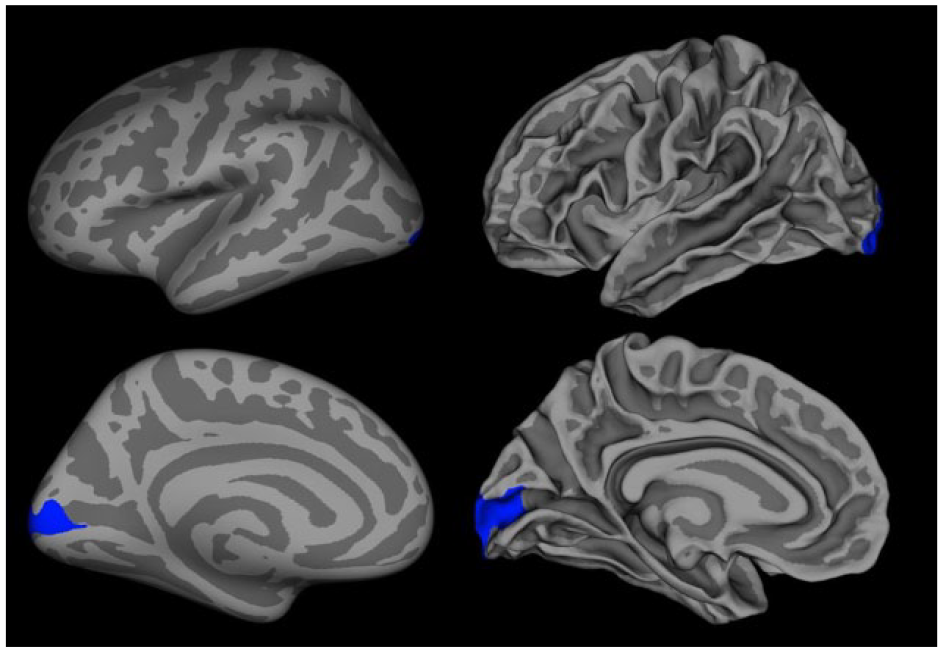
Significant FreeSurfer white matter cluster, corrected for multiple comparisons, indicating the regional difference between Schizophrenia with OB and Schizophrenia without OB. Left images show the inflated brain and right images show white matter.
There were no significant gray or white matter volumetric differences in left or right Heschl’s gyrus or planum temporal between schizophrenia patients with and without OB.
SPSS analysis of subcortical volumes
There were no significant differences in any subcortical region between schizophrenia patients with and without OB, regardless of whether potential confounding variables were controlled for.
SPSS analysis of occipital lobe volumes and lateral ventricular volumes
Left and right lateral ventricular volumes were not statistically different between these groups, nor was the L:R ratio of lateral ventricular volumes. Total occipital lobe volumes were not statistically different either.
There were no significant relationships between angle of OB, curvature indexes, occipital lobe volumes (or their L:R ratio) or lateral ventricular volumes (total brain volume normalized or their ratios) or age or sex (p > 0.05).
Discussion
The prevalence and degree of bending observed in the occipital regions are consistent with our hypothesis that OB prevalence would be greater in schizophrenia patients than in controls and that rightward OB would be more prevalent than leftward OB, consistent with OB studies in schizophrenia (Deutsch et al., 2000; Zhao et al., 2009), major depression (Maller et al., 2014b) and bipolar disorder (Maller et al., 2015).
Our findings are commensurate with a study of OB in major depression (Maller et al., 2014b), which reported OB prevalence of 35.3% among those with major depression and 12.5% among controls, and also consistent with our finding of 34.3% OB prevalence among patients with bipolar disorder (Maller et al., 2015). OB prevalence (and direction) is therefore similar in three psychiatric disorders investigated thus far. The finding of greater OB prevalence in males with schizophrenia is consistent with our findings of OB within bipolar disorder.
Although those with schizophrenia had larger lateral ventricular volumes than controls, which is consistent with the literature, we did not find a relationship between OB and asymmetrical ventricular volume. This is in contrast to our previous hypothesis that rightward OB may be caused by left ventricle enlargement. However, as those with schizophrenia had such enlarged left and right lateral ventricular volumes, the difference between them was not statistically significant and, therefore, was not statistically associated with OB. Hence, the OB in our sample of schizophrenia patients is unlikely to be due to ventricular enlargement. A possible explanation is that OB is a result of shape change rather than volume (Savadjiev et al., 2014).
Interestingly, this study did find a number of regions to be significantly larger among those with schizophrenia, although most were in the occipital lobe, including the pericalcarine sulcus, which is precisely the region expected to be involved in OB. When comparing schizophrenia patients with and without OB, we found statistically greater white matter volume in the left occipital lobe in the region where the OB occurs (pericalcarine). However, volume does not necessarily equate to function. That is, a greater volume may represent reduced neural pruning and less integrity (Foster et al., 1999; Van Petten, 2004), and as such, the hypothesis of compromised left occipital lobes in those with rightward OB is consistent with findings of reduced white matter microstructure in the left occipital lobe in patients with psychiatric disorders (e.g. Benedetti et al., 2011, 2013; Bruno et al., 2008; Sui et al., 2011). It is possible that a developmental abnormality could lead to an excess production of neurons (Selemon et al., 1995). Furthermore, larger volumes in the left occipital cortex than normal support the possibility that there is applied pressure to the immediate tissue which displaces the occipital lobe over to the right hemisphere. We found greater white matter volume in Heschl’s gyrus among those with schizophrenia when compared with control subjects. While this is in contrast to white matter volume findings reported in the literature (e.g. Beasley et al., 2009; Kim and Jeong, 2015), our SPSS results were not corrected for multiple comparisons and, therefore, must be treated with caution.
