Abstract
Objective:
Information on the rates and predictors of polypharmacy of central nervous system medication in older people with intellectual disability is limited, despite the increased life expectancy of this group. This study examined central nervous system medication use in an older sample of people with intellectual disability.
Methods:
Data regarding demographics, psychiatric diagnoses and current medications were collected as part of a larger survey completed by carers of people with intellectual disability over the age of 40 years. Recruitment occurred predominantly via disability services across different urban and rural locations in New South Wales and Victoria. Medications were coded according to the Monthly Index of Medical Specialties central nervous system medication categories, including sedatives/hypnotics, anti-anxiety agents, antipsychotics, antidepressants, central nervous system stimulants, movement disorder medications and anticonvulsants. The Developmental Behaviour Checklist for Adults was used to assess behaviour.
Results:
Data were available for 114 people with intellectual disability. In all, 62.3% of the sample was prescribed a central nervous system medication, with 47.4% taking more than one. Of those who were medicated, 46.5% had a neurological diagnosis (a seizure disorder or Parkinson’s disease) and 45.1% had a psychiatric diagnosis (an affective or psychotic disorder). Linear regression revealed that polypharmacy was predicted by the presence of neurological and psychiatric diagnosis, higher Developmental Behaviour Checklist for Adults scores and male gender.
Conclusion:
This study is the first to focus on central nervous system medication in an older sample with intellectual disability. The findings are in line with the wider literature in younger people, showing a high degree of prescription and polypharmacy. Within the sample, there seems to be adequate rationale for central nervous system medication prescription. Although these data do not indicate non-adherence to guidelines for prescribing in intellectual disability, the high rate of polypharmacy and its relationship to Developmental Behaviour Checklist for Adults scores reiterate the importance of continued medication review in older people with intellectual disability.
Introduction
The prescription rates of central nervous system (CNS) medications to people with intellectual disability (ID) far exceed those within the general population. During 2011, within the Australian general population around 10% of people aged 45–54 years had accessed a CNS medication, increasing to around 25% in those aged 75 years and over – the largest rate across all age groups (ABS, 2011). In comparison, rates of CNS medication use are as high as 60% in adults with ID (Sachdev, 1991; Tsiouris et al., 2013), with limited representation within these samples of those aged 45 years and over. International studies also consistently report high rates of CNS medication polypharmacy (defined as more than one CNS medications) in ID, particularly of antipsychotics and anticonvulsants (Doan et al., 2013, 2014; Edelsohn et al., 2014; Hill et al., 1985; Sachdev, 1991; Thomas et al., 2010; Tsiouris et al., 2013). These findings raise concerns given the common adverse effects of these medications, which, especially in people with ID, can further exacerbate physical, mental, neurological and behavioural issues and markedly affect the quality of life (Allen, 2008; Matson et al., 2003; Thomas et al., 2010). Indeed, polypharmacy of CNS medication (as defined by the Monthly Index of Medical Specialties [MIMS]) in people with ID has been associated with an increased susceptibility to these side effects (Habetaler et al., 2014; Mahan et al., 2010).
Compared with the general population, people with ID have been found to have higher rates of psychiatric diagnosis, especially psychotic and affective disorders (Cooper et al., 2007) and neurological diagnoses, especially seizure disorders (Bowley and Kerr, 2000; McGrother et al., 2006; Santosh and Baird, 1999). The high prevalence of these diagnoses indeed warrants CNS medication use; however, studies have shown that CNS medications in people with ID are commonly associated with treatment of challenging behaviours (McGillivray and McCabe, 2004; Matson and Neal, 2009). Challenging behaviours are defined as Culturally abnormal behaviours of such an intensity, frequency or duration that the physical safety of the person or others is likely to be placed in serious jeopardy or behaviour which is likely to seriously limit use of or result in the person being denied access to ordinary community facilities. (Emerson, 1995)
In a large Dutch study (n = 2373), about one-third of adults with ID were on an antipsychotic medication: for almost 60% of participants it was prescribed for challenging behaviours, with only 22% having a diagnosed psychiatric disorder and the remaining 18% with no reason specified for the CNS medication use (De Kuijper et al., 2010). Fleming et al. (1996) found that 69% of their sample (n = 118) was receiving CNS medication for challenging behaviour, whereas only 8% had a psychiatric diagnosis (Fleming et al., 1996). Similarly, in a more recent Australian study of 117 adults with ID, antipsychotics were the most commonly prescribed medication class, and the presence of challenging behaviours and a psychiatric illness was significant predictors of their use (Doan et al., 2013).
Prescription of CNS medication for challenging behaviour in the absence of a psychiatric diagnosis lacks consistent evidence to support its use (Deb and Unwin, 2007; Thomas et al., 2010; Tsiouris et al., 2013; Tyrer et al., 2009). This practice has occurred at least partly in response to the frequent occurrence of challenging behaviour in the ID population (Cooper et al., 2007), often presenting as non-specific symptoms requiring comprehensive multidisciplinary assessment and targeted intervention (WPA (SPID), 2010). Furthermore, impairments in communication and conceptual thought in ID present difficulties for psychiatric assessment and diagnosis and can lead to missed diagnosis and misdiagnosis (Torr, 2013). A recent Australian commissioned report recommended that medications should be kept to a minimum, with a review for adverse effects and efficacy conducted after 3–4 months and be removed from the treatment plan as safely and as soon as possible (Thomas et al., 2010).
There is limited research into prescription of CNS medications for older people with ID, despite the increased life expectancy in this group (Patja et al., 2000; Torr and Davis, 2007) and a dearth of evidence available to inform policy and practice. A study that has preliminarily addressed this issue indicated that CNS medication appears particularly prevalent in the oldest age bracket, with 51% of participants aged 47 years and over on a psychotropic medication (Doan et al., 2013). Pary (1993) noted that rates of CNS medications increase with age from the 20–39 year age bracket to those over 55 years. This evidence is supported by reports indicating that once people with ID are placed on CNS medication, they are often continued with that prescription indefinitely without review (Thomas et al., 2010).
