Abstract
While much emphasis has been placed on the cortex as a site of interest in schizophrenia, cortical function is dependent on connections with subcortical structures such as the thalamus and basal ganglia. We have previously hypothesized that the basal ganglia and thalami may be key structures underlying the neurobiology of schizophrenia, particularly given the proposed parallels between the frontal-subcortical dementias and schizophrenia [1] and the role of frontal-striato-thalamic circuits in this disorder [2, 3].
Recent authors have focused on the thalamus, a key relay station for processing of sensory and other information, as a potential site of structural abnormality in schizophrenia (e.g. [4–7]). The thalamus also receives input from the hippocampus and amygdala, which are also abnormal in schizophrenia [7, 8]. In addition, there have been neuropathological reports of reduced cell count in the dorsomedial thalamic nucleus [9, 10]. However, functional imaging literature is sparse and mixed. Some studies have identified thalamic hypofunctioning in schizophrenia [11–13], hyperfunctioning [14], no change [15, 16], or abnormal patterns compared to normals [17].
The basal ganglia are components of neural circuits that involve the thalami and have connections to cortical and limbic regions. In addition to their well-established role in the control of movement, the basal ganglia are thought to play an important role in cognitive functions such as attention and the regulation of affect [18]. Functional imaging studies in schizophrenia have reported changes in the basal ganglia, but these are somewhat contradictory (for a review, see [19]). In a study of 18 never-medicated patients, Buchsbaum and colleagues [20] found diminished metabolic rates in the basal ganglia. These data are also supported by Siegel et al. [15] in a positron emission tomography study of 70 unmedicated patients. However, studies in medicated patients usually report hypermetabolism in the basal ganglia [11, 16, 21], suggesting that this is an effect of antipsychotic medication. Therefore, the few available functional imaging studies in unmedicated or drug-naïve patients suggest that the basal ganglia are hypofunctional in schizophrenia, with a similar, though weaker tendency for the thalamus.
Structural imaging studies have also identified thalamic and caudate volumetric abnormalities in schizophrenia. The finding of increased caudate volume has generally been attributed to the effects of medication, which is supported by the few studies in drug-naïve patients that have reported increases in caudate volume following neuroleptic treatment [22, 23], and a study by Keshavan and colleagues [24] that reported bilaterally smaller caudate volume in 16 neuroleptic naïve first episode patients compared to normal controls. Further, a recent longitudinal study [25] found a reduction of caudate volume when patients were switched from classical neuroleptics to the atypical antipsychotic clozapine. In addition to the medication issue, the variability of findings that exist in the literature may be attributed to factors such as poor matching of patients and control subjects or the use of small subject numbers.
While the literature tends to report increased caudate volumes in schizophrenia, reports tend to indicate decreased thalamic volume [5, 6, 26–28] or no change [7, 29–34]. Of the two studies which report data from drug naïve patients in comparison with controls, one [13] found no difference in thalamic area, while another [6] found a trend toward lower volumes in neuroleptic naïve patients. Further, in a recent study thalamic volumes were smaller than controls in a familial high-risk sample premorbidly, but not in a group of first-episode schizophrenia patients [7].
An important problem in structural imaging studies is the difficulty in the mensuration of the thalamus. The thalamus is a grey matter structure with numerous white matter afferents and efferents, resulting in an unclear boundary when viewed by magnetic resonance imaging (MRI). Adequate boundary delineation is made more difficult through the use of thick slices, resulting in partial volume artefact. Additionally, the lateral borders are particularly difficult to define when using T1-weighted images, as is common in recent studies (e.g [5]).
In an effort to address the issues of poor delineation of these subcortical grey matter structures and resulting low reliability, we developed a novel method for volumetric estimation of both the caudate and thalami using algebraically manipulated double echo MRI sequences. Summing or subtracting images (i.e. simple signal averaging) acquired at different echo times (TE) can differentially enhance fluid/tissue or grey/white contrast [35]. Addition of the paired T2 and proton density (PD) images has been shown to enhance grey/white matter contrast, while subtraction enhanced tissue/fluid contrast (see [35]).
This paper reports on the optimal method for volumetric estimation of these two structures, through the evaluation of the relative volumes and reliability differences in measures of thalamic and caudate volumes produced from four image types: the originally acquired double echo PD and T2-weighted images, and the images resulting from the algebraic summation and subtraction of these images. The issue of different volumetric estimates from different image types is important, since it is frequently assumed that measurements from MRI are equivalent to those obtained from actual tissue, with little or no regard to contrast or partial volume effects.
