Abstract
Objectives:
This clinical practice guideline for treatment of DSM-5 feeding and eating disorders was conducted as part of the Royal Australian and New Zealand College of Psychiatrists (RANZCP) Clinical Practice Guidelines (CPG) Project 2013–2014.
Methods:
The CPG was developed in accordance with best practice according to the National Health and Medical Research Council of Australia. Literature of evidence for treatments of anorexia nervosa (AN), bulimia nervosa (BN), binge eating disorder (BED), other specified and unspecified eating disorders and avoidant restrictive food intake disorder (ARFID) was sourced from the previous RANZCP CPG reviews (dated to 2009) and updated with a systematic review (dated 2008–2013). A multidisciplinary working group wrote the draft CPG, which then underwent expert, community and stakeholder consultation, during which process additional evidence was identified.
Results:
In AN the CPG recommends treatment as an outpatient or day patient in most instances (i.e. in the least restrictive environment), with hospital admission for those at risk of medical and/or psychological compromise. A multi-axial and collaborative approach is recommended, including consideration of nutritional, medical and psychological aspects, the use of family based therapies in younger people and specialist therapist-led manualised based psychological therapies in all age groups and that include longer-term follow-up. A harm minimisation approach is recommended in chronic AN. In BN and BED the CPG recommends an individual psychological therapy for which the best evidence is for therapist-led cognitive behavioural therapy (CBT). There is also a role for CBT adapted for internet delivery, or CBT in a non-specialist guided self-help form. Medications that may be helpful either as an adjunctive or alternative treatment option include an antidepressant, topiramate, or orlistat (the last for people with comorbid obesity). No specific treatment is recommended for ARFID as there are no trials to guide practice.
Conclusions:
Specific evidence based psychological and pharmacological treatments are recommended for most eating disorders but more trials are needed for specific therapies in AN, and research is urgently needed for all aspects of ARFID assessment and management.
Expert reviewers
Associate Professor Susan Byrne, Dr Angelica Claudino, Dr Anthea Fursland, Associate Professor Jennifer Gaudiani, Dr Susan Hart, Ms Gabriella Heruc, Associate Professor Michael Kohn, Dr Rick Kausman, Dr Sarah Maguire, Ms Peta Marks, Professor Janet Treasure and Mr Andrew Wallis.
Introduction
This guideline for the clinical management of eating disorders is a project of the Royal Australian and New Zealand College of Psychiatrists (RANZCP). The guideline represents the work of a core working group of health care academics and professionals and wide consultation with key stakeholders and the community.
The guideline is intended to provide current evidence based guidance on the assessment and treatment of people with eating disorders by psychiatrists and other health professionals in the Australian and New Zealand context and includes identifying further research needs. It is written with reference to other international guidelines such as those of the American Psychiatric Association and the United Kingdom National Institute for Health and Care Excellence guidelines and is intended to address both broad but also specific issues, such as those relevant to Māori and Pacific, and Aboriginal and Torres Strait Islander peoples.
Overview
Eating disorders are characterised by disturbances of eating behaviours and a core psychopathology centred on food, eating and body image concerns. Early reports of an anorexia nervosa-like illness date to the 1600s (Silverman, 1983) and anorexia nervosa as a diagnostic entity was described first in 19th century medical reports (Gull, 1874; Lasegue, 1873). In contrast, bulimia nervosa and binge eating disorder were not described until the 20th century. The American Psychiatric Association DSM-5 diagnostic criteria for anorexia nervosa (APA, 2013) include self-imposed or maintained weight loss such that the person is underweight (for age and height) and associated overvaluation of shape and weight (see Table 1). Two subtypes of anorexia nervosa are specified: restrictive type (with or without compulsive exercise); and binge eating/purging type, with binge eating (uncontrolled overeating) and purging (vomiting, laxative or diuretic misuse). Severity is specified according to BMI (kg/m2) status.
Comparative clinical features of DSM-5 eating disorder diagnostic groups.
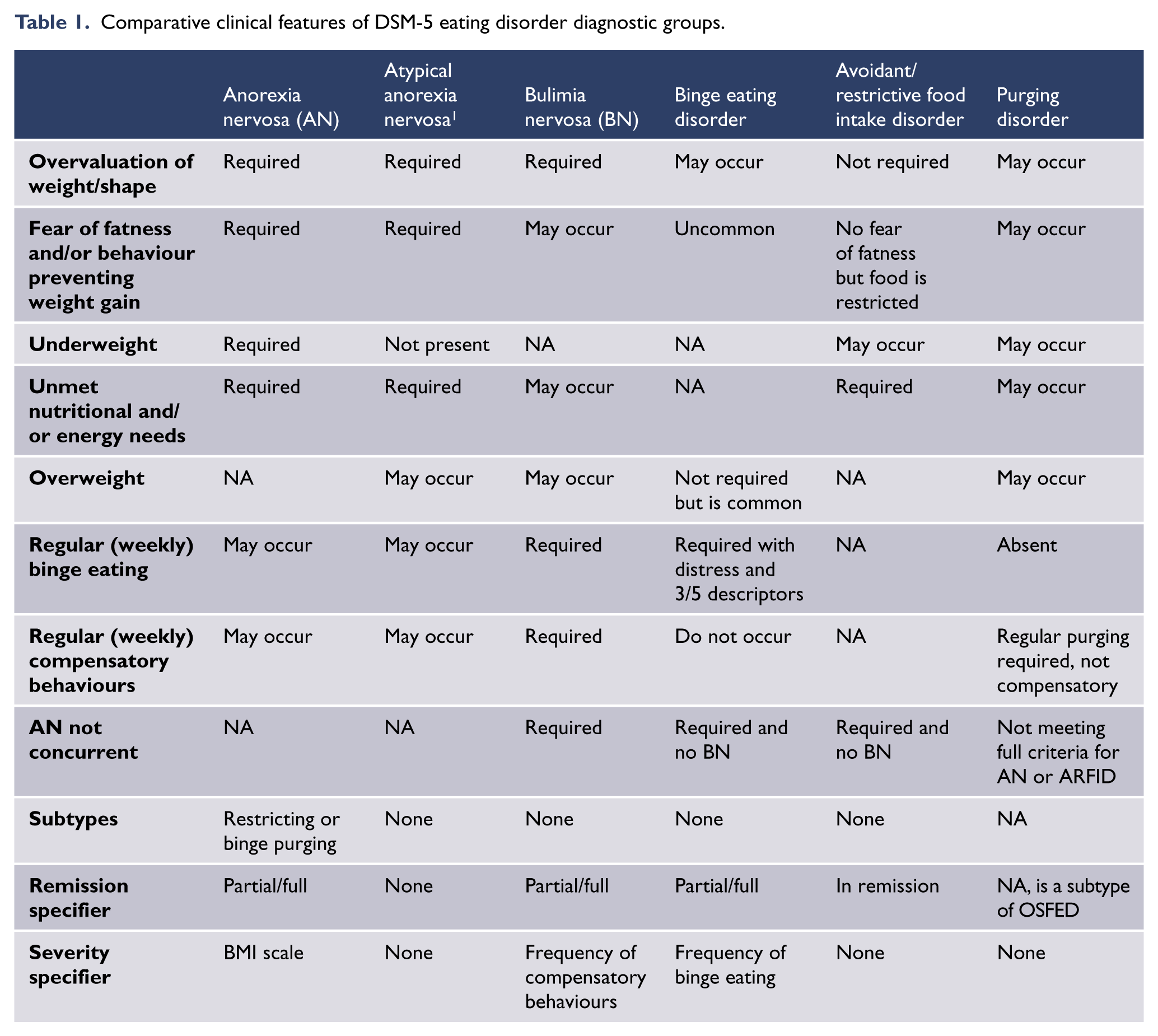
Bulimia nervosa and binge eating disorder are both defined in the DSM-5 by having regular and sustained binge eating episodes. People with bulimia nervosa also compensate for binge eating with regular extreme weight control behaviours (such as purging). As they do not engage in such compensation regularly, people with binge eating disorder are likely to be overweight or obese. People with bulimia nervosa also have a self-view that is unduly influenced by weight and shape overvaluation. Other specified and unspecified feeding and eating disorders (OSFED and UFED) in the DSM-5 include atypical anorexia nervosa (where BMI may be within the normal range) and sub-threshold forms of bulimia nervosa and binge eating disorder on the basis of insufficient frequency and/or duration of disordered eating behaviours. 1 A new disorder added to DSM-5 is avoidant/restrictive food intake disorder (ARFID) which, like binge eating disorder, and in contrast to anorexia nervosa and bulimia nervosa, is not characterised by body image disturbance. This departure from weight/shape overvaluation as a key feature of all eating disorders is the subject of discussion in the field and likely to also be found in the ICD-11 revision (Al-Adawi et al., 2013). Readers interested in this debate are referred to Russell (2013) and Hay (2013a).
Since the classic writings of Hilde Bruch (Bruch, 1978) the ‘face’ of anorexia nervosa and eating disorders has changed dramatically. Eating disorders are not the ‘preserve’ of females, the wealthy or ‘westerners’. In the general population, lifetime prevalence of anorexia nervosa is around 1% in women and < 0.5% in men, bulimia nervosa around 2% in women and 0.5% in men, and binge eating disorder around 3.5% in women and 2.0% in men (Favaro et al., 2003; Hudson et al., 2007; Keski-Rahkonen et al., 2007; Lewinsohn et al., 2000; Oakley Browne et al., 2006; Preti et al., 2009; Raevuori et al., 2009; Striegel-Moore et al., 2003; Wade et al., 2006). Point (three-month) prevalence in Australia is estimated at around 1% for bulimia nervosa, 2% for binge eating disorder (using the DSM-5 criteria of weekly frequency of binge eating and extreme weight control behaviours) and 3% for other eating disorders (specified or unspecified according to the new DSM-5 criteria) (Hay et al., 2008). The gender ratio in bulimia nervosa is similar to that of anorexia nervosa but binge eating disorder has a more even gender distribution (Hudson et al., 2007). Accurate point prevalence has not been estimable for anorexia nervosa in Australia but 12-month prevalence in the New Zealand survey was <1% (Wells et al., 2006).
Eating disorders are associated with notable quality of life impairment and impact on home, work, personal, and social life (Jenkins et al., 2011; Mitchison et al., 2012; Mond et al., 2012) and economic cost (Butterfly Foundation, 2012). Eating disorders also frequently co-occur with other mental health disorders, particularly anxiety disorders and depression (Hudson et al., 2007). The peak age of onset of anorexia nervosa is in early to mid-adolescence but may occur at any age, including in childhood, where the gender balance is more even (Madden et al., 2009). The reasons for the greater number of boys presenting in childhood years are unclear. In bulimia nervosa and binge eating disorder onset is more commonly in later adolescence and young adulthood (Stice et al., 2013) and binge eating disorder is more likely a mid-life disorder with a much more even gender frequency. It is important to be aware that all eating disorders can, and do, arise at any age, and in both females and males. In addition, eating disorders often go undiagnosed and untreated. Thus it is common for adults to present for treatment many years after onset, even into late middle-age (Bulik et al., 2012; Hart et al., 2011b). However, most people make a sustained recovery with treatment. This includes people with anorexia nervosa, where up to 40% of adults (and a higher percent of adolescents) will make a good five-year recovery, a further 40% a partial recovery and those with persistent illness may yet benefit from supportive therapies. For those with bulimia nervosa at least 50% fully recover and the outcomes with treatment are also good if not better for binge eating disorder (Steinhausen, 2002; Steinhausen and Weber, 2009; Steinhausen et al., 2003).
Aetiology
Socio-cultural, biological and psychological factors all contribute to the aetiology of eating disorders (Mitchison and Hay, 2014; Smink et al., 2012; Stice, 2002). The strongest socio-demographic risk factor for having an eating disorder continues to be being of female gender and being from the developed world where the ‘thin ideal’ prevails. Migrants from the developing world seem to be at particular risk. Also at risk are those living in urban areas and undertaking life pursuits where body image concerns predominate, for example, competitive gymnastics and fashion modelling.
In all eating disorders there is an increased genetic heritability and frequency of a family history. A family history of ‘leanness’ may be associated with anorexia nervosa and a personal or family history of obesity with bulimic eating disorders. Early menarche (controlling for body weight) also increases risk. Also likely important are epigenetic changes to DNA structure that are not encoded by the DNA sequence itself but which nonetheless result in enduring changes in gene expression and which are transmitted to subsequent generations. These can occur following periods of food deprivation (e.g. the Dutch starvation in World War 2), food repletion, or severe environmental stress (Campbell et al., 2011).
