Abstract
Objective:
This review provides a brief account of the clinically relevant functional neuroanatomy of the thalamus, before considering the utility of various modalities utilized to image the thalamus and technical challenges therein, and going on to provide an overview of studies utilizing structural imaging techniques to map thalamic morphology in the spectrum of neurodegenerative disorders.
Methods:
A systematic search was conducted for peer-reviewed studies involving structural neuroimaging modalities investigating the morphology (shape and/or size) of the thalamus in the spectrum of neurodegenerative disorders.
Results:
While the precise role of the thalamus in the healthy brain remains unclear, there is a large body of knowledge accumulating which defines more precisely its functional connectivity within the connectome, and a burgeoning literature implicating its involvement in neurodegenerative disorders. It is proposed that correlation of clinical features with thalamic morphology (as a component of a quantifiable subcortical connectome) will provide a better understanding of neuropsychiatric dysfunction in various neurodegenerative disorders, potentially yielding clinically useful endophenotypes and disease biomarkers.
Conclusion:
Thalamic biomarkers in the neurodegenerative disorders have great potential to provide clinically meaningful knowledge regarding not only disease onset and progression but may yield targets of and perhaps a way of gauging response to future disease-modifying modalities.
Essentially, the cortex must view the world through the thalamus: that is the only view the cortex has.
Introduction
Framing the thalamus in the context of endophenotypes
There has been a marked shift in the approach to characterizing and understanding neuropsychiatric disease with a focus upon endophenotypes that has driven change in research paradigms (Gottesman and Gould, 2003). The concept of intermediate phenotypes (or endophenotypes) has been described as ‘measurable components unseen by the unaided eye along the pathway between disease and distal phenotype’ (Gottesman and Gould, 2003). Accordingly, correlation between genetic factors and measurements of the neural circuit substrates of cognition, emotion, motor function and behaviour may constitute steps towards establishing a genotype to morphotype relationship. The morphotype thus derived may then determine the phenotype (see Looi and Walterfang, 2012).
In this paper, we apply the framework of the endophenotypes and its antecedents in the endomorphotypes (morphology) and ultimately (although not currently) the endogenotype to understand the involvement of a crucial network hub in the human brain, the thalamus, in neurodegenerative disease. We argue that the thalamus serves as a crucial hub in the extensive topographically distinct neuronal networks relevant to human brain disease and its manifestations; the morphology (shape and volume) of the thalamus can be quantified in vivo; quantified morphology (morphometry) may be correlated to endophenotypic features of the disease such as impairments in cognition, emotion, motor function and behaviour, and therefore, morphometry of the thalamus may be used to measure disease progression as a potential biomarker.
Basic and clinical neuroanatomy of the thalamus
Historical aspects
The term ‘thalamus’ is from the Greek word thalamos (θάλαμος), referring to an inner-most room but also used to describe a storeroom and a sleeping or bridal chamber; the word was used commonly in ancient Greek texts, such as Homer’s Odyssey. The first documented neuroanatomical usage of the word was by Galen in the second century AD when using as an analogy the floor plan of a Greek house, but it is quite likely he was referring to a part of the ventricular system (possibly as a ‘storeroom’ of pneuma), rather than what is referred to as the thalamus today; Thomas Willis, in the mid to late 1600s, was the first to refer to the large mass of grey matter in the dorsal part of the diencephalon as the thalamus (see Jones, 2007).
Embryology and gross anatomy of the thalamus
During development, the primitive forebrain gives rise to the telencephalon (or end-brain), which has as its derivatives the cerebral cortex, striatum, amygdala and associated structures, and the diencephalon (or between-brain), which gives rise primarily to the thalamus and hypothalamus. It is likely that connections between the thalamus and cortex occur after subcortical afferents have innervated the thalamus, with corticothalamic connections preceding thalamocortical connections. Hence, maturation of the thalamus and cortex are related intimately, such that an abnormality in the early neurodevelopmental period of thalamic organization could impair normal cortical development, and vice versa; consistent with a neurodevelopmental model, it has been proposed that pathology of the thalamus may play an important role in the neurobiology of schizophrenia (see Byne et al., 2009).
While the diencephalon can be generally divided into the thalamus and hypothalamus, it includes a number of other structures (such as the habenular nuclei of the epithalamus). The thalamus itself can be divided into the dorsal and ventral divisions, based on their relative position in the lateral diencephalic wall during development, and also on the basis of their connectivity (Jones, 2007). The dorsal thalamus is composed of nuclei having reciprocal connections with the cerebral cortex and striatum, while the ventral thalamus (despite having direct input from the cortex and, in part from the basal ganglia) does not project to the cortex. The ventral thalamic nuclei include the reticular thalamic nucleus (RTN), zona incerta (ZI) and ventral lateral geniculate nucleus (LGN), and a shared feature of these nuclei appears to be projections to and a close anatomical and functional relationship with the dorsal thalamus (Kolmac et al., 2000; Mitrofanis, 2005; Mitrofanis and Guillery, 1993; Power et al., 1999). From a neuroanatomical and neuroimaging perspective, the RTN and ZI assist with the lateral and inferior boundary definition, respectively, throughout much of the rostral–caudal extent of the dorsal thalamus (Power et al., 2015). This review will focus primarily on the dorsal thalamus unless otherwise stated.
Occupying approximately the volume of a Brazil nut (Bertholletia excelsa) in the human (~7000mm3 in a healthy young adult), the two dorsal thalami are located strategically at the very ‘inner-most’ region of the brain, between other subcortical structures and cortex (see Figure 1). Gross anatomical landmarks defining the boundaries of the thalamus are as follows: the third ventricle serves as the medial boundary, and the internal capsule separates the thalamus from the basal ganglia laterally; superiorly, the thalamus is bound by the lateral ventricle, and inferiorly, a number of distinctive structures abut the thalamus (including the ZI, substantia nigra, subthalamic nucleus, red nucleus). The dorsal thalamus has anterior, medial and lateral subdivisions, defined by a curved sheet of myelinated fibres called the internal medullary lamina (Figure 2). These subdivisions are composed of distinct thalamic nuclei, defined on the basis of their cytoarchitecture, patterns of connectivity and functionality (see Table 1, Figures 1 and 2); however, these nuclei share many features in common as outlined below. The anterior subdivision is defined by the anterior nucleus (AN) and the medial subdivision by mediodorsal nucleus (MD). The lateral subdivision has a dorsal and ventral tier of nuclei: the dorsal tier contains the lateral dorsal nucleus (LD) and the pulvinar; the ventral tier contains the ventral anterior (VA), ventral lateral (VL) and ventral posterior (VP) nuclei. Sometimes considered as a caudal extension of the ventral tier are the LGN and medial geniculate nucleus (MGN) (which are found inferior to the pulvinar). The internal medullary lamina itself also encloses the intralaminar nuclei (IL); the midline nuclei (ML) cover portions of the medial surface of the dorsal thalamus.
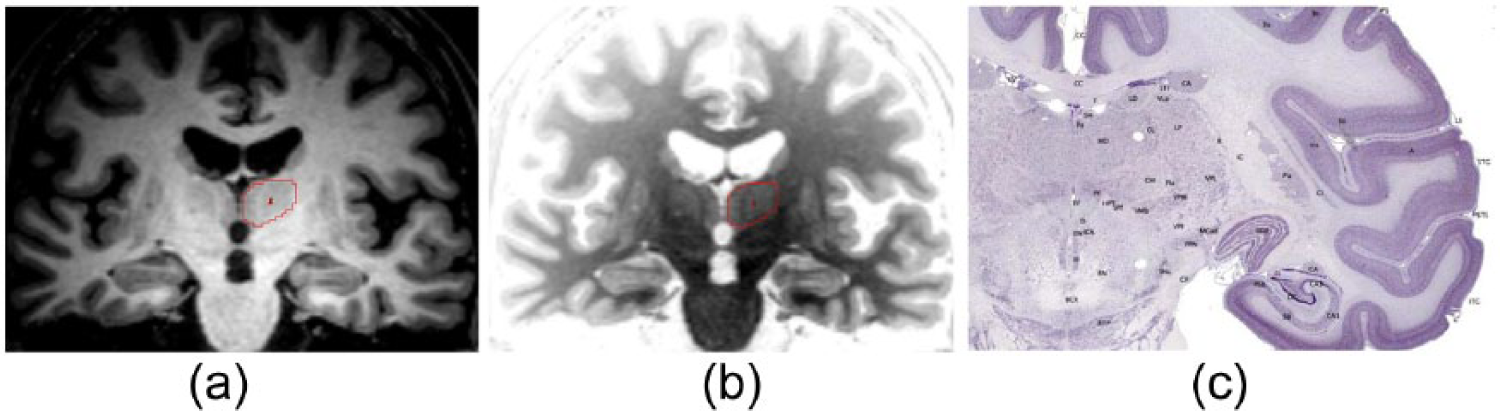
Figure showing sections through the thalamus. (a) and (b) are magnetic resonance imaging (MRI) T1 weighted images of the same section of human thalamus (traced in red), with intensity settings inverted (using ANALYZE 11.0 software); images provided by the authors. (c) is a Nissl stained coronal section through Macaca Mulatta thalamus.
