Abstract
Objective:
Studies of overall cancer incidence and mortality in psychiatric patients have had mixed results. Some have reported lower than expected cancer incidence or mortality, while others have found no association or an increased risk depending on sample, psychiatric diagnosis, cancer site and methodology. Few studies have compared cancer incidence and mortality using the same population and methodology.
Method:
A population-based record-linkage analysis to compare cancer incidence and mortality in psychiatric patients with that for the general Queensland population, using an historical cohort to calculate age- and sex-standardised rate ratios and hazard ratios. Mental health records were linked with cancer registrations and death records from 2002 to 2007.
Results:
There were 89,992 new cancer cases, of which 3349 occurred in people with mental illness. Cancer incidence was the same as the general population for most psychiatric disorders. Rates were actually lower for dementia (hazard ratio = 0.77; 95% confidence interval = [0.67, 0.88]) and schizophrenia (hazard ratio = 0.84; 95% confidence interval = [0.72, 0.98]). By contrast, mortality was increased in psychiatric patients (hazard ratio = 2.27; 95% confidence interval = [2.15, 2.39]) with elevated hazard ratios for all the main psychiatric diagnoses.
Conclusions:
Lifestyle, such as alcohol or tobacco use, would not explain our findings that people with mental illness are no more likely than the general population to develop cancer but more likely to die of it. Other factors may be the difficulty in differentiating medically explained and unexplained symptoms, greater case fatality or inequity in access to specialist procedures. The study highlights the need for improved cancer screening, detection and intervention in this population.
Introduction
The physical health of psychiatric patients has attracted increasing attention over the last 10 years with around 85% of the excess mortality related to natural causes in contrast to 15% to suicide and other unnatural causes (Lawrence et al., 2013). To date, most attention has been on cardiovascular risk factors rather than other causes such as cancer. This is surprising given that people with mental illness engage more frequently in behaviours that are associated with cancer risk. This includes higher rates of smoking, alcohol and substance use (Kisely et al., 2000; Regier et al., 1990). In addition, a common side effect of antipsychotic medications is hyperprolactinaemia, which, in turn, is associated with an increased risk of breast cancer in women and possibly prostate cancer in men (Tworoger et al., 2007).
In spite of these risk factors, studies of overall cancer incidence and mortality in psychiatric patients have had mixed results. Some authors have reported lower than expected cancer incidence or mortality in psychiatric patients, depending on site, while others have found no association (Black and Winokur, 1986; Kisely et al., 2008, 2013; Lawrence et al., 2000a; Osborn et al., 2007, 2013; Saku et al., 1995). Still others have found an increased risk of incidence or mortality (Brown et al., 2000; Kisely et al., 2008; Lichtermann et al., 2001). Within Australia, two population-based studies of overall mortality in psychiatric patients also found conflicting results (Drew, 2005; Lawrence et al., 2000b). One from Western Australia reported that mortality from chronic physical illness, including cancer, was significantly greater (Lawrence et al., 2000b), while another from the Australian Capital Territory (ACT) found no association other than for suicide (Drew, 2005).
Some of this discrepancy may be explained by differences between psychiatric diagnoses. Schizophrenia has particularly been associated with a reduced incidence of cancer such as melanoma and prostate cancer (Barak et al., 2005; Cohen et al., 2002; Dalton et al., 2005; Grinshpoon et al., 2005; Mortensen, 1994). These results were confirmed by a recent meta-analysis of cancer incidence rates in patients with schizophrenia that combined 16 data collections from 15 studies (Catts et al., 2008). However, these findings have not been universal. For example, a database study from Israel described a mixed picture with possible increased rates of breast cancer and respiratory cancer in some groups of people with schizophrenia (Grinshpoon et al., 2005). Another from the United Kingdom found that people with schizophrenia had statistically significantly increased risks of incident breast cancer and colon cancer (Hippisley-Cox et al., 2007). In the case of bipolar or affective disorder, three studies found an increased risk of cancer (BarChana et al., 2008; Carney and Jones, 2006; Ösby et al., 2001) while another three did not (Lawrence et al, 2000a; Levav et al., 2009; Osborn et al., 2007). There are fewer studies that consider a broader range of other psychiatric disorders.
The use of different methodologies and cancer outcomes may also account for some of the conflicting results (Bushe and Hodgson, 2010). Some studies are restricted to patients in hospital. The reporting of cohort age at study conclusion may be an additional critical confounder. Populations where patients are not followed up to an age when cancer incidence and mortality is maximal may underestimate cancer mortality and incidence, making standardised mortality ratios (SMRs) complex to interpret. Differences in study power may also be a factor in the differing results from Western Australia (Lawrence et al., 2000b) and the ACT (Drew, 2005).
As previously noted, psychiatric diagnoses other than schizophrenia or affective disorder have been studied in less detail and few studies have compared the rates of different diagnoses using the same population and methodology (Lawrence et al., 2000a; Kisely et al., 2008, 2013). One example was a study of an inception cohort showing how psychiatric patients in general were more likely to have metastases at diagnosis, and less likely to receive specialised interventions (Kisely et al., 2013). There was also a reduced incidence of cancer in patients with dementia and schizophrenia (Kisely et al., 2013). However, this study was from a jurisdiction of only 2.5 million inhabitants, and therefore limited by small cell sizes for certain cancers and psychiatric diagnoses. As a consequence, the study may have been under-powered to detect some differences in, say, rarer cancer sites. This limitation is especially relevant given that overall cancer incidence may not be the correct metric as there are numerous cancer types, each with their own causes (Bushe and Hodgson, 2010). Rather than measure cancer as an entity, it may be more appropriate to consider individual cancer types (Bushe and Hodgson, 2010).