Psychological factors include a ‘milieu’ of weight concern in formative developmental years and specific personality traits, mostly notably low self-esteem (all eating disorders) and high levels of clinical perfectionism for those with anorexia nervosa, and impulsivity for bulimic disorders. Adverse experiences including emotional and sexual child abuse increase personal vulnerability, most likely through impeding a robust sense of self-worth and adaptive coping. The eating disorder then provides a sense of improved self-esteem and self-control for the individual (Stice, 2002).
Aim and scope
This clinical guideline will aim to provide guidance in the clinical treatment of people with eating disorders, namely anorexia nervosa, bulimia nervosa, binge eating disorder, ARFID. The clinical practice guideline (CPG) may have clinical utility to corresponding forms of other specified or not specified eating disorders that fail to meet DSM or ICD diagnostic criteria for anorexia nervosa, bulimia nervosa, binge eating disorder or ARFID. It will focus on two age groups in anorexia nervosa: (1) adults and older adolescents (18 years and above) and (2) children and adolescents living at home, as treatment and outcomes differ between these groups. One special population, people who are obese or overweight with an eating disorder, will also be addressed. 2
There is much more space given to anorexia nervosa in this CPG than other disorders. This does not reflect the prevalence of disorders but rather the added complexities of assessment and management for anorexia nervosa compared to bulimia nervosa and binge eating disorder, and the paucity of knowledge in ARFID. In contrast to bulimia nervosa and binge eating disorder, anorexia nervosa presents more frequently in children and adolescents as well as in adults, it is more likely to become severe and enduring and has more extensive medical co-morbidities. We decided not to separate the two disorders of recurrent binge eating (bulimia nervosa and binge eating disorder) as there is much overlap in assessment and treatment approaches and evidence for a transdiagnostic approach.
Method
This guideline was developed as part of the Royal Australian and New Zealand College of Psychiatrists, Clinical Practice Guidelines Project 2013–2014. It was developed in accordance with best practice as outlined by the National Health and Medical Research Council (NHMRC, 2007).
The literature review focused on recent systematic reviews that would include relevant treatment trials since the RANZCP guidelines were written and subsequently reviewed and updated for the consumer guidelines in 2009 (Beumont et al., 2003; RANZCP, 2004; RANZCP, 2009). A comprehensive literature review was thus conducted with dates 2008–2013 to systematically identify and synthesise all studies that were potentially relevant to the guideline. The search was undertaken using PubMed and the search terms ‘anorexia nervosa’ OR ‘bulimia nervosa’ AND ‘treatment guidelines’ OR ‘systematic review’ OR meta-analysis’. Reference lists of identified systematic reviews were also searched for relevant empirical studies on which the CPG recommendations are based. Forty-nine papers were generated and inspected for relevance and quality (including level of evidence grade according to NHMRC categories). Twenty-seven potentially relevant systematic reviews and empirical trials were reviewed by each of two members of the working group for inclusion, and 21 papers were included (members being ineligible to review literature that they had authored or co-authored themselves). Inclusion criteria were reaching a gradable level of evidence according to NHMRC categories of at least level III or higher. Five papers (three from expert reviewers, one from a working group member and one by a member of the public who was consulted) had also been identified by members of the working group and reviewed according to the same process. The results of the search are depicted in Figure 1.
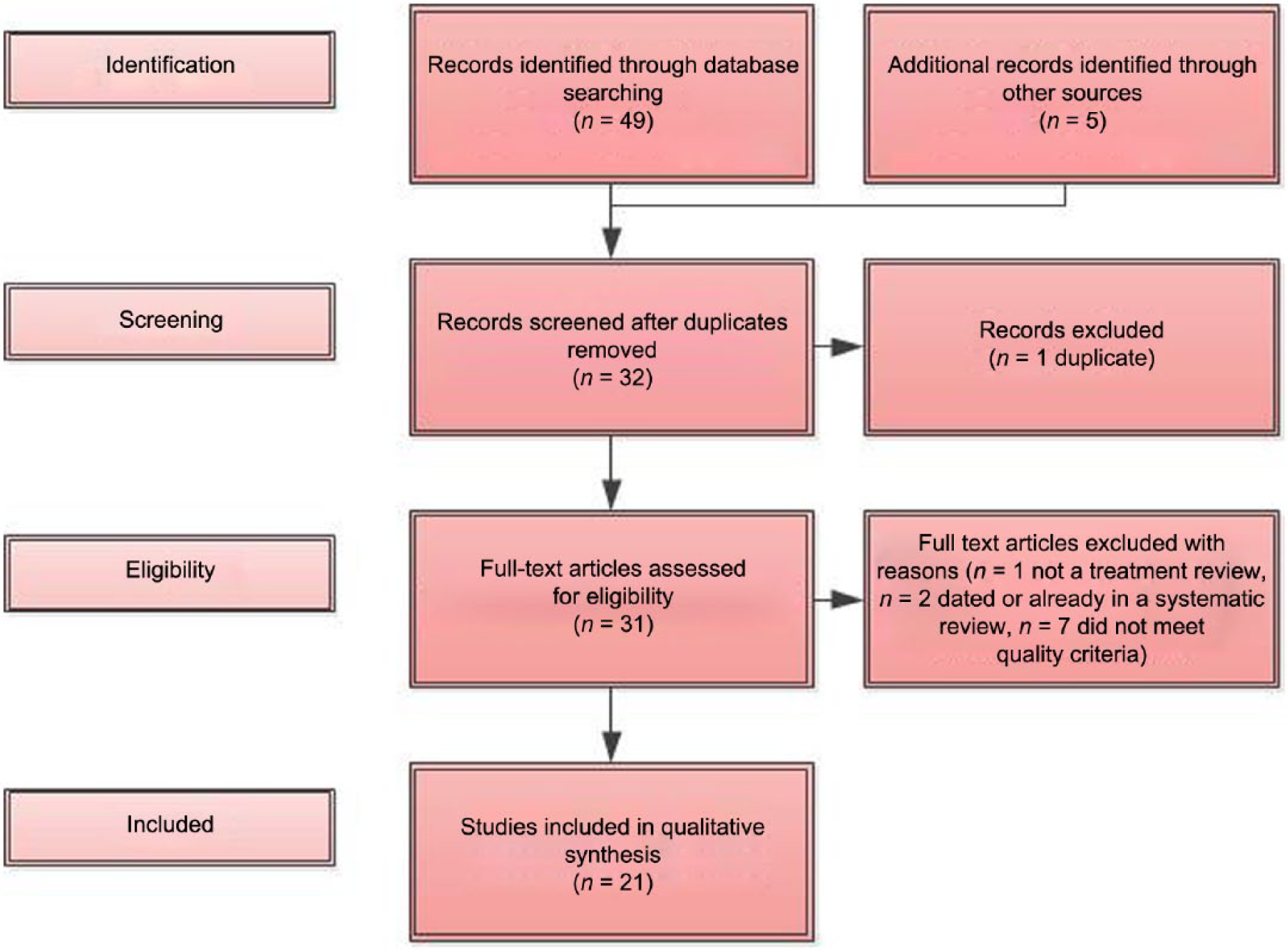
PRISMA flow diagram.
Evidence based recommendations (EBR) were formulated after appraising the evidence using the NHMRC levels of evidence ratings (see Table 2). Where evidence was weak or lacking, consensus based recommendations (CBR) have been formulated. A consensus based recommendation is the lowest level of evidence. It is the consensus of a group of experts in the field and is informed by their agreement as a group according to their collective clinical and research knowledge and experience. In this process level IV articles were considered where higher level evidence was lacking and they informed the CBR.
National Health and Medical Research Council designations of level of evidence based recommendation (EBR).

A series of drafts were then prepared and refined by the working group. The final draft was then reviewed by national and international expert advisers, professional bodies (medicine, psychology, dietetics, nursing, social work and occupational therapy) and special groups (consumer, carer, Aboriginal and Torres Strait Islander, Māori and Pacific and migrant) prior to an extensive community consultation process. A full list of people and groups consulted and tables of included and excluded studies are available on the RANZCP web site.
General principles of treatment for all eating disorders
Person-centred informed decision-making
Safe and empirically supported treatment options based on available research and expert consensus should be discussed with the individual and their family. These options should be centred wherever possible on an informed decision made with the person and (where appropriate) their family. For children and adolescents the decision balance will be age appropriate and will involve their parents or legally appointed guardian.
Involving family and significant others
Unless there are contraindications or the individual is opposed, family or significant others should be enlisted as partners in the assessment and treatment process. Given the considerable burden on family members it is important that the family is provided with appropriate support and information.
Recovery-oriented practice
Care for people with eating disorders should be provided within a framework that supports the values of recovery-oriented care (Australian Health Ministers Advisory Council, 2013). Recovery-oriented practice encapsulates mental health care that:
recognises and embraces the possibilities for recovery and wellbeing created by the inherent strength and capacity of all people experiencing mental health issues
maximises self-determination and self-management of mental health and wellbeing
assists families to understand the challenges and opportunities arising from their family member’s experiences
provides evidence-informed treatment, therapy, rehabilitation and psychosocial support that helps people to achieve the best outcomes for their mental health, physical health and wellbeing
works in partnership with consumer organisations and a broad cross-section of services and community groups
embraces and supports the development of new models of peer-run programs and services
maximises choice
supports positive risk-taking
recognises the dignity of risk, i.e. the individual’s right to make treatment choices that the treating health care team might not see as being the most effective decision
takes into account medico-legal requirements and duty of care
promotes safety.
Least restrictive treatment context
Where possible, treatment should be offered in the setting that is least restrictive and best suited to the individual’s needs and preferences. Many people with eating disorders are difficult to engage as they are ambivalent, or fear giving up their eating disorder behaviours, or deny the illness or the seriousness of their symptoms. Offering options and control can help with therapeutic engagement. Legislation in both New Zealand and Australia allows for involuntary assessment or treatment if a person with anorexia nervosa has impaired decision-making capacity, and is unable or unwilling to consent to interventions required to preserve life. Although involuntary treatment may provide the opportunity to prevent fatal complications, the potential adverse effects on therapeutic alliance needs to be considered (Carney et al., 2007). The short-term weight gain response of involuntary patients with anorexia nervosa has been shown to be comparable to those admitted voluntarily (Watson et al., 2000). Many of those who are treated on an involuntary basis later agree that treatment was necessary and remain therapeutically engaged (Guarda et al., 2007; Tan et al., 2010; Watson et al., 2000).
Multidisciplinary approach
Expert consensus and clinical cohort studies (e.g. NICE, 2004b) support a multidisciplinary approach to ensure that the individual gets access to the combined medical, dietetic (Dietitians Association of Australia, 2012) and psychological interventions required to maximise the chances of a full recovery. Ideally, team members will have specialist knowledge, skills and experience in the area of eating disorders, and be situated in the same location or at least in places easily accessible for those being treated and their families. Although team members may have differing perspectives, a united approach in delivering treatment is critical. The general practitioner is often in the best position to be the key coordinating clinician, especially if the treating specialists are not co-located.
Stepped and seamless care
Ideally, a range of options including outpatient, intensive outpatient with meal support, day program, and inpatient treatment should be available. As many people will not be seen by a specialist service, specialists should build strong links with primary care, general hospital and community providers in order to facilitate access and smooth transitions of care between general practice, emergency departments, medical wards, mental health settings, private clinicians and specialist services (House et al., 2012).
A dimensional and culturally informed approach to diagnosis and treatment
Establishing the presence of the core syndrome of an eating disorder is crucial in terms of informing treatment, but significant symptom variability occurs within and between individual experiences of anorexia nervosa, bulimia nervosa and binge eating disorder and other eating disorders. A rigid approach to diagnosis should be avoided (Pike, 2013). Empirically supported definitions of severity are still in development (Maguire et al., 2008) and although physical measures (e.g. BMI and more specific indicators of malnutrition such as amenorrhoea, hypotension, bradycardia, hypothermia and neutropaenia) are commonly used as markers of severity, psychological symptoms and clinical history should also inform severity formulations (Maguire et al., 2012). Comparative studies (EBR III) highlight possible cultural and ethnic variability in the presentation of anorexia nervosa or other eating disorders (Soh et al., 2006). There are insufficient studies on gender differences to inform the need for major differences in assessment approaches or treatment delivery for males (Murray and Touyz, 2013).