The potential for polypharmacy in older adults is perhaps exacerbated by increased comorbidities. Compared with the general population, people with ID are at increased risk of a range of age-related health disorders, including frailty (Evenhuis et al., 2012), seizure disorders (McCarron et al., 2014), multimorbidity (Hermans and Evenhuis, 2014) and dementia (Cooper, 1997; for a review, see Evans et al., 2013). Given their multiple risk factors for medication side effects, the issue of medication use patterns in elders with ID warrants attention.
The aim of this study was to examine CNS medication use in an Australian sample of older people with ID, with a particular focus on predictors of polypharmacy. We hypothesised that there would be a high rate of CNS medication use and polypharmacy among our older sample. We also hypothesised that challenging behaviours and psychiatric or neurological diagnoses would be significant predictors of CNS medication polypharmacy.
Methods
Participants
The study was carried out in accordance with the Declaration of Helsinki and was approved by the Human Research Ethics Committees of The University of New South Wales, Monash University and La Trobe University. When appropriate, written consent for the study was obtained from the person with ID. Otherwise, written consent by proxy was obtained from the person’s legal guardian or closest relative, with verbal consent obtained from the person with ID wherever possible.
In Wave 1 of the Successful Ageing in Intellectual Disability (SAge-ID) Study, 118 participants with ID were recruited from areas within metro Sydney (Randwick, Botany Bay, Rockdale, Waverley, Woollahra, City of Sydney, Leichhardt, Marrickville, Ashfield, Burwood, Strathfield, Canterbury, Hurstville, Kogarah, Canada Bay, Sutherland Shire, Campbelltown, Camden, Liverpool, Fairfield and Bankstown), Illawarra and Shoalhaven areas in New South Wales (NSW) and the Loddon Mallee region of Victoria, Australia. The expected size of the people with ID over the age of 40 in our NSW catchment was expected to be 3350, based on the Department of Social Services (formally Department of Families, Housing, Community Services and Indigenous Affairs) estimate of the number of people receiving a disability pension with a primary medical condition of ID in these areas (Department of Families, Housing, Community Services and Indigenous Affairs, Disability support pensioners with primary medical condition of intellectual/learning by gender and selected LGAs – September Quarter 2012, 23 April 2013, personal communication). Multiple recruitment strategies were employed: (1) mail out to eligible participants via the databases of government disability service providers (Ageing, Disability and Home Care in NSW and Department of Human Services in Victoria); (2) direct approach to community group homes; (3) direct approach to hostels and aged care facilities; (4) mail-out and newsletter promotion through local carer support organisations, respite services and assisted employment services and (5) mail out to general practitioners (GPs) and psychiatrists.
After the return of expression of interest forms, potential participants and carers were contacted. Following explanation of the study, assessment of inclusion/exclusion criteria and verbal consent, people with ID and their primary carers were posted participant information and consent forms along with a baseline questionnaire to complete.
Participants were included in the study if they had ID ranging from mild to profound, were aged above 40 years and had an informant who knew them well and agreed to complete a questionnaire. Exclusion criteria included terminal illness and current residence in a correctional facility. Of the recruited participants, 114 with available medication data comprised the sample for this analysis. The other four persons were excluded because medication data were missing.
Measures
For this analysis, we used baseline questionnaire data from the SAge-ID study. An informant (a family member, paid carer or friend) completed a questionnaire about the person with ID, including detailed structured family history, adaptive behaviour, challenging behaviour, medical and psychiatric history, medication use, lifetime and current levels of physical activity, leisure/mental activity, social activity and occupational activity. The questionnaire took approximately 2 hours to complete.
Specifically, the questionnaire included items regarding the person with ID’s age, gender, living status, known causes of ID (if any) and lifetime diagnosis of an autism spectrum disorder. The Scales of Independent Behaviour–Revised (SIB; Bruininks et al., 1996) were used to assess adaptive function. Questions addressed physical characteristics, such as the presence of high cholesterol, high blood pressure, diabetes and any sensory or physical disability in the person with ID, and the person’s height and weight. Respondents were also asked to list all current medications taken by the person with ID, including over-the-counter medications, and to provide details of the dose, frequency and length of medication use.
Respondents were asked to provide details about any psychiatric or neurological diagnoses and the Developmental Behaviour Checklist for Adults (DBC-A). Diagnosis was classified as disorders for which there was a primary or secondary indication for any of the included CNS medications: schizophrenia or psychotic disorder, depression, anxiety and bipolar disorder as a psychiatric diagnoses, and epilepsy or seizure disorder and Parkinson’s disease as neurological diagnoses. The DBC-A is a 107-item carer-report checklist designed to assess emotional and behavioural disturbance in people with ID aged over 18 years, with total scores above 51 indicative of clinically significant levels of psychopathology (Mohr et al., 2011). Additionally, six analytically derived subscales can be scored: disruptive behaviours, communication and anxiety disturbance, self-absorption, antisocial, depression and social relating (Einfeld and Tonge, 2002; Mohr et al., 2005).
Data analysis
A researcher trained in pharmacy (K.M.C.) reviewed all data to identify CNS medications and grouped them into categories according to MIMS Annual (2009). According to this system, medication Category 3 represents the agents for the CNS, which are further subcategorised as sedatives/hypnotics (3a), anti-anxiety agents (3b), antipsychotic agents (3c), antidepressants (3d), CNS stimulants (3e), movement disorder medications (3f), anticonvulsants (3g) and anti-emetics and anti-nauseants (3h). Anti-emetics and anti-nauseants were excluded from analysis as it was not relevant to our study question. Once the medications were coded, a psychiatrist (J.N.T.) examined all CNS medications and reported diagnoses to determine whether a condition was present for which the CNS medication could be considered either a first- or second-line treatment according to standard clinical practice. For this study, CNS medication polypharmacy was defined as current use of more than one CNS medication and multi-class polypharmacy was defined as current use of more than one class of CNS medication. Medications used pro re nata (PRN) were included.