Further methodological issues in the evaluation of subtle structural abnormalities in schizophrenia relate to difficulties in matching patients and control subjects. There is an inherent difficulty comparing volumetric differences between patients and controls due to the high degree of overlap between the two groups. One strategy to address this issue is the use of monozygotic twins discordant for schizophrenia. The use of monozygotic (MZ) twins has previously been successful in identifying subtle structural changes in patients with schizophrenia [36]. Because each pair of twins shares a common genome and similar educational, economic and family backgrounds the degree to which these factors contribute to subcortical volume is minimized. To date, there have been no studies examining thalamic volumes using twin methodology. We report on a group of MZ twins discordant for schizophrenia, using the subsample of those originally studied by Suddath and colleagues [36] who had dual echo scans.
Method
Subjects
The subject sample consisted of 13 sets of MZ twins, eight discordant for schizophrenia (three female, five male) and five normal (one female, four male), which were recruited as part of a larger study at the National Institute of Mental Health, Washington DC [37]. Zygosity was determined by physical similarity and a likeness questionnaire [38], and confirmed by analysis of 19 red-cell antigens and, when necessary, by fingerprinting and HLA typing. The minimum length of discordance was 4 years. The study was approved by the institutional ethics committee; subjects provided written informed consent to participate in the study.
The diagnosis in both the discordant and normal sets of twins was assessed using the Structured Clinical Interview for DSM-III-R (SCID-III-R) [39]. The affected twins of the discordant pairs met the dianostic criteria for schizophrenia, while none of the twin pairs met criteria for a schizophrenia spectrum disorder (schizophrenia, schizoaffective disorder, schizophreniform psychosis or delusional disorder). None of the study participants met criteria for substance abuse either at the time of testing or in recent years.
The mean age (± SD) for the discordant group was 34.1 ± 6.8 years (range 25–41) and for the normal group was 31.4 ± 5.3 years (range 27–41). The mean age of illness onset for the affected twins in the discordant group was 20.6 ± 4.9 years (range 14–30) and the mean illness duration at assessment was 13.5 ± 6.5 years (range 8–27). All were receiving neuroleptic medication (mean total lifetime dose in fluphenazine equivalents = 34 375 mg; range = 3 000–112 000 mg). Only one had received a course of electroconvulsive therapy.
MRI acquisition and image processing
The same 1.5T Signa scanner (General Electric Medical systems) and the same scanning sequences were used for all subjects. A double echo sequence with time-to-echo (TE) = 20, 80 ms and time-torepetition (TR) = 2000 ms was used to obtain simultaneous protondensity (PD) and T2-weighted images. For each echo, 18 trans-axial slices parallel to the orbito-meatal line were obtained with 6.0 mm thickness with an interslice gap of 0.5 mm (field-of-view = 240 mm, 256 · 256 matrix). The data were transferred to a Unix workstation running ANALYSE (Biomedical Imaging Resource, Mayo Foundation/ Clinic, Rochester, MN MAYO clinic) for subsequent processing and analysis.
Image processing
Each T2 slice was stripped of skull and scalp using a semiautomated procedure to produce a series of 1-bit masks. Multiplication of the PD images by the mask was used to strip the PD images, thus producing two identically stripped images per slice. Subsequent addition of the paired T2 and PD images enhanced grey/white matter contrast, while subtraction enhanced tissue/fluid contrast (see [35]; Fig. 1).
