Abstract
Background:
There is increasing evidence that children with autism spectrum disorder are accompanied by specific anatomical alterations. However, the anatomical abnormalities in adults with autism spectrum disorder are poorly understood. This study was aimed to identify the neuroanatomical substrates underlying the pathophysiology of adults with autism spectrum disorder. We also investigated the relationship between neuroanatomical alterations and clinical and demographic characteristics.
Methods:
A total of 13 datasets were enrolled, of which 12 studies compared whole-brain differences of 382 adult patients with autism and 393 healthy control subjects. We conducted a meta-analysis to quantitatively estimate regional gray matter volume abnormalities in individuals with autism using the effect-size signed differential mapping.
Results:
The voxel-wise meta-analysis revealed that relative to controls, adults with autism spectrum disorder had significantly increased gray matter volume in the middle temporal gyrus, superior temporal gyrus, postcentral gyrus and parahippocampal gyrus, and reduced gray matter volume in the anterior cingulate cortex and cerebellum. Variations in gray matter volume were significantly associated with the mean age and mean total IQ score of the patients, as well as with the percentage of male patients with autism.
Conclusion:
These findings confirmed that the neuroanatomical alterations in the fronto-temporal cortices, limbic system and cerebellum in adult individuals with autism were different from the children and young adolescent’s autism. The effects of demographic characteristics on the brain morphological changes allow us to further clarify the neurobiological mechanisms and developmental trajectory in adult population with autism spectrum disorder.
Introduction
Autism spectrum disorder (ASD) is considered as a lifelong neurodevelopmental disorder and is characterized by deficits in communication and social interaction as well as repetitive behavior or restricted interest (Diagnostic and Statistical Manual of Mental Disorders–Fifth Edition [DSM-5], American Psychiatric Association, 2013).Since first behavioral symptoms appear early in life and the diagnosis of autism is commonly made by 2–4 years of age, the literature on the autism has largely focused on children and young adolescents. For those diagnosed with ASD in childhood, most symptoms would tone down or even disappear in several typical childhood autistic traits when they become adults, but leaving core autism features, such as limited reciprocity in communication and social interaction and a very restricted range of self-chosen behaviors (Billstedt et al., 2007). In contrast to children with autism, adults with autism often show significant disability, as well as psychiatric and behavioral comorbidities (Marriage et al., 2009). Moreover, comorbid conditions, medication and other factors can complicate and change the ASD symptom profile, and thus bring difficulty in obtaining an accurate diagnosis of adult with autism.
Although the etiopathogenesis of autism has yet to be determined, it has been suggested that brain structure abnormalities play an important role in the development of autism. Studies have shown age-specific anatomical alterations in brain size and volume in ASD (Courchesne et al., 2011; Redcay and Courchesne, 2005). Both cortical gray matter (GM) and white matter (Abell et al., 1999; Carper et al., 2002), as well as several subcortical structures, such as amygdala (Sparks et al., 2002) and hippocampus (Sparks et al., 2002), are observed to be expanded in autism. However, these gross anatomical differences are most prominent during early postnatal life and childhood and may be less apparent during adolescence and adulthood. A variety of postmortem and meta-analyses also validated that such enlargements are present only in children and adolescents with ASD (Stanfield et al., 2008). Although a great percentage of the literature on autism anatomy has addressed the anatomical abnormalities that are found in the beginning stages, the older and mature autistic neural structural abnormalities that underlie the emergence of autistic behavior in adults have not yet been addressed.
Although some neuroimaging studies recruited older adolescents or adults with autism, the results of these adult studies were less consistent than findings for children. For example, volume increases and decreases have been reported in temporal lobe, limbic system, as well as cerebellum (Abell et al., 1999; Craig et al., 2007), and those regions are hypothesized to be critically involved in ASD. The heterogeneity of subjects, variability of outcome measures used and other fundamental problems of research design have led to discrepancies in describing the etiology of the disorder.
There are many aspects of the medical, environmental and socio-demographic domains that are associated with or even cause neuroanatomical abnormalities in the brain. ASD is a kind of neurodevelopmental disorder, and therefore, related changes in age and IQ are crucial to understanding the neurobiological mechanisms associated with autistic behavior. It appears that the adult autistic populations included in past adult population studies were far from homogeneous with respect to age range and cognitive development (Murphy et al., 2011). Therefore, knowledge of the influence of confounding variables such as age, gender and IQ, as well as clinical characteristics on brain anatomical changes of ASD, is needed. To delineate the characteristic abnormalities associated with ASD in the context of normative longitudinal changes in neuroanatomy, demographic and clinical domains properly require large and complex studies.
Currently, a novel coordinate-based meta-analytic technique called effect-size signed differential mapping (ES-SDM) has enabled the identification of the most spatially consistent brain changes within the literature through the use of the coordinate information reported in each study. ES-SDM can also allow us to investigate the potential effect of several relevant socio-demographic and clinical variables by means of simple linear regression (Radua et al., 2012). This method has been successfully applied to identify the most consistent structural brain changes in autism (Aoki et al., 2013; Cauda et al., 2011, 2014; Redcay and Courchesne, 2005; Stanfield et al., 2008), but to our knowledge, no study has focused on brain changes in adults with autism. Only one meta-analysis study has split categorically into pediatric and adolescents samples and adults samples, but in these 10 adult datasets, 3 were not pure adult sample (Hyde et al., 2010; Kosaka et al., 2010; Rojas et al., 2006); for example, in Rojas’ study, the mean age was 20.78 years, but the age range was 7.8–44 years (Rojas et al., 2006). The different age stage included in this mixed sample which included both adolescents and adults will work as a confounding factor, which makes it difficult to identify the neurobiological mechanisms underlying adults with autism.
Here, we use ES-SDM to identify the most consistent brain structural changes found in voxel-based morphometry (VBM) studies involving individuals with adult ASD and typically developing individuals. Furthermore, by combining these techniques with meta-regression methods, we were able to characterize the impact of key clinical variables on brain structure in adults with ASD.
Materials and methods
Study selection
A systematic search strategy involving two steps was performed to identify relevant studies. First, a literature search of articles published from 1 January 1999 through 14 January 2015 was conducted using the PubMed, ScienceDirect, Medline and Web of Science database. We used the following search terms: ‘autism or Asperger, or adult autisms plus VBM, morphometry, voxel based or voxel wise’. Review articles and reference lists of included studies were checked to identify further studies for inclusion.