Indigenous care
Clinicians must demonstrate the appropriate knowledge, skills, awareness, and attitudes (‘cultural competence’) when working with people with eating disorders from indigenous and other cultural backgrounds. Assessment and treatment of eating disorders in Aboriginal and Torres Strait Islander and Māori and Pacific peoples should ascribe to the broader principles of mental health care as recommended in the RANZCP online training guide (RANZCP, 2012).
Approaches to care of people of Aboriginal and Torres Strait Islander background should be informed by an understanding of their history and culture even though there is little written about specific approaches to treatment for those who suffer from an eating disorder. However, epidemiologic evidence indicates that Aboriginal and Torres Strait Islander people are no less likely, and may be more likely, to experience an eating disorder as non-indigenous Australians (Hay and Carriage, 2012). In particular, poor nutrition and health-related consequences such as Type II diabetes are well recognised problems in this population. The reasons for this are complex, but the loss of traditional values and practices in food choices and replacement with foods of less nutritional value may be part of a broader process of cultural dispossession combined with social disadvantage. In addition, Aboriginal and Torres Strait Islander adolescents do have body image concerns which appear to focus around a desired ‘muscular’ shape (Cinelli and O’Dea, 2009) and may be less concerned about being slim or losing weight (McCabe et al., 2005; Ricciardelli et al., 2004).
Epidemiological data are scarce in Māori and Pacific peoples, with older data suggesting a lifetime prevalence of 3.1% (0.7% anorexia nervosa; 2.4% bulimia nervosa) and 3.9% respectively (Baxter et al., 2006). However, there are high levels of obesity in both Māori and Pacific peoples compared with the New Zealand adult population overall (Ministry of Health, 2012a, 2012b). Thus, whilst the prevalence of binge eating disorder is unknown, because of an associated risk with obesity it is possible that Māori and Pacific peoples are at increased risk of binge eating disorder.
In New Zealand ‘cultural competence’ explicitly includes application of te Tiriti o Waitangi (the Treaty of Waitangi) principles of partnership, participation and protection. This may require different assessment practices, for example the presence of wider whānau (family) at consultations, styles of treatment engagement and the communication of treatment information. These competencies are further outlined by the Medical Council of New Zealand and the New Zealand Psychologists Board (MCNZ, 2006a, 2006b, 2010; NZPB, 2011). In every aspect, working with different cultural groups requires respect for different world views, values and meanings. Clinicians should seek cultural advice and leadership from cultural support staff/whānau advisers to maximise engagement and therapeutic alliance.
Section one: anorexia nervosa
Anorexia nervosa in adults
Introduction
The following section outlines core assessment and treatment guidelines for adults with anorexia nervosa. There is a lack of high quality evidence to guide the clinician in the treatment of adults who have anorexia nervosa. Methodologically robust studies are small in number and inconclusive, meaning that conflicting results are common. Many studies are limited by methodological issues including small samples, low quality design, and short follow-up periods (Watson et al., 2010). There have however been improvements in the evidence base for treatments for adults with anorexia nervosa since the 2003 RANZCP Clinical Practice Guidelines (Beumont et al., 2003; Watson and Bulik, 2012). Clinical practice is as well best informed by considering recent systematic reviews and accessing empirically investigated treatments in conjunction with consensus opinions of experts in the field (Beumont et al., 2003; RANZCP, 2004; Watson et al., 2010; Yager et al., 2006).
Assessment
A comprehensive assessment of the individual and their circumstances should be undertaken to confirm the diagnosis of anorexia nervosa and any comorbid psychiatric or medical diagnoses, to evaluate medical and psychiatric risks, and to develop a biopsychosocial formulation. Collecting assessment information is an ongoing task as clinical issues and priorities unfold throughout treatment. Comprehensive initial assessment of adults should include the following components:
Collating a thorough history including the various symptoms of anorexia nervosa which include but are not limited to: dietary restriction; weight loss; inability to restore weight; body image disturbance; fears about weight gain; binging; purging; excessive exercise; early satiety; constipation; and the use of laxatives, diuretics, or medications to lose or maintain low weight (APA, 2013). Other symptoms may include disturbed eating behaviours, e.g. eating apart from others and ritualistic patterns of eating such as prolonged meal times and division of food into very small pieces (Wilson et al., 1985). It is important to accurately assess nutritional and fluid intake, with specific enquiries made as to the adequacy of main meals and snacks consumed. Where possible, collateral sources such as family members and other clinicians involved in the person’s care should be utilised. The perspective of others is especially important given that symptom minimisation, poor insight or genuinely poor understandings of the seriousness of symptoms are common aspects of anorexia nervosa (APA, 2013).
Investigating any medical complications and the current level of medical risk. This is essential and should include a brief physical examination including measurement of weight, height, calculation of BMI, seated and standing pulse rate to detect resting bradycardia and/or tachycardia on minimal exertion due to cardiac deconditioning, blood pressure (seated and standing) and temperature. These findings are needed to determine if immediate hospital admission is required (see Table 3). Investigations should include serum biochemistry to detect hypokalaemia, metabolic alkalosis or acidosis, hypoglycaemia, hypophosphataemia, and hypomagnesaemia, serum liver function tests, serum prealbumin levels and a full blood examination looking for evidence of starvation-induced bone marrow suppression such as neutropaenia and an electrocardiogram (ECG). A bone mineral density scan should be performed routinely if the person has been underweight for six months or longer with or without amenorrhea and thereafter every two years whilst still struggling with an eating disorder (Mehler et al., 2011). The assessment should also include any history of fainting, light-headedness, palpitations, chest pain, shortness of breath, ankle swelling, weakness, tiredness and amenorrhoea or irregular menses
Assessing psychiatric comorbidity, e.g. anxiety, depression, substance misuse, suicidality, personality disorders, anxiety disorders and deliberate self-harm. Comorbidity in people with anorexia nervosa is common and therefore assessment for such should be routine. Lifetime prevalence of comorbidity has been reported ranging from 55% in community adolescent samples to 96% in adult samples (Godart et al., 2007; Madden et al., 2009; Milos et al., 2003; Swanson et al., 2011). All forms of anxiety disorder may occur and in one study the most common was social phobia (42%), followed by post-traumatic stress disorder (26%) and generalised anxiety disorder (23%) (Swinbourne et al., 2012). Furthermore, social anxiety is not only related to eating in public but may pre-date the onset of the eating disorder. Rates of comorbidity are similar in men and women (Raevuori et al., 2009). However, clinicians should be aware that depression, obsessional thinking, anxiety and other psychiatric symptoms can represent the reversible effects of starvation on the brain (Keys et al., 1950).
Assessing cognitive changes due to starvation such as slowed thought processing, impaired short-term memory, reduced cognitive flexibility and concentration and attention difficulties (Hatch et al., 2010). Whilst brain imaging is not routine, these problems reflect consistent findings of reduced grey matter volumes that often do not reverse following weight recovery (Phillipou et al., 2014).
Considering possible predisposing and precipitating factors including a family history of eating disorders, early attachment and developmental difficulties, premorbid obesity, interpersonal problems, and dieting or other causes of rapid weight loss (Mitchison and Hay, 2014; Stice, 2002). Rapid weight loss from any cause, including physical illness, can trigger cognitive changes including obsessive thinking about food, in turn precipitating and perpetuating the symptoms of anorexia nervosa (Keys et al., 1950).
Indicators for consideration for psychiatric and medical admission for adults.
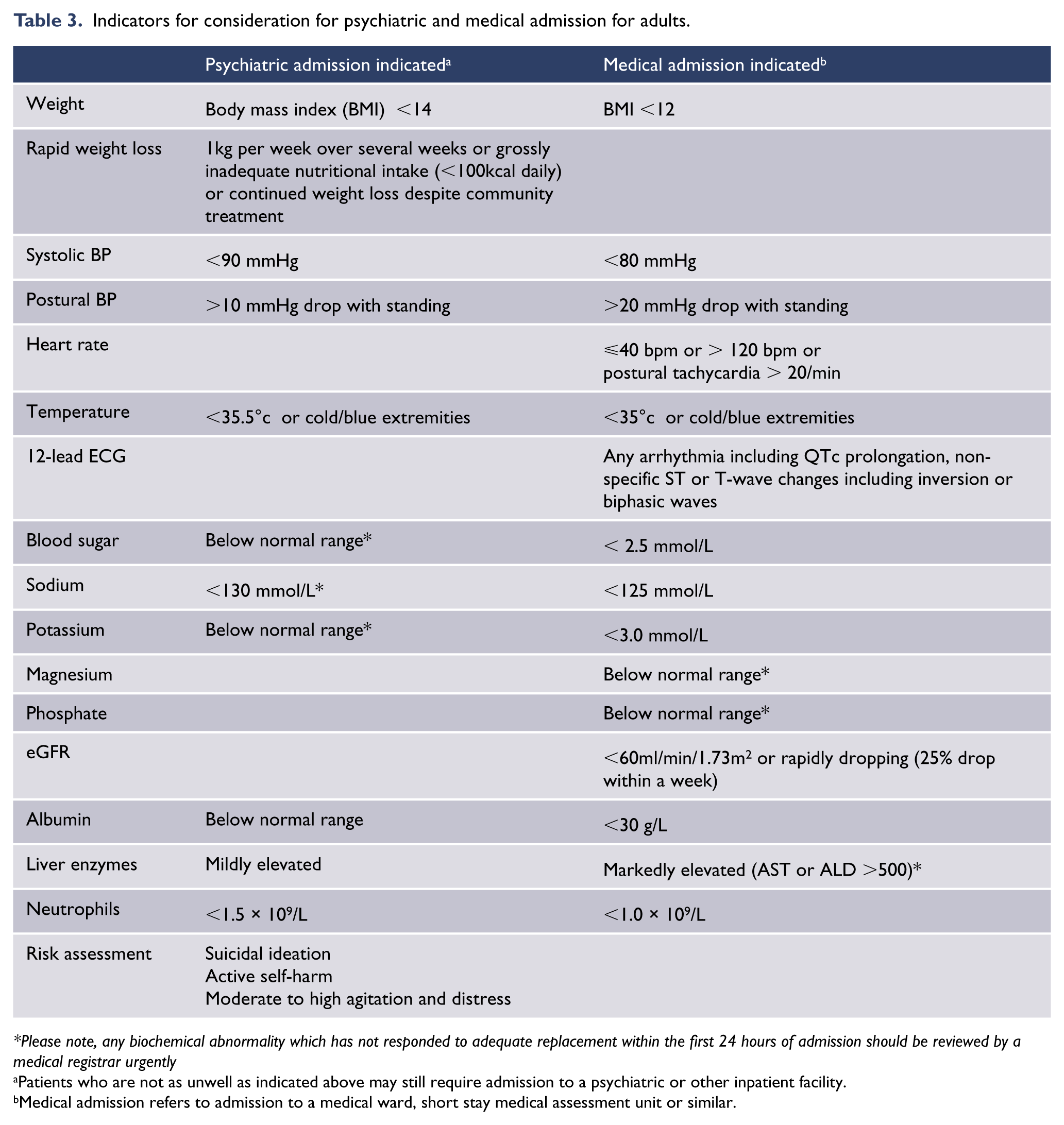
Please note, any biochemical abnormality which has not responded to adequate replacement within the first 24 hours of admission should be reviewed by a medical registrar urgently
Patients who are not as unwell as indicated above may still require admission to a psychiatric or other inpatient facility.
Medical admission refers to admission to a medical ward, short stay medical assessment unit or similar.
Treatment
Setting priorities
Thorough assessment should lead to a working diagnosis or diagnoses, risk assessment and case formulation setting immediate treatment priorities. The case formulation should include preliminary hypotheses about predisposing, precipitating and maintaining factors, as well as noting the individual’s strengths and protective factors. Given the typically prolonged time between onset and presentation, and the diverse pathways into anorexia nervosa, early firm statements about causal factors pertaining to an individual should be avoided. It is generally agreed that treatment priorities should be set as follows: engagement, medical stabilisation, reversal of the cognitive effects of starvation, and provision of structured psychological treatment. These are discussed in more detail below.
Engagement
Engaging with the individual with anorexia nervosa and their family at the first appointment can be challenging as there is typically extreme anxiety at the prospect of increased nutrition and weight gain, which are essential for recovery. To enable therapeutic engagement it is crucial that the clinician take a non-judgemental, inclusive, empathetic and non-threatening stance. Although lacking level I empirical evidence, sensible practices for improving engagement have included psychoeducation, enlisting the support of the family (Treasure et al., 2007), motivational interviewing around what the individual themselves reports as important, and appealing to the ‘healthy’ part of the person (Nordbø et al., 2008; Vitousek et al., 1998).