Original and modified MR images: (a) proton density (PD); (b) T2-weighted; (c) summed (PD + T2); (d) subtracted (PD-T2)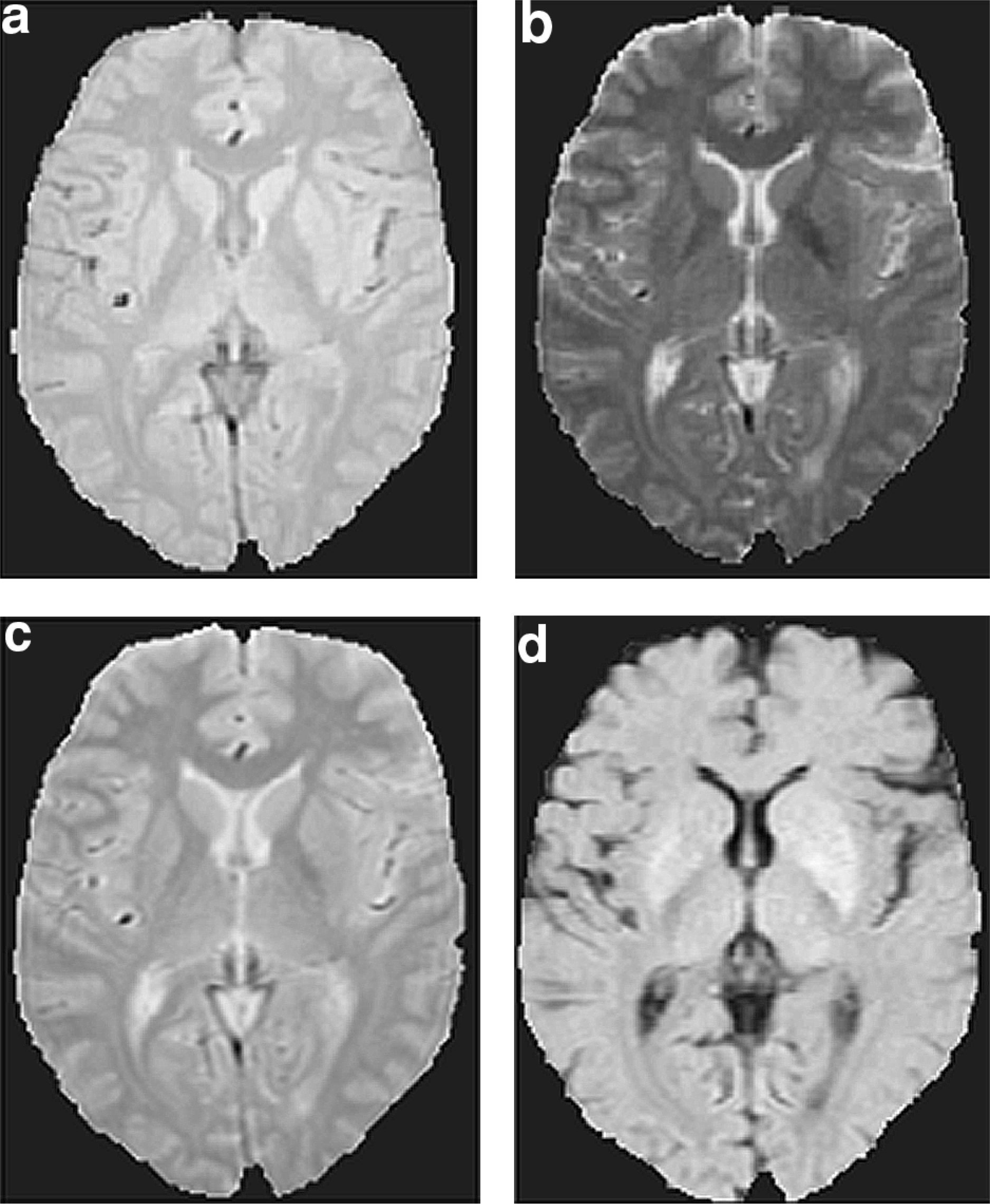
Measurements
Scans were coded so that raters were blind to all subject characteristics. The raters traced the region of interest in the axial plane on every slice in which it could be visualized (typically 2–4 slices). Some thresholding (increasing the image contrast by manipulating the pixel intensity maxima and minima) was used to enhance unclear borders.
The definitions of the landmarks used for the caudate and thalamus are as follows. We used the subcallosal fasciculus as the anterior boundary of the caudate, the stria terminalis as the posterior boundary, the lateral ventricle as the medial boundary and the anterior limb of the internal capsule as the lateral boundary. The tail of the caudate was not traced, as it was not readily identified on these sequences. The anterior boundary of the thalamus was defined as the frontal horn of the lateral ventricle, the posterior border as the occipital horn of the lateral ventricle superiorly and a cerebrospinal fluid (CSF)-filled cistern inferiorly, the medial boundary as the third ventricle, and the lateral boundary as the genu and posterior limb of the internal capsule.