We therefore undertook a study of data from Queensland, which at 4.7 million inhabitants, has a population that is almost double that of Western Australia.
Method
This was a population-based record-linkage analysis from Queensland, using a historical cohort to calculate rate ratios (RRs). Ethics approval was received from the relevant University and Queensland Health Human Research Ethics Committees. We used administrative data on mortality, inpatient, outpatient and community psychiatric contacts to identify psychiatric patients with cancer registrations, admissions or death records (Table 1).
Queensland data sources.

QHPADC: Queensland Hospital Admitted Patients’ Data Collection; CESA: Client Event Services Application.
We used the case definition for psychiatric disorder validated by the Public Health Agency of Canada (PHAC) for use in administrative databases (Kisely et al., 2009b). This is any contact with a health service with an International Classification of Diseases (ICD)-9 diagnosis of 290–319 or ICD-10 equivalent. We also included post-partum mental disorders (ICD-9 648.4), suicide and non-accidental injury (ICD-9 E950–959), as well as contact with the mental health system not covered by the above ICD-9 Chapter 5 diagnoses to ensure comparability with previous work (Kisely et al, 2013).
Data covering all the administrative databases were only available from 2002. Patients were censored at the occurrence of the event under study, or 31 December 2007. We compared their outcomes to the general population through the calculation of age- and sex-standardised rates, which were then adjusted for confounders using multivariate analyses (see section ‘Statistical methods’).
Data quality
This can be assessed through the precision of the disease estimate, the control of confounders and the degree of selection and information bias (Mortensen, 1995). In this study, the precision of the disease estimate was maximised through the size of the databases, which cover 4.7 million residents. It was also maximised through coverage of all public-sector inpatient, outpatient and community contacts within Queensland Health. Information bias was reduced through the use of aliases and phonetic spelling in probabilistic linkage protocols to minimise linkage failures due to name changes and spelling variants. Cross linkage with other databases in the system further aided the identification of identity errors. In terms of data quality checks, demographic features such as indigenous status were correctly identified in about 89% of cases when checked with other sources. In addition, a manual review of records identified as having ‘definite links’ using a software package (ChoiceMaker) found only 0.28% false matches. Finally, linkage of different databases covering the same population increased the chance that variables missing in one database may be available in another, thereby increasing the ability to control for confounders.
Classifications of mental disorders and cancers
Among the psychiatric cases, we used a hierarchy of last available versus earlier diagnoses, and inpatient versus outpatient care, reflecting increasing data reliability. A principal psychiatric diagnosis was assigned to each patient who had contact with mental health services using the following procedure. The final diagnosis in a care episode was taken to allow for revision of preliminary diagnoses during a period of observation or treatment. The last occurring psychiatric diagnosis across the episodes was then assigned as the principal diagnosis according to a diagnostic hierarchy. If an earlier diagnosis was higher in the hierarchy than the last recorded diagnosis, the earlier diagnosis was taken as the principal diagnosis. The hierarchy gave precedence to organic and psychotic disorders, allowed for conditions such as substance dependence to be considered as potential comorbidities and then gave preference to conditions within Chapter 5 in ICD-9, or equivalent. Non-specific disorders outside Chapter 5 in ICD-9, or equivalent, came last.
Preference was given to diagnoses made in inpatient treatment units over diagnoses from outpatient clinics or psychiatric residential units. The most recent inpatient diagnosis took precedence over the most recent outpatient diagnosis even if the admission predated any outpatient contact. These procedures were designed to allow more specific psychiatric diagnoses to take precedence over less specific diagnoses, and to favour an underlying condition rather than a non-specific symptom or event.
Cancers were classified using the ICD-O classification of diseases at the three-digit level. This follows the same system used for classifying cancers by the Cancer Registry. The specific sites selected for further analysis were the most frequent cancers in males and females.
Statistical methods
We initially calculated the age- and sex-standardised rates per 100,000 person years for mortality and cancer incidence for anyone meeting the case definition of psychiatric disorder (Kisely et al., 2009b).
Five-year age groups were used up to age of 85 years and above. The latter were considered as a single group because of small numbers. The directly standardised rate represented what the crude rate would have been in the study population if it had the same distribution as the standard population with respect to the variables for which standardisation was carried out (Last, 1988). This approach allowed comparisons of mortality and cancer incidence given each group was adjusted to the same standard. However, recognising that direct standardisation can become unstable when working with very small cell sizes, we did sensitivity analyses of the effect of using indirect standardisation. In this procedure, the specific rates in the standard population are averaged, using as weights the distribution of the study population (Last, 1988).
Rates of cancer incidence and mortality for the non-psychiatric population were calculated in the same way, using the file of all cancer registrations from July 2002 to December 2007. Denominators were taken from estimated resident population data.
RRs were then calculated to compare the age- and sex-standardised rates for patients with mental health service contact prior to cancer diagnosis, with those in the general population who did not have psychiatric contact. Rates were also calculated for each cancer site. In addition, we calculated RRs for each psychiatric diagnosis and for cases that had ever been inpatients.
Regression models
Cox proportional hazards regression was used to examine risk factors for cancer among psychiatric patients. Results were expressed as hazard ratios (HRs), risk periods being calculated in the same manner as for the analysis of rates. Factors in the model were principal psychiatric diagnosis, age, sex, residential location, cumulative length of stay in inpatient care and socio-economic status. As in previous work, socio-economic status was assigned to the collection district (CD) or residential postcode of each patient using the ‘Socioeconomic Indices for Areas’ (SEIFA) produced by the Australian Bureau of Statistics (ABS) (Lawrence et al., 2000a). CDs, to which over 80% of addresses in the study were geocoded, were used in preference given they are the smallest geographical area used by the ABS. They therefore have greater validity than postcodes. SEIFA gave a ranking to each CD or postcode based on census data.