Medical stabilisation
Admission to hospital is indicated if the person is at imminent risk of serious medical complications, or if outpatient treatment is not working (Beumont et al., 2003). Indicators of high medical risk requiring consideration for admission (see Table 3) include any one of the following: heart rate <40 bpm or tachycardia on standing due to cardiac deconditioning with >20 bpm increase in heart rate, blood pressure <90/60 mm Hg or with >20 mm Hg drop on standing, hypokalaemia, hypoglycaemia, hypophosphataemia, temperature <35.5°C, or BMI < 14 kg/m2. It is important to note that patients can report feeling well even when the risk of cardiac arrest is high. Admission is also indicated if there is rapid weight loss, several days of no oral intake, supervision required for every meal, uncontrolled purging or exercise, or suicidality.
Ideally, whenever possible and practicable, people with anorexia nervosa requiring admission should be admitted to a specialist eating disorders unit. There is debate about rates of weight gain in inpatient settings with recommended rates ranging from 500–1,400g/week (NICE, 2004b; Yager et al., 2006). There is evidence that where weight gain is the key prioritised treatment goal, inpatient treatment is superior to less intense forms of treatment due to the faster weight gain in those settings (Hartmann et al., 2011). However, inpatient beds are unfortunately scarce and can be difficult to access. In such situations, another model that has been effective in managing acute problems is admission to general medical or psychiatric beds, with support provided to generalist clinicians by a specialist eating disorder consultation-liaison service (EDOS, 2011). Where patients are at very high medical risk (e.g. with BMI < 12 or significant medical complications), they will need to be admitted to a medical setting with input from psychiatry consultation-liaison services until medically stabilised, before being transferred to a psychiatric or eating disorders specialist unit for ongoing nutritional rehabilitation and psychiatric treatment. Discharge from hospital should only occur when the person is medically stabilised, has had enough nutrition to reverse any cognitive effects of starvation so that she or he can benefit from outpatient or day patient psychotherapy (often several weeks of nutrition are required to achieve this), has had trials of leave to demonstrate that she or he can eat outside hospital, and has a direct link in with appropriate outpatient monitoring, support and treatment (EDOS, 2011).
Following discharge to less intense treatment settings, there is insufficient evidence to point to the best means of maintaining weight (Hartmann et al., 2011). One small randomised control trial found a manualised cognitive behavioural therapy for anorexia nervosa (CBT-AN) was superior to nutritional counselling and usual care (all treatment including nutritional counselling delivered by psychologists) in preventing relapse (Pike et al., 2003). Nutritional counselling alone should not be the sole treatment for people with anorexia nervosa; a multidisciplinary approach is more supported (Dietitians Association Australia, 2003) and there is strong clinical consensus that continuity of treatment should be provided. For people with severe and enduring eating disorders, a more flexible and patient-centred approach may need to be taken when considering criteria for admission and discharge (see separate section on severe and enduring eating disorders in this clinical practice guideline).
Medical complications and their treatment
Table 4 lists the common medical complications and their management. If the medical complication is secondary to malnutrition, effective treatments must always include regular and adequate nutrition. It is important to ensure that prescribed nutrition is being consumed by the individual, either through supervision and support during mealtimes by staff in hospital or family/carers out of hospital; or if the person is well enough, through self-recording of oral intake discussed at outpatient treatment sessions. Similarly, if medical complications are due to purging or other behaviours, measures need to be taken to address these.
Physical and laboratory findings and their management.
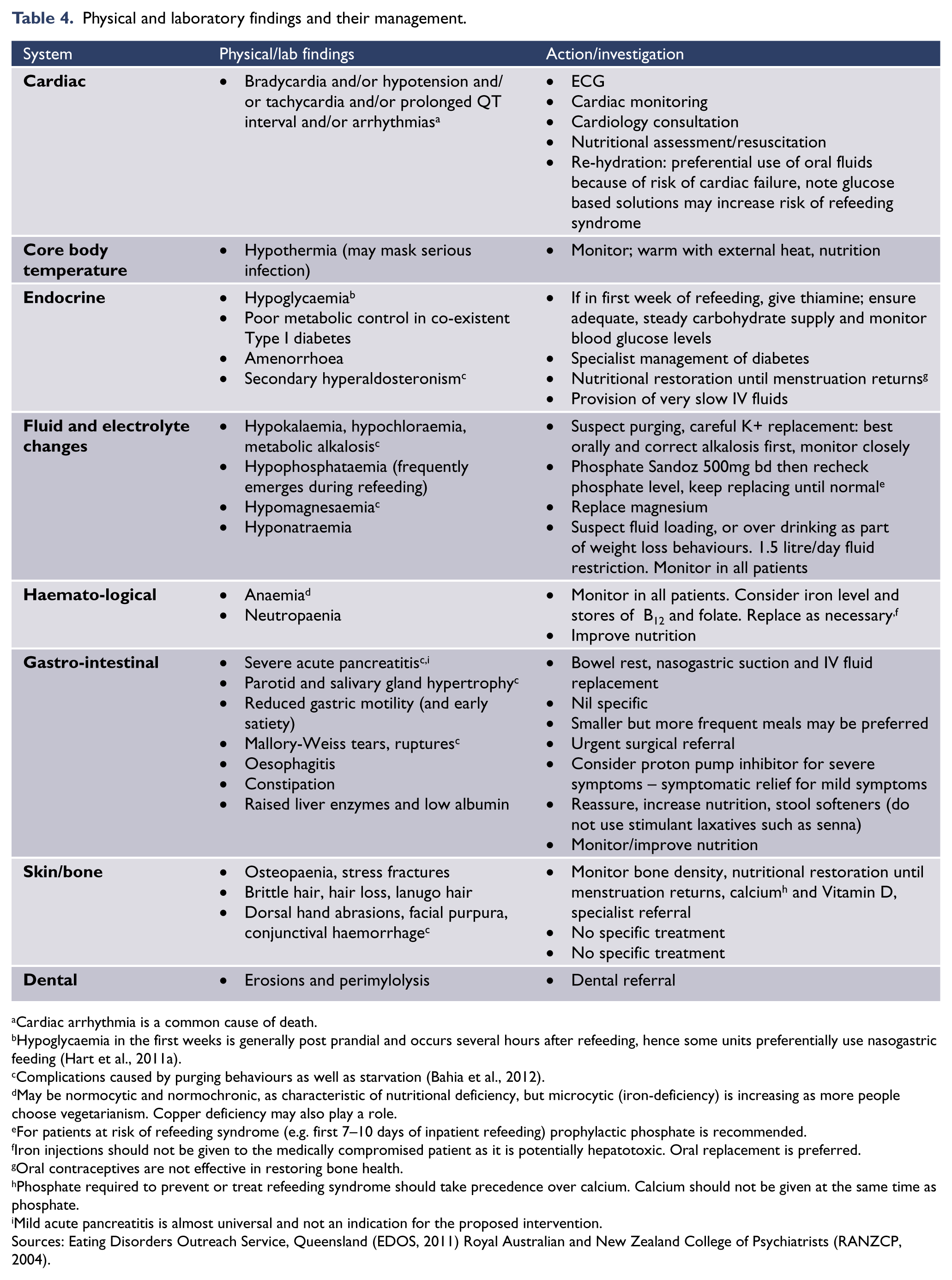
Cardiac arrhythmia is a common cause of death.
Hypoglycaemia in the first weeks is generally post prandial and occurs several hours after refeeding, hence some units preferentially use nasogastric feeding (Hart et al., 2011a).
Complications caused by purging behaviours as well as starvation (Bahia et al., 2012).
May be normocytic and normochronic, as characteristic of nutritional deficiency, but microcytic (iron-deficiency) is increasing as more people choose vegetarianism. Copper deficiency may also play a role.
For patients at risk of refeeding syndrome (e.g. first 7–10 days of inpatient refeeding) prophylactic phosphate is recommended.
Iron injections should not be given to the medically compromised patient as it is potentially hepatotoxic. Oral replacement is preferred.
Oral contraceptives are not effective in restoring bone health.
Phosphate required to prevent or treat refeeding syndrome should take precedence over calcium. Calcium should not be given at the same time as phosphate.
Mild acute pancreatitis is almost universal and not an indication for the proposed intervention.
Sources: Eating Disorders Outreach Service, Queensland (EDOS, 2011) Royal Australian and New Zealand College of Psychiatrists (RANZCP, 2004).
Refeeding syndrome
Refeeding syndrome is a serious and potentially fatal medical complication of aggressive refeeding of an individual who has been malnourished for a lengthy period. Refeeding syndrome is understood to be due to the switch from fasting gluconeogenesis to carbohydrate-induced insulin release triggering rapid intracellular uptake of potassium, phosphate and magnesium into cells to metabolise carbohydrates (Kohn et al., 2011). This, on top of already low body stores of such electrolytes due to starvation, can lead to rapid onset of hypophosphataemia, hypomagnesia and hypokalaemia. In addition, insulin-triggered rebound hypoglycaemia can occur, exacerbated by the fact that such patients have depleted glycogen stores. The risk factors for refeeding syndrome include the degree of malnutrition and adaptation to this state, the levels of serum minerals and electrolytes such as phosphate and potassium and the rate of provision of carbohydrate in relation to other nutrients (Gentile et al., 2010; Kohn et al., 2011; Mehler et al., 2010; O’Connor and Goldin, 2011; Ornstein et al., 2003).
There is a wide range of opinion as to ideal starting doses of nutrition for adults with anorexia nervosa, with often little evidence to support the varied opinions (Gaudiani et al., 2012; Katzman, 2012; Kohn et al., 2011). Traditionally, it has been thought that the risk of refeeding syndrome can be reduced by ‘starting low’ and ‘going slow’ with nutrition, and monitoring serum phosphate, potassium and magnesium daily for the first 1–2 weeks of refeeding, and replacing these electrolytes immediately if they fall below normal range (Beumont et al., 2003; NICE, 2004b; Yager et al., 2006).
All authors agree on the importance of regularly monitoring and replacing phosphate, potassium and magnesium. However, traditional recommendations for refeeding designed to prevent refeeding syndrome are now seen by many to be too conservative, and unnecessarily put the severely malnourished person at risk of ‘underfeeding syndrome’ and further medical deterioration. Findings from case series studies range from those reporting large numbers of adolescents being fed on relatively high initial rates of up to 8400kJ of low-carbohydrate continuous nasogastric feeds with supplemental phosphate without causing refeeding syndrome (Kohn et al., 2011; Whitelaw et al., 2010), to those of severely malnourished adults reporting that 45% of participants developed significant refeeding-induced hypophosphataemia with much lower mean initial refeeding doses of 4000kJ/day (Gaudiani et al., 2012). Thus, refeeding syndrome has been observed even with very low initial feeding doses, and initial dose has not been shown to be a predictor of refeeding hypophosphataemia or refeeding syndrome (Gaudiani et al., 2012; O’Connor and Goldin, 2011). It is unclear whether the recent literature supporting safe use of higher refeeding doses in adolescents (for more information, see the section in these guidelines on treating anorexia nervosa in children and adolescents) is applicable to adults who may have been more severely malnourished for much longer periods, theoretically putting them at higher risk of refeeding syndrome.
In light of the conflicting and inadequate literature, the CPG group recommend taking a ‘middle path’ with adults, commencing refeeding at 6000kJ/day. This should be increased by 2000kJ/day every 2–3 days until an adequate intake to meet the person’s needs for weight restoration is reached. This diet should be supplemented by phosphate at 500mg twice daily and thiamine at least 100mg daily for the first week, and thereafter as clinically indicated for people at high risk of refeeding syndrome (e.g. BMI <13). For people at high risk of refeeding syndrome, commencing with continuous nasogastric feeding with low-carbohydrate preparations (i.e. 40–50% of energy from carbohydrates) seems prudent to avoid triggering postprandial rebound hypoglycaemia due to insulin secretion in people with inadequate glycogen stores. The most important aspects of preventing refeeding syndrome are a heightened physician awareness of the syndrome, and regular monitoring of the person’s clinical status, including physical observations and biochemical monitoring, especially to guide phosphate prophylaxis or supplementation.