Regions of interest were initially measured on a subset of 13 randomly selected individuals from the total sample of 26. These 13 individuals consisted of five discordant pairs, one normal pair and one single schizophrenia subject. The thalamus was measured on all four image types (T2, PD, summed and subtracted), and the caudate on T2, PD, and summed only as it was not visible on subtracted images. Based on these analyses the summed images were chosen as the optimal method to assess the caudate and thalamic volumes for the entire data set (see below for details).
We also estimated whole brain volumes on the 1-bit masked images using a semiautomated technique which excluded cisterns, sulcal cerebrospinal fluid and extracortical tissue. Volumes were calculated on all slices beginning from the inferior slice above the level of the orbits (typically 9–11 slices).
Reliability of different image types
One rater (NB) performed all measurements on the subset of 13 patients to assess intrarater reliability, while interrater reliability was determined between NB and two other investigators. Summed images produced the highest reliabilities, as measured by intraclass correlation coefficients (ICC), for both caudate and thalamus (see Table 1). When this sample was retraced for a third time, approximately 12 months later, reliability remained high (ICC = 0.90).
Caudate and thalamic reliabilities (intraclass correlation coefficients) of volumes produced by different image types
Results
Comparison of image types
The volumes determined for each image type and for each structure are presented in Fig. 2. Proton density images produced larger caudate volumes than T2 or summed images (PD: 6937 ± 1026, T2: 4932 ± 956, and summed: 4513 ± 998 mm3) while T2 images produced larger thalamic volumes than PD, summed or subtracted images (T2: 17 530 ± 1904, PD: 15 357 ± 2211, summed: 15 530 ± 2019 and subtracted: 14 731 ± 1764 mm3).
Thalamic (dotted lines) and caudate (solid lines) volumes (mm3) of a subset of 5 discordant twin pairs produced from T2, proton density (PD) and summed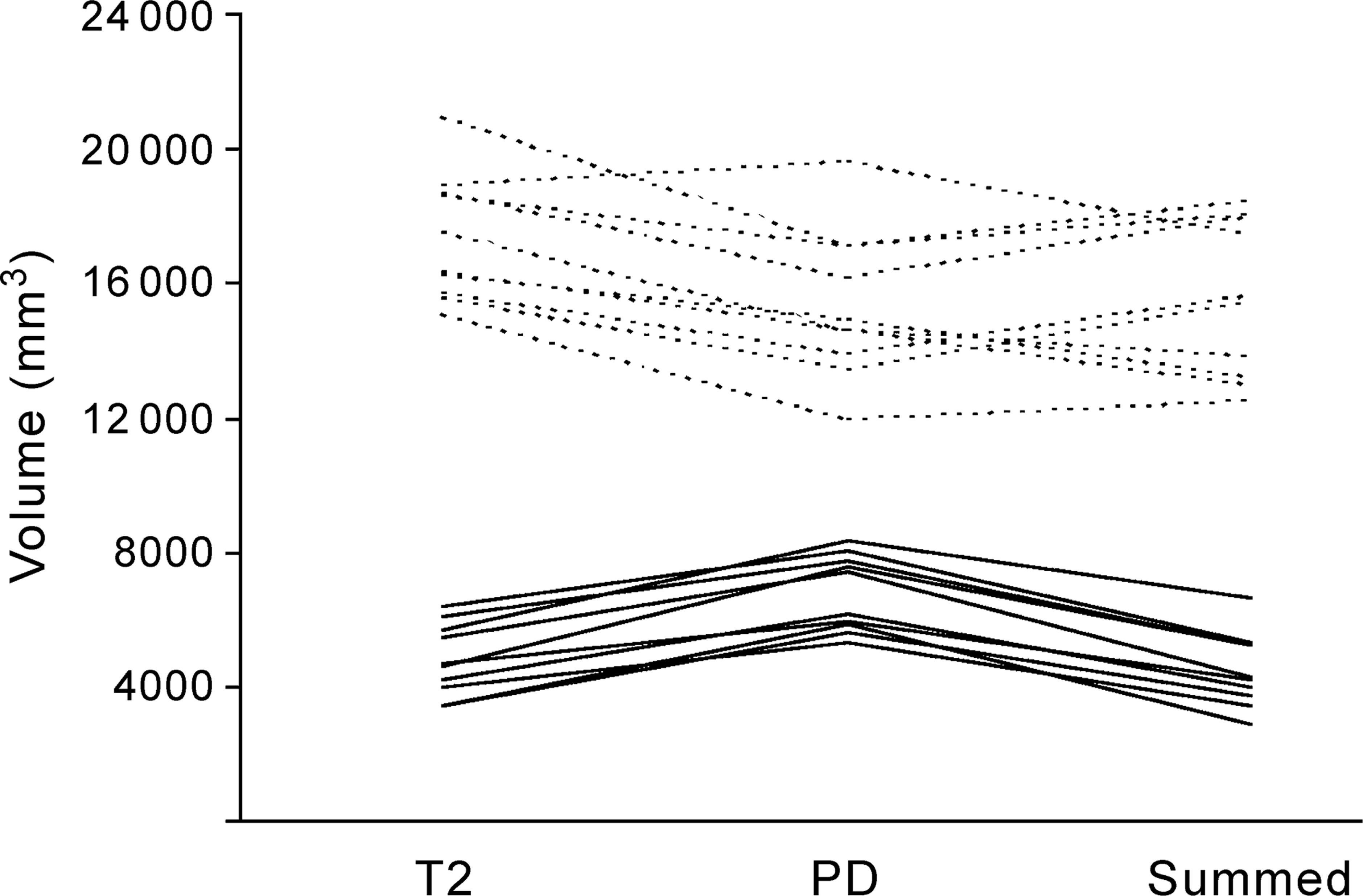
A 3-way repeated measures ANOVA (region by image type by diagnosis) was performed on the five pairs of twins discordant for schizophrenia from the subset of the 13 randomly selected individuals. There was no 3–way interaction, suggesting that the volumes obtained for each structure and for each method were independent of diagnosis. However, there was a significant image type by region interaction (Wilks’ λ = 0.069, F[2, 3] = 20.35, p = 0.018) (see Fig. 2).
Because the summed images were the most reliable (see Table 1) for both caudate and thalamic measurements, they were chosen to measure thalamic and caudate volumes on all 26 subjects.
Wilcoxon Signed Rank Tests (WSRT) were used for subsequent analyses because of the small sample and non-normal distribution of the measured volumes. The WSRT tests differences between the medians of distributions which are unaffected by outliers. Empirical sampling distributions were computed by SPSS Exact to give exact P-values.
Whole brain volumes and laterality effects