Proportional hazards regression was also used to examine case fatality, adjusting for the same variables as incidence. In this model, all patients, both psychiatric and non-psychiatric, who had a cancer diagnosed between July 2002 and December 2007, were included. Risk commenced at the date of cancer diagnosis, and patients were censored at death or 31 December 2007.
For all the analyses, we ran separate models for psychiatric diagnosis and severity (ever an inpatient or outpatient only).
Results
Incidence
There were 89,992 new cases of cancer, of which 3349 occurred in people with mental illness. The psychiatric patients who developed cancer had a median age of 67 years (interquartile range [IQR] = 54–79 years) compared with a median age of 65 years (IQR = 55–75 years) among cancer patients with no psychiatric history. Totally, 1879 (56%) were male. Patients with cancer who had been in contact with mental health services were more likely to live in rural districts or in areas of greater socio-economic disadvantage than those who had not received psychiatric treatment (Table 2).
Place of residence and socio-economic status of patients with cancer with and without contact with mental health services.
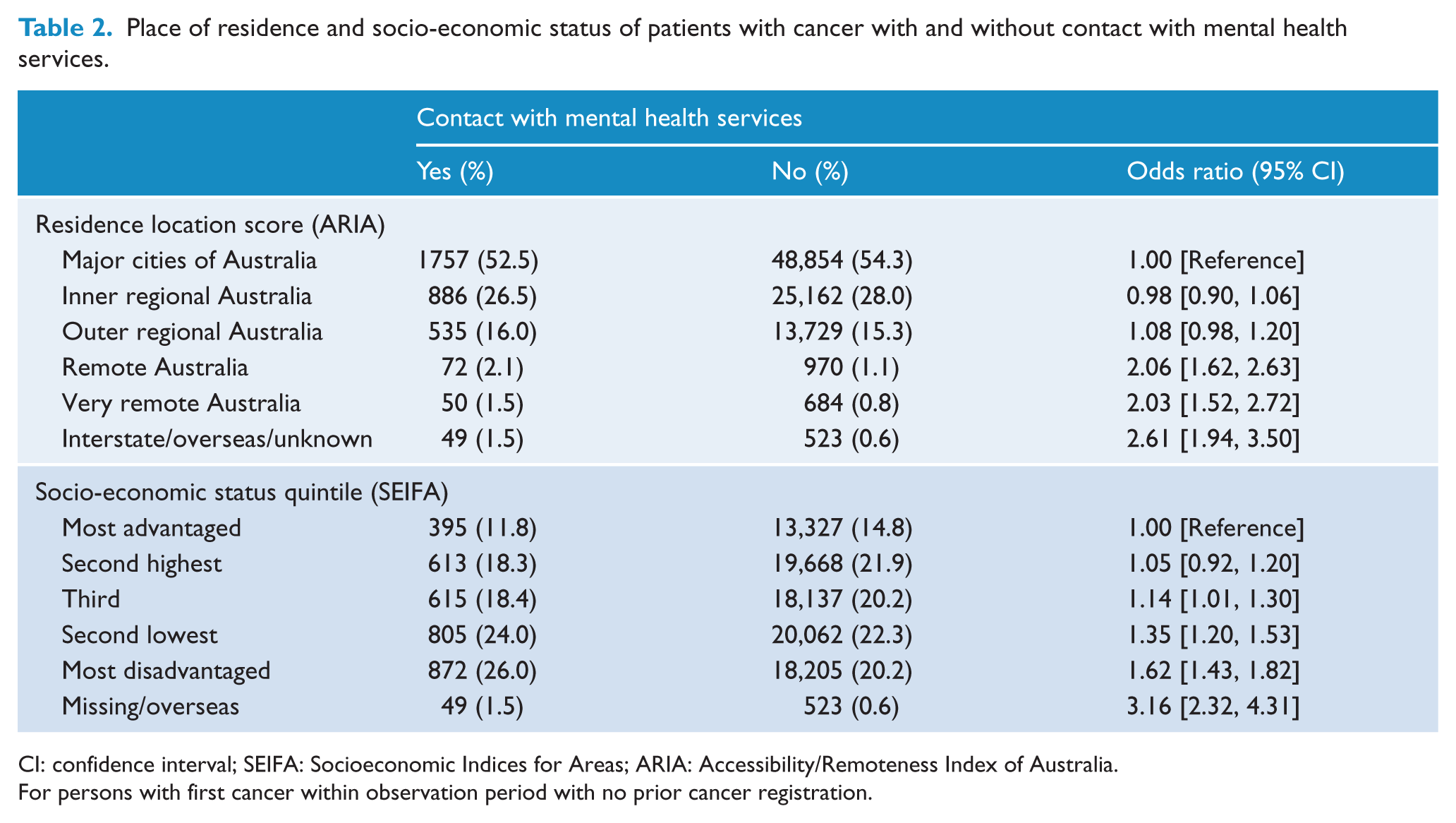
CI: confidence interval; SEIFA: Socioeconomic Indices for Areas; ARIA: Accessibility/Remoteness Index of Australia.
For persons with first cancer within observation period with no prior cancer registration.
Overall cancer incidence for psychiatric patients was lower than for the general population (Table 3). This was also true for the main cancer sites of prostate, breast, colorectal and melanoma, as well as cancers of the uterus, thyroid and urinary tract (Table 3). By contrast, the incidence was greater for cancer of the lung, mouth oesophagus and liver, as well as lymphoma. We found a similar pattern for those cancers that are not specific to either sex when males and females were considered separately. For the purposes of this analysis, breast cancer was considered to be specific to women although it can occur rarely in males.
Cancer incidence and mortality ratios for mental health patients compared to the general population (age-adjusted).