Studies were selected for the meta-analysis using the following inclusion criteria: (1) an adult autistic disorder or Asperger’s disorder diagnosed based on the Diagnostic and Statistical Manual of Mental Disorders–Third Edition, Revised (DSM-III-R), Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition (DSM-IV) or International Statistical Classification of Diseases and Related Health Problems–10th Revision (ICD-10) diagnostic criteria; (2) use of the whole-brain VBM method to investigate differences in whole brain; (3) comparisons of individuals with autistic disorder or Asperger’s disorder with a healthy comparison group; (4) age range of patient group and control group is 18–60 years; (5) clearly reported the three-dimensional coordinates of changes in stereotactic space. The exclusion criteria for the final meta-analysis were as follows: (1) literature reviews or any other meta-analysis results; (2) subjects were children or adolescent, or subjects’ ages ranged between adolescent and adulthoods; (3) patients group or control group comprising less than 10 individuals; (4) studies that re-analyzed previously published data; (5) non-English or unavailable full-text record studies. Meta-analysis of Observational Studies in Epidemiology guidelines were followed in the study (Stroup et al., 2000).
Broad searches yielded 951 hits, 831 of which did not meet the inclusion criteria based on inspections of their titles and/or abstracts. These searches initially identified 35 candidate autism-related articles for possible inclusion after careful assessment for methodology consideration (Figure 1). Recent studies have identified age-specific brain morphological changes in autism (Courchesne et al., 2007, 2011), which means that the brain impairments may differ at different ages (e.g. overgrowth of the amygdala and temporal and frontal cortex emerges at young ages but not at older ages) (Courchesne et al., 2011; Stanfield et al., 2008) and that defects present at younger ages may not be present at older ages and vice versa. Because of these age-specific changes in anatomical pathology, 16 studies that enrolled patients who were younger than 18 years were discarded (Boddaert et al., 2004; Brieber et al., 2007; Cheng et al., 2011; Kaufmann et al., 2013; Ke et al., 2008; Kurth et al., 2011; Kwon et al., 2004; McAlonan et al., 2005, 2008; Mengotti et al., 2011; Riva et al., 2011, 2013; Salmond et al., 2005, 2007; Uddin et al., 2011; Waiter et al., 2004). Eight studies also had to be excluded because subjects’ age ranges are between adolescents and adults (Bonilha et al., 2008; Freitag et al., 2008; Greimel et al., 2013; Hyde et al., 2010; Kosaka et al., 2010; Rojas et al., 2006; Spencer et al., 2006; Waiter et al., 2004). One article was identified in one review article (Riedel et al., 2014). Therefore, a total of 12 high-quality datasets were finally included in this meta-analysis (Abell et al., 1999; Craig et al., 2007; Ecker et al., 2012; Itahashi et al., 2015; Lai et al., 2013; McAlonan et al., 2002; Mueller et al., 2013; Riedel et al., 2014; Schmitz et al., 2006, 2008; Toal et al., 2010; Wilson et al., 2009).
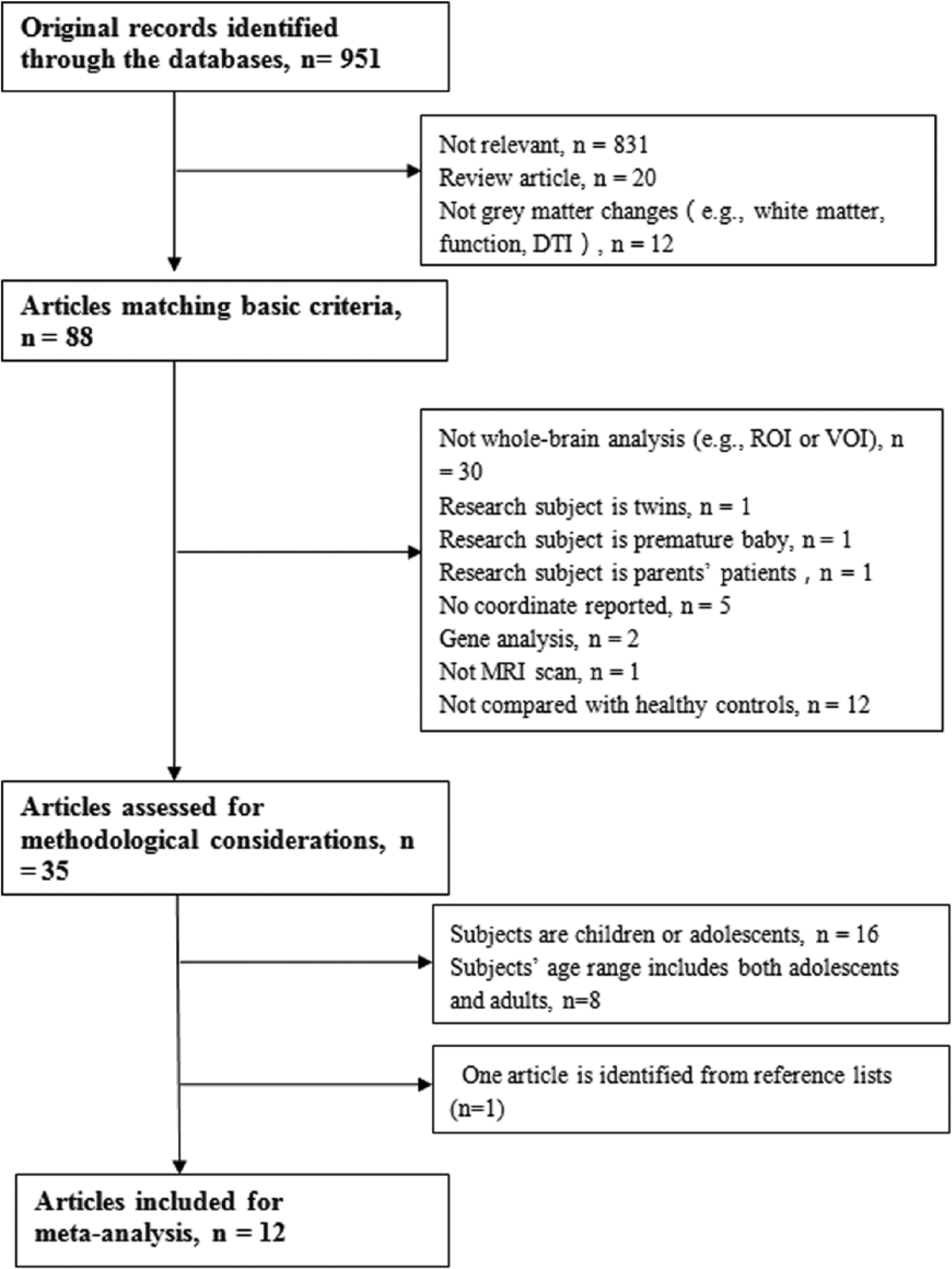
Flow diagram of inclusion and exclusion process of selected articles of VBM studies in patients with ASD.