We found no difference in whole brain volumes (see Table 2) between twins for either the discordant group (WSRT [exact], p = 0.473) or the normal group (WSRT [exact], p = 0.188). Further, there were no significant differences between either left and right thalamic or caudate volumes when compared for each twin in the three groups. There were also no significant differences in the differences between left and right caudate (WSRT [exact], p = 0.95) or thalamic (WSRT [exact], p = 0.742) volumes when comparing affected twins with their unaffected cotwin. Therefore we report total, absolute volumes.
Mean thalamic, caudate, and wholebrain volumes (mm3) of monozygotic twins discordant for schizophrenia and normal twins
Group analysis of caudate and thalamic volumes
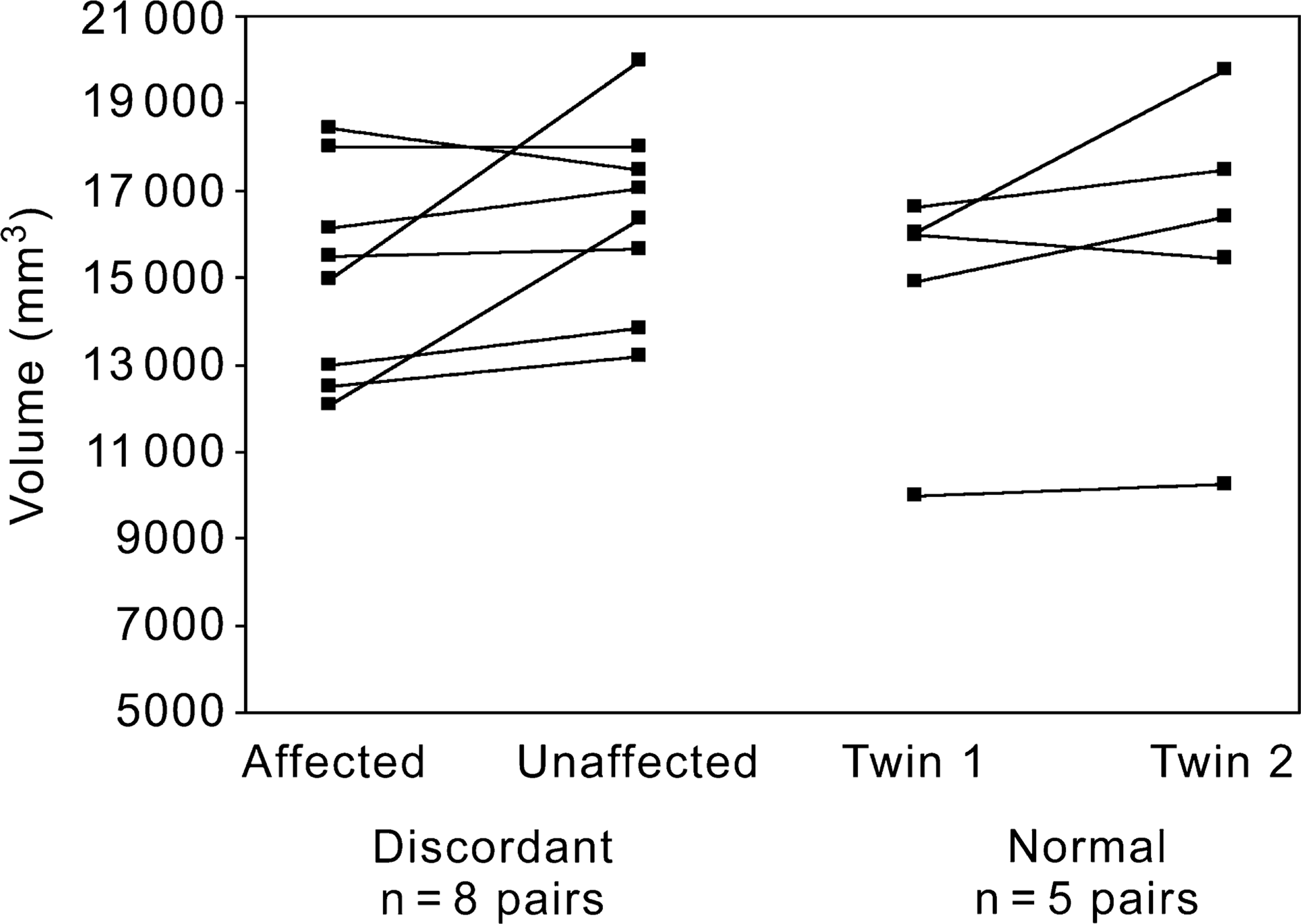
Figures 3 and 4 and Table 2 show the total caudate and thalamic volumes of both groups of twins. When the affected twins were compared with their unaffected cotwins, significantly larger caudate volumes were found in the affected twins (WSRT [exact], p = 0.012), while normal twins did not differ (WSRT [exact], p = 0.125). There were no differences in the thalamus between affected and unaffected twins (WSRT [exact], p = 0.109) nor between normal twins (WSRT [exact], p = 0.188). In addition, we examined the correlation between total caudate volume of the affected group and lifetime medication dose, which, while not significant, did indicate a slight trend for larger caudate volumes to be associated with larger total lifetime doses (r s = 0.56, p = 0.073).