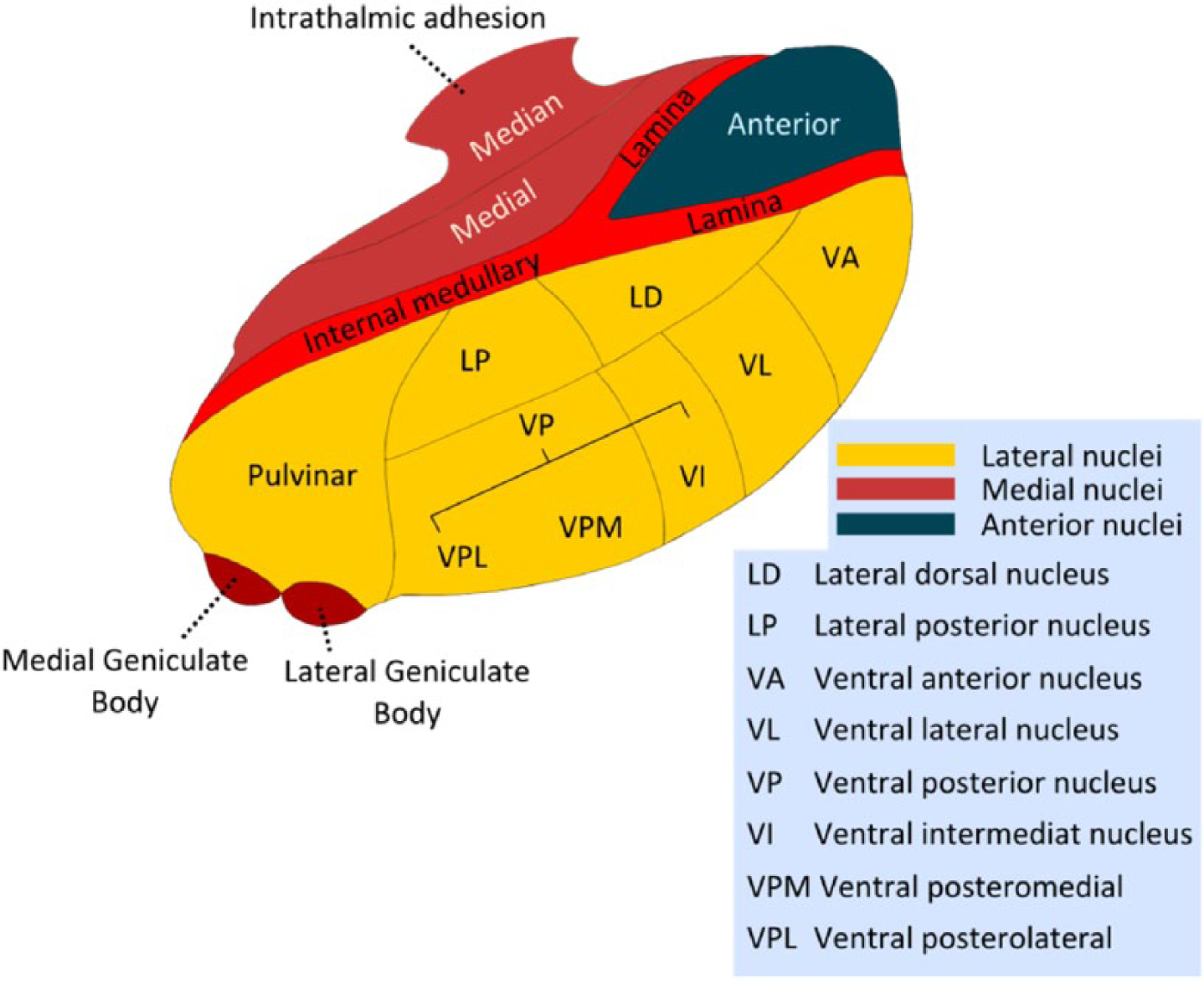
Schematic diagram of the thalamus (right) showing subdivisions and nuclei.
Classification, connections and functions of the dorsal thalamic nuclei (first order and higher order).
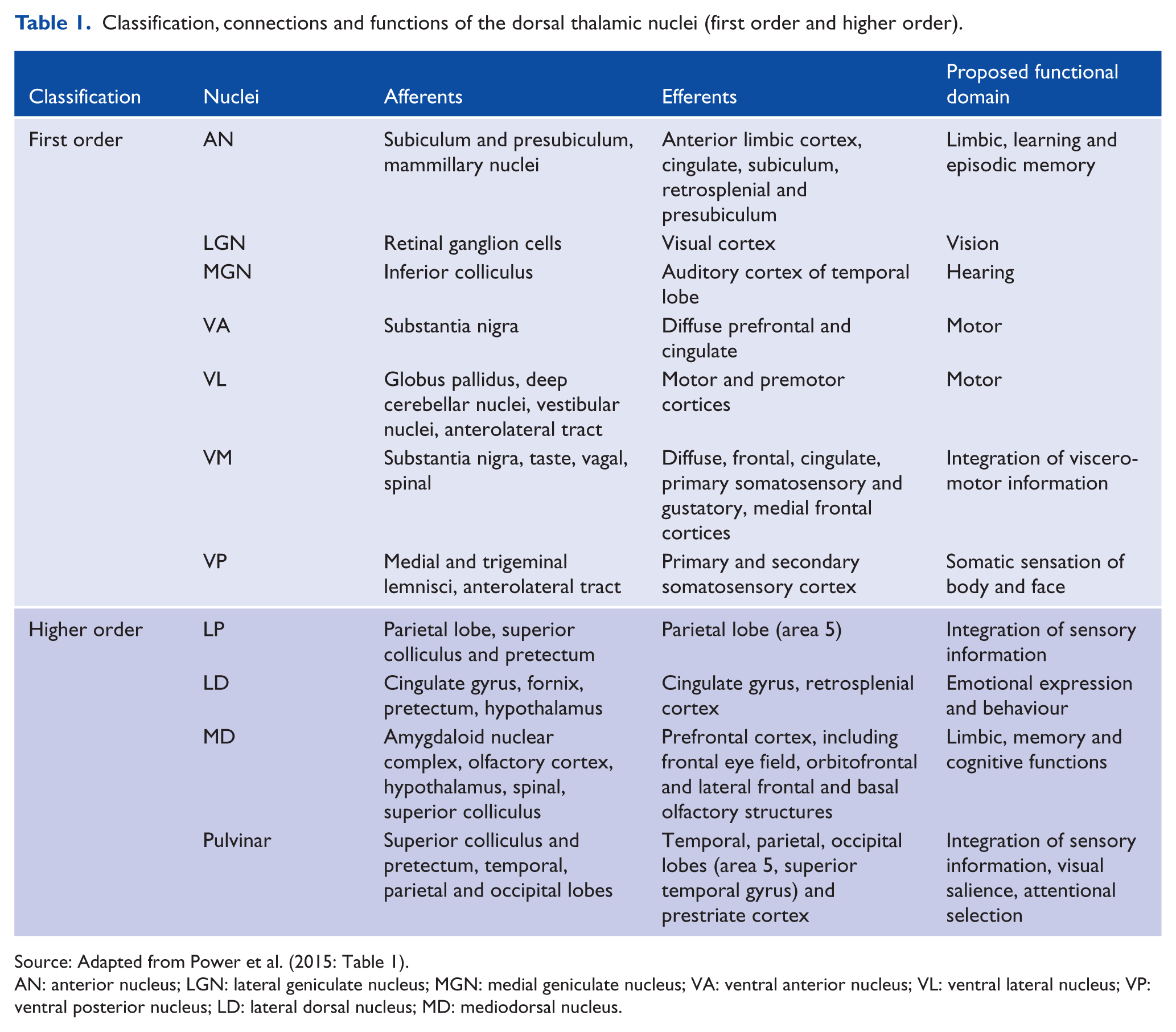
Source: Adapted from Power et al. (2015: Table 1).
AN: anterior nucleus; LGN: lateral geniculate nucleus; MGN: medial geniculate nucleus; VA: ventral anterior nucleus; VL: ventral lateral nucleus; VP: ventral posterior nucleus; LD: lateral dorsal nucleus; MD: mediodorsal nucleus.
Genetic factors involved in neurodevelopment
The neurodevelopment of the thalamus has been summarized extensively (see Blackshaw et al., 2010; Jones, 2007), and the following is a brief synopsis to establish that there are significant genetic factors involved in development. A relatively small number of gene sets are thought to be expressed in the developing diencephalon, with three major compartments identified (p1, p2 and p3) (Puelles and Rubenstein, 2003), and a mid-diencephalic organizer (Scholpp et al., 2009) releasing several signalling factors crucial to thalamic organization, including Sonic hedgehog (SHH) and fibroblast growth factor (FGF) (Bulfone et al., 1993).
A recent study reported that genetic variation within the Human Leukocyte Antigen locus (previously associated with schizophrenia) influences the volume of the left thalamus (Brucato et al., 2015). Further studies will need to ascertain the genetic factors associated with thalamic volume. A correlation between genetic factors and thalamic volume may constitute a step towards establishing a genotype to morphotype relationship. The morphotype thus derived may then determine the phenotype (see Looi and Walterfang, 2012).