Voxel-based meta-analysis
Prior to conducting the voxel-based meta-analysis, the reported peak coordinates were used to recreate a statistical map of the differences between patients and controls for each study. For each article included in the meta-analysis, we recorded the following variables: sample size, sex, mean age of the participants, diagnosis, IQ scores and performance in behavioral tests.
The ES-SDM (www.sdmproject.com/software), version 4.13, for meta-analysis was conducted. As an intrinsically whole-brain technique, this method does not restrict analyses to a priori regions of interest, thereby providing a regionally unbiased assessment of neuroanatomical differences on a point-by-point basis across the entire brain. This new meta-analytic technique uses the reported peak coordinates to recreate maps of the effect size of group differences. It has been widely applied in neuropsychiatric disorders including schizophrenia and attention-deficit/hyperactivity disorder (ADHD) (Bora et al., 2012; Hart et al., 2013; McCarthy et al., 2014), described in detail elsewhere (Radua and Mataix-Cols, 2009) and only briefly summarized here. First, we ensured that the same threshold throughout the whole brain was used within each included study, in order to avoid biases toward liberally thresholded brain regions (Radua and Mataix-Cols, 2009). Second, a map of the differences in GM is separately recreated for each study. Third, the mean map is obtained by voxel-based calculation of the mean of the study maps, weighted by the squared root of the sample size of each study so that studies with large sample sizes contribute proportionally more to the final map. The standard ES-SDM thresholds (uncorrected p < 0.005, extent threshold of clusters >10 voxels) which were proposed to optimally balance the sensitivity and specificity and to be an approximate equivalent to corrected p-value < 0.05 in ES-SDM for effect size were used.
Sensitivity analysis and heterogeneity analysis
A systemic whole-brain voxel-based jackknife sensitivity analysis was conducted to test the replicability of results in different studies. The analysis was repeated 20 times, with a different study being excluded each time. If the findings of previous SDM significant results could be replicated in all or most of the included studies, the findings might be very replicable and conclusive.
The statistical heterogeneity of individual clusters was examined using a random effects model with Q statistics (χ2 distribution converted to z values and tested with a permutation approach [p < 0.005, uncorrected for false discovery rate [FDR]; peak height z = 1; cluster extent = 10 voxels]); the same approach was used in our previous study (Chen et al., 2015). To control for any possible methodological differences between the studies, the subgroup analysis was repeated several times and included only those studies that were methodologically homogeneous (see Supplementary Appendix).
Meta-regression of confounding biases
We enrolled all the possible confounding factors that might influence the analytic results, such as the mean age of the patients and controls, the gender ratios of both groups, percentage of medicated patients, scores of clinical rating scales, onset age, family history, duration of illness and comorbidities. Meta-regression steps were performed for these factors to determine whether they were correlated with meta-analytic GM results (see Supplementary Appendix for details of meta-regression method).
Results
As shown in Figure 1, the search strategy identified 951 studies; of these, 12 studies compared whole-brain differences of 382 adult patients with autism and 393 healthy control subjects (Abell et al., 1999; Craig et al., 2007; Ecker et al., 2012; Itahashi et al., 2015; Lai et al., 2013; McAlonan et al., 2002; Mueller et al., 2013; Riedel et al., 2014; Schmitz et al., 2006, 2008; Toal et al., 2010; Wilson et al., 2009). The percentage of male subjects in autism (306 patients, 80.1%) and control (316 control subjects, 80.4%) samples was not statistically different (χ2 = 6.286, p = 0.615). The mean age of the autism (32.40 years) and the control subjects (32.24 years) was not significantly different (t = 0.098, p = 0.923). However, the mean IQ of the healthy control was marginally significantly higher than that of the patient group (t = −2.023, p = 0.054). The clinical and demographic data of the participants from all recruited studies are presented in Table 1.
Demographic characteristics of voxel-based morphometry datasets included in the meta-analysis.
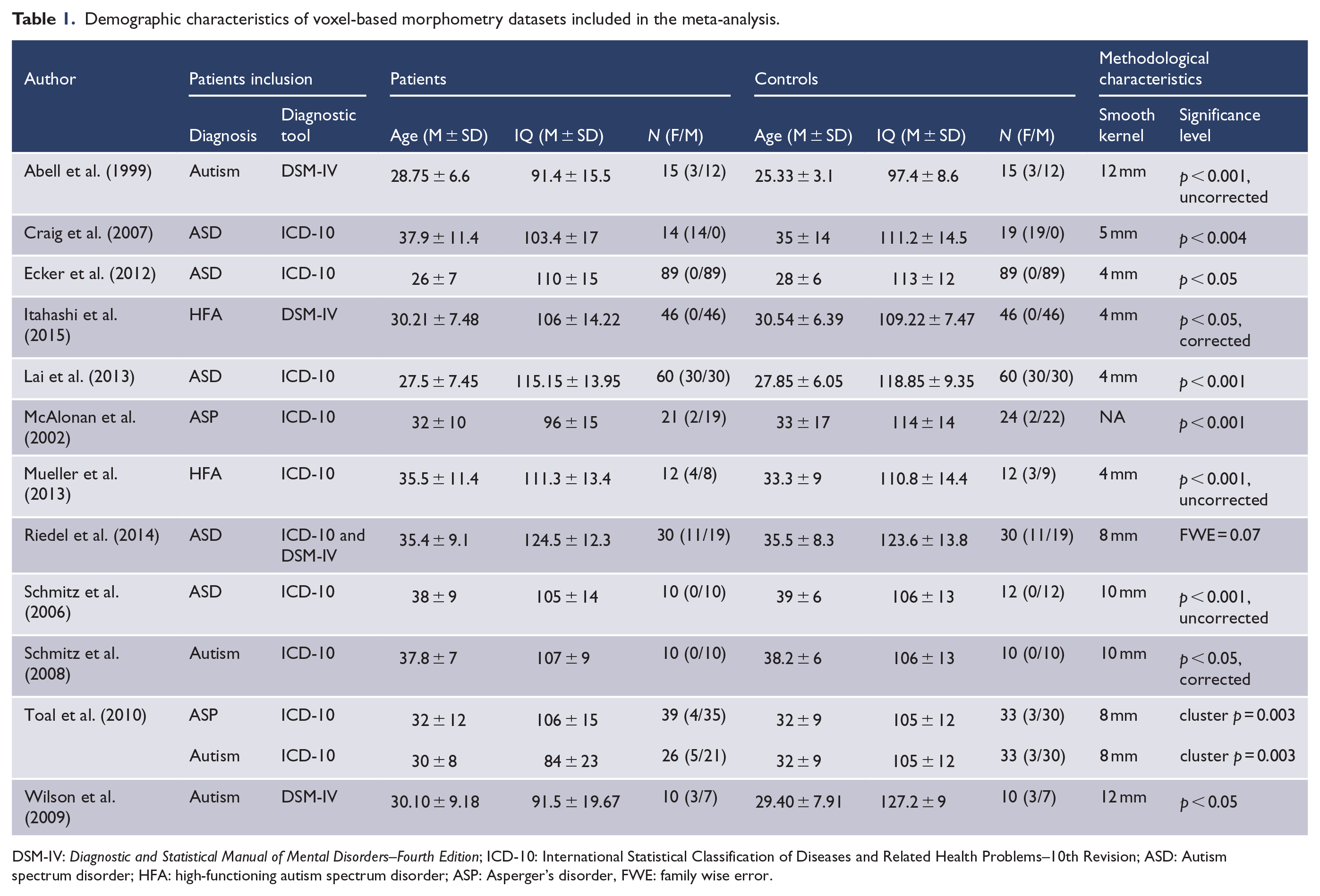
DSM-IV: Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition; ICD-10: International Statistical Classification of Diseases and Related Health Problems–10th Revision; ASD: Autism spectrum disorder; HFA: high-functioning autism spectrum disorder; ASP: Asperger’s disorder, FWE: family wise error.