Data were analysed using SPSS for Windows 22.0 (SPSS Inc., Chicago, IL, USA). Descriptive statistics were obtained for the sample for demographic characteristics and use of medication. To describe adaptive behaviour, standard scores for the SIB-R broad independence subscale were grouped into the following categories: <25, 25–39, 40–54, 55–69, 70–84 and over 85. For inferential statistics, the weighted score (W-score) of the SIB-R was used (Bruininks et al., 1996). Expectation minimisation (EM) imputation was used for DBC-A scores in cases where fewer than 10% of data were missing.
Group differences in demographics, body mass index (BMI), weighted SIB-R broad independence score, comorbid psychiatric diagnosis and genetic cause of ID between those who were in the medicated group (those taking a CNS medication) and those who were in the non-medicated group (those who were not taking a CNS medication) were assessed using t-tests (for continuous variables) or χ2 tests (for categorical variables). Homogeneity of variance was determined using the Levene test.
Predictors of CNS medication prescription/polypharmacy use were analysed via linear regressions for two dependent variables: (1) number of CNS medications and (2) number CNS medication classes. The predictors for both analyses included any psychiatric diagnosis (no = 0, yes = 1), any neurological diagnosis (no = 0, yes = 1), weighted SIB-R broad independence score (continuous), DBC-A total score (continuous), age, gender (male = 0, female = 1), residential living status (not living in a residential facility = 0, living in a residential facility = 1) and autism (no autism = 0, autism = 1).
To attempt to control for the high proportion of people in the sample who were using anticonvulsants for a seizure disorder, we then re-ran these regressions with adjusted CNS medication values, whereby if an anticonvulsant was used by a person who had a seizure disorder, this medication was not included in the total number of CNS medications or total number of CNS medication classes. We also re-ran the correlations completely without CNS medications most commonly indicated for neurological diagnosis (i.e. anticonvulsants and movement disorder medications). Post hoc analysis was conducted to assess the interaction of gender and DBC-A total score on number of prescribed CNS medication classes via addition of the interaction coefficient to the regression model and assessment of its significance.
Multicollinearity of predictors was assessed by the variance inflation factor (VIF). Cook’s distance was used to assess the impact of influential cases and standardised residuals were inspected to identify outliers. For robust analysis, a simple bootstrapping method based on 1000 samples was used to obtain bias-corrected and accelerated (BCa) 95% confidence intervals and alpha for t-tests and linear regression.
Results
Sample characteristics
Data from the 114 participants with ID (54.3% male, average age = 51.0 (7.5) years, range = 40–76 years) were included in the analysis. In all, 52 (45.6%) participants were living in a residential facility, 38 (33.3%) with their family, 17 (14.9%) were living independently and 6 (5.3%) in an aged care facility. The regional distribution of participants was as follows: 54 (47.4%) from metropolitan Sydney, 38 (33.3%) from regional areas of NSW and 22 (19.3%) from a selected regional/rural area of Victoria.
In terms of aetiology of ID, 18 (15.8%) reported a known genetic cause for their ID (12 with Down syndrome and 1 each with Fragile X, Angelman, Bloch–Sulzberger, Turner, Lennox–Gastaut and Noonan syndromes). Six (5.3%) reported a genetic cause for their ID, but the precise genetic diagnosis was unknown. A total of 15 participants (13.2%) had been diagnosed with an autism spectrum disorder and 42 (36.8%) with a seizure disorder.