Physiology
There are two types of neurons in dorsal thalamic nuclei which can be distinguished by their unique morphology and chemoarchitecture: locally acting interneurons immunoreactive for γ-aminobutyric acid (GABA), and glutamatergic relay cells projecting outside of the thalamus (which have a variety of voltage dependent membrane properties). The physiological properties of thalamic relay cells suggest that they have two main types of response modes – either tonic or burst – which will determine the nature of the message relayed to the cortex, and these response modes are thought to underpin changes in behavioural states. A burst response mode is utilized for signal detection, or salience (for instance, in response to novel stimuli or sudden changes in the environment), whereas a tonic response mode is believed to be utilized by the thalamic relay cells for more accurate signal analysis (Crick, 1984; Sherman and Guillery, 2006). The response mode of thalamic relay cells (i.e. shifting from signal analysis to signal detection, and vice versa) is thought to be modulated by brainstem and cortical inputs to the thalamus; it has been suggested that disturbance of this process may underpin the aberrant salience seen in neuropsychiatric disease (for instance, see Byne et al., 2009).
Drivers and modulators
The nuclei of the dorsal thalamus are innervated by two types of afferent fibres, classified as either drivers or modulators; this classification is regardless of whether the fibres originate from cortical or subcortical structures (see Figure 3). Afferent fibres can be defined on the basis of their synaptic morphology (e.g. drivers have large terminals on proximal dendrites forming multiple contacts, whereas modulators have small terminals on distal dendrites forming single contact) and postsynaptic actions (e.g. drivers activate ionotropic receptors and cause large excitatory postsynaptic potentials [EPSP], whereas modulators also activate metabotropic receptors, and cause small EPSP). The key feature of the modulator axon is that its terminals are less well-grouped than those of the driving axon terminals; furthermore, modulator afferents send a branch to innervate the RTN, whereas driver afferents do not (Bourassa et al., 1995; Darien-Smith et al., 1999). A key feature of driver afferents is that they determine the thalamic relay cell receptive field, whereas modulators do not. Drivers can therefore be conceptualized as those afferents to the thalamus that carry the message to be passed on by the thalamic relay cell, while the modulators can be conceptualized as those afferents to the thalamus that influence how this message is passed on by the thalamic relay cell. In other words, the drivers may ‘generate’ the message, while the modulators ‘adjust’ the message.
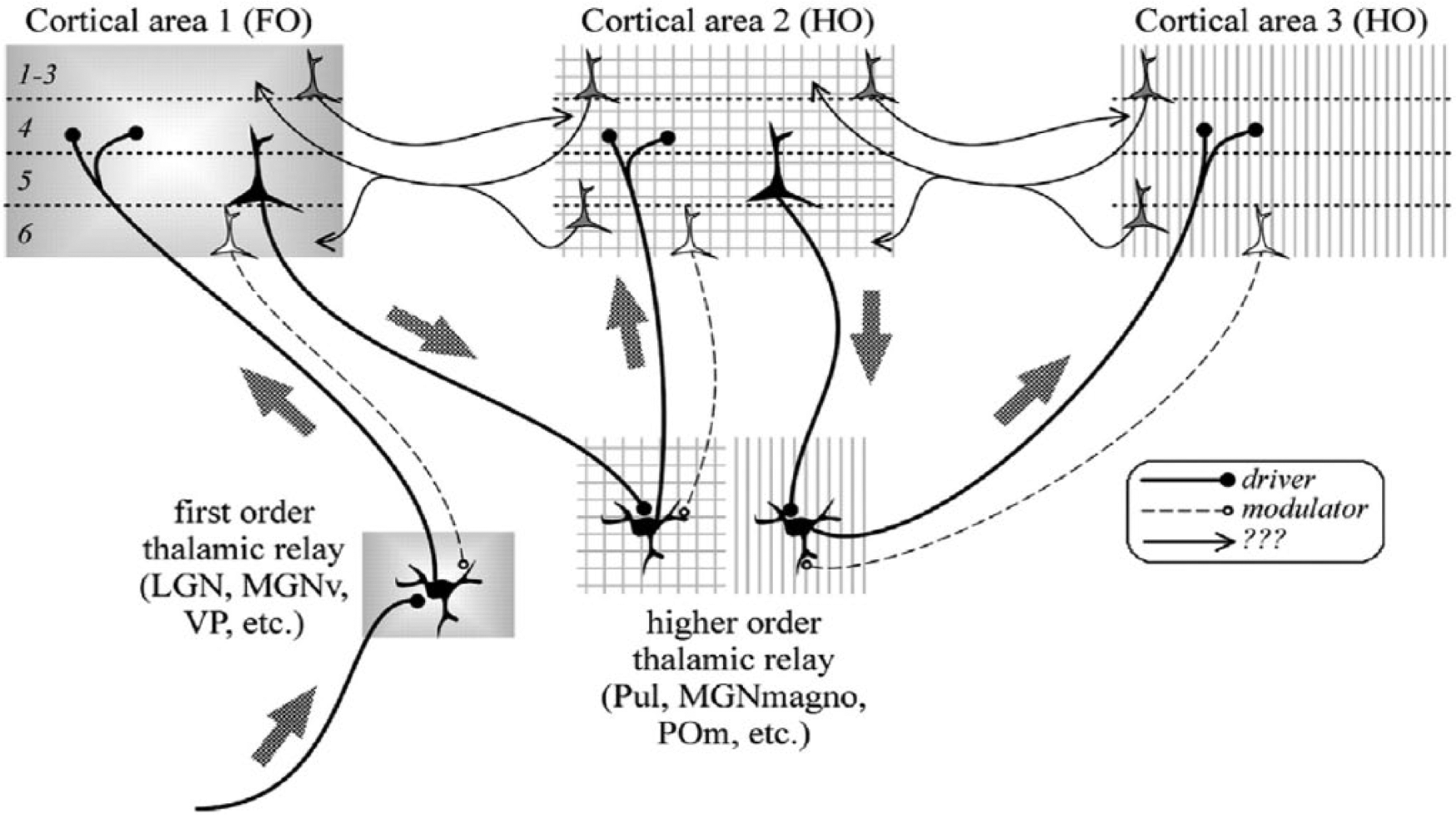
Schematic diagram showing first order and higher order relays. A first order thalamic relay represents the first relay of peripheral or subcortical information of a particular type to a first order or primary cortical area. A higher order relay relays information from layer V of one cortical area to another cortical area; this can be between first order and higher order cortical area or between two higher order cortical areas. The difference is the driver input, which is subcortical for a first order relay and from layer V of cortex for a higher order relay.
First order and higher order nuclei
The nuclei of the dorsal thalamus can be categorized according to the afferent fibres they receive (i.e. driver or modulator fibres) (see Figure 3). The cells of first order nuclei (or ‘primary’ nuclei; e.g. LGN, MGN, VP) are innervated by drivers originating from a peripheral or subcortical structure, and receive feedback inputs (modulator afferents) from pyramidal cells in layer VI of the ipsilateral cortex (these cortical afferents also send branches to the RTN and this circuitry appears to have a modulatory function). In the configuration of this connectivity, visual, auditory and somatosensory afferent fibres bring peripheral information pertaining to their respective sensory modality to the first order nuclei of the thalamus (Figure 3).
In contrast to first order nuclei, higher order nuclei (or association nuclei; e.g. pulvinar, MD) receive their primary excitatory inputs (or drivers) from pyramidal cells in layer V of the ipsilateral cortex. These particular inputs to the higher order nuclei resemble the main peripheral or subcortical inputs to the first order nuclei, not only in the nature of their structure and synapses they make but also in that they lack a branch to the RTN (Guillery, 1995). In the configuration of this connectivity, cells of higher order nuclei are involved in the passage of information (for instance, with regard to the current output of the driving cortical area) from one cortical area to another via a transthalamic pathway (Figure 3); at least half of the thalamus is thought to be involved with this kind of signalling. While this challenges the traditional understanding of corticocortical communication, it is suggested that direct corticocortical pathways may be providing some other function (e.g. modulatory; Sherman and Guillery, 2006). Hence, it has been proposed that the higher order nuclei, which are well developed in humans, have a significant role in higher cortical functions, with the implication that impairments of such signalling could lead to dysfunctional cortical processing (see Guillery, 1995; Sherman and Guillery, 2006).
Maps in the thalamus
Some of the early studies of visual pathways reported rapid retrograde cell degeneration in a restricted column of visual thalamus (LGN) following localized lesions in the visual cortex, together with localized visual field losses (Kaas et al., 1972); subsequent trans-neuronal retrograde degeneration in the corresponding retinae were noted. These findings suggested there was a map of the visual field not only in the visual cortex but also in the visual thalamus. It is now well established that many of the afferent and efferent connections of the thalamus are mapped (see example of sensorimotor mapping in Figure 4). This not only includes driver afferents to first order nuclei (such as visual afferents to LGN, auditory afferents to MGN and somatosensory afferents to VP) but also conservation of these maps in thalamic circuitry, with topographically organized first and higher order thalamocortical and corticothalamic pathways.