Refeeding protocols should, however, be individualised where necessary to minimise both the risk of refeeding syndrome and complications due to underfeeding, and involve the input of a dietician experienced in the treatment of eating disorders. Methods of nutritional provision include supervised meals, high energy high protein oral liquid supplements and nasogastric feeding. On very rare occasions where the above methods are unable to be utilised, parenteral nutrition may be indicated. The least intrusive and most normal method of nutrition that can be reliably provided should be used.
Monitoring progress and reviewing priorities
In assessing whether an individual’s nutritional heath has been adequately restored, weight is only an approximate indicator. Other indicators of normal physiological functioning should be considered, including normal blood glucose levels, reversal of hypotension and bradycardia, normal blood cell counts with bone marrow suppression reversed, return of menstruation and normal cognitive functioning.
Assessing psychological progress is more difficult given that many features of anorexia nervosa involve internalised symptoms and behavioural deficits. However, such assessment should include monitoring dietary intake, compensatory behaviours and body image disturbance and dissatisfaction. Absence of progress in treatment after reasonable trial periods should prompt a treatment review and consideration for changing interventions and/or increasing treatment intensity.
Specific treatments
Providing psychoeducation, support and building a therapeutic relationship are all crucial activities at all stages of treatment. The more intense structured psychological therapies should generally be initiated only after the individual is sufficiently stabilised and cognitively improved from the acute effects of starvation. It is important that the treatment plan is individualised and addresses any comorbid conditions.
Individual therapies
There are major shortcomings in the literature, making any robust and direct comparison between commonly used psychological treatments (e.g. cognitive behaviour therapy, interpersonal psychotherapy, or psychodynamic psychotherapy) very difficult. These limitations include small numbers of trials and lack of statistical power.
Cognitive behavioural therapy (CBT) and its many forms, for example CBT-Enhanced (Fairburn, 2008), are frequently recommended approaches for anorexia nervosa. Common to these approaches are the activities of directly challenging anorexia-related behaviours, cognitions and patterns of thinking, especially symptoms that maintain the disorder. In inpatient settings, less restrictive behavioural approaches are likely to be more effective than strict regimens which are often perceived by the individual as punitive and demeaning (Touyz et al., 1984). Despite the popularity of CBT and improving evidence of its effectiveness, further investigation is required (Bulik et al., 2007; Hay, 2013b; Zipfel et al., 2014). There is no empirical evidence that guided self-help CBT (CBT-GSH) is a useful treatment for anorexia nervosa, with some concluding that it is contraindicated (Wilson and Zandberg, 2012).
Specialist supportive clinical management (SSCM) (McIntosh et al., 2006) has been shown in one trial to be more effective than CBT or interpersonal therapy (McIntosh et al., 2005). SSCM has as its primary focus resumption of normal eating and the restoration of weight, but it also allows a flexible approach to addressing life issues impacting on the eating disorder.
The Maudsley model of anorexia nervosa treatment for adults (MANTRA), not to be confused with the ‘Maudsley’ model of family based therapy, is a recently developed manualised individual therapy for adults with anorexia nervosa, drawing on a range of approaches including motivational interviewing, cognitive remediation and flexible involvement of carers. It aims to address the obsessional and anxious/avoidant traits that are proposed as being central to the maintenance of the illness (Schmidt et al., 2012). In a randomised controlled trial (RCT) it was, however, shown to be no more effective than SSCM, with recovery rates low in both arms of the trial (Schmidt et al., 2012).
Motivation-based therapies (motivational interviewing, motivational enhancement etc.), either as the main treatment or in conjunction with another therapy, have been adapted to eating disorders (Casasnovas et al., 2007; Nordbø et al., 2008; Treasure et al., 2007) including anorexia nervosa. Meaningful engagement in therapy is a crucial component in all treatments for anorexia nervosa, as are techniques to enhance change. Recent critical reviews question whether purely motivation-based therapies improve treatment efficacy in anorexia nervosa (Knowles et al., 2012; Waller, 2012). This does not dismiss the crucial importance of challenging resistance to change and targeting behavioural changes alongside wider psychological changes: these processes underpin most psychotherapies.
Other individual approaches that may be helpful in adults with anorexia nervosa include interpersonal psychotherapy, cognitive analytic therapy, focal psychoanalytic and other psychodynamic therapies (Watson, 2010; Zipfel, 2014), although again the level of evidence for each of these is modest, and generally limited to a very small number of trials. Interpretation of findings where specific psychological therapies are compared with other therapies is hampered by methodological problems (Hartmann et al., 2011). Web-based therapies for anorexia nervosa have not been sufficiently investigated by EBR II research (Aardoom et al., 2013). A single RCT has shown that a CBT-based program delivered on the internet may reduce the risk of relapse after hospitalisation (Fichter et al., 2012). It is advised that any treatment via the internet poses unique risks in providing responsible care. There is increased likelihood of miscommunications and there are significant regulatory issues if the practice occurs across different jurisdictions. Given the very limited evidence of effectiveness, these additional factors contribute to a high degree of caution if using internet-based therapies to treat anorexia nervosa.
Family therapies
There is moderate research-based evidence for family therapies in younger people with anorexia nervosa up to their late teens, living with family and with an illness duration of less than three years, with evidence that family therapy is more effective than individual treatment (Russell et al., 1987). Whilst family therapy approaches vary in their focus and etiological stances, a common theme is the involvement of family in treatment to help recovery especially in regard to interventions to restore weight.
Recent evidence reviews have reported that family therapy and a specific form of family therapy termed ‘family based treatment’ (FBT) remains the most well-studied treatment for young people with anorexia nervosa, and has been associated with persistent positive outcomes on physical and psychological parameters (Keel and Haedt, 2008; NEDC, 2010; Watson and Bulik, 2012); however, views are mixed (Couturier et al., 2013; Fisher et al., 2010; Strober, 2014). One of these reviews (Couturier et al., 2013) found that although FBT was not superior to individual treatment at the end of treatment, there did appear to be significant benefits at the 6–12 month follow-up. It is important to note that FBT has not been shown to be an effective treatment for anorexia nervosa in adults older than 18 years.
There is wider clinical consensus that families play an important role in assessment and overall treatment processes for younger people. Thus, unless contraindicated, family functioning should be assessed and appropriate support provided to manage the burden to families of anorexia nervosa. Other promising models of family interventions have been developed for adults, such as skills-based training for family members (Treasure et al., 2007) and couples work with partners (Bulik et al., 2011), but these are yet to be evaluated in well-designed studies. Further research and guidance with regards to family therapy in children and adolescents is discussed later in this guideline.
Pharmacotherapy
Recent systematic reviews of RCTs and meta-analyses of the pharmacological treatment of anorexia nervosa suggest weak evidence for the use of any psychotropic agents (Aigner et al., 2011; Flament et al., 2012; Hay and Claudino, 2012; Kishi et al., 2012). Prescribing for comorbid conditions (e.g. anxiety or mood disorders) is best left until it is clear that such symptoms are not simply secondary to starvation, although low doses of antipsychotics such as olanzapine may be helpful when patients are severely anxious and demonstrate obsessive eating-related ruminations, but more trials are needed (Hay and Claudino, 2012). Caution is required for any psychotropic medication, as physical problems secondary to anorexia nervosa may place individuals at greater risk of adverse side effects. Evidence for the effectiveness of medications to reverse bone density loss is lacking (Mehler et al., 2011).
Other treatments
A variety of other treatments have been proposed as primary treatments or as an adjunct to treatment of anorexia nervosa, but not established because of inadequate evidence to support their routine use. Zinc deficiency is common in anorexia nervosa and may be associated with dermatological change (Kim et al., 2010; Lask et al., 1993). In a single small double-blind trial, zinc supplementation was associated with a more rapid rate of body mass increase (Birmingham et al., 1994). Zinc supplementation is, however, not a routine component of therapy, because assessment of zinc status is difficult, the purported benefit limited, and deficiency will correct with general nutritional improvement (Lask et al., 1993).
Clinical observations and animal studies of decreased thermogenic activity with application of external heat have led to the incorporation of warming as a component of some treatment programs (Bergh et al., 2002; Birmingham et al., 2004; Gutierrez and Vazquez, 2001). A controlled trial of warming for three hours a day failed to demonstrate any advantage for weight gain compared with ‘treatment as usual’. Biofeedback focused on satiety and rate of eating underpins the Mandometer, a computer-based device that attempts to retrain eating patterns, has support from small observational studies and a small RCT compared to a wait-list control group (Bergh et al., 1996, 2002), but not from another larger controlled study (van Elburg et al., 2012). Recently, stereotactic surgery and deep brain stimulation have been considered for the management of refractory anorexia nervosa (Lipsman et al., 2013b). A pilot study of six adult patients with chronic refractory anorexia nervosa underwent implantation of electrodes to the sub-callosal cingulate region of their brain and then long-term deep brain stimulation (Lipsman et al., 2013a). While there may be some benefits to this approach, assessment has been inadequate and the wider response has been cautious (Hutton, 2013; Treasure and Schmidt, 2013).
Relapse prevention
Prospective and retrospective studies show varying rates of relapse, but it is generally agreed that the rates are substantial. A recent prospective study (Carter et al., 2012) showed a relapse rate of 41%, with the highest risk for relapse within 4–9 months after treatment. There is insufficient evidence about treatments to reduce relapse, but well-designed studies are consistent in finding that those with anorexia nervosa-binge purge subtype have higher rates of relapse, meaning that these individuals should receive more intense follow-up.
Outcomes and prognosis
Steinhausen (2002) reported that 70% of a multicentre European cohort with anorexia nervosa had fully recovered at follow-up at a mean of 6.4 years, and around 75% had no other evidence of psychiatric illness. Strober and colleagues documented in detail the follow-up of a cohort of 95 individuals with anorexia nervosa followed longitudinally. Eighty-seven percent recovered at least partially (loss of three of four diagnostic criteria of anorexia nervosa) at a mean of 59 months from assessment, and 76% experienced full recovery at a mean of 79 months. In an adolescent cohort, it was reported that recovery was still occurring 11 years after initial assessment (Strober et al., 1997). Steinhausen also reported lower mortality rates than ‘all-age’ data, with a 2.9% crude mortality rate in an adolescent European cohort (Steinhausen et al., 2003).