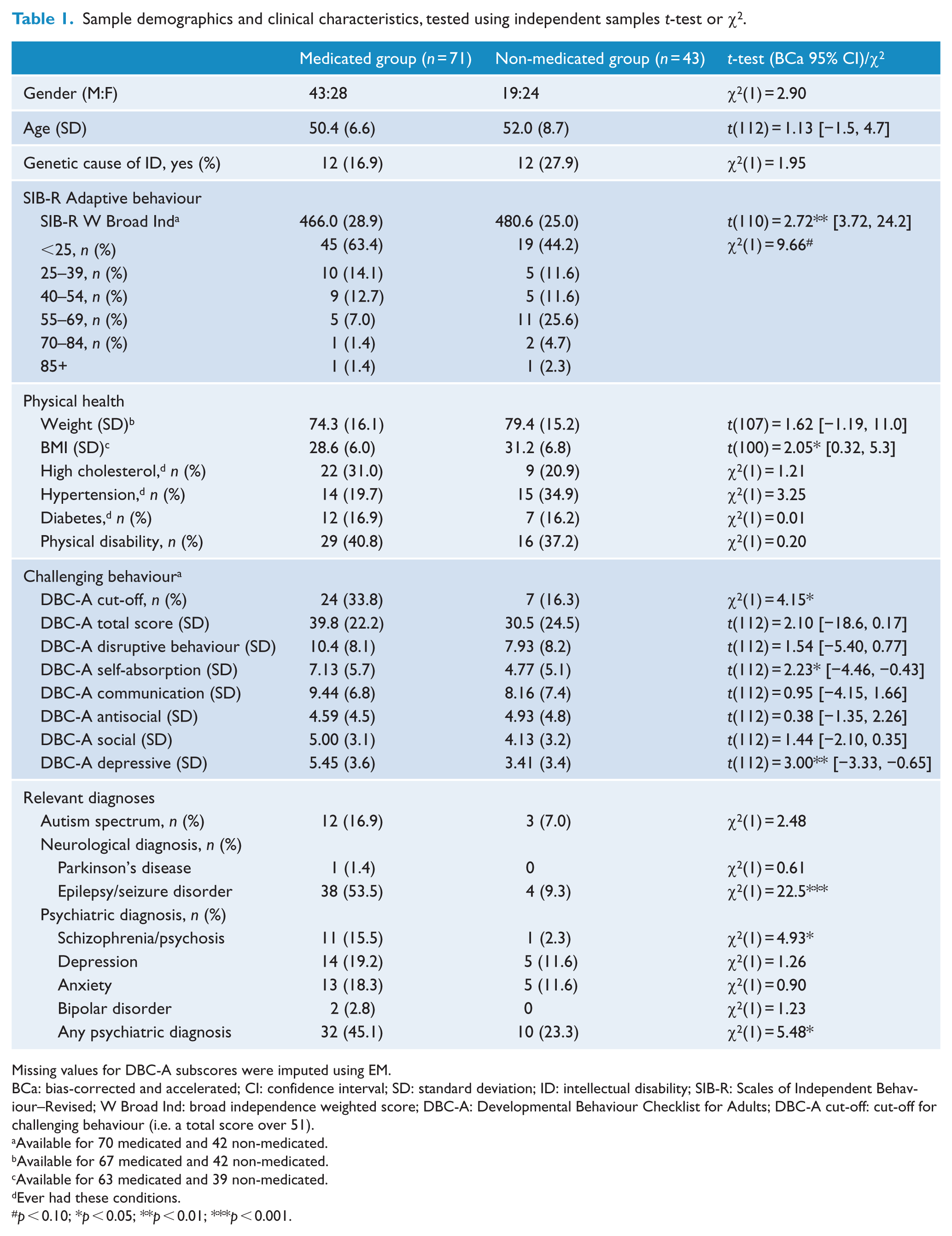
In all, 71 (62.3%) participants were reported to be taking at least one form of CNS medication. Demographics and health profiles for the medicated and non-medicated groups are detailed in Table 1. The groups did not differ significantly in terms of gender, age, weight, lifetime prevalence of high cholesterol, hypertension or diabetes, presence of physical disability, diagnosis of Parkinson’s disease, depression, anxiety, bipolar disorder or autism spectrum disorder (p > 0.05 for all analyses). In comparison with the non-medicated group, the medicated group had a significantly higher proportion of epilepsy or seizure disorder (χ2(1) = 22.5, p < 0.001), diagnosis of a psychotic disorder (χ2(1) = 4.93, p < 0.05) and any overall psychiatric diagnosis (χ2(1) = 5.48, p < 0.05).The medicated group had a significantly lower weighted SIB-R broad independence score (t(110) = 2.72, p < 0.01) and BMI (t(100) = 2.05, p < 0.05).
Sample demographics and clinical characteristics, tested using independent samples t-test or χ2.
Missing values for DBC-A subscores were imputed using EM.
BCa: bias-corrected and accelerated; CI: confidence interval; SD: standard deviation; ID: intellectual disability; SIB-R: Scales of Independent Behaviour–Revised; W Broad Ind: broad independence weighted score; DBC-A: Developmental Behaviour Checklist for Adults; DBC-A cut-off: cut-off for challenging behaviour (i.e. a total score over 51).
Available for 70 medicated and 42 non-medicated.
Available for 67 medicated and 42 non-medicated.
Available for 63 medicated and 39 non-medicated.
Ever had these conditions.
p < 0.10; *p < 0.05; **p < 0.01; ***p < 0.001.
In comparison with the non-medicated group, there were more participants in the medicated group who were above the cut-off for challenging behaviour as determined by the DBC-A total score (χ2(1) = 4.15, p < 0.05); however, there was only a trend difference in total score between the groups (t(112) = 2.10, p < 0.1). In terms of DBC-A subscores, the medicated group scored significantly higher on the self-absorbed (t(112) = 2.23, p < 0.05) and depression subscales (t(112) = 3.00, p < 0.01) than the non-medicated group. There were no other significant differences on the DBC-A.
Gender did not have a bearing on total DBC-A score (t(112) = 0.144, p = 0.886), broad independence score (t(110) = −0.383, p = 0.702), diagnosis of schizophrenia (χ2(1) = 0.104, p = 0.747), bipolar disorder (χ2(1) = 0.016, p = 0.900), depression (χ2(1) = 0.028, p = 0.866), anxiety (χ2(1) = 0.012, p = 0.914) or seizure disorder (χ2(1) = 1.515, p = 0.218).
CNS medication use and polypharmacy
A list of the CNS medications used by sample participants is supplied in the supplementary materials (see Supplementary Table 1). The most common prescribed CNS medication category was anticonvulsants (41.2% of sample), followed by antipsychotics (32.5%), antidepressants (23.7%), anti-anxiety medications (10.5%), movement disorder medications (3.5%), sedatives/hypnotics (1.8%) and CNS stimulants (0.9%).
Of those on a CNS medication, 76.1% of participants were on more than one (either from the same class or from different medication classes), and 59% were on more than one CNS medication class, with 12 participants (10.5% of the total sample) taking three or more classes of medication. Figure 1 is a Venn diagram providing details of the CNS medication polypharmacy within the sample. The most common form of polypharmacy that occurred within the same class was anticonvulsants (n = 21). There was only one participant who was prescribed more than one antipsychotic. The most common multi-class polypharmacy was the combination of an antipsychotic and an anticonvulsant (n = 19).