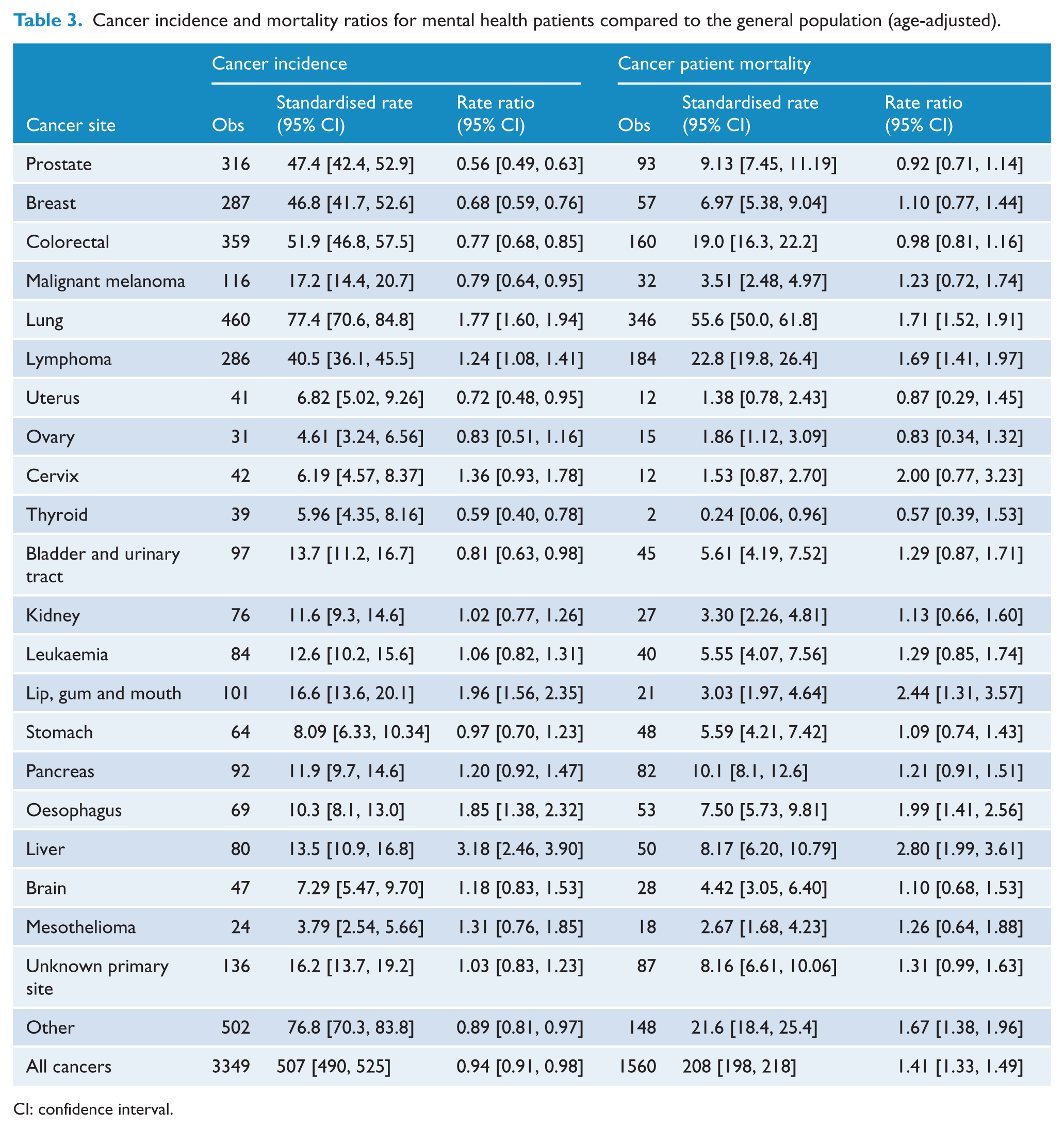
CI: confidence interval.
In terms of specific psychiatric diagnoses, cancer rates were generally the same as for the general population (Table 4). The only exceptions were that males and females with alcohol or drug disorders had a greater cancer incidence than the general population (Table 4), while females with depressive disorders had a lower incidence (RR = 0.84; 95% CI = [0.69, 0.99]). Patients of both sexes with ‘other mental disorders’ also had a lower incidence (Table 4).
Cancer incidence and mortality by most severe psychiatric disorder (age-adjusted).