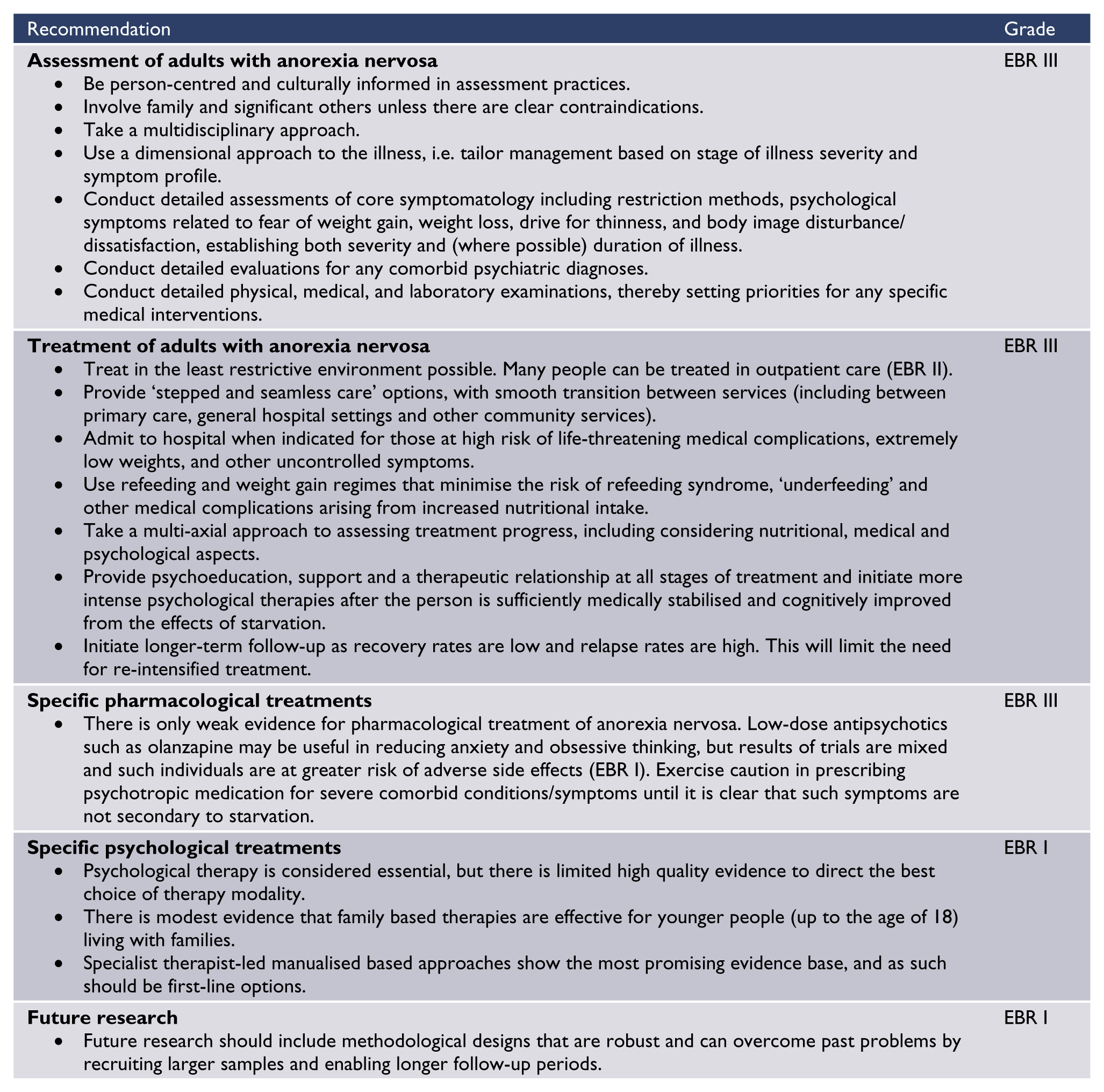
Summary of recommendations with level of evidence base (EBR) or consensus (CBR)
Anorexia nervosa in adolescents and children
Introduction
Anorexia nervosa is the third most common chronic disorder affecting adolescent girls, with a mean mortality rate of 5% in adults and 2% in adolescents (Steinhausen et al., 2002). The mortality rate is up to 18 times greater than in non-affected women aged 15–24 years (Steinhausen et al., 2003). The peak age of onset for anorexia nervosa is 15 to 19 years, accounting for approximately half of all presentations (73.9 to 270 females per 100,000 person years and 6.4 to 15.7 males per 100,000 person years) with nearly all people with anorexia nervosa presenting between 10 and 29 years (Herpertz-Dahlmann, 2009). Individuals between 10 and 14 years account for approximately one in five new presentations of anorexia nervosa (Keski-Rahkonen et al., 2007; Lucas et al., 1999; Raevuori et al., 2009). The incidence of early onset (children aged 5–13 years) eating disorders in Australia is estimated to be 1.4–2.8 per 100,000 children (Madden et al., 2009).
Lifetime prevalence rates for anorexia nervosa are higher in younger women compared with older age women, with rates consistently higher in more recent studies. This is consistent with the findings of Lucas et al. (1999), who demonstrated that the incidence of anorexia nervosa in 15 to 24 year old women has increased over the past 50 years. In the 10 to 14 year old age group, rates of anorexia nervosa also appear to have increased since the 1960s (Lucas et al., 1999), though the limited number of studies that have looked at this group make it difficult to be definitive. What can be clearly stated about this younger age group is that males make up a far greater proportion of those with anorexia nervosa, making up between one in six and one in four presentations (Madden et al., 2009; Nicholls et al., 2011b) compared with between one in eight and one in 15 presentations in adults (Hoek and van Hoeken, 2003; Lucas et al., 1999; Miller and Golden, 2010; Preti et al., 2009).
While two thirds of children aged less than 12 years with weight loss due to an eating disorder present with similar psychological symptoms to older adolescents and adults with anorexia nervosa, there are also differences. Children in this age group are less likely to report fear of weight gain or fatness (Lask and Bryant-Waugh, 1992; Madden et al., 2009; Nicholls et al., 2011b; Walker et al., In Press), more likely to fail to appreciate the severity of their illness (Fisher et al., 2001), more likely to present with non-specific somatic symptoms (Blitzer et al., 1961; Madden et al., 2009; Nicholls et al., 2011b), more likely to be diagnosed with other specified or unspecified feeding or eating disorder or ARFID, more likely to be boys, less likely to report vomiting or laxative abuse, more likely to have lost weight more rapidly and more likely to have a lower percent ideal body weight (%IBW) than older individuals with anorexia nervosa (Madden et al., 2009; Nicholls et al., 2011b; Peebles et al., 2006; Walker et al., In Press).
Complications from malnutrition and compensatory behaviours associated with anorexia nervosa include growth retardation, osteoporosis, infertility and changes in brain structure (Katzman, 2005) as well as psychological complications including depression, anxiety, obsessive compulsive disorder and cognitive impairment (Hatch et al., 2010). These complications are greatest during early adolescence due to disruption of critical periods of physical, psychological and social development (Golden et al., 2003; Katzman, 2005).
Assessment
There is limited evidence to guide the assessment of children and adolescents with anorexia nervosa with all recommendations based on expert consensus. At assessment, every child or adolescent suspected of having anorexia nervosa needs a comprehensive review of psychological and physiological signs and symptoms. Ideally assessments should be multidisciplinary and include professionals with expertise in psychiatric diagnosis, medicine and dietetics. Assessment should involve both children and their families or carers unless this is contraindicated due to safety concerns such as abuse or domestic violence (Mariano et al., 2013). Assessments in children and adolescents should be developmentally informed.
Psychological assessment should include a review of both eating disorder symptoms and comorbid psychiatric symptoms. The most common comorbid illnesses in adolescents with anorexia nervosa are anxiety disorders, including obsessive compulsive disorder (OCD) and major depressive disorder (MDD) (Milos et al., 2003; Steinhausen, 2002; Swanson et al., 2011). As in adult care, minimising mortality associated with anorexia nervosa is key to initial medical care. Some people will require admission for medical compromise (see Tables 3, 4 and 5). Medical instability is the key indicator for acute hospitalisation.
Guidelines for inpatient admission for children.

NB: These are a guide only and do not replace the need for individual clinical judgement.
For children, admission would generally be to a medical ward.
People may also require admission for: Uncontrolled eating disorder behaviour. Failure to respond to outpatient treatment. Severe psychiatric comorbidity.
Treatment
Current evidence supports outpatient care as the first-line treatment in adolescent anorexia nervosa, and there is a growing body of evidence to guide such care, particularly the role of family treatment (Lock, 2011; Smith and Cook-Cottone, 2011). There are, however, few studies into the role of inpatient treatment in anorexia nervosa. Hospitalisation of adolescents with anorexia nervosa for the management of acute medical instability (e.g. hypothermia, hypotension, electrolyte abnormalities and cardiac arrhythmias) is thought to be essential in preventing mortality associated with anorexia nervosa (Golden et al., 2003; Katzman, 2005).
Treatment, especially of children and adolescents with more severe disease, should be multidisciplinary, and include focused psychological therapy of the eating disorder and comorbid psychological problems. It should typically include psychoeducation of families, nutritional and medical therapy (at times pharmacotherapy) and may require case management involving schools and other agencies.
A total of nine RCTs have looked at treatment of anorexia nervosa exclusively in adolescents, and 12 RCTs have included adolescents (see Table 6). (Ball and Mitchell, 2004; Crisp et al., 1991; Eisler et al., 2000; Godart et al., 2012; Gowers et al., 2007; le Grange et al., 1992; Lock et al., 2005, 2010; Rhodes et al., 2008; Robin et al., 1994, 1999; Russell et al., 1987). All 12 of these studies included an outpatient intervention and all but one of these trials (Gowers et al., 2007) included an evaluation of some form of family intervention, including four trials that have compared different structures or doses of family interventions (Eisler et al., 2000; le Grange et al., 1992; Lock et al., 2005; Rhodes et al., 2008). Eight of the trials have looked at individual psychological interventions in anorexia nervosa, five in a direct comparison with family treatment (Ball and Mitchell, 2004; Lock et al., 2010; Robin et al., 1994, 1999; Russell et al., 1987), one comparing individual treatment with individual treatment augmented with family treatment (Godart et al., 2012) and two trials comparing individual outpatient treatment to inpatient treatment (Crisp et al., 1991; Gowers et al., 2007).
Randomised controlled psychotherapy trials in adolescent anorexia nervosa.
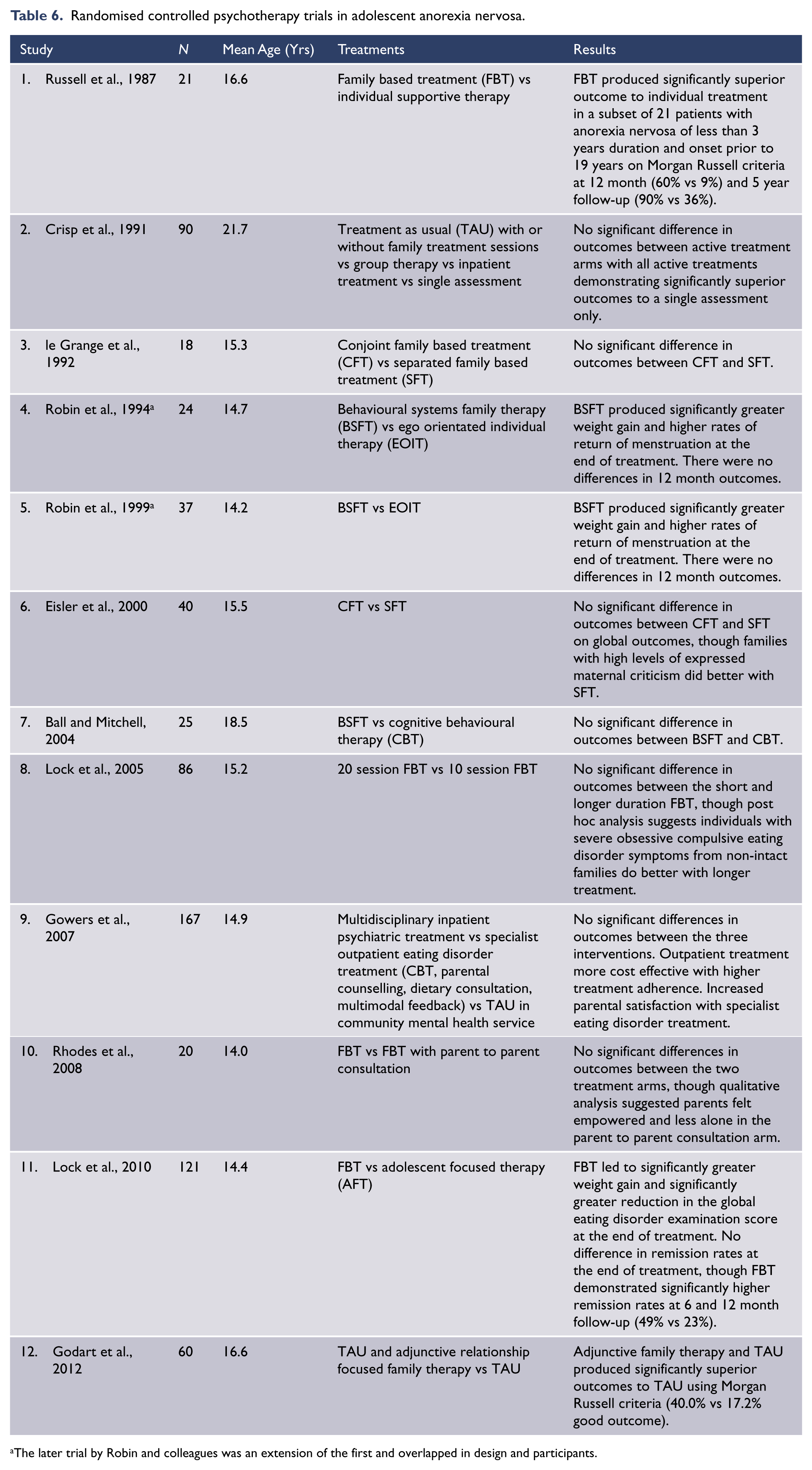
The later trial by Robin and colleagues was an extension of the first and overlapped in design and participants.
Psychological treatment
Family therapy
There have been 11 RCTs in adolescent anorexia nervosa that have included family interventions, though of these interventions only the one created by investigators from the Maudsley Hospital in the 1980s (Family Based Treatment – FBT) has been systematically investigated (Lock, 2011). Based on the outcomes of these RCTs and a recently published systematic review (Hay, 2013b), there is a clear and growing body of evidence that supports the efficacy of family treatment in adolescent anorexia nervosa, in particular family treatment that focuses on eating disorder behaviours and weight gain. Of these treatments FBT has been the most extensively studied and has not only demonstrated efficacy in the treatment of adolescent anorexia nervosa but also superiority to some types of individual therapy. There is a general consensus that FBT is now the first-line treatment for adolescents with anorexia nervosa who are aged less than 19 years and have a duration of illness of less than three years (le Grange et al., 2010; Lock, 2011; Russell et al., 1987).