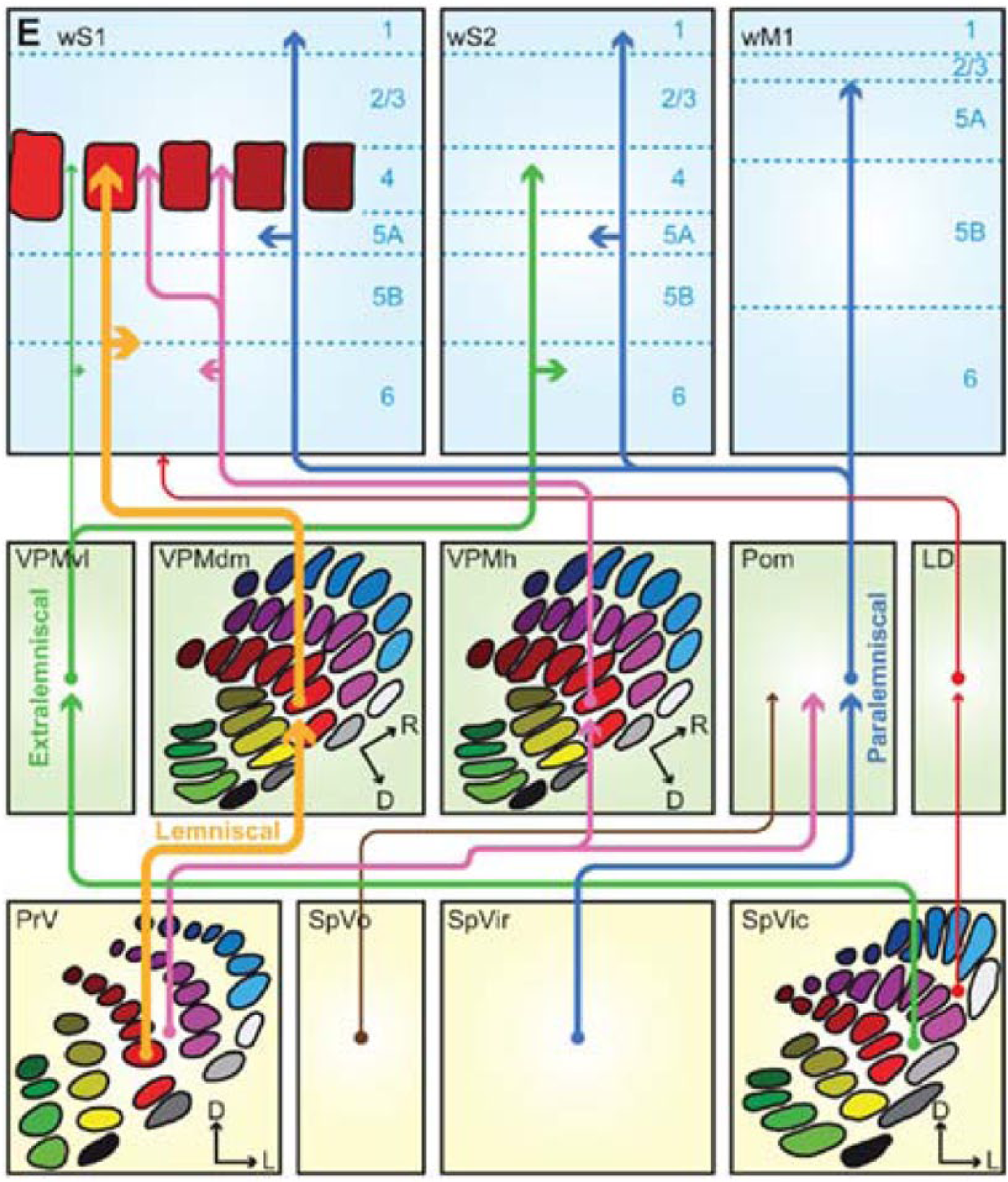
Maps in the thalamus. Schematic representation of the trigemino-thalamo-cortical pathways. The arrowheads indicate the termination areas of the axons. Note that (in the cerebral cortex) the postsynaptic cells may have their somata in other layers. The line thickness indicates the relative importance of the pathways. The barreloids in VPM are indicated in an oblique coronal slice, the barrelettes of the trigeminal nuclei in coronal slices.
One suggested mechanism in neurodegenerative disease is trans-synaptic neurodegeneration: it is proposed that deafferentation from damaged cortical neurons causes synaptic dysfunction, and this process leads to a neuronal propagation of disease along the related and therefore susceptible neural circuits (Jindahra et al., 2012; Looi and Walterfang, 2012; Palop and Mucke, 2009). Trans-synaptic degeneration could be one of a number of processes that leads to subcortical pathologies seen in the striatum in Huntington’s disease (HD), frontotemporal dementia (FTD) and Alzheimer’s disease (AD), for instance, where there is subcortical neuronal loss (Looi et al., 2012; Looi and Walterfang, 2012). A parallel explanation may also exist in the form of the putative propagation of proteopathies (protein misfolding or prions) along the same neural circuits, also resulting in topographically distinct patterns of neural loss in most age-related neurodegenerative disease (Warren et al., 2013). Indeed, it is also possible that proteopathies may also contribute to trans-synaptic neurodegeneration, although of course this does not exclude deafferentation phenomena. Similar mechanisms are likely to be at play in the other circuit structures, or hubs (such as the thalamus) in neurodegenerative disorders (Looi et al., 2014). Given the topographical relationship that the thalamus has with the cortex, one would envisage that investigating thalamic morphology may provide disease-specific maps of structural change in the cortical afferent pathways in various neurodegenerative conditions (Power et al., 2015; Power and Looi, 2014). Not surprisingly, the effect of cortical lesions in animal studies has been shown to have different effects on the receptive field properties of cells in the first order vs higher order thalamic nuclei: receptive fields of cells in first order nuclei (which receive corticothalamic modulator afferents) survive, whereas those of the relay cells of the higher order nuclei (which receive corticothalamic driver afferents) are lost (see Sherman and Guillery, 2006).
A critical hub in the subcortical connectome
There are numerous well-defined neural circuits involving the thalamus in addition to those already described, such as the fronto-subcortical circuits which are proposed to underpin motor activity and behaviour in humans (see Figure 5); furthermore, fronto-subcortical circuit dysfunction may also explain the likeness of behavioural changes seen in a plethora of frontal cortical and subcortical disorders (Cummings, 1993; Haber and Calzavara, 2009; Looi et al., 2012; Tekin and Cummings, 2002).
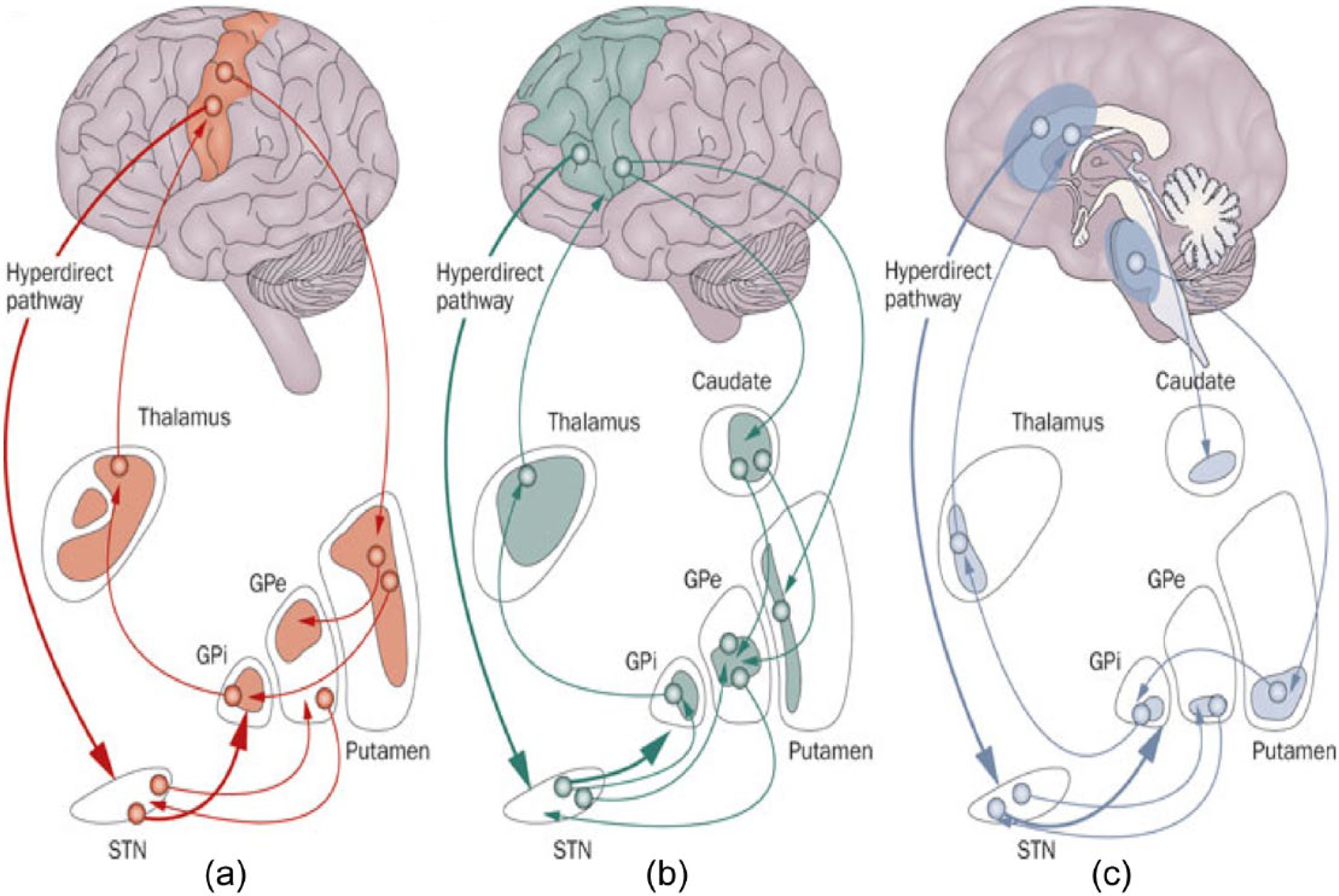
Fronto-subcortical loops involving the thalamus. Schematic diagram illustrating the organization of the fronto-subcortical loops within human brain: (a) motor loops, (b) associative loops and (c) limbic loops.