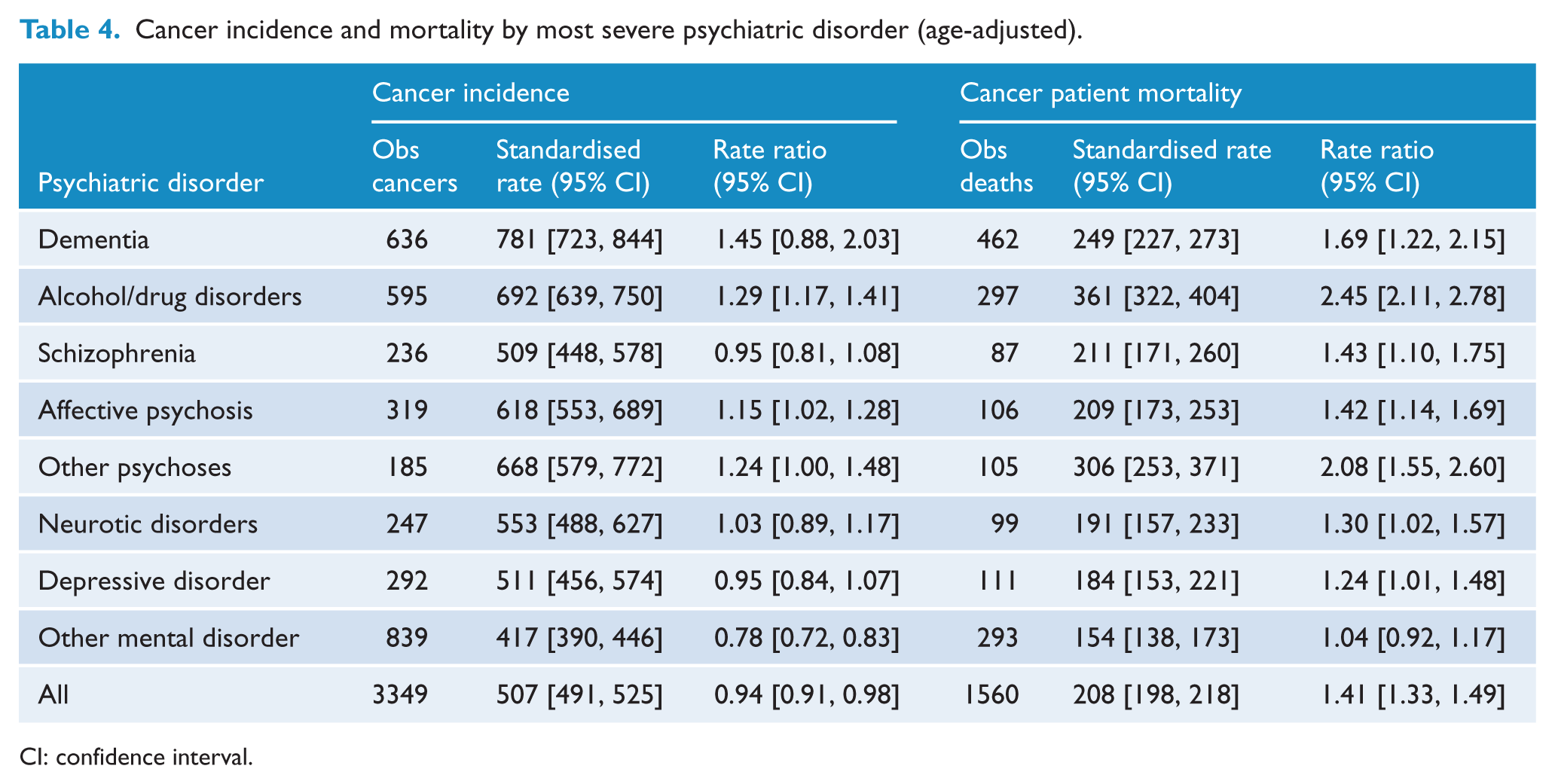
CI: confidence interval.
Table 5 presents the results of the Cox regression. The HR measures the association between being diagnosed with cancer and each variable in the table (e.g. contact with psychiatric services), taking into account person-time and the effect of the other variables. The risk of cancer was higher in male than female psychiatric patients (HR = 1.28; 95% CI = [1.19, 1.37]), and cancer risk increased with age (Table 5). We found no significant association of cancer incidence with socio-economic status. Patients with dementia had a reduced risk of cancer as did those with schizophrenia (Table 5). By contrast, patients with alcohol or drug disorders had an increased risk (Table 5). Similarly, patients who had spent any time as an inpatient had a greater risk ofcancer incidence compared with those treated as outpatients only (Table 5).
Proportional hazards regression of the risk of being diagnosed with cancer for mental health patients.
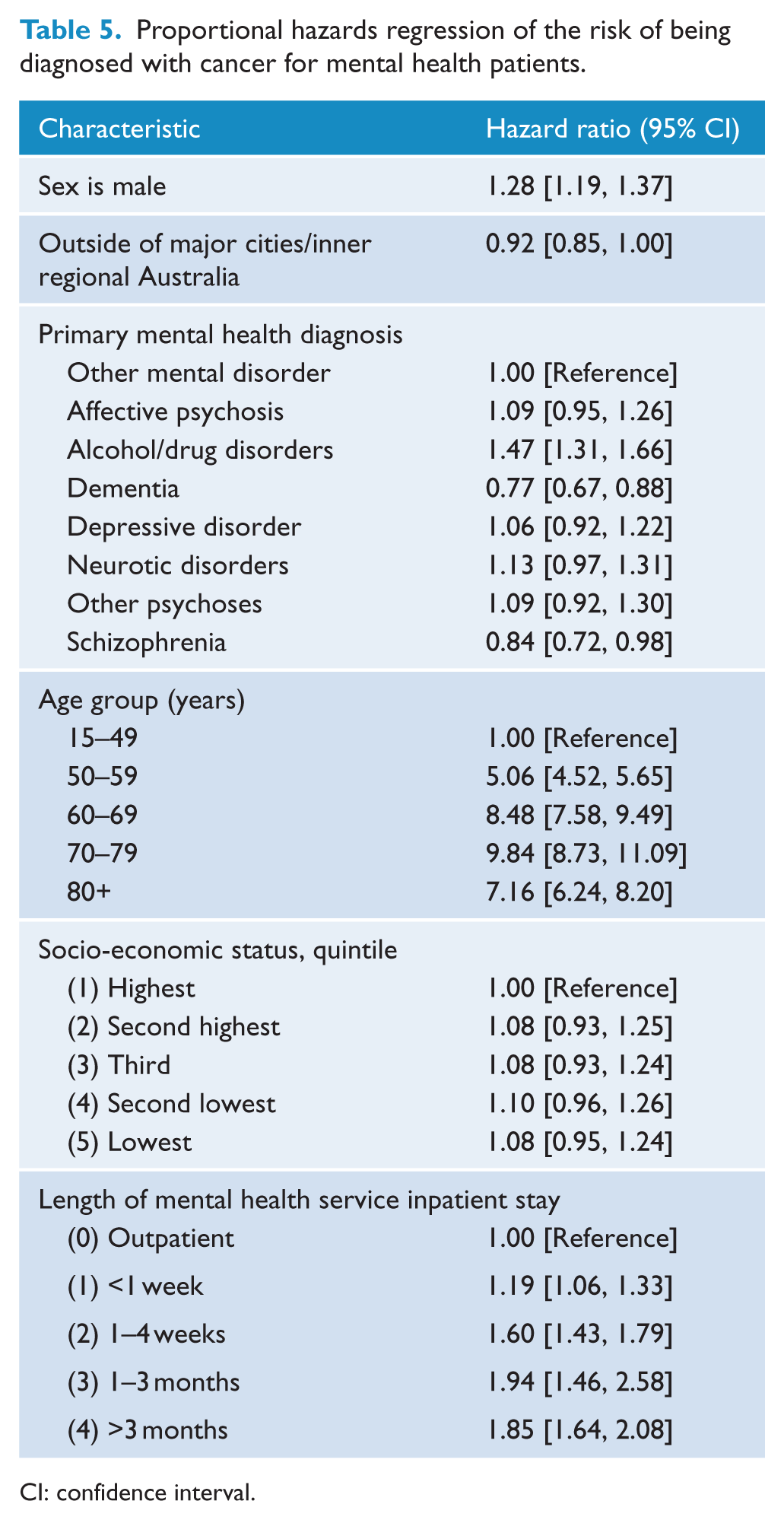
CI: confidence interval.