Individual therapy
A total of eight RCTs in adolescent anorexia nervosa have included individual therapy interventions, including five that have compared individual therapy with family treatment. The three largest of these trials compared family treatment with ego orientated individual therapy (EOIT) or a modification of it called adolescent focused therapy (AFT). Both therapies are psycho-dynamically informed, individual, adolescent psychotherapies focusing on issues of adolescent development including autonomy, self-efficacy, individuation, assertiveness and psychological barriers to eating (Lock et al., 2010; Robin et al., 1995, 1999). While family treatment led to significantly higher weight gain and menstruation at treatment completion and improved remission rates 12 months after treatment completion in comparison to EOIT/AFT (Lock et al., 2010), individual treatment did lead to improvements in weight, menstruation and eating disorder pathology. In the two remaining RCTs comparing family and individual treatment the individual interventions included a supportive or ‘treatment as usual’ (TAU) intervention (Russell et al., 1987) and an eating disorder specific CBT intervention (Ball and Mitchell, 2004). This second study was hampered by small numbers and demonstrated no difference between CBT and family therapy. A recently published case series has suggested that CBT-E (enhanced cognitive behavioural therapy) may be effective in adolescent anorexia nervosa (Dalle Grave et al., 2013).
TAU has been examined in three RCTs. It has been described as supportive, educational and problem centred, focusing on both eating disorder specific issues and those related issues considered to prolong eating disorder behaviours. In the two studies comparing TAU with either FBT or TAU and family treatment, outcomes from TAU were inferior (Godart et al., 2012; Russell et al., 1987). In the third trial, outcomes from TAU were equivalent to specialist inpatient care (Crisp et al., 1991).
Positive prognostic factors for FBT are early weight gain (approximately 2kg in the first four weeks), while individuals with more severe eating disorder symptoms (assessed using the eating disorder examination (Fairburn, 2008)) and/or comorbid OCD appear to have a poorer outcome or are more likely to need additional sessions of FBT (Lock et al., 2005). In addition, high expressed emotion families were shown to do better with separated FBT (Eisler et al., 2000).
There is little evidence to guide clinicians as to which adolescents may do poorly with family therapy and better with an individual approach. Lock et al. (2010) have emphasised the important of compliance. Thus, a relative contraindication to family therapy is inability on the part of the family to commit to the treatment for whatever reason, including parental illness (Lock et al., 2010). While there is clearly much need for further studies of individual therapy in adolescents, particularly CBT, evidence to date suggests that in those adolescents and their families who do not respond to, or are unable to engage in, FBT, options for individual interventions that may be considered would include AFT and eating disorder specific CBT, with little evidence to support TAU.
Inpatient treatment
Hospitalisation of adolescents with anorexia nervosa for the management of acute medical instability (e.g. hypothermia, hypotension, electrolyte abnormalities and cardiac arrhythmias) is thought to be essential in preventing associated mortality (Golden et al., 2003; Katzman, 2005). However, the benefits of inpatient weight restoration and the assumption that hospital is the best venue for refeeding once medical stability has been achieved remain unsupported by current evidence.
There is little evidence to guide the role of inpatient care in adolescent anorexia nervosa. Expert consensus currently recommends outpatient therapy as the first-line treatment (NICE, 2004b). This position is supported by two RCTs comparing inpatient treatment for anorexia nervosa with a number of individual outpatient interventions (Crisp et al., 1991; Gowers et al., 2007). In both of these trials, adolescents with anorexia nervosa were admitted to psychiatric units with experience in treating eating disorders, though not restricted to the treatment of individuals with eating disorders. In both trials there was no significant difference in outcomes between inpatient treatment and outpatient individual therapy, though Gowers et al. (2010) reported improved treatment adherence and cost effectiveness with outpatient treatment. Patient satisfaction was highest with specialist treatment, either inpatient or outpatient (Gowers et al., 2010). Previous findings have suggested that outpatient care costs approximately 10% of the cost of inpatient care (Katzman et al., 2000).
Pharmacotherapy
There is insufficient evidence to recommend psychotropic medication in adolescents with anorexia nervosa. The use of anxiolytic or antidepressant medications to relieve symptoms should be done with caution.
Nutritional and medical treatment
There is a need to formulate appropriate nutritional goals for weight regain. There are, however, widespread views and practices in relation to weight goals in treatment. A UK and European survey of services identified a 24kg range of target-weights for a 14 year old girl of average height (Roots et al., 2006), albeit that prediction of physiological ‘normality’ is imprecise. BMI centiles can be utilised to predict the weight at which endocrinological normality will be achieved, but they need to be interpreted in the light of other physical assessments. Golden recommends a ‘target-weight’ between the 14th and 39th BMI percentile for age (Golden et al., 2008). Key and colleagues (2002) have promoted the use of pelvic ultrasound demonstration of ovarian follicles as an indicator of normal weight. This is likely to be achieved between the 13th and 30th BMI centile (Allan et al., 2010; Key et al., 2002; Madden et al., 2009). It is essential to note that as recommended healthy weight for height changes with age, BMI centile charts for children and adolescents must be utilised when determining weight goals in treatment. Charts are freely available from the World Health Organization and Centers for Disease Control and Prevention websites (CDC, 2013; WHO, 2013), as are the freely available computer program EpiInfo (CDC, 2008) and AnthroPlus (WHO, 2007), which can be used for calculating age related centiles.
Nutritional therapy in children and adolescents may be provided using regular food or special supplements and delivered orally or via nasogastric tubes to ensure timely provision of adequate nutrients (Rigaud et al., 2007; Zuercher et al., 2003). Gastrostomy feeding and parenteral nutrition have been utilised but should not be part of routine therapy (Diamanti et al., 2008; Findlay et al., 2011; Melchior and Corcos, 2009; Silber, 2008).
As with adults, initiation of nutritional therapy in significantly malnourished adolescents has risks, and should be undertaken carefully, by experienced clinicians who are cognisant of the risk of refeeding syndrome (MARSIPAN, 2011). While there is disagreement and lack of clear evidence regarding the optimal rate of feeding (orally or by nasogastric tube), it is generally accepted that for medically unstable adolescents the process should proceed cautiously, that ‘full feeds’ for longer-term weight recovery should be achieved within 5–7 days of initiation, usually with the use of nasogastric tube feeding to ensure that nutrients are delivered, and that the risk of hypoglycaemia and electrolyte shifts is minimised (Kohn et al., 2011; MARSIPAN, 2011; O’Connor and Goldin, 2011). Many groups routinely supplement with phosphate and thiamine during this period (Kohn et al., 2011; MARSIPAN, 2011). Monitoring of serum electrolytes and minerals is important during initiation of feeding. 3
Nutritional therapy needs to continue after the achievement of a healthy weight and discharge. This needs to involve regular monitoring of nutritional status (anthropometry, assessment of physiological function through measuring temperature, pulse, blood pressure and capillary refill, as well as intermittent measurement of biochemical parameters such as hormonal profiles and vitamin D). This should involve an experienced dietician who can assess nutrient intake and aid in setting appropriate dietary goals (MARSIPAN, 2011).
Sustained malnutrition in childhood and adolescence may be associated with a range of complications such as growth failure, pubertal delay, osteopaenia and osteoporosis, and in the longer-term increased risk of obesity, hypertension and heart disease are common sequelae. In order to minimise these risks long-term maintenance of healthy weight is important. Osteopaenia and osteoporosis are most likely to develop in girls who become malnourished early in pubertal development, and those with prolonged malnutrition and amenorrhoea (Swenne and Stridsberg, 2012; Turner et al., 2001). Worryingly, recovery from anorexia nervosa does not ensure resolution of osteopaenia (Wentz et al., 2007). It has been recognised that restoration of normal hormonal function via restoration and maintenance of normal weight is the best way of dealing with this problem (Misra and Klibanski, 2011). The common practice of prescription of the oral contraceptive pill in malnourished adolescent girls with amenorrhoea is not recommended because it does not improve bone density, and may provide false reassurance about physiological normality (Golden et al., 2002). Recently, use of physiological levels of oestrogen (via a hormonal patch) and progesterone in an RCT was associated with improvement in bone density, and may have a place in therapy (Misra and Klibanski, 2011; Misra et al., 2011).
Outcomes and prognosis
A small Swedish cohort of patients with adolescent-onset anorexia nervosa who were followed for 18 years experienced full recovery in 88%, despite more than a third having a persisting identifiable psychiatric disorder (Wentz et al., 2007). The outcomes for young onset eating disorders appear generally better than for older adolescent and adult onset eating disorders.
Transitioning from child and adolescent services into adult streams can be a potentially stressful and destabilising time for adolescents and their families or carers. Careful planning and appropriate levels of support are necessary to ensure this changeover proceeds smoothly.
Summary of recommendations

Severe and long-standing anorexia nervosa
Introduction
People with severe and long-standing anorexia nervosa 4 have one of the most challenging disorders in mental health care (Strober et al., 2010; Wonderlich et al., 2012). They have the highest mortality rate of any mental illness with a marked reduction in life expectancy (Steinhausen et al., 2002; Arcelus et al., 2011) and impose a heavy burden on health and other public services. Furthermore, they are often under or unemployed, on sickness benefits, suffer multiple medical complications (renal, liver, cardiac failure and osteoporosis), have repeated admissions to general and specialist medical facilities and are frequent users of primary care services with considerable strain on carers and families.
Robinson (2009) has argued that those with a severe and enduring eating disorder (SEED) need to be considered as having a serious illness which comprises not only psychiatric and medical sequelae but family, social and occupational complications as well. He conducted a series of qualitative studies in which he found that people with SEED scored similarly to severely depressed people on quality of life measures. More worrisome was the finding that life skill scores were on a par with people with schizophrenia. As a result he advocates a psychiatric rehabilitation model that comprises long-term follow-up, crisis intervention, specific psychological interventions, and attention to substance misuse. He also includes basic self-care needs which pay attention to nutrition, housing, financial issues, recreational activities as well as occupational ones (Robinson, 2009).
Evidence of efficacy for treatment approaches for people with severe and enduring anorexia nervosa (SE-AN) is very limited (Hay et al., 2012). To date, there has only been one RCT (Touyz et al., 2013) that specifically tested two psychological treatments for such individuals. Touyz and colleagues (2013) compared two standard treatments (SSCM and CBT) which were modified for those patients who suffered from a profound and persistent disorder. The findings suggest that CBT for severe and enduring (SE) illness was superior in reducing core symptoms at follow-up, but that both CBT-SE and SSCM-SE contributed to improvements over time in health-related quality of life, body weight, depression and motivation to change. These findings should not only provide hope for those suffering from severe and enduring anorexia nervosa but also stimulate interest in developing new psychosocial treatments.
Clinical and research implications
People with severe and enduring anorexia nervosa require a special treatment paradigm as they have usually experienced multiple treatment failures and present with a myriad of mental health and medical problems (Wonderlich et al., 2012). Goals of therapy need to be reconceptualised. The general clinical wisdom to date has been to reduce the focus on changing eating disorder symptoms and instead work collaboratively with the individual in a measured manner to reduce harm, maintain symptom stability and in particular enhance their quality of life. Because of their history of negative treatment experiences and repeated treatment failures, both the clinician and patient often share the experience of hopelessness and despair about the likelihood of meaningful change.
Treatment
Paradigm of management for severe and long-standing eating disorders
Adapted from approaches discussed by Strober et al. (2010), Williams et al. (2010), and Wonderlich et al. (2012).
It is important to collaboratively agree and articulate goals with the patient and (where appropriate) significant others, so as to create an environment of support and comfort. The individual is best served by a safe and secure treatment strategy that allows them to feel contained but yet allows for very gradual change. Elements of this framework include the following:
A prolonged period of assessment allowing the development of a shared understanding of the maintaining factors for their eating disorder and the identification of simple achievable goals that are embarked upon using extremely small steps.
Focus on improved adaptive function as a primary goal. Restoration of a normal weight or BMI may not be a primary focus of treatment unless desired by the individual. Refeeding is a collaborative enterprise so as to avoid unnecessary distress and further evasion or avoidance of therapy.