Regional differences in GM
In the pooled meta-analysis, patients with adult autism had increased GM mainly in left middle temporal gyrus (MTG, extend to superior temporal gyrus [STG] and parahippocampal gyrus), right postcentral gyrus and left parahippocampal gyrus when compared with healthy controls. We also observed decreased GM primarily in the right cerebellum and left anterior cingulate cortex (ACC) (Table 2; Figure 2).
Regional differences in gray matter between adult autism patients and HC subjects identified by the present meta-analyses (voxel-wise p < 0.005).
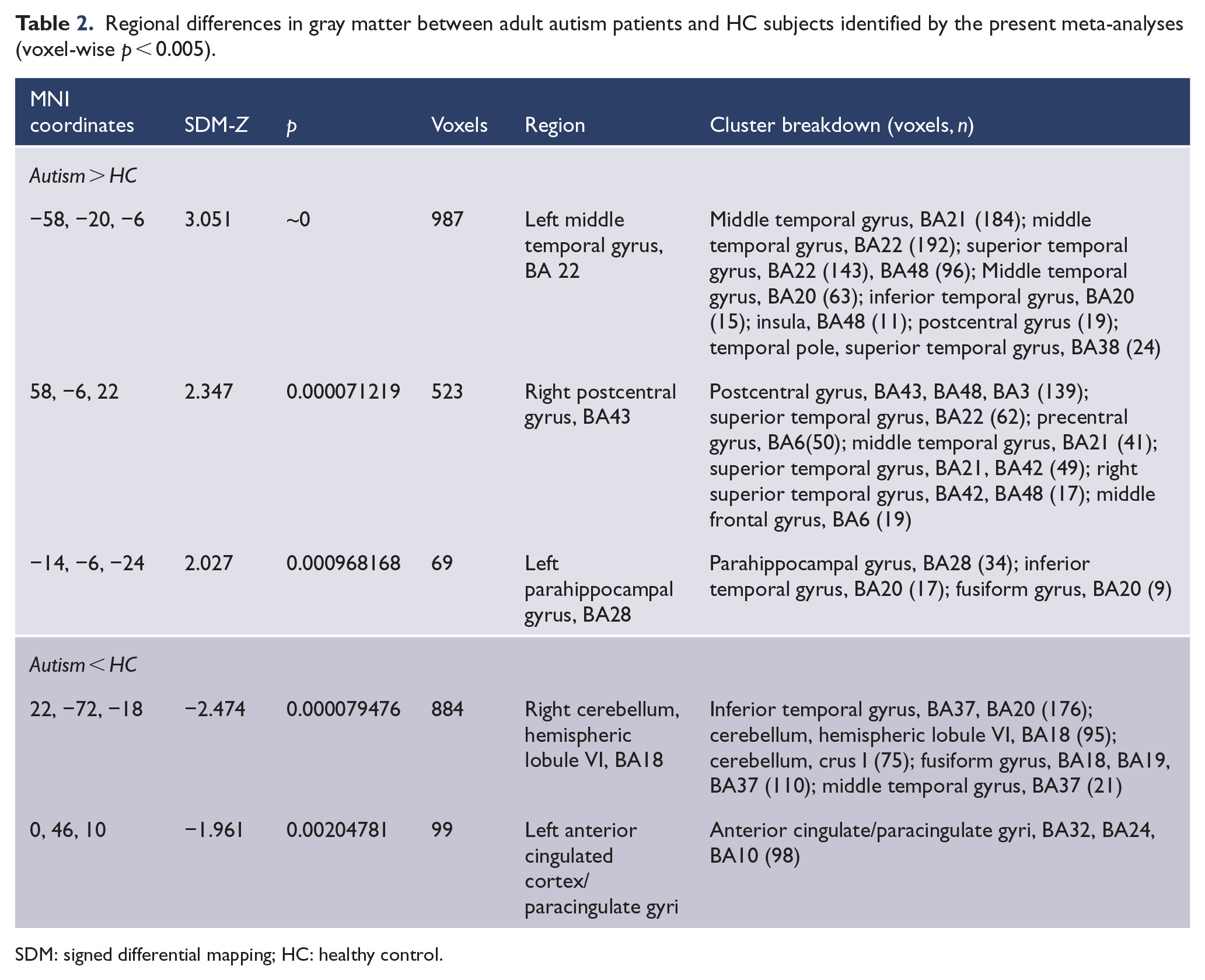
SDM: signed differential mapping; HC: healthy control.