Fronto-subcortical circuits are named according to their function or site of origin in the cortex (e.g. ‘motor’ circuits: motor, oculomotor; and ‘cognitive’ circuits: dorsolateral prefrontal, orbitofrontal and anterior cingulate) (see Figure 5); importantly, the functional topography of cortex is maintained throughout these circuits. The circuits share a similar organization, originating in the prefrontal cortex, then projecting to the striatum (caudate, putamen, ventral striatum), having connections with the globus pallidus and substantia nigra, and thence to the thalamus, which ‘closes’ the loop by projecting back to the prefrontal cortex (Figure 5). There are also ‘open’ connections of each of these circuits, which are thought to integrate information from functionally related sites. Within each of these circuits, there are both direct and indirect pathways, which can modulate input to the thalamus, the function of which is thought to modulate overall circuit activities in response to different inputs (Tekin and Cummings, 2002). There is also emerging evidence that fronto-subcortical circuits can influence each other (see Haber and Calzavara, 2009), suggestive of integrative processing. This schema has provided a useful approach to neurodegenerative conditions on a neural circuit basis, on the rationale that delimited subsets of neuropsychiatric dysfunction may share a common neural circuit basis (Cummings, 1993; Looi et al., 2012). In neurodegenerative disease, it is proposed that cortical or subcortical lesions may disconnect fronto-subcortical circuits, and it is the circuit dysfunction that underpins the neuropsychiatric presentation for that disease. Such fronto-subcortical circuit dysfunction is thought to mediate the cognitive and behavioural disturbances manifest in FTD, for instance (Looi et al., 2012; Looi and Walterfang, 2012).
In a similar framework, a large body of structural and functional imaging studies have defined the cerebellothalamocortical circuits, linking functional areas of cerebellar cortex through the thalamus with both motor and non-motor cerebral cortex, as indicated in Figure 6 (see Middleton and Strick, 1994). Functional imaging during cognitive tasks together with tractography demonstrates cerebellar projections through motor and intralaminar thalamus to prefrontal cortex, supporting the role of these circuits not only in motor but also cognitive processing (Prevosto and Sommer, 2013). It has been proposed that dysfunction of cerebellothalamocortical circuits may underpin the manifest emotional and cognitive dysfunction in a range of neuropsychiatric conditions (Andreasen and Pearson, 2008; Konarski et al., 2005; Schmahmann, 2004).
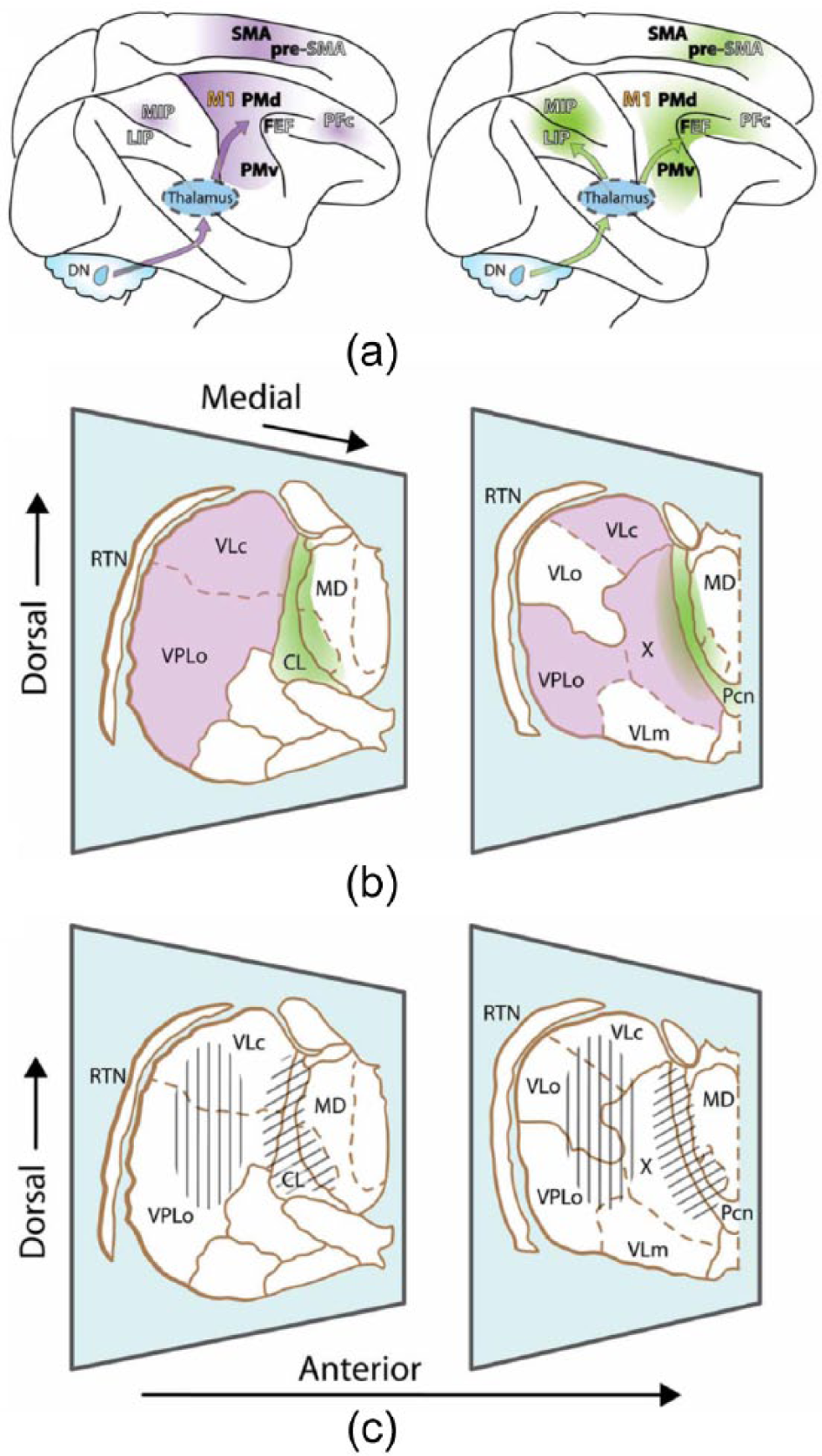
Cerebellothalamocortical circuits. (a) Lateral view of the rhesus monkey brain with hemispheres separated to expose mesial wall on top. Arrows schematically represent cerebello-cortical pathways relayed via cerebellar territories in lateral (left, violet) and central thalamus (right, green). (b) Two representative sections of the thalamus of Macaca mulatta, viewed from a lateral anterior perspective. Cerebellar domains in lateral and central thalamus are presented in violet and green, respectively. (c) Same sections as (b), but illustrating effector-related functional, as opposed to hodological, compartments. Hatching shows the rough somatotopic locations of regions related to arm (vertical) and eye (diagonal) movements.
As such, the thalamus may be considered a crucial hub in the network of subcortical structures that underpins significant intra- and inter-hemispheric circuit connectivity, known collectively as the subcortical connectome (see Looi et al., 2014). Therefore, further investigating the thalamus and its topographically distinct morphology is likely to yield important information across the broad spectrum of neuropsychiatric conditions.
Thalamic function and dysfunction
Classically, the thalamus has been referred to as ‘the gateway’ to the cortex as it was thought that the dorsal thalamus was limited to functioning as a relay of information; in this respect, it has been regarded as a group of cells that are concerned with passing information on to the cerebral cortex about the activity of either the periphery or other parts of the central nervous system. The thalamus has also been linked to many more functions, e.g., in transferring information between different cortical areas (Guillery, 1995), influencing states of arousal through its connections with the brainstem and basal forebrain (Steriade and Deschenes, 1984), sharpening or synchronization of input from the cortex (Sillito et al., 1994) and generating salience (Crick, 1984).