We estimated cranial and brain volumes and found significant differences in the mean of these volumes between our schizophrenia patients and healthy controls, such that those with schizophrenia had smaller brains but disproportionately smaller cranial size. That is, their heads contained proportionally less CSF than healthy controls. There are consistent reports of reduced brain volume among those with schizophrenia, but such analyses do not always normalize for this factor. Hence, it is not surprising that we did not find schizophrenia volumes to be smaller when normalized by total brain volume. If we had normalized by cranial size and not total brain volume, it is likely that we would have found widespread volumetric reductions among those with schizophrenia. We previously demonstrated that the impact upon results is vastly different depending upon whether data are normalized (Maller et al., 2014a). As for the reduced total brain volume, as previously suggested (Maller et al., 2014b), insufficient neural pruning may initiate an alteration in cortical inhibition, an underlying feature in patients with schizophrenia (Lewis, 2014; Radhu et al., 2015). Restricted neural pruning may force the brain to torque and lead to one occipital lobe warping around the other.
Reduced subcortical volumes are reported to be reduced in schizophrenia patients. We also found this to be true in our sample; however, the hippocampus was not among these regions. In a different schizophrenia sample, we found hippocampal volumes to be significantly reduced when compared with matched controls; however, those results were derived from manual segmentation which is considered the Gold Standard in estimating hippocampal volumes. By contrast, this study estimated hippocampal volumes via an automated routine (FreeSurfer) which has been reported to have up to 29% variability when compared with manual hippocampal segmentation (Cherbuin et al., 2009; Pipitone et al., 2014).
Statistical analyses in Qdec showed that normalized white matter volume was reduced throughout the brain in the schizophrenia sample, but most of these regions did not reach significance following multiple corrections. The only regions to survive multiple corrections were the superior parietal and insular regions. Nevertheless, the overall pattern of findings suggests that normalized gray matter is increased in schizophrenia but normalized white matter is reduced; therefore, the overall finding is one of no difference in normalized brain volume. There are a number of studies demonstrating altered white matter microstructure, as expressed by diffusion-weighted indexes such as fractional anisotropy (Wagner et al., 2015; White et al., 2013), indicating reduced connectivity in the brains of individuals diagnosed with schizophrenia. It is possible that increased gray matter is part of the plastic nature of the brain’s attempt to compensate for this reduced connectivity.
There are recent meta-analyses of magnetic resonance imaging (MRI) studies (Bora et al., 2011, 2012) that have revealed an interesting interaction between sex and laterality and form of psychosis in relation to bipolar illness and schizophrenia in tissue volumes in insula and cingulate gyrus. It would be interesting for future studies to investigate how this might relate to OB.
OB is more prevalent in major depression, bipolar disorder and schizophrenia than in healthy control subjects. However, how the various aspects of OB may differ between the groups, the extent to which other parts of the brain may relate to this phenomenon and examining the potential of that information to aid in diagnosis or screening of psychiatric disorders are yet to be elucidated.
Limitations
Our study did not collect information regarding medications, number of episodes, IQ, education, lifestyle and personality, childhood disorders/abuse or socioeconomic status. All these factors may influence the prevalence or extent of OB. Future investigations will also consider family history of schizophrenia. We also did not formally assess mental illness in controls. Furthermore, handedness was not assessed; people with schizophrenia are more likely to be non-right handed (Hirnstein and Hugdahl, 2014). It has been reported that left-handedness in a group of patients with schizophrenia will vary depending on the percentage of milder forms of schizophrenia in the sample (Luchins et al., 1979).
Results in SPSS were not corrected for multiple comparisons; hence, those results must be treated with caution.
The current study adopted a simple present or absent assessment of OB. Future investigations should aim to quantify displacement in three dimensions. Furthermore, future work should focus on prevalence of bending in other cortical lobes/interhermispheric fissure.
Conclusion
This study investigated OB in a sample of patients diagnosed with schizophrenia and controls. We found that OB is nearly three times more prevalent among patients with schizophrenia and is most commonly rightward. Future research in larger samples, incorporating other neuroimaging measures such as diffusion and spectroscopy, would advance our understanding of the underpinnings and associations of OB. While the origins and implications of OB are unclear, this study suggests, along with previous studies, that OB may be a feature of psychiatric illness.
Footnotes
Acknowledgements
We thank all the patients and volunteers who agreed to participate in this study and the staff of the MRI facility at the Alfred Hospital, Melbourne, Victoria, Australia, and at the Centre for Addiction and Mental Health, University of Toronto, Canada. We would also like to thank Dr Stuart Lee for his assistance with statistical analysis.
Contributors
J.J.M. designed the study, wrote the protocol, carried out measurements, conducted the statistics and wrote the first draft. R.J.A. carried out measurements. R.H.T. performed statistical analyses. Z.J.D. designed the study and performed psychiatric assessments. J.V.R. assisted in the designing the protocol. P.B.F. designed the study and performed psychiatric assessments. All authors contributed to editing the manuscript.
Declaration of Conflicting Interests
In the last 5 years, Z.J.D. received research and equipment in-kind support for an investigator-initiated study through Brainsway Inc. Z.J.D. has also served on the advisory board for Hoffmann-La Roche Limited and Merck and received speaker support from Sunovion and Eli Lilly.
Funding
J.J.M. is funded by the National Health and Medical Research Council (NHMRC) as a Career Development Fellow; the funding agency had no further role in study design; in the collection, analysis and interpretation of data; in the writing of the report; or in the decision to submit this paper for publication. P.B.F. is supported by an NHMRC Practitioner Fellowship (6069070).