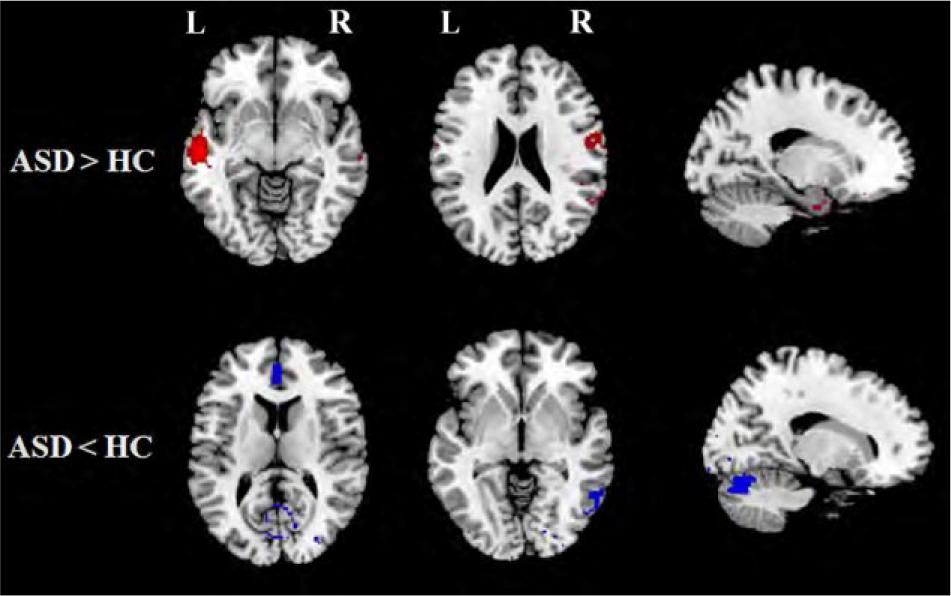
Regions showing increased and reduced gray matter in patients with adult autism compared with healthy controls.
Reliability analysis and heterogeneity analysis
As shown in Table 3, a whole-brain jackknife sensitivity analysis of the pooled meta-analysis showed that the findings of GM increases in patients in left middle temporal gyrus and right postcentral gyrus were preserved throughout all 13 combinations of datasets. The results of increased GM in the left parahippocampus gyrus and decreased GM in right cerebellum remained significant in all but one combination, and those with decreased GM in the left ACC in all but three combinations.
Subgroups and sensitivity analyses.
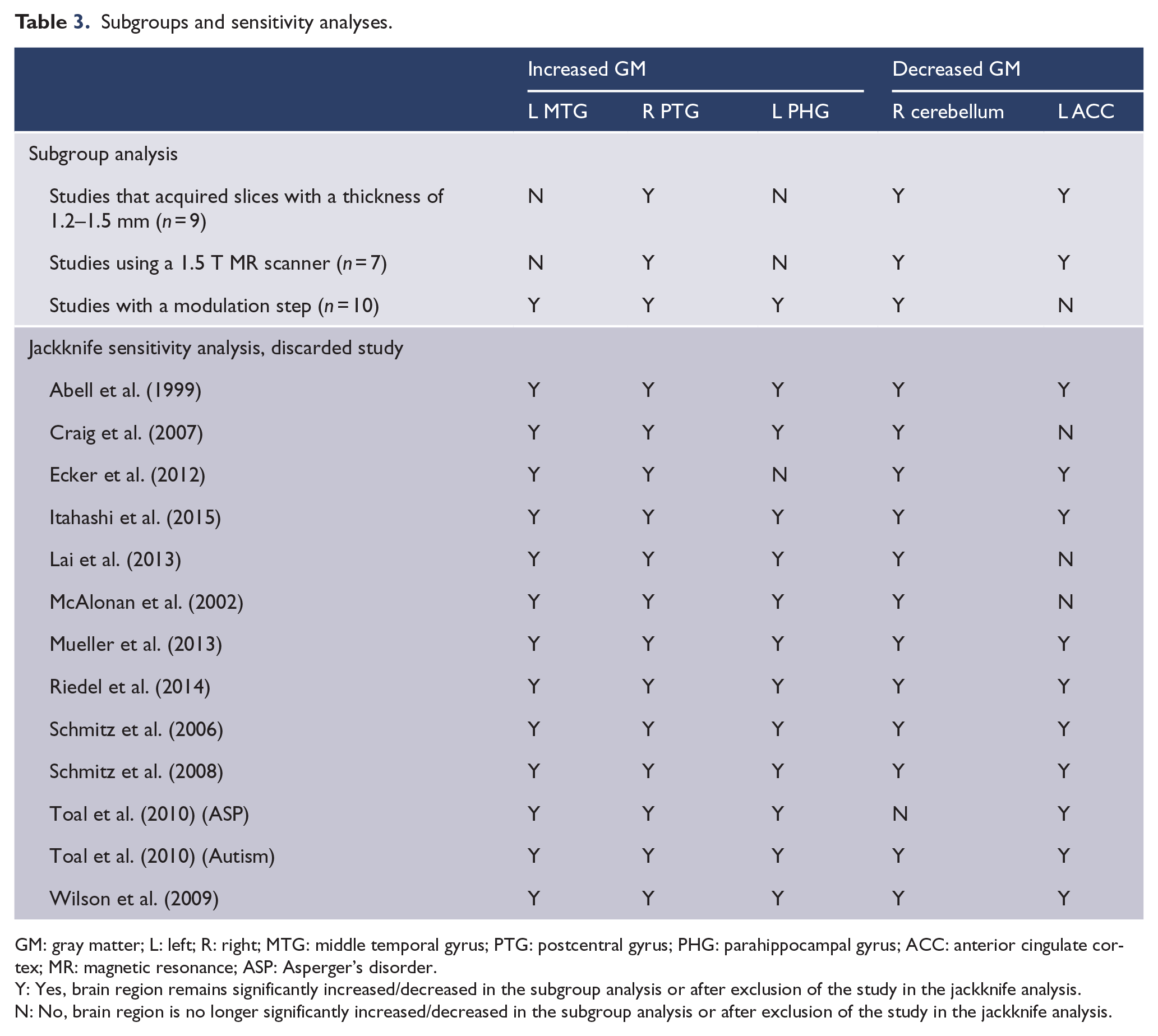
GM: gray matter; L: left; R: right; MTG: middle temporal gyrus; PTG: postcentral gyrus; PHG: parahippocampal gyrus; ACC: anterior cingulate cortex; MR: magnetic resonance; ASP: Asperger’s disorder.
Y: Yes, brain region remains significantly increased/decreased in the subgroup analysis or after exclusion of the study in the jackknife analysis.
N: No, brain region is no longer significantly increased/decreased in the subgroup analysis or after exclusion of the study in the jackknife analysis.