Mortality
In all, 1560 psychiatric patients diagnosed with cancer died during the study period. This rate was 41% higher than that of the general population (Table 3). Cancer patient all-cause mortality was elevated in both male (RR = 1.42; 95% CI = [1.32, 1.52]) and female psychiatric patients (RR = 1.39; 95% CI = [1.27, 1.51]). We found similar results when we considered cancer-specific mortality (RR = 1.31; 95% CI = [1.23, 1.40]). As for incidence, mortality rates were highest for cancer of the lung, mouth, oesophagus, liver and lymphoma (Table 3). Patterns for males and females were similar.
In terms of specific psychiatric diagnoses, cancer mortality rates were elevated in all diagnoses except ‘other mental disorders’ where the rate was the same as for the general population (Table 4). Patterns for males and females were again similar.
On Cox regression, psychiatric patients had a significantly higher mortality risk (HR = 2.27; 95% CI = [2.15, 2.39]) (Figure 1). Male gender, older age and greatersocio-economic disadvantage were associated with higher all-cause mortality (Table 6). The highest risk for all-cause mortality occurred in patients with alcohol and drug disorders, dementia, schizophrenia and other psychoses, as well as those who had ever been inpatients (Table 6). There was a similar pattern for cancer mortality.
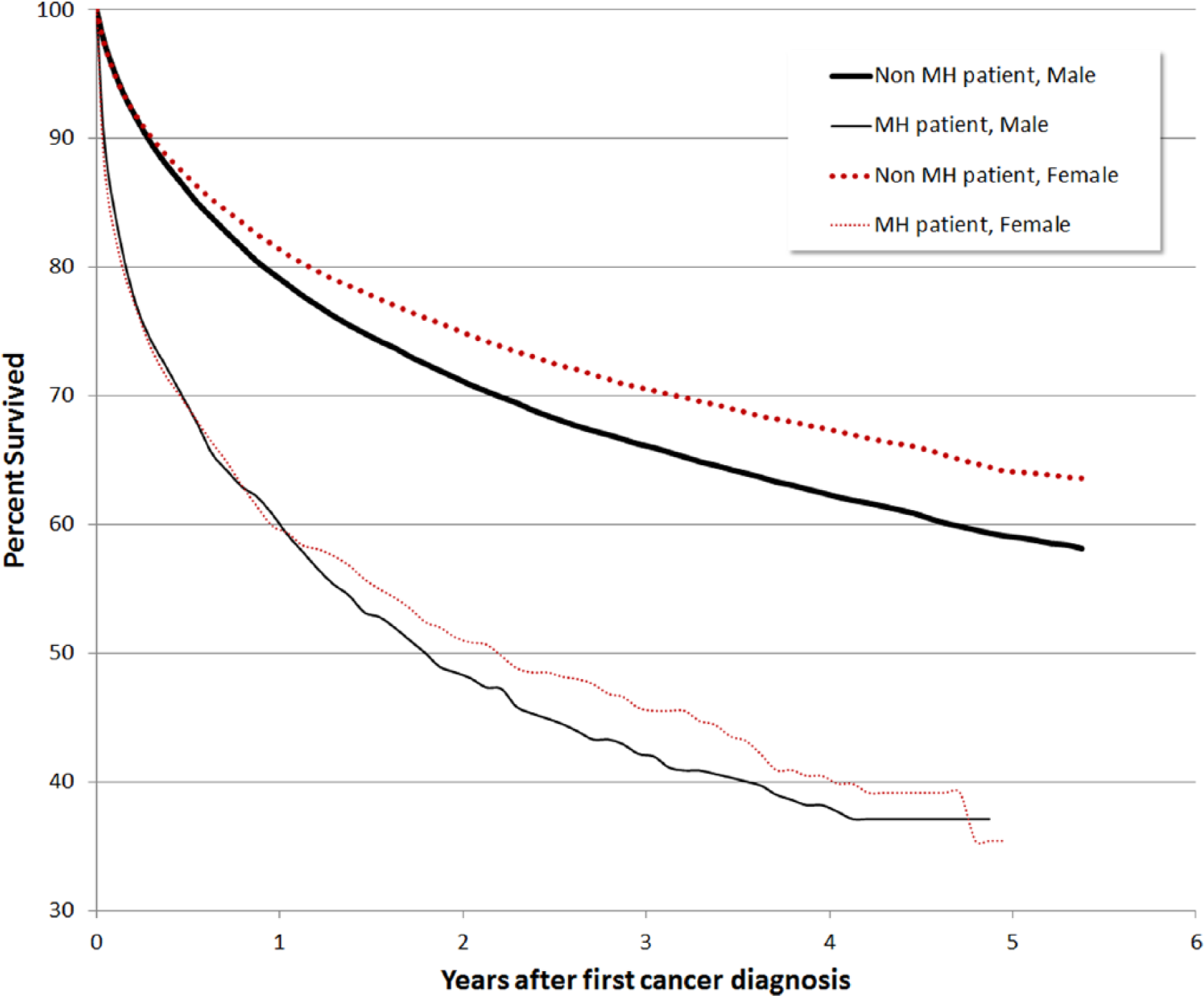
Survival since diagnosis, all cancers, by contact with mental health services.