Total caudate volumes (mm3) of monozygotic twins discordant for schizophrenia and normal monozygotic twin pairs Total thalamic volumes (mm3) of monoygotic twins discordant for schizophrenia and normal monoygotic twin pairs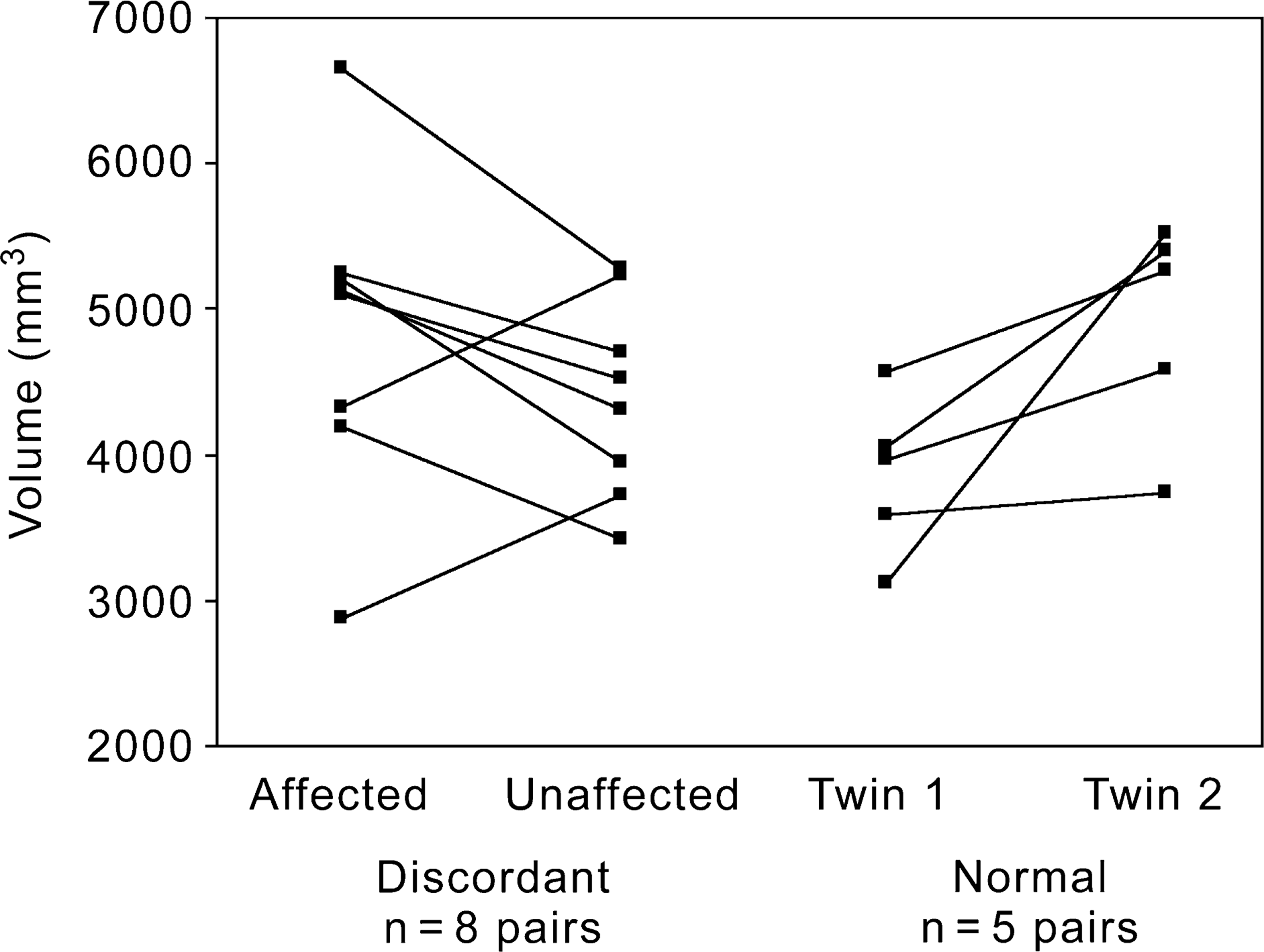
Discussion
In this study, we developed a novel and reliable method for estimating the volume of the thalami and caudate nuclei in a sample of twins discordant for schizophrenia. The main findings of this study were of increased caudate volumes but no thalamic volume differences in affected twins compared to their unaffected cotwins.
Further, using the different image types (PD, T2, summed and subtracted) to estimate thalamic and caudate volumes we found significantly different volumes for each image type, and an interaction of structure (thalamus or caudate) by image type. This finding raises methodological issues about using MRI to examine different brain structures.
Our finding of increased caudate volume in affected twins compared to their unaffected cotwins is probably due to the effects of typical antipsychotic medication. This has been demonstrated by Chakos et al. [25], who showed significant caudate volume reduction when chronically treated schizophrenia patients were switched from classical neuroleptics to the atypical antipsychotic clozapine. Although the correlation between caudate volume and total lifetime medication in the present study did not reach significance, the trend supports previous work [6]. However, it is clear from Fig. 3 that two of the discordant twin pairs had caudate volumes which were the reverse of the expected relationship (i.e. the ill twin had smaller caudate nuclei). In addition, two of the normal twin pairs had extremely large intra-twin differences in their caudate volumes. While this may reduce the confidence with which we ascribe the enlarged caudate to medication, we feel that the strong findings from previous studies support our conclusion.