Analysis of heterogeneity revealed that a number of regions (right amygdala, right postcentral gyrus, left superior frontal gyrus and left precentral gyrus) with altered GM exhibited significant statistical heterogeneity among the studies (p < 0.005).
Subgroup analysis
The results described above remained largely unchanged when the analyses were limited to methodologically homogeneous groups of studies. Only one additional significant cluster in the right amygdala (Montreal Neurological Institute [MNI] coordinates x = 32, y = 0, z = −26, SDM Z = −1.541, p = 0.0018) emerged in the subgroup analysis of studies that acquired slices with a thickness of 1.2–1.5 mm and of studies that used a 1.5-T magnetic resonance (MR) scanner.
Meta-regression analysis
We explored the mean age, mean total IQ score and the percentage of male patients with autism, which were available for all 382 patients in the 13 datasets, using meta-regression. The mean age of the patients group was negatively associated with GM in the left MTG (r = −0.661, permutation-derived p < 0.0001; Figure 3). The mean total IQ score of the patients was positively correlated with GM in the left parahippocampal gyrus (r = 0.607, permutation-derived p < 0.0001; Figure 4). The percentage of male patients with autism was significantly associated with GM changes in the right ACC (r = 0.890, permutation-derived p < 0.0001) and right cerebellum (r = 0.521, permutation-derived p < 0.0001) (see Figure 5). The following variables could not be studied because data were available for fewer than 10 studies: mean age at onset, illness duration and percentage of medicated participants. We were also unable to assess whether autism symptom severity was associated with the reported structural changes because the included studies used a wide range of different measures.
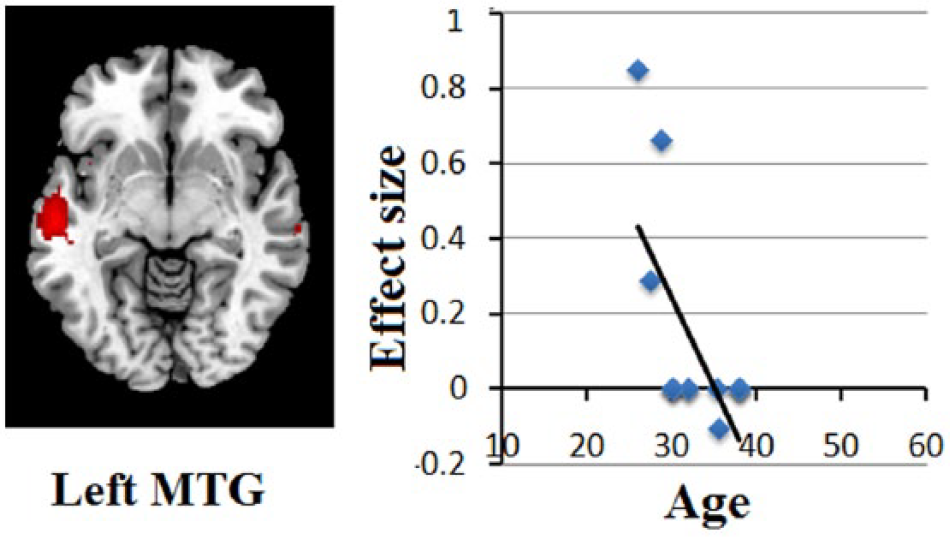
Results of the meta-regression analysis showing a significant inverse association between age and gray matter volumes in the left middle temporal gyrus.
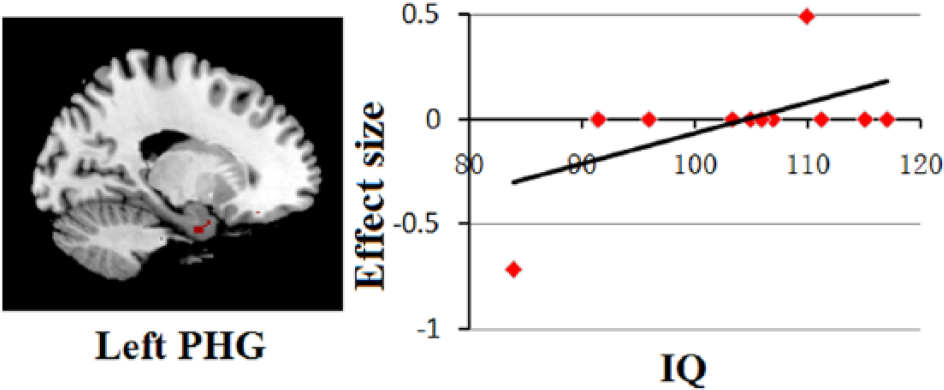
Results of the meta-regression analysis showing a significant association between total IQ score and gray matter volumes in the left parahippocampal gyrus.
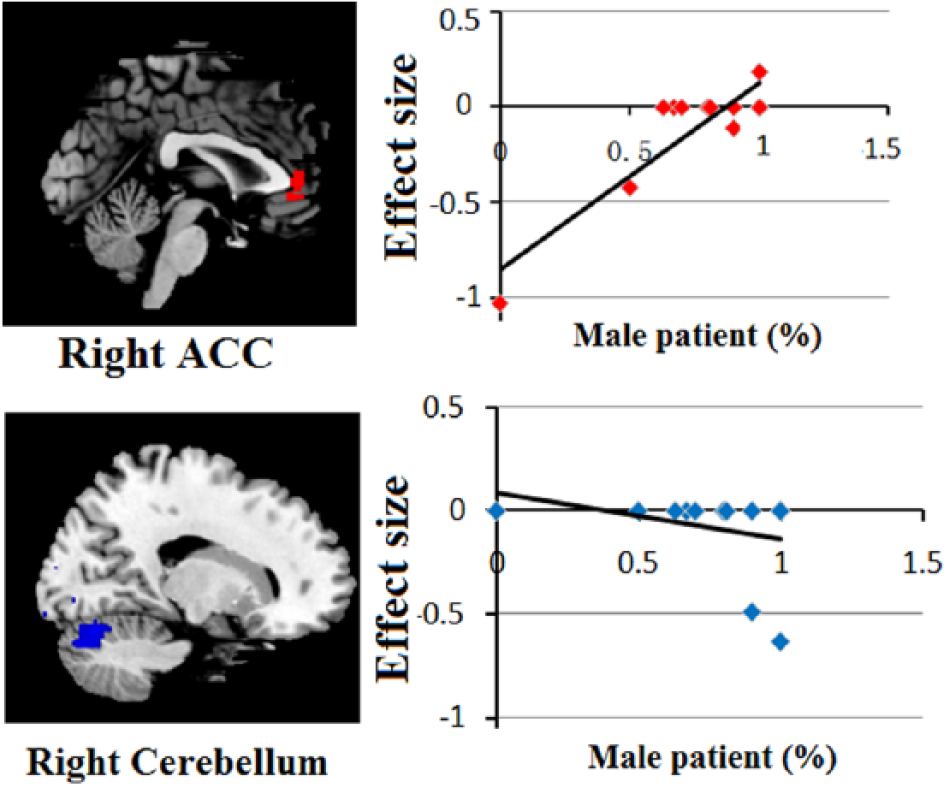
Results of the meta-regression analysis showing a significant association between the percentages of male patients with autism and gray matter volumes in the right anterior cingulate and right cerebellum.