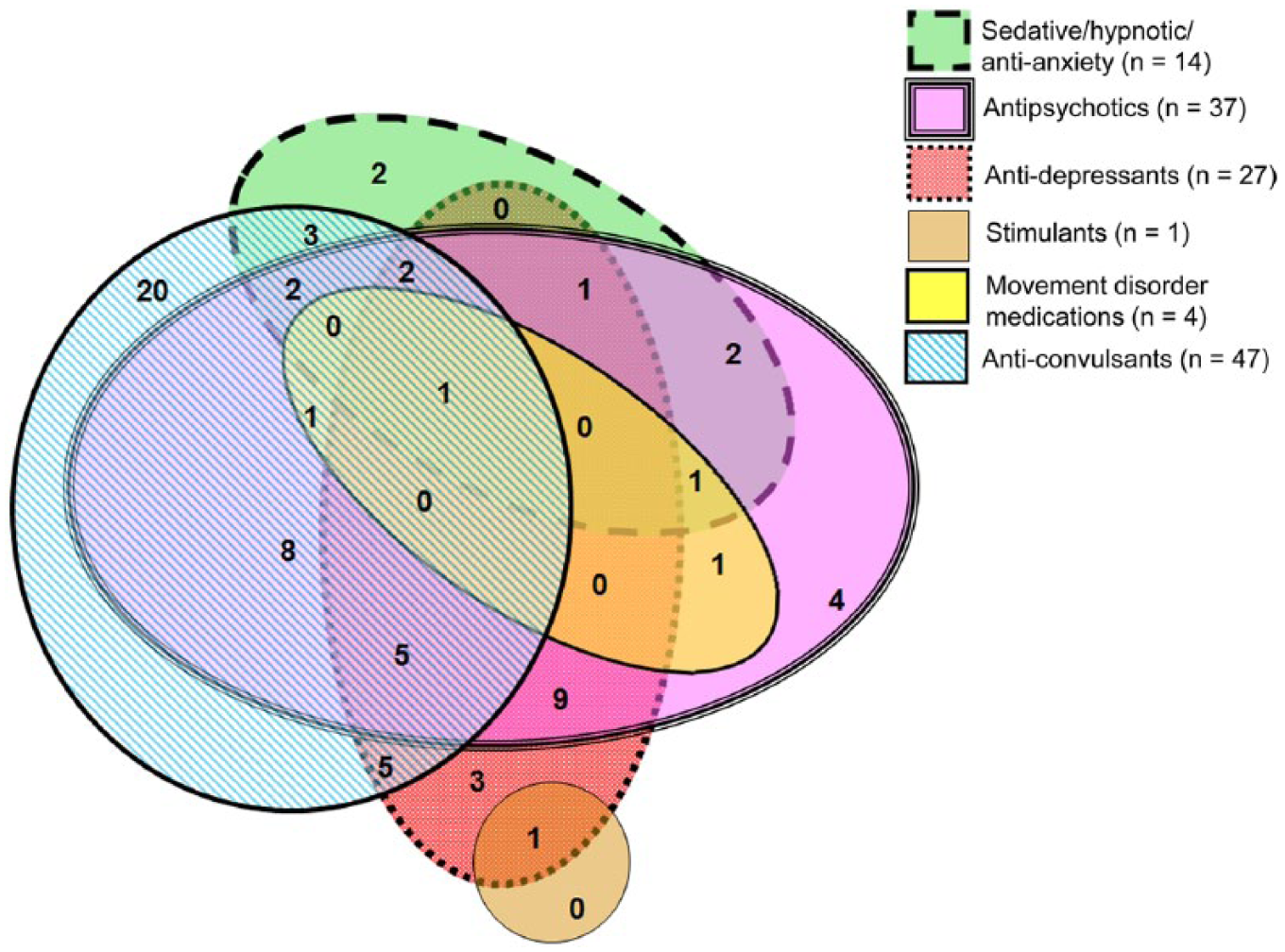
Venn diagram detailing the CNS medication polypharmacy within the sample. The key for different medication classes is as follows: solid green (sedatives/hypnotics/anti-anxiety medications), solid pink (antipsychotics), red with white dots (antidepressants), solid tan (stimulants), solid yellow (movement disorder medications) and white and blue stripped (anticonvulsants).
Prescribing patterns
According to the reported psychiatric or neurological diagnosis, the number of participants for which the CNS medication could be considered either a first- or second-line treatment was as follows for each medication category: 39 (83.0%) of those taking anticonvulsants, 7 (58.4%) of those taking anti-anxiety agents, 14 (51.9%) of those taking antidepressants, 15 (40.5%) of those taking antipsychotics and 1 (25.0%) of the participants taking movement disorder medications. Neither the two participants taking sedatives nor the one participant taking a stimulant had a corresponding reported indication.
Linear regression
The results from the linear regression predicting number of CNS medications used are shown in Table 2. The regression was significant overall (F(8, 101) = 7.75, p < 0.001). Significant predictors of CNS medication polypharmacy were the presence of neurological diagnosis and DBC-A total score, whereby a participant with a higher DBC-A score was more likely to be prescribed a greater number of CNS medications than someone with a lower score. VIF values were between 1.0 and 1.1. Cook’s distance for variables was less than 1, and standardised residuals were within 3 standard deviations (SDs).
Linear regression assessing predictors of CNS medication polypharmacy (either from the same medication class or from different classes).
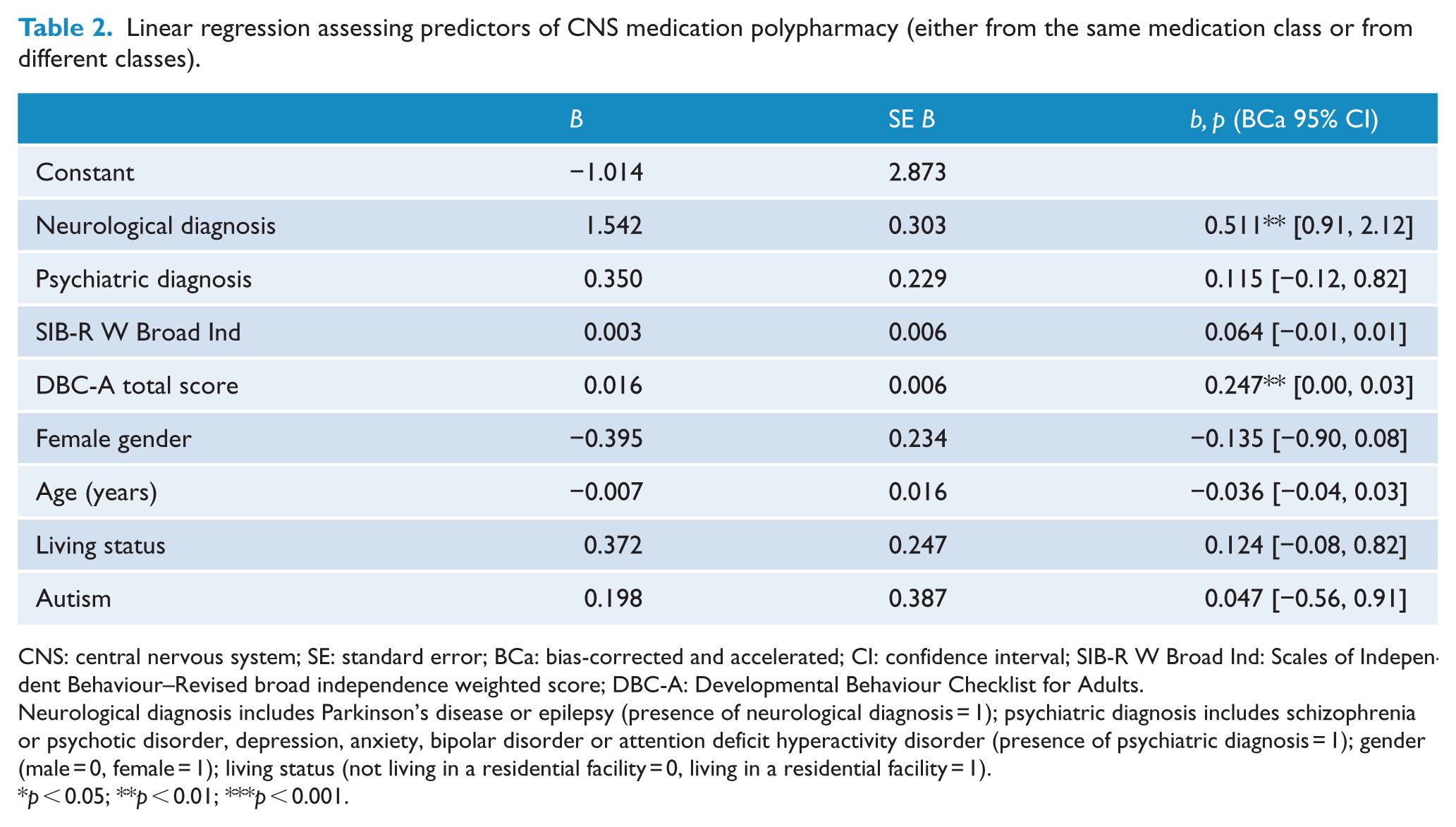