The current conceptualization of thalamic function is as a modulatory gate, with thalamic relay cells conveying the message from their driving afferent fibre (either cortical or subcortical) to the cortex. Transmission of the message occurs with the thalamic relay cell either in burst or tonic modes, without a significant change in the message from the driving afferent. The relay of information is influenced by a range of modulatory inputs, adjusting the way in which the information is relayed rather than altering the nature of the relayed information. One of the many but key challenges facing neuroscientists investigating the thalamus is to clarify precisely what benefit is obtained by sending information through the thalamus before it reaches the cortex. As postulated by Sherman and Guillery (2006), an advantage of higher order nuclei and transthalamic corticocortical communication could be that ‘the thalamus could serve as the organ of reflection of cortical activity back upon itself’; they argue that the role of the thalamus in perceptual processing is likely to be significant and speculate that a function of thalamocortical inputs to cortex (either from relay cells of first order or higher order thalamic nuclei) could be to keep the cortex updated on the most recent motor commands (Sherman and Guillery, 2006).
Taken together, when considering the role of the thalamus in neurological and psychiatric disorders, one could therefore consider dysfunction of the neural circuits involving the thalamus and a failure of modulatory gating, rather than neuropsychiatric symptoms somehow arising directly from the thalamus itself.
Neuroimaging the thalamus
Neuroimaging has been used as a means to visualize and measure the thalamus in vivo, and we propose that this can lead towards a quantitative morphology, otherwise known as morphometry.
Modalities utilized for imaging the thalamus
While a range of functional imaging modalities can be utilized to estimate thalamic function, structural imaging techniques have a higher spatial resolution than functional imaging methods, ideal for investigating diminutive subcortical structures. With the evolution of structural magnetic resonance imaging (MRI) modalities and advanced image analytic methods, more precise quantification of changes in the size and shape in subcortical structures such as the thalamus can be achieved. In terms of structural MRI, meta-analyses have reported thalamic atrophy in schizophrenia (Adriano et al., 2010) and major depressive disorder (Du et al., 2012), but not in bipolar disorder (Hallahan et al., 2011). Thalamic atrophy has been reported in both sporadic AD (De Jong et al., 2008) and the pre-symptomatic stage of familial AD (Lee et al., 2013; Ryan et al., 2013), as well as in progressive supranuclear palsy (PSP) (Whitwell et al., 2011), FTD (Cardenas et al., 2007; Chow et al., 2008) and HD (Kassubek et al., 2005).
Analytical methods (e.g. spherical harmonic point distribution method [SPHARM-PDM] [Styner et al., 2006]; radial distance mapping [Madsen et al., 2010]) measure the surfaces of neural structures using three-dimensional models (or mathematical ‘meshes’) allowing comparison of surface regions of a structure between diagnostic groups. These methods can expose statistically significant regional volume differences in the thalamus, which can provide important information regarding which underlying subdivisions or nuclei of the thalamus could be affected in the disease state being investigated (see McKeown et al., 2008).
Thalamic nuclei can also be defined by their patterns of thalamocortical connectivity (see Table 1); hence, diffusion tensor imaging (DTI) and tractography are useful modalities that enable one to visualize discrete white matter neural tracts, allowing the subdivisions and nuclei of thalamus to be imaged according to preselected cortical areas. DTI has been utilized to divide the thalamus into thalamocortical domains (Jakab et al., 2012), and this modality has the potential to yield specific localization of functional activations or lesions to putative thalamic nuclei. Given that most of the afferent and efferent fibres of the thalamus are mapped, DTI will be pivotal in further exploring the role of the thalamus in neurodegenerative disorders. Mapping of the thalamus within widespread networks will also be greatly facilitated by advances in resting state functional magnetic resonance imaging (fMRI), and better understand the distinct spatio-temporal courses of various neurodegenerative disorders (Zhou et al., 2012).
Furthermore, merging these newer techniques (e.g. generated statistical shape models and connectivity maps) and advanced modalities (e.g. either DTI or positron emission tomography [PET] and matching sophisticated MRI scanning and advanced analytical techniques) has the potential when correlated with clinical features to further enhance our understanding of various neuropsychiatric disorders (see McKeown et al., 2008; Whitwell et al., 2011). Hence, advances in neuroimaging modalities will increasingly enable researchers to more precisely quantify the disease effect on thalamic morphology.
Technical challenges
There are a number of challenges faced when attempting morphological analysis of the thalamus, namely, associated with thalamic boundary definition within the constraints of the resolution afforded by current imaging technologies. Defining the inferior boundary is particularly problematic along the intercommissural distance, and utilizing distinctive nuclei (such as the red nucleus and subthalamic nucleus) and white matter tracts as landmarks is imperative (Figure 1); the nuclei of the ventral thalamus (in particular the RTN and ZI) that partly encapsulate the dorsal thalamus throughout its rostral–caudal extent can also enhance inferior and lateral boundary definition (rather than being included in morphological analysis with the dorsal thalamus) (see Power et al., 2015) (Figure 1). The rostral pole of the dorsal thalamus is particularly challenging, and in order to exclude it from the adjacent hypothalamus and capture the full extent of this boundary, it has been suggested thalamic tracing should ideally occur with brain slices no greater than 1 mm thickness and proceed in a caudal to rostral direction (the caudal pole being readily identifiable as the pulvinar) (see Power et al., 2015). A number of automated approaches to mapping thalamic morphology, while having research utility as they can deal efficiently with a large numbers of brain scans, have thus been limited in their ability to precisely define thalamic boundaries (see Andreasen et al., 1994); studies utilizing automatic segmentation protocols often report larger mean thalamic volumes (Ryan et al., 2013; Zarei et al., 2010). Manual segmentation of the thalamus has therefore been utilized as the preferred approach, particularly in the setting of significant inter-individual variability of the thalamus and its component nuclei (see Spinks et al., 2002; Uylings et al., 2008).
A number of groups have published validated protocols for manual segmentation of the thalamus using structural MRI, however, the majority lack detailed information regarding precise boundary definition (see Power et al. (2015) for review). Many studies employ the approach published by Portas et al. (1998) who detailed an approach manual segmentation in 20–21 consecutive coronal 1.5 mm slices in a rostral–caudal direction, while providing a comprehensive description of adjacent landmarks, there is little information to guide manual tracing of the challenging inferior boundary and rostral pole, for instance. There are similar limitations in the protocol published by Spinks et al. (2002), involving 12 coronal 1.5 mm slices, however, an advancement of their technique was the utilization of three imaging modalities (T1, T2 and PD weighted images) and functions within the software package (BRAINS2) to assist with boundary definition. Our own group has recently published a protocol of manual segmentation using 28–30 consecutive 1 mm coronal slices using ANALYZE 11.0 software addressing many of the limitations and building on some of the advances of previous works (Power et al., 2015).
It is anticipated that advanced neuroimaging protocols for manual segmentation of the thalamus using structural MRI will drive the emergence of technologies towards more precise automated segmentation techniques of the human thalamus, which would improve efficiency in both the clinical and research setting. For the time being, further research is required in order to investigate the correlation of newer protocols with existing automated techniques. Whatever strategies emerge, they will need to be widely accessible, inexpensive (e.g. using software freely available on the web) and generalizable (i.e. use techniques that can be incorporated across a variety of software packages) in order for them to be relevant.
Thalamic morphology in neurodegenerative disorders
Conceptualization of neurodegenerative disorders on a neural circuit basis
Two key features characterize most age-related neurodegenerative disorders, the first being abnormal folding and accumulation of disease-specific proteins (sometimes termed ‘proteopathic’ agents) such as tau, beta amyloid or synuclein, and the second being progressive pathological change across the neuraxis (Braak and Braak, 1991; Braak et al., 2003; Deng et al., 2004; Kril and Halliday, 2011) (see Figure 7). The mechanism of disease ‘spread’, particularly by extant neuronal networks, has become an important research focus, on the rationale that by better understanding such mechanisms, one could potentially develop treatment modalities to halt the spread of disease throughout the brain.