Discussion
The primary objective of this study was to identify distinct GM abnormalities in adult populations with autism. We used quantitative ES-SDM meta-analytic methods to synthesize findings of 12 structural neuroimaging studies of adults with autism. The study is timely given that a sufficient number of high-quality studies have only recently become available. The most robust GM increases were identified in MTG, STG, parahippocampal gyrus and postcentral gyrus, while the most evident reductions were located in the cerebellum and ACC. Furthermore, the variations in GM volume (GMV) were significantly correlated with the mean age and mean total IQ score of patients, as well as the percentage of male patients with autism; these correlations may reflect the effects of demographical variation on the brain morphological changes of ASD.
Regional increases in GM regions
It is well known that lesions in the STG, MTG and parahippocampal gyrus are associated with marked impairment in social perception (Zilbovicius et al., 2006), emotional processing and theory of mind (Redcay, 2008). Many neuroimaging studies have revealed brain abnormalities that are consistent with the primary pathological findings that have been identified in the temporal region in autism. Magnetic resonance imaging (MRI) volumetric studies have revealed significant enlargement of GM in the frontal and temporal lobes in autism when compared to controls (Abell et al., 1999; Ecker et al., 2012; Waiter et al., 2004). Additionally, increases in GM in this region were correlated with increased social symptom severity in autism (Ecker et al., 2012). However, adult autistic patients are characterized by hypoactivation within the temporal lobe during sustained attention (Murphy et al., 2014) and decreased functional connectivity when processing emotional body language (Libero et al., 2014). These findings are in line with resting functional magnetic resonance imaging (fMRI) studies that have shown reduced spontaneous brain activity and connectivity in temporal regions (Itahashi et al., 2014, 2015). Considering that social perception and empty function have been associated with this region and that improved social communication skill has been reported in adult autistic patients (Howlin et al., 2004; Tobin et al., 2014), we tentatively assume that increased temporal GMV or density in adult autism may be a compensatory mechanism to alleviate communication-related symptoms in ASD. In contrast to adults with autism, autistic children show significant reduction GM concentration (Boddaert et al., 2004) and functional hypoperfusion (Zilbovicius et al., 2000) in the STG, which has been implicated in the social communication skills that are characteristically impaired in children and adolescents with autism. This finding may contribute to understanding of the growth trajectory in social perception impairment that is characteristic of autism.
We also observed regional increases in GMV in the postcentral gyrus. As part of the somatosensory cortex, the postcentral gyrus receives its afferents mainly from the ventral posterior nucleus and from the ventral posteromedial nucleus of the thalamus (Young et al., 2003). A previous fMRI study revealed that the pre- and post-central sulci were activated by observation of human movement only in autistic individuals but not in controls, demonstrating atypical impairment in autism (Martineau et al., 2010). This area has also been implicated in cognitive control, particularly in executive function (Koechlin et al., 2003), which is known to be specifically impaired in ASD. Our finding of increased GMV in the fronto-temporal cortices and limbic system lends support to theories implicating these regions in the pathophysiology of autism.
Regional decreases in GM regions
One of most consistently reported regions of GM deficits in ASD is the ACC, which serves as an integration hub for many different types of executive function, affect and socio-emotional behavior. There is compelling evidence that altered metabolism (Levitt et al., 2003) and reduced volume of the ACC were found in autism (Haznedar et al., 1997). Likewise, microscopic study revealed that increased cell packing density and decreased cell size in several sub-regions within the ACC were observed in many autistic individuals (Simms et al., 2009). There are also evidence of abnormal ACC activation and functional connectivity in autism (Agam et al., 2010; Assaf et al., 2010; Thakkar et al., 2008). Importantly, these deficiencies in function have been associated with inflexible and repetitive symptoms in ASD (Thakkar et al., 2008). Thus, impairment in the development of this system may constitute a neural substrate for the socio-cognitive deficits present in autism.
Individuals with ASD had a significant decrease in GMV located in cerebellum (mainly in posterior lobules VI, crus I) in this study, which is consistent with some previous meta-analyses of VBM in ASD (Cauda et al., 2011; DeRamus and Kana, 2014). GM abnormalities within the cerebellum are well documented in the ASD literature (Abell et al., 1999; Riva et al., 2013; Rojas et al., 2006). Traditional evidence indicates cerebellar dysfunction is associated with motor impairments in autism (Mostofsky et al., 2009). Recent studies indicate that the cerebellum plays an important role in non-motor functions as well. Townsend et al. (1999) reported smaller cerebellar vermal lobules VI–VII were associated with greater attention-orienting deficits. The presence of motor and cognitive deficits in autism may be related to the cerebellar dysfunction in autism.
In contrast, structural neuroimaging studies in pediatric and adolescent patients with ASD have reported more widely spread volumetric impairments, which include not only the ACC and cerebellum (McAlonan et al., 2008; Riva et al., 2011, 2013) but also the amygdala–hippocampal complex and medial parietal cortex (Brieber et al., 2007; Ke et al., 2008; McAlonan et al., 2005; Riva et al., 2013). It is clear that the amygdala and its adjacent structures are implicated in social cognition and emotional processing (Adolphs, 2010; Emery et al., 2001) and that the medial parietal cortex is involved in self-awareness and self-representation (Lombardo and Baron-Cohen, 2010); thus, the much more widely distributed brain impairment may reflect the more severe autistic symptoms in children and adolescents. Further longitudinal and cross-sectional studies with a larger number of different age cohorts of patients and comparison subjects are needed to observe the effect of age on autism more directly.