Proportional hazards regression of mortality risk for cancer patients.
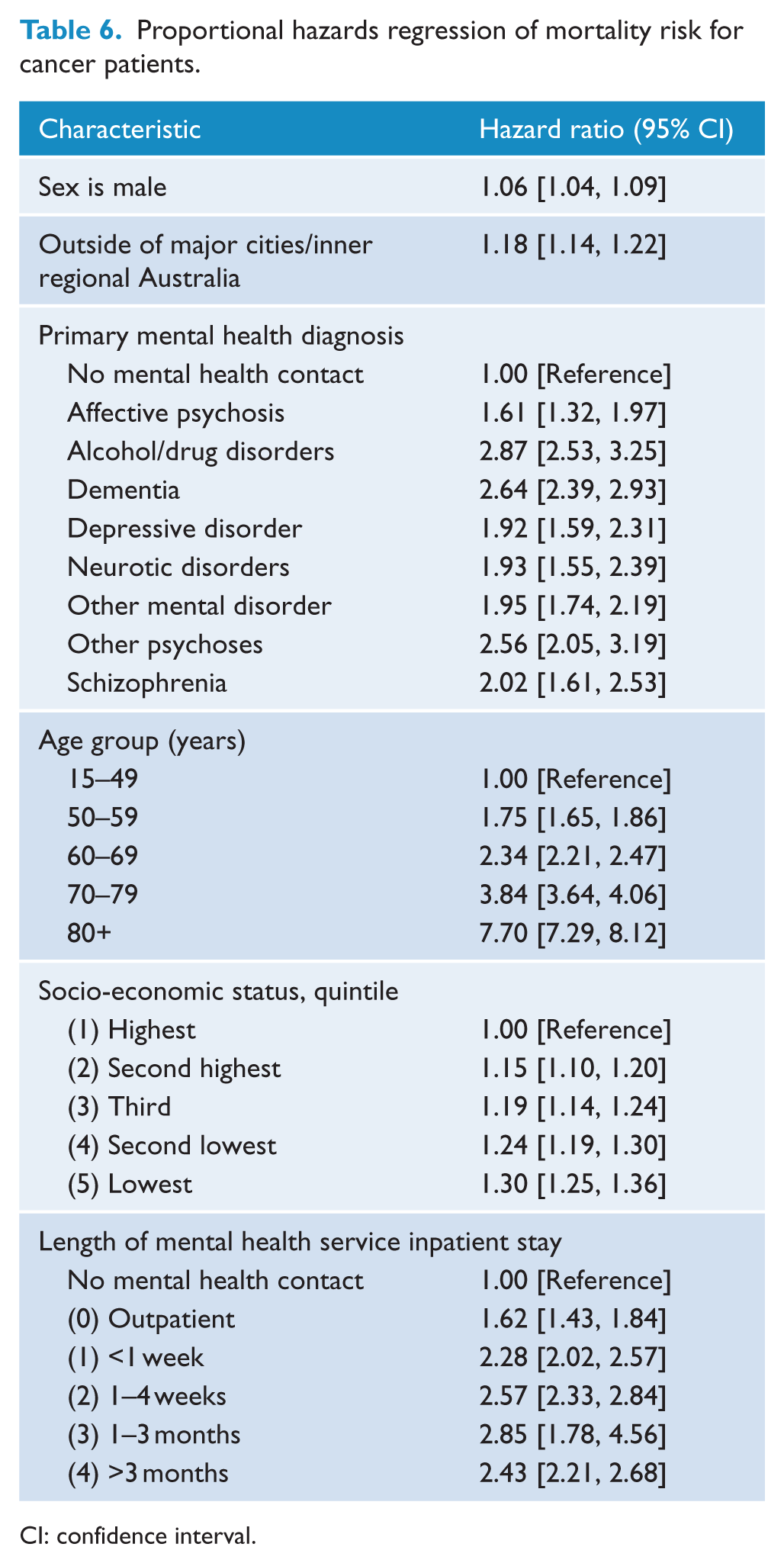
CI: confidence interval.
We found similar results when using indirect as opposed to direct standardisation. For instance, the indirectly standardised incidence rate for all cancers was 0.83 (95% CI = [0.81, 0.86]) and for mortality it was 1.34 (95% CI = [1.28, 1.41]).
Discussion
There are conflicting data from Australia on whether people with psychiatric disorder have increased mortality from physical illness. Some of the difference has been ascribed to the use of an inception cohort in Western Australia and an historical cohort in the ACT (Drew, 2005; Lawrence et al., 2000b). This is because inception cohorts of psychiatric cases presenting to services for the first time, as opposed to historical cohorts of prevalent cases, may be more sensitive to the increased mortality risk faced by patients given evidence that much of the excess mortality occurs within the first 7 years after contact with mental health services (Lawrence et al., 2005). Our study suggests that irrespective of sample (historical or inception cohort), psychiatric patients have increased mortality from chronic physical illnesses such as cancer. Differences in the experiences of people with mental illness between Australian jurisdictions may therefore be real and so warrant further exploration.