CNS: central nervous system; SE: standard error; BCa: bias-corrected and accelerated; CI: confidence interval; SIB-R W Broad Ind: Scales of Independent Behaviour–Revised broad independence weighted score; DBC-A: Developmental Behaviour Checklist for Adults.
Neurological diagnosis includes Parkinson’s disease or epilepsy (presence of neurological diagnosis = 1); psychiatric diagnosis includes schizophrenia or psychotic disorder, depression, anxiety, bipolar disorder or attention deficit hyperactivity disorder (presence of psychiatric diagnosis = 1); gender (male = 0, female = 1); living status (not living in a residential facility = 0, living in a residential facility = 1).
p < 0.05; **p < 0.01; ***p < 0.001.
The results from the linear regression predicting number of classes of CNS medications used are shown in Table 3. The regression was significant overall (F(8, 101) = 6.23, p < 0.001). Significant predictors were diagnosis of neurological or psychological illness, DBC-A total score and gender. Participants with a higher DBC-A score and male gender were more likely to be prescribed more than one class of CNS medication. VIF values were between 1.0 and 1.1. Cook’s distance for variables was less than 1. There were two standardised residuals that fell outside of 3 SDs. Removal of these potential outliers did not change the significance of the model nor the predictors. Post hoc regression including the gender by DBC-A total score interaction coefficient was not significant (p = 0.99).
Linear regression assessing predictors of CNS medication class polypharmacy.
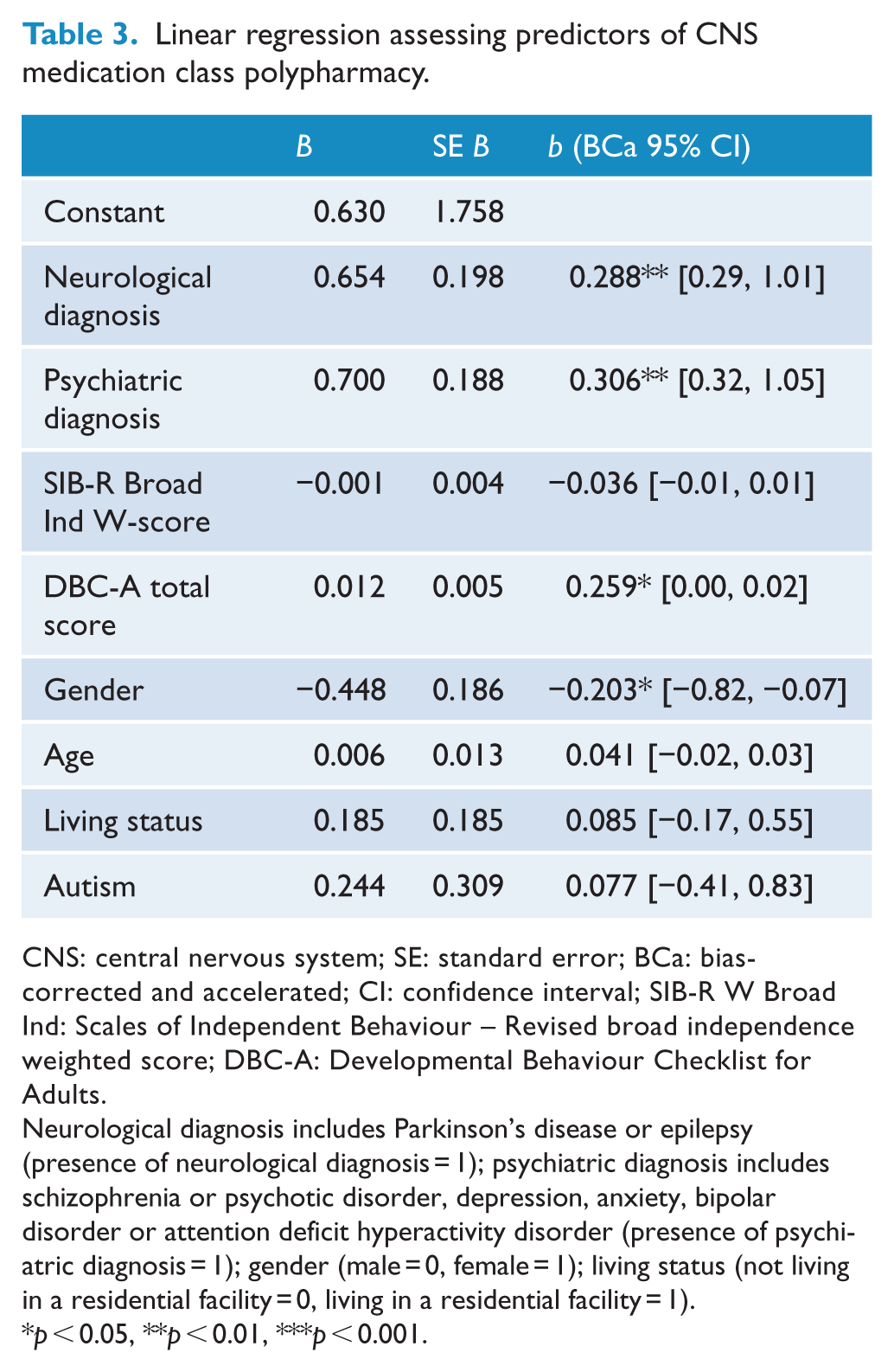
CNS: central nervous system; SE: standard error; BCa: bias-corrected and accelerated; CI: confidence interval; SIB-R W Broad Ind: Scales of Independent Behaviour – Revised broad independence weighted score; DBC-A: Developmental Behaviour Checklist for Adults.