Effects of demographical variation on the structural changes
The mean age of the patient group was negatively associated with GM in the left MTG, which indicated that the older adult autistic individuals tended to have smaller MTG. Prior studies have observed thinner cortices in the STG and adjacent areas with increasing age in participants with ASD relative to typically developing controls (Doyle-Thomas et al., 2013; Scheel et al., 2011; Wallace et al., 2010). Maturational brain changes in this region have been implicated in multimodal sensory integration and cognitive functions are believed to be associated with improvement in these functions (Chao et al., 1999; Onitsuka et al., 2004). Thus, deviation from the typical developmental trajectory of GM in temporal brain regions may underlie age-related impairments in social and communication functions in ASD.
Past researches suggest that autistic symptoms varied during cognitive development (Mayes and Calhoun, 2004; Miller and Ozonoff, 2000), that is, as IQ increases, autistic symptoms decrease. Consistent with this, we found disturbed GM in the parahippocampal gyrus was positively related to the mean total IQ of this study population. The increased GMV in the parahippocampal gyrus with higher IQ level in autism patients may indicate higher mind theory ability and other related function implicated in this area. Furthermore, some researches hold that a discrepancy between verbal and nonverbal intellectual abilities may be a particularly informative phenotype of ASD (Black et al., 2009). However, few of the included articles reported the verbal and nonverbal IQ subscale scores, which limited our ability to perform further regression analysis. Therefore, further studies taking IQ and subscale IQ variables into account are needed for a more complete understanding of the brain abnormalities in patients with autism.
The observed sex-related increase in the ACC and cerebellum was also well documented by Lai (Lai et al., 2013) and Takahashi (Takahashi et al., 2011) among adult males on the autism spectrum, which suggested the male autism tends to increase GMV. The sex difference in the ACC may contribute to superior rational cognitive functions in males. As it is consistent with what is known as the ‘extreme male brain’ theory, it posits autism represents an extreme of the male pattern with enhanced systemizing ability (Baron-Cohen, 2002; Baron-Cohen et al., 2005). Sex differences are also seen in epidemiological and clinical aspects of autism. Male individuals show a greater incidence of autism (4.3 for a mean male/female sex ratio) than females (Fombonne, 2003). Males are found to show more externalizing behavioral problems than females, such as aggressive behavior, hyperactivity, reduced prosocial behavior and increased repetitive/restricted behaviors and interests (Werling and Geschwind, 2013). These evidences could explain the biological basis of the male bias in ASD.
Although MRI studies have shown some evidence for brain enlargement in early ages of children that may be related to degeneration and decline during adolescence and young adulthood, it is still unclear how anatomical pathology alters with age after these early years. Our study suggests that in adult autism, abnormal neural and anatomy processes may be different from children: brain enlargement in adult autism seems to be most prominent in the frontal, temporal and limbic area, but brain atrophy is mainly located in the cerebellum and anterior cingulate area. Consistent with this, Raznahan et al. (2010) reported that the ratio of cerebral lobar volume to overall total brain volume follows an abnormal trajectory across adolescence and adulthood, suggesting that in autism, abnormal neural processes continue to occur during adulthood. In older samples, clinical studies have observed part of reduced symptom severity but keep some core autism symptom with increasing age in participants with ASD relative to typically developing controls (Howlin et al., 2004; Tobin et al., 2014). Thus, evidence of current meta-analysis in the autistic person may reflect complex change with age and secondary remodeling processes (possibly both brain degeneration and enlargement) after the neuroinflammatory processes (Morgan et al., 2010). These evidences suggest that cortical dysmaturation in ASD is not restricted to childhood but extends across the lifespan, which led to the theory of age-specific anatomical abnormalities in autism.
The finding of associations between these brain structural changes and several demographical variations provides further evidence for different neurobiological processes in autistic samples. Both the group analysis and regression technique revealed that people with ASD had significant age-related differences in brain anatomy compared with the typically developing individuals, which reflect the different progresses of disease phases of autism. Although relatively little is known of the factors that shape age-related changes in autism during aging, individual differences in maturation of cortical thickness have been related to IQ (Shaw et al., 2006). This neuroanatomical correlation with IQ in adult autism was also found in the present meta-regression analysis. In other words, differences could be accounted for by preexisting differences in IQ. Furthermore, gender differences associated with GMV changes may help explain the increased vulnerability of males to ASD. These findings will help us to understand the neurobiology and developmental trajectory in adult autism. However, further longitudinal studies are needed to fully elucidate the influences of demographic profiles on brain morphometry and the processes underpinning the age-related brain differences in adults with autism.
Limitations
Despite our success in demonstrating consistencies in findings across studies, specific methodological limitations should be considered with respect to the current structural neuroimaging literature examining adult subjects with autism. First, peak-based meta-analyses are based on summarized (i.e. coordinates from published studies) rather than full statistical image from the original studies, and this may result in inaccurate results (Salimi-Khorshidi et al., 2009). Also, different studies used different statistical thresholds which would bias the results. Future studies of larger and more diverse samples are needed to determine whether this approach is justifiable. Third, the heterogeneity analysis showed that a number of regions with altered GM exhibited significant statistical heterogeneity among studies. To examine the moderator variables that contributed to the heterogeneity of the findings, we conducted meta-regression analyses and subgroup analyses (Radua and Mataix-Cols, 2009). Fourth, the included studies varied in terms of the data acquisition, analysis techniques, patient characteristics and clinical variables. For example, the patients with autism or Asperger’s syndrome are both included in our study. As different subtypes of the classical autistic spectrum, autism and Asperger’s syndrome may have different etiologies, which could influence the results of our findings. It would also be valuable to analyze the effect of subtypes or comorbidities; however, there were no sufficient studies to carry out these subgroup analyses. The differences between GM volume and GM concentration in the included studies might also influence our results. Further longitudinal follow-up studies are necessary to understand the natural progression of autism, as well as its different anatomy and cognitive markers that characterize the differential stages of autism.
Conclusion
In summary, we used the ES-SDM method to identify regions that reliably exhibited anatomical deficits in adults with autism. Our findings indicate that adult autistic patients have significant and robust GM changes in the fronto-temporal cortices, limbic system and cerebellum, which is different from childhood autism. These brain structural changes, together with the results of correlation with demographic characteristics, may clarify the neurobiology and developmental trajectory in adult population with ASD.
Footnotes
Acknowledgements
Xun Yang and Lihua Qiu contributed equally to this work.
Declaration of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This study was supported by the National Natural Science Foundation (Grant Nos. 81371536 and 91132720, and 81271532), the Fundamental Research Funds for the Central Universities, Southwest University for Nationalities (2014NZYQN39), and the China Postdoctoral Science Foundation (Grant No. 2015M572479).