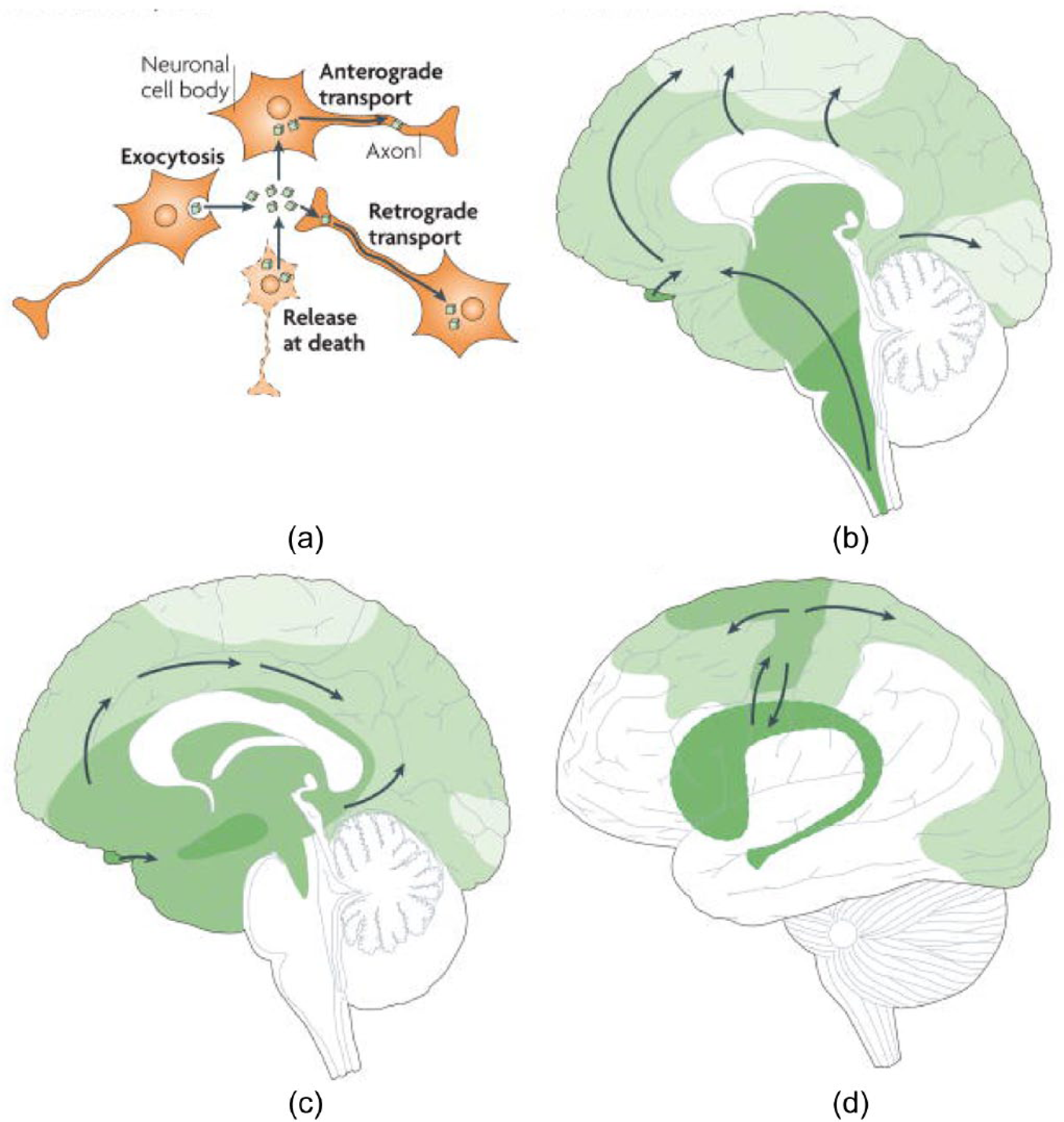
Conceptualization of neurodegenerative disorders on a neural circuit basis. (a) Intracellular protein aggregates can be released from neurons by exocytosis or cell death. The aggregates are taken up by, for example, adjacent neuronal cell bodies and are either retained in the cell soma (local spread of pathology) or transported anterogradely by axons. Alternatively, they are taken up by axon terminals and transported retrogradely to the cell soma. The protein aggregates can spread between-brain regions by axonal transport. (b–d) Three drawings propose principles for how neuropathological changes in Parkinson’s, Alzheimer’s and Huntington’s diseases spread spatiotemporally during disease progression. The earlier the neuropathology develops in a given brain region, the darker the shading in the diagram. As only one view (mid-sagittal for Parkinson’s and Alzheimer’s diseases; lateral for Huntington’s disease) of the brain is depicted for each disorder, not all relevant anatomical structures and details of the spreading patterns (indicated by arrows) are presented.
Stemming from advances in the understanding of the mechanism by which abnormal prion proteins lead to neurodegenerative processes (Aguzzi and Polymenidou, 2004; Prusiner, 1982), similar frameworks have been applied to investigating neurodegeneration in a range of disorders. Researchers have attempted to ‘seed’ proteopathic agents such as beta amyloid and tau in animal models, and various lines of evidence have suggested that spread of seeded protein aggregates occurs in distinct spatial (i.e. anatomical) and temporal patterns (for review, see Walker and LeVine, 2012). Researchers subsequently proposed that different neurodegenerative disorders have patterns of disease spread that is characteristic, with the clinical correlate being a distinctive clinical syndrome for that disorder (i.e. a characteristic temporal constellation of cognitive, emotional, behavioural and motor features) (see Brundin et al., 2010; Polymenidou and Cleveland, 2012). Following advances in methods such as graph theory to better understand neural circuitry (Bullmore and Sporns, 2009), and combined with structural and functional neuroimaging, a more recent conceptualization of neurodegenerative disorders on a neural circuit basis has emerged (Brundin et al., 2010) (see Figure 7). Researchers propose that proteopathic agents take seed in a specific part of the neuraxis and then spread throughout the brain’s intrinsic pathways over time, causing neural damage and/or dysfunction through a range of mechanisms in both cortical and subcortical structures (Raj et al., 2012; Seeley et al., 2009; Warren et al., 2013; Zhou et al., 2012).
Neuroimaging studies in the common neurodegenerative disorders have tended to focus on dysfunctional neocortical rather than subcortical circuits, the most parsimonious explanation being that neocortex dysfunction was thought to underpin the cognitive dysfunction that often presents early in disease. There is a burgeoning body of literature identifying the important role of subcortical structures in these circuits and contributing to cognitive disturbance in neurodegenerative disorders, such as the striatum in AD, behavioural variant FTD, progressive nonfluent aphasia (PNFA) and stroke (De Jong et al., 2008; Looi et al., 2009; Looi et al., 2012; Seeley et al., 2009; Zhou et al., 2012). To date, relatively few studies have investigated thalamic morphology in the neurodegenerative disorders; as a pivotal structure in the subcortical connectome, such investigations are likely to be fruitful (see Looi et al., 2014; Power et al., 2015; Power and Looi, 2014).
Thalamic morphology in healthy ageing
In a study of 57 healthy subjects ranging in age from 21 to 82 years who underwent neurocognitive testing (Stroop test and Pencil Memory Scanning Test) and structural MRI, a decrease in thalamic volume was reported across the lifespan (Van der Werf et al., 2001); this decrease in thalamic volume was reported to be independent of the decrease in total brain volume, and found to occur prior to it. Thalamic atrophy was found to start early in the lifespan and have a linear association with advancing age; the same study reported that thalamic atrophy correlated with diminished performance on tests of processing speed (Van der Werf et al., 2001). However, more normative data are needed regarding thalamic morphology across the lifespan.
Thalamic morphology in Alzheimer’s dementia
Thalamic atrophy on MRI has been reported in sporadic AD relative to controls (Callen et al., 2001; De Jong et al., 2008; Zarei et al., 2010). Furthermore, thalamic atrophy in AD is associated independently with cognitive deterioration using the Cambridge Cognitive Assessment–Revised (CAMCOG-R) and Mini-Mental State Examination (MMSE) in a cohort of memory clinic patients diagnosed with probable AD (n = 69) relative to those without objective memory deficits (‘memory complainers’, n = 70), when controlled for age, gender, educational level, intra-cranial volume and neocortical grey matter volume (De Jong et al., 2008). Combining shape and connectivity analysis in 16 patients with AD compared with 22 healthy controls, Zarei et al. (2010) reported bilateral atrophy in the regions of the MD nuclei of the thalamus (in regions connected with prefrontal and temporal cortex, and hippocampus), as well as bilateral atrophy in anterodorsal regions of the thalamus (which they attributed to possible internal medullary lamina atrophy). Their findings are, in part, consistent with reports of amyloid deposits and neurofibrillary tangles in the limbic nuclei of the thalamus, especially the MD and sub-regions of the AN, and involving the IL nuclei (Braak and Braak, 1991; Xuereb et al., 1991).
In familial AD, thalamic atrophy (Lee et al., 2013; Ryan et al., 2013) and increased fractional anisotropy of the thalamus bilaterally (Ryan et al., 2013) have been reported in the pre-symptomatic stage of disease, consistent with amyloid imaging studies demonstrating that amyloid deposition first occurs in the striatum and thalamus in pre-symptomatic cohorts (see Knight et al., 2011). Longitudinal studies of thalamic morphology in both sporadic and familial AD are warranted.
Thalamic morphology in Parkinsonian disorders
In the early stages of PSP and PD, these disorders can be difficult to distinguish, and definitive diagnosis is established by postmortem observation of characteristic histological abnormalities and regional pathology. While neuronal loss is seen postmortem in the intralaminar thalamic nuclei in both PSP and PD, degeneration in motor thalamus is reported in PSP, but not PD (Halliday et al., 2005; Henderson et al., 2000); the authors speculate that dysfunction of cerebellothalamic pathways in PSP may be an important distinguishing feature. Hence, the identification of distinguishing features on MRI between PSP and PD (and other parkinsonian disorders, such as corticobasal syndrome [CBS]) may improve diagnostic accuracy in vivo.