Neurological diagnosis includes Parkinson’s disease or epilepsy (presence of neurological diagnosis = 1); psychiatric diagnosis includes schizophrenia or psychotic disorder, depression, anxiety, bipolar disorder or attention deficit hyperactivity disorder (presence of psychiatric diagnosis = 1); gender (male = 0, female = 1); living status (not living in a residential facility = 0, living in a residential facility = 1).
p < 0.05, **p < 0.01, ***p < 0.001.
We re-ran the regressions with altered CNS medication total and CNS medication class total values, adjusting so that anticonvulsant use for seizure disorder was not included in the total. The results from the linear regression predicting number of CNS medications used were again significant (F(8, 101) = 4.08, p < 0.001) as was regression predicting number of classes of CNS medications used (F(8, 101) = 4.74, p < 0.001). For both models, significant predictors included the presence of psychiatric diagnosis, higher DBC-A total score and male gender.
The regressions were re-run again with altered CNS medication total and class values, excluding medications most commonly indicated for neurological diagnosis (i.e. anticonvulsants and movement disorder medications). The results from the linear regression predicting number of CNS medications used were again significant (F(8, 101) = 4.00, p < 0.001) as was regression predicting number of classes of CNS medications used (F(8, 101) = 4.78, p < 0.001). For both models, significant predictors included the presence of psychiatric diagnosis, higher DBC-A total score and male gender.
Discussion
This study was motivated by the lack of data about the CNS medication prescribing patterns of older adults with ID. As expected from previous research in younger adults, the findings in our sample highlight the high rate of CNS medication prescription and polypharmacy. Polypharmacy was associated with the presence of neurological diagnosis and DBC-A total score, and multi-class polypharmacy was further associated with psychiatric diagnosis and male gender. The robustness of the findings is emphasised by the results yielded after re-running the regressions to adjust for the high rate of seizure disorder (and the corresponding medications required), which demonstrated that diagnosis of a psychiatric disorder, higher DBC-A score and male gender remained significant predictors of polypharmacy.
The finding that over 60% of our sample was prescribed at least one CNS medication is at odds with prescribing that occurs within the general population of similar age range (between 10 and 25%; ABS, 2011) and reflects the higher rates of psychiatric and neurologic diagnosis that occurs in the ID population (McGrother et al., 2006; Smiley et al., 2007). Furthermore, the most common type of CNS medication used is also disparate across our sample and that of the general population. In this sample, anticonvulsants were the most common drugs prescribed, followed by antipsychotics (cf. antidepressants in the general population; ABS, 2011). These discrepancies are indicative of the large differences in morbidities between older people without versus those with ID.
Indications for the medications used in the sample were based on those known and approved for the medication classes included. This approach is limited, especially given that the timeframe for different questions is varied such that respondents are asked about current medications and behaviours versus lifetime diagnoses. Using this methodology, however, the results suggest that there was adequate rationale for CNS medication prescription related to anticonvulsants, but a rationale for the prescription psychotropic medications was less clear, especially for antipsychotics. A neurological diagnosis (the majority of which were seizure disorders) was the strongest predictor of CNS medication polypharmacy, and accordingly, anticonvulsants were the most common medication class taken and the most common combination within-class polypharmacy. It is noteworthy to mention here that with the introduction of newer antiepileptic drugs, monotherapy for epilepsy treatment in ID is preferred over polytherapy when possible (Santosh and Baird, 1999).
Higher DBC-A score was a predictor of CNS medication polypharmacy in older adults. This analysis was limited in that there was inadequate power to assess DBC-A subscales within the regression models. Nonetheless, t-tests revealed that the medicated group scored higher on the depression and self-absorbed subscale of the DBC-A (Table 1). From this cross-sectional analysis, it is impossible to determine whether these differences represent traits that increase the propensity for CNS medication prescription (i.e. the behaviour is being managed with the medication, possibly ineffectively), whether these are side effects of the CNS medication (i.e. the behaviour disturbance is an adverse effect) or whether the medication is being used for a neuropsychiatric disorder that tends to be associated with these traits.