There are important methodological limitations to many previous studies, some of which we have tried to address. While caudate measurement reliability is usually high, reliability estimates for thalamic volumes have been low (e.g [5]), or not reported. Of 10 thalamic studies reviewed for this paper, only two report high reliability [30, 33], although the latter paper assessed reliability on only three cases. Our method of using summed double echo MR images appears to have overcome this problem, as well as producing caudate reliability estimates on par, or better than previous studies. This was despite the use of thick (6 mm) slices.
Another strength of the current study was the use of MZ twins. Some previous studies have either not matched their patient and control samples or have used suboptimal matching strategies (e.g. [13, 32, 40]). When assessing small volume differences between patients with schizophrenia and controls, the large overlap between the groups can mask subtle differences if they are not adequately matched. The study of twins is considered to reduce the degree of variation between patients and controls and therefore increase discriminative power. While our sample was small and the images used relatively thick, significant differences in caudate volumes were identified between the discordant twin pairs. This suggests that our technique was able to detect a 10% difference in volume. However, larger subject samples together with thinner image slices may be necessary to detect more subtle volume differences in the thalamus [7].
An equally important, yet unexpected finding in the present study was an interaction between the mensuration method used (image type) and the structure in question. Caudate volumes were larger when PD or summed images were used in comparison with T2 images, while in contrast, thalamic volumes were larger on T2 images. However, there was no interaction with diagnosis, suggesting that comparison between patients with schizophrenia and controls was valid regardless of the method used.
The likely explanation for this methodological finding is the effect of partial volume at tissue boundaries. The caudate has CSF on its medial border, while the thalamus has little surrounding CSF but a number of fibre tracts from the internal capsule that blur its lateral boundary. When estimating caudate volume on T2 images, CSF is bright and is unlikely to be mistakenly included as caudate, resulting in a lower volume. On PD images (particularly in the superior aspect), the frontal periventricular white matter is brighter than surrounding white matter and may be included in the caudate measurement, resulting in a larger reported volume. When estimating thalamic volume on T2 images, there is an apparent ‘lightening’ of white matter in the posterior border of thalamus (the internal capsule), making it appear more like grey matter. Therefore, there is a tendency to include this tissue in thalamic measurements, resulting in a larger reported volume. This ‘lightening’ of white matter may be a result of striatal cell bridges in the posterior limb of the internal capsule. It may be that the use of thinner slices would reduce this effect and result in smaller thalamic volumes reported from T2 images.
If this is the explanation for the effects observed it becomes important to examine such interactions, as brains with more or less CSF may produce different volumes for certain structures. That is, while there was no interaction between method, structure and diagnosis in our study, such interactions need to be excluded in conditions in which CSF spaces are increased, such as schizophrenia. Comparisons of the various techniques with post-mortem specimens that have been scanned may be useful to determine the validity of different method(s) and would be necessary if significant interactions with diagnosis are found.
In summary, the present study reports an improved method for measuring the volumes of subcortical structures, with greater reliability than previous methods, particularly for the thalamus. Our findings showed that caudate volumes were larger in patients with schizophrenia in comparison with their unaffected twin, presumably due to treatment with typical antipsychotic medication, while no differences were identified in the thalamic volume. Further studies that carefully assess the validity of the methods used are necessary to examine the nature of thalamic changes in schizophrenia.
Acknowledgements
Initial collection of the twins data was supported in part by a grant (MH 41176) from the National Institute of Mental Health to the Friends Medical Science Research Center and by funds from the Theodore & Vada Stanley Foundation. The imaging analyses were conducted in Australia and were supported by Jack Brockhoff Foundation, Ian Potter Foundation, L.E.W. Carty Trust and Percy Baxter Charitable Trust, and the Australian Communications and Computing Institute. Dennis Velakoulis was supported as a National Health and Medical Research Council research officer. Christos Pantelis's initial work on the twin data at National Institute of Mental Health leading to this work was supported by the Lilly Travel Fellowship from the Royal College of Psychiatrists (UK) and a travel fellowship from the King Edward's Hospital Fund for London. We thank Michael Wong for his contribution during the preparation of the text and collection of the data, and Geoff Stuart and Paul Dudgeon for their valuable statistical advice.