This study is also one of the few studies to assess both cancer incidence and mortality using a standardised methodology in the same population. In some respects, it found a similar pattern to Western Australia and Canada in that mortality from cancer was higher in psychiatric patients, but cancer incidence no greater, and sometimes lower, than that in the general population (Kisely et al., 2008; Lawrence et al., 2000a). However, in other respects, there were differences especially when considering individual cancer sites. In the other two jurisdictions, the highest mortality RRs were for cancer of the brain in both sexes, cancer of the prostate or of the urinary tract in males, and cancers of unknown primary site in males and females (Kisely et al., 2008; Lawrence et al., 2000a). However, in the case of Queensland, both incidence and mortality were highest for cancer of the lung, mouth and oesophagus, liver and lymphoma. A possible explanation is either alcohol or tobacco use. However, tobacco use cannot be the sole reason for the difference in results with Western Australia as there is no evidence that smoking among people with mental illness varies across Australia (Cooper et al., 2012; Morgan et al., 2011). Other possibilities include differences between jurisdictions, for which we could not adjust such as population distribution, the proportion of indigenous residents and access to, or use of, health services.
Limitations
Ethnicity, marital status, education level, comorbidity and disability are not recorded on the cancer registry and so could not be included in our models. There was also no information on medication. Although we adjusted for socio-economic status, our results may not be generalisable to people who receive private psychiatric treatment, especially outpatients. In addition, socio-economic status was assigned by residential CD or postcode rather than individual-level information. In addition, country of birth was not included in this linked dataset.
Administrative data may also be subject to recording bias, especially for diagnosis and particularly in secondary fields. We therefore emphasised overall psychiatric morbidity, not sub-categories or secondary diagnoses, to minimise possible bias. We were unable to study the effects of lifestyle such as diet, smoking or alcohol and substance use. It is therefore possible that incidence rates for some cancers that are related to lifestyle may have been confounded by the absence of these data. However, it is likely that overall cancer incidence might have been still lower had we been able to adjust for lifestyle for these cancers and so, if anything, this would strengthen our conclusion that people with psychiatric disorders are no more likely to develop cancer but more likely to die of it. Finally, usable data covered fewer years and therefore fewer subjects than anticipated, and only extended to 2007. However, in the case of the latter, evidence from mortality rates for other physical disorders such as ischaemic heart disease suggests that the gap in life expectancy between the psychiatric and general population has increased over time (Lawrence et al., 2003, 2010). If this were also true for cancer, any bias would be to underestimate any difference between psychiatric patients and community controls.
Implications
There may be several explanations for the increased case fatality in people with mental illness. One might be reduced access to screening services. A narrative review of 17 studies showed a 20–30% reduced likelihood of breast, cervical and colorectal cancer screening for patients with mental illness in the majority of studies, with a 60% reduced likelihood for those with severe mental illness (Happell et al., 2012). However, these findings may be less relevant for this study given that the greatest differences were for cancers that are not commonly the subject of screening. There may also be delays in the time it takes to be diagnosed with cancer after presenting with symptoms. In a study of patients with oesophageal cancer, O’Rourke et al. (2008) found that cancer patients who had a Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) diagnosed psychiatric comorbidity waited a median of 90 days between reporting symptoms and receiving a diagnosis, compared to 35 days for patients without mental illness. They were also more likely to have advanced disease at the time of diagnosis (37% vs 18%). These results may be particularly relevant for our finding of especially high mortality rates for oesophageal cancer in people with mental illness. This is consistent with other work on the presence of metastases at presentation that showed statistically significant but rather small differences in absolute terms between people with and without psychiatric disorder (Kisely et al., 2013). However, such differences are unlikely to be the sole explanation, findings reinforced by another recent study suggesting that the elevated mortality associated with cancer is unlikely to be primarily the result of the cancer being more advanced by the time they are diagnosed (Chang et al., 2014).
Following diagnosis, psychiatric patients have a reduced likelihood of surgery after diagnosis for all types of cancer, and for those who do have surgery, the length of time between cancer diagnosis and surgery is longer. For example, men are less likely to have a colorectal resection, and women are less likely to have surgery for colorectal, breast and cervical cancer even after adjusting for the presence of metastases at presentation. They are also less likely to receive surgical therapy for oesophageal cancer (38% vs 59%) (O’Rourke et al., 2008). These findings are consistent with documented disparities in access to cardiovascular treatments faced by people with mental illness (Kisely et al., 2007, 2009a).
Our results may therefore indicate greater action to improve cancer outcomes of psychiatric patients. Because of the complex, multifactorial nature of the association between psychiatric disorder and cancer, multipronged approaches will be required to tackle these inequalities, in the same way that multipronged approaches have been used to reduce the mortality associated with cancer in the general population. These strategies should include both individual and population-based components. Given findings that 72% of excess deaths occur in patients who have only ever seen their family doctor for their psychiatric problems (Kisely et al., 2005), collaborative arrangements with primary care may be helpful. One example is Western Australia’s strategy to improve the physical health of people with severe mental illness (Healthright) (Stanley and Laugharne, 2011). This strategy engages the public mental health system, general practice and specialist medical and psychiatric services in the physical health care of people with psychiatric disorders.
Footnotes
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Funding
The research reported in this paper was supported by funding from a grant from Cancer Council Queensland (grant number 631521). The data used for this study were made available through the Population Health Research Network funded by the Australian Government’s National Collaborative Research Infrastructure Strategy.