Psychiatric diagnosis was a significant predictor of multi-class polypharmacy, that is, prescription of more than one class of drug (e.g. antipsychotic combined with anticonvulsant). In the treatment of comorbid symptom dimensions, this practice may be fully justified (Royal Australian and New Zealand College of Psychiatrists, 2005). Overall, polypharmacy (i.e. multiple drugs across any class or within the same class) was not predicted by psychiatric diagnosis, except when the high rates of treatment of seizure disorders were controlled for. Similarly, when both anticonvulsants and movement disorder medications were excluded from the analysis altogether, psychiatric diagnosis emerged as a significant predictor of overall polypharmacy. The regression models in this study also highlight a propensity for males to be prescribed a greater number of CNS medication classes than are females. This is a finding that has not been reported in previous studies, although another study reported that males were more likely to receive restrictive intervention, including chemical restraint (Webber et al., 2011). Previously, it has been found that males are more likely to exhibit challenging behaviours as aggression (McClintock et al., 2003) and, hence, may be perceived as more threatening and requiring chemical intervention (Webber et al., 2011). Conversely, in this study, there was no difference across genders in the DBC-A disruption subscale, indicating that aggressive behaviour was not rated higher in males or that the medications were successfully treating previous aggressive behaviour. While there was a significant difference in the social-relating disturbance DBC-A subscale between genders, there were no differences in patterns of neurological or psychiatric diagnosis, broad independence score and DBC-A total score, and the interaction between gender and DBC-A score was not significant. These findings suggest that DBC-A score and gender were independent predictors of multi-class polypharmacy. It is unclear why males would be more likely to be prescribed more than one class of CNS medication in this sample, but future investigation is warranted.
The finding of higher BMIs in the non-medicated group is contrary to the evidence that psychotropic medication use has a negative impact on weight and BMI in ID (De Kuijper et al., 2013), although this may relate to the noted differences in broad independence between the two groups. Indeed, increased independence is often associated with poor diet and weight gain in people with ID (Melville et al., 2008).
Limitations
There were some limitations to consider when interpreting the findings of this study. First, inherent limitations have been associated with questionnaire studies, including the potential for inaccurate data. Of particular relevance to the current results is the possibility that inaccurate medication information was reported and missed, or diagnoses were under-reported. In order to minimise this risk, we asked that the main carer of the person with ID take responsibility for completing the questionnaire, thereby ensuring the most accurate information available was recorded. In addition, carers providing questionnaires with missing data were followed up by telephone.
The limited sample size meant that exploring other factors that may have predicted CNS medication use (e.g. specific diagnosis of ID, specific medication classes that more commonly used for challenging behaviours or specific DBC-A categories are more predictive of CNS medication polypharmacy) was beyond the scope of the study.
In NSW, less than 5% of potential participants in the areas targeted were recruited into the study. Data were not available regarding the potential number of people in the Victorian region. This was despite a broad-based approach to recruitment. Furthermore, adaptive behaviour levels were skewed in the direction of a more severely affected sample, the rate of seizure disorders was consistent with rates found in severe cases of ID (Santosh and Baird, 1999) and psychotic disorders reported in our sample are much higher than those reported previously (Smiley et al., 2007). Under-ascertainment of people with mild ID is common in studies of this population (Lennox et al., 2005). There are a number of factors that may have influenced this sampling bias in this study. It may be a result of recruiting predominantly through ID services or that people with milder ID are less likely to have a carer readily available to answer the questionnaires compared to those with a more severe ID who are likely receiving a higher level of care. Furthermore, it is possible that those who were most affected by age-related issues may have been more likely to agree to participate or, conversely, that those with the greatest number of health and psychosocial issues may have chosen not to participate due to time constraints. The results should therefore be interpreted with this caveat in mind. Notable strengths of our strategy included sampling from a number of different socio-economic areas within urban and rural locations across NSW and Victoria, as well as recruiting across a broad spectrum of ID services as well as family homes. A future area of enquiry would be to monitor the use of CNS medications across these locations in light of differences in policy, practice and service provision across states and regions.
Finally, it was only when controlling for the high rates of treatment of seizure disorders, the regressions revealed that psychiatric diagnosis was a significant predictor of overall polypharmacy. These results suggest that the high rates of medicating seizure disorders in this sample may be diluting the results, and adjusting for these comorbidities should be taken into consideration in future studies.
Conclusion and recommendations
Given the high rate of CNS medication use and polypharmacy and the obvious nexus between CNS medication use, behaviour and psychiatric diagnoses, these findings in older adults have relevance to current policy practice. Greater awareness of, and adherence to, national recommendations (Trollor, 2012) and international guidelines (WPA (SPID), 2010) is required. Concerted training and education of primary care providers and psychiatrists in this area would also be of benefit. Regular medication reviews, similar to those afforded to other ageing patients, will be likely to minimise the burden of polypharmacy and its associated potential harms. A key challenge for the National Disability Insurance Scheme (NDIS) is to improve access to appropriate behavioural support for people with ID and challenging behaviour. Such measures, together with the development of enhanced multidisciplinary frameworks which include both disability and mental health professionals, are likely to lead to a more considered approach, particularly to psychotropic medication prescription in people with ID.
It appears timely to reiterate the current medication guidelines for ID in the context of challenging behaviours. The main recommendations from this study are to use CNS medications once other non-pharmacological interventions have been considered. If the decision to prescribe a CNS medication has been made, then appropriate physical examinations, investigations and regular monitoring are required. Prescribing only one medication at a time also enables the opportunity to safely and rationally reduce the dose and use of CNS medications (Thomas et al., 2010; Torr, 2013; WPA (SPID), 2010).
Footnotes
Acknowledgements
The authors wish to thank the individuals and their families, carers, guardians and friends who participated in the SAge-ID study. Thanks also to the organisations that assisted with recruitment. We thank all the funders for their contributions to this valuable research.
Declaration of interest
The authors declare that there is no conflict of interest.
Funding
Core funding for this project was received from The NSW Government Department of Family and Community Services, Ageing, Disability and Home Care (ADHC). Other funding to support the broader SAge-ID Study came from the Dementia Collaborative Research Centres (DCRC), a La Trobe University Faculty of Health Sciences Grant and The Mason Foundation’s Medical and Scientific Research Grants.