Thalamic atrophy on MRI has been reported in cohorts with PSP relative to controls, but not in patients with PD, multisystem atrophy, CBS or dementia with Lewy bodies (DLB) (Boxer et al., 2006; Cordato et al., 2005; Messina et al., 2011; Watson et al., 2009); it should be noted that at least some DTI data have suggested different topographical involvement of the thalamus in CBS relative to PSP (Erbetta et al., 2009; Hess et al., 2014), but these studies are difficult to interpret.
A number of multimodal studies, both cross-sectional and longitudinal, have reported involvement of particular thalamic nuclei in PSP. In a small cross-sectional study involving 14 patients with ‘mild’ PSP and 14 healthy controls undergoing DTI and structural MRI, Padovani et al. (2006) reported thalamic atrophy involving the pulvinar, MD and AN bilaterally. In a multimodal imaging study, 18 patients with probable PSP and 18 healthy controls underwent resting state fMRI, DTI and structural MRI; atrophy in the thalamus in the PSP group on MRI was reported, with reduced thalamic functional connectivity using fMRI, and DTI data implicating the VL (or motor) thalamus (Whitwell et al., 2011). Moreover, a PET and fMRI study in 16 patients with PSP suggested imbalance and falls correlated with thalamic dysfunction (Zwergal et al., 2011). In a longitudinal study by Whitwell et al. (2012), 16 patients with ‘probable’ PSP had serial MRIs and clinical measures (including the PSP rating scale) at 6 months and 12 months of follow-up; progressive thalamic atrophy over time was observed, however, no correlation between thalamic atrophy and clinical measures was observed.
Recent imaging data suggest there may be subtle nuances with regard to thalamic morphology in PD. Automated MRI data suggest left thalamic atrophy in PD with dementia compared to those with PD without dementia (Summerfield et al., 2005), and furthermore, using spherical harmonic-based representations shape (but not volume) changes have been reported in the thalami in PD (McKeown et al., 2008). McKeown et al. (2008) suggest these shape differences may represent selective non-dopaminergic degeneration reported in the caudal IL nuclei of the thalamus in PD (Henderson et al., 2000), and that detecting these subtle changes may be limited by the resolution of MRI (McKeown et al., 2008).
Thalamic morphology in other neurodegenerative disorders
Thalamic morphology has been investigated in a range of additional neurodegenerative disorders. Thalamic atrophy has been reported in patients with FTD compared with controls (Cardenas et al., 2007; Chow et al., 2008). Few studies, however, have investigated whether these morphological features characterize the spectrum of FTDs, and whether thalamic morphology could assist in delineating various subtypes of this entity. Rohrer et al. (2010) investigated 18 FTD patients who had antemortem MRI and postmortem confirmation of the TDP-43 subtype of FTD (positive immunoreactive inclusions), and using voxel-based morphometry reported that thalamic atrophy was useful in distinguishing FTD TDP-43 type 3 relative to types 1 and 2. Sharon et al. (2012) investigated patterns of brain atrophy in FTD patients who were carriers for chromosome open reading frame 72 (C9ORF72) expansions, and reported greater thalamic atrophy (left > right) in carriers vs non carriers of this expansion.
Thalamic atrophy on MRI has been reported in HD in the regions of MD, and the VL and IL nuclear groups bilaterally (Kassubek et al., 2005), consistent with postmortem findings (Heinsen et al., 1999). However, not all studies have reported thalamic atrophy in HD (Rosas et al., 2003). In a study with 44 HD patients (genetically confirmed) and 22 controls, using the Stroop colour word and Digit Symbol tests, thalamic atrophy correlated with cognitive impairment but not motoric dysfunction in the early clinical stages of disease (Kassubek et al., 2005).
Thalamic morphology in multiple sclerosis
Thalamic atrophy appears to be an important feature in multiple sclerosis (MS) (for review, see Minagar et al., 2013) and has been correlated postmortem with neuronal loss or altered cytoarchitecture in the thalamus, in particular in the MD and LGN (Cifelli et al., 2002; Vercellino et al., 2009). Thalamic atrophy has been reported in paediatric populations with MS (Kerbrat et al., 2012), and in adults with MS, it is one of the earliest and most prominent features of subcortical grey matter pathology when patients present with clinically isolated syndrome (CIS) (Calabrese et al., 2011). Thalamic atrophy has been demonstrated in the spectrum of MS subtypes and has been correlated with cognitive and functional impairment in MS (Batista et al., 2012; Houtchens et al., 2007; Mesaros et al., 2011). Multimodal longitudinal studies are warranted to elucidate the interplay of these findings in MS, and better define the utility of thalamic morphology in MS from a clinical perspective.
Towards thalamic endophenotypes and biomarkers in neurodegenerative disease
Towards endophenotypes and biomarkers
The concept of intermediate phenotypes (or endophenotypes) has been described as ‘measurable components unseen by the unaided eye along the pathway between disease and distal genotype’ (Gottesman and Gould, 2003). This concept has already been applied to the morphology of subcortical structures in neurodegenerative disorders, such as the striatum (Looi and Walterfang, 2012). As outlined, there is a growing body of literature reporting thalamic involvement across a broad range of neurodegenerative disorders. While further studies are required to ascertain whether genetic factors are associated with thalamic volume, there is evidence of distinct patterns of altered thalamic morphology in many neurodegenerative disorders, and some evidence of an association between morphology and clinical features in those disorders. Should further evidence converge suggesting that distinctive genetic, morphological and clinical features are associated with the thalamus, this may provide a basis for thalamic endomorphotypes. Leading on from this argument, investigating thalamic morphology (as a measurable characteristic of disease) therefore has the potential to yield neuroimaging biomarkers in the neurodegenerative disorders.
Potential utility of the thalamus as a morphological biomarker in neurodegenerative disease
Neuroimaging investigation of subcortical structures is likely to contribute knowledge regarding how the morphology (shape and volume) of structures, such as the thalamus, underpins the neural circuits sub-serving cognition, emotion, movement and behaviour. Utilizing thalamic morphology in neurodegenerative disorders has the potential not only to assist with diagnostic clarification, but is likely to contribute to better understanding the spatio-temporal course in various diseases. Furthermore, characterizing the disease effects on this strategic ‘hub’ in the subcortical connectome will enable neuroimaging to be utilized to map the onset, progression and, ultimately (when disease-modifying agents are accessible), response to treatment modalities in a form directly related to the neuropsychiatric symptoms and signs of disease (e.g. cognitive, emotional, behavioural and motor dysfunction).
Future directions and challenges
Further multifaceted studies will be required to advance the frontiers of knowledge of the subcortical connectome in order to better comprehend the complexity of brain disease on a neural circuit basis. With regard to the thalamus as a central hub in the connectome, additional studies are required to better understand the genetic basis of thalamic morphology, including processes of neurodevelopment, neurogenesis and apoptosis in the thalamus. Neuroimaging studies will need to investigate associations of thalamic morphology with a range of clinical markers in health and disease, including neurocognitive function, emotion and behaviour, and motoric function. Longitudinal studies are warranted to ascertain the temporal and spatial effects of disease processes on thalamic morphology, particularly in patient cohorts with well-established genetic vulnerabilities.
Given the challenges of thalamic boundary definition and variability of methods employed between neuroimaging studies, widely available and validated protocols for segmentation of the thalamus on MRI using sophisticated imaging techniques and brain analysis software will need to be established in order for morphological studies to progress and be reproducible. These protocols may well lead to more reliable automated segmentation techniques, in essence rendering automated techniques redundant in the future. Multimodal neuroimaging approaches will be essential (such as combining structural MRI, with DTI and shape analysis) in order to fully appreciate the impact of disease processes on different thalamic nuclei, and (through the well-established topographical relationships of the thalamus) better appreciate diseased brain circuitry.
In the Odyssey, Book 19 comes an early usage of thalamos: ‘ἡ δ’ ἴεν ἐκ θαλάμοιο περίφρων Πηνελόπεια, Ἀρτέμιδι ἰκέλη ἠὲ χρυσέῃ Ἀφροδίτῃ’, translated by Fagles (1996) (‘Now down from her chamber came reserved Penelope, looking for all the world like Artemis or golden Aphrodite’). Emerging from the chamber of thalamic morphology, we may come upon the vista of neural circuit dysfunction in neurodegenerative disorders and, given its particular relationship with cortex, perhaps it may be one of the most panoramic.
Footnotes
Acknowledgements
B.D.P. conceived and wrote the first draft of the manuscript, and is guarantor for this paper; J.C.L.L. co-conceived the work as the research network coordinator. All authors contributed to the final version of the manuscript. Given the restrictions on the number of references, only limited citations are provided on the section Basic and Clinical Neuroanatomy of the Thalamus.
Declaration of interest
The authors declare that there is no conflict of interest. The authors alone are responsible for the content and writing of the paper.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors. B.D.P.’s academic time is supported by the School of Medicine Fremantle, The University of Notre Dame Australia, and J.C.L.L.’s academic time by the Research Centre for the Neurosciences of Ageing, Academic Unit of Psychiatry and Addiction Medicine, Australian National University Medical School.