Changes to eating behaviours that improve nutrition and that are emphasised. This is done cognisant that most people with severe and long-standing anorexia nervosa can increase their caloric content to 1200 kilocalories per day without resultant weight gain or loss due to reduced energy metabolism or adaptations to starvation, although most people would lose weight on 1200 kilocalories. Careful encouragement so that any endeavours in this regard are recognised and the fear involved understood. There should be no reproach if aborted as this challenge can easily be visited again.
Assessment and encouragement of improved interpersonal function and social or other activity that enables the experience of feelings of pleasure or mastery. This can also stimulate the individual’s cognitive function. Independence and autonomy are actively fostered.
Careful monitoring of physical health by an empathic medical practitioner with decisions being made in a supportive and respectful manner, maintaining safety and avoiding crises wherever possible. Short-term admissions for medical stabilisation can, however, be life-saving.
Inclusion of meetings with family members and pertinent others providing education and ongoing support, with an aim to minimise anger and negative affect displayed towards the person with anorexia nervosa. It is also very important to give permission to loved ones to take leave of absence.
Some patients benefit from multidisciplinary case management services offered through public hospitals or others who work with other long-term psychiatric patients.
Hospitalisation and partial hospitalisation
Many people with long-standing disorders have multiple previous negative experiences of inpatient care (La Puma et al., 2009). Hospitalisation should be in order to achieve realistic, collaboratively agreed goals of care or for achievement of medical rescue based on achieving well defined medical parameters such as systolic BP above 90mmHg, pulse rate above 50 bpm, normal white cell count or albumin, etc. General inpatient psychiatric hospitals tend to be ill suited for this type of patient. Refeeding, if required, should be undertaken by a medical team with both the knowledge and experience of treating such patients (George et al., 2004). George et al. have reported a pilot day hospital program designed specifically for those with severe and enduring anorexia nervosa. Such programs that cater specifically for the needs of people with severe and enduring symptomatology are not only able to retain them in longer-term treatment but result in clinically significant changes, for example unexpected but encouraging requests to be transferred to a traditional day program as greater changes in weight and lifestyle are desired.
Countertransference
Treating someone with severe and enduring anorexia nervosa is an entirely different experience to more conventional treatment and the work can be long-term and not immediately rewarding. However, clinicians who work with people who have long-standing eating disorders consistently report in the literature the importance of never giving up hope or expectation of improvement (Theander, 1985).
In the view of the authors of this CPG, no therapist should ever be placed in a position to take on people who have long-standing eating disorders unless there is a clear will to want to do so, as this often turns into a formidable task with the therapist’s patience, anxieties and energies challenged, especially when the individual may be close to death. Outpatient teams usually comprise a psychiatrist and/or clinical psychologist, physician and dietitian. It is an absolute imperative that there is regular communication amongst team members and any medical and/or non-medical decisions are negotiated carefully with the individual. Therapists are reminded that the words of Tom Main in ‘The ailment’ (1957) remain relevant today: ‘The sufferer who frustrates a keen therapist by failing to improve is always in danger of meeting primitive human behaviour disguised as treatment’ (Main, 1957: 9).
Summary of recommendations

Section two: bulimia nervosa and binge eating disorder
Introduction
This section addresses management guidelines for the major eating disorders found in those who are not underweight, namely bulimia nervosa and binge eating disorder. In the DSM-5 (APA, 2013), those who do not meet full diagnostic criteria for bulimia nervosa or binge eating disorder because of low frequency and/or duration of behaviours may be categorised under ‘other specified’ or if there is another reason for not meeting criteria (e.g. ‘binge’ episodes are not objectively large) the diagnosis may be ‘unspecified feeding or eating disorder’. Whilst there is little evidence base for these related disorders it is likely that strategies effective for the full disorder may also be effective for sub-threshold disorders.
Several systematic reviews published in the past decade are in agreement on the evidence base for psychosocial and pharmacological treatments in bulimia nervosa and binge eating disorder and where more research is required (Aigner et al., 2011; Bulik et al., 2007; Hay, 2013b; Hay and Claudino, 2012; Hay et al., 2009; NICE, 2004b; Wilson and Zandberg, 2012).
Assessment
Assessment of people with bulimia nervosa or binge eating disorder should include inquiry into characteristic eating disorder: (a) behaviours, namely binge eating (uncontrolled episodes of overeating large amounts of food), weight control behaviours that may or may not be compensatory for binge eating (self-induced vomiting, laxative, and/or diuretic misuse), dietary restriction and/or fasting, compulsive or driven exercise and others such as insulin misuse in diabetic patients or misuse of diet pills or illicit stimulant drugs such as methamphetamine; and (b) cognitions of weight and/or shape overvaluation, and body image and eating preoccupations. People should be assessed for a past history of other eating disorders, especially anorexia nervosa, as this may be associated with increased likelihood of relapse and a poorer outcome in some (but not all) studies (Eckert et al., 1995; Goldschmidt et al., 2013; Mitchison et al., 2013; Vaz-Leal et al., 2011). Other common psychiatric co-morbidities are anxiety and mood disorder(s), impulse control and substance use disorder (Hudson et al., 2007; Lacey and Evans, 1986).
Physical examination is important as there is evidence of an increased risk of medical co-morbidities including, but not exclusive to, those associated with obesity, notably Type II diabetes, mellitus and hypertension (Kessler et al., 2013). Assessment should include measurement of weight, height, pulse rate and blood pressure and calculation of BMI. Serum biochemistry should be done to check for hypokalaemia and dehydration (effects of purging behaviours). Other assessments such as random glucose and cardiovascular examination and ECG should be done as medically indicated. Where primary psychological treatment is provided by a therapist without medical training, a general practitioner will need to assist with medical assessment and/or ongoing care.
Treatment
Psychological therapies
First-line treatment for bulimia nervosa and binge eating disorder in adults is an individual psychological therapy. The best evidence for such therapy is for CBT. CBT has been found to be superior consistently to wait-list control and most other psychological therapies for bulimia nervosa (NICE, 2004b). The evidence is weaker due to fewer trials in binge eating disorder where behavioural weight loss management is also effective in the short (Hay, 2013b; Hay et al., 2009) but not longer-term (Wilson et al., 2010).
A specific transdiagnostic enhanced therapy (CBT-E) developed by Fairburn (Fairburn, 2008) has been found more efficacious than other psychological approaches (Fairburn et al., 2009), although the specificity of CBT-E requires more evidence (Spielmans et al., 2013). As CBT-E is a well delineated and manualised form of CBT it is described here in detail. However, in accordance with evidence based practice, clinicians may apply variations of CBT and/or use other evidence based psychological therapies according to their expertise and individual preference. CBT/CBT-E has four well defined stages over 20 weeks. It begins with psychoeducation and a CBT informed formulation of the processes maintaining the person’s disorder, and uses it to identify problems to be targeted in therapy. This is followed by the introduction of monitoring of key behaviours, establishment of regular meals and snacks, and within session weighing (sessions 1–7 over one month). The second stage (sessions 8 and 9, weeks 5 and 6) is a ‘taking stock’, or reflection and review phase with revisiting and modification of the formulation as appropriate. The third stage (sessions 10–17, weeks 7–14) is a personalised program where the main mechanisms maintaining the eating disorder are addressed. This includes the utilisation of behavioural experiments to reduce problematic behaviours, particularly those associated with weight/shape overvaluation such as body checking, and an additional module addressing a core maintaining factor, namely mood intolerance. Stage 4 (sessions 18–20, weeks 15–20) looks to the future, ensuring improvements are maintained and includes relapse prevention. A broad version (CBT-Eb) has been developed to address additional core maintaining factors with three optional modules addressing interpersonal deficits, clinical perfectionism and low self-esteem if applicable. CBT-Eb has been found to have an advantage over the original ‘focused’ CBT-Ef for people with comorbid personality disorder or other complex psychopathology (Fairburn et al., 2009).
Self-help and scalability of CBT
Where access to a therapist is delayed or there are costs or other barriers, CBT can be provided as a first-step, or stand-alone therapy in guided self-help form. An example of such an evidence based self-help book that has been evaluated for delivery within 10 half-hour session times by Australian general practitioners (Banasiak et al., 2005) is Bulimia Nervosa and Binge Eating: A Guide to Recovery (Cooper, 1995). Pure or unguided self-help may be effective in binge eating disorder; however, it has poorer outcomes compared to guided self-help CBT or specialist provided CBT in bulimia nervosa (Hay, 2013b).
Wilson and Zandberg’s (2012) systematic review similarly supported self-help CBT as an effective, accessible and time and cost-efficient alternative to specialist delivery of CBT. Furthermore, it has been translated into delivery via telemedicine and the internet. They noted, however, that most CBT-guided self-help books have not kept up to date with developments in CBT such as CBT-Eb (see above).
Other psychological therapies
There is a small and weak evidence base for interpersonal psychotherapy and dialectical behaviour therapy in both bulimia nervosa and binge eating disorder, and mindfulness in binge eating disorder (Kristeller and Wolever, 2011). Where therapists have expertise in these therapies and not in CBT and a CBT-trained therapist is not accessible then it may be appropriate to use either of these for adults. Findings are mixed for FBT in older adolescents or adults and, unlike in anorexia nervosa, FBT would not be first-line in bulimia nervosa or binge eating disorder (le Grange et al., 2007; Schmidt et al., 2007).
Pharmacotherapy
RCTs and meta-analysis have found that tricyclic antidepressants may be efficacious for people with bulimia nervosa (Flament et al., 2012; Hay and Claudino, 2012) but adverse effects limit clinical utility. In contrast, high dose fluoxetine (60mg/day) is effective for people with bulimia nervosa and this or other SSRI antidepressants are effective for both bulimia nervosa and binge eating disorder. The antiepileptic topiramate also is effective in both conditions and is associated with weight loss. However, topiramate may cause problematic side effects such as paresthesias and taste perversion (Arbaizar et al., 2008; Hay and Claudino, 2012).
Where psychological therapy is not available, antidepressants or antiepileptic medication such as topiramate 5 may be used (Flament et al., 2012; Hay and Claudino, 2012). However, trials of drug alone treatments have seldom followed participants up in the long-term, and therefore how long the medication should be continued for is unclear. High attrition rates and binge eating abstinence rates have consistently been found to be lower for drug alone treatments than when combined with CBT (Flament et al., 2012; Hay and Claudino, 2012). Trials in bulimia nervosa and binge eating disorder (Flament et al., 2012; Hay and Claudino, 2012) find an additive benefit for combined psychological and pharmacological treatment, but findings are inconsistent. Thus, when people have limited response to psychotherapy alone, or they have a comorbid mood disorder such as depression, pharmacotherapy may have a role as an adjunctive treatment.
Indicators for admission
The majority of people with bulimia nervosa may be treated as outpatients. Although evidence is lacking, people who are not responding to outpatient care or otherwise have an increased risk (e.g. because of suicidality or pregnancy) may benefit from the increased intensity of therapy and eating supervision available in an inpatient or day patient unit.
Medical assessment
Most medical problems in people with bulimia nervosa occur as a result of purging behaviours. Table 4 (above) lists these and their management. A dental evaluation should be considered if self-induced vomiting has been a prominent symptom. An increasing problem is comorbid weight disorder and metabolic syndrome, both of which may require further medical assessment and treatment (see below).
Management of weight disorder
Many people who have a binge eating disorder and increasing numbers with bulimia nervosa are also obese with consequential medical complications. Behavioural weight loss therapy for those with bulimia nervosa may be as effective as CBT in reducing binge eating and more effective in attaining weight loss in the short-term, but not the longer-term (Wilson et al., 2010). Similarly, topiramate and/or orlistat may aid weight loss and binge eating in the short-term (Arbaizar et al., 2008; Golay et al., 2005; Grilo et al., 2005).
Where comorbid obesity is problematic some people may benefit from weight loss management strategies but evidence is weak for any specific approach (Bulik et al., 2012).
Outcomes for bulimia nervosa and binge eating disorder
Most people with bulimia nervosa, binge eating disorder or other specified feeding or eating disorders (OSFED) experience a good outcome in long-term follow-up studies, with 50% or more free of symptoms at five years or more (Fairburn et al., 2000; Steinhausen and Weber, 2009). Steinhausen and Weber conducted a quantitative analysis of outcome data from 79 studies of bulimia nervosa (Steinhausen and Weber, 2009). He reported a recovery rate of 45%, whilst 27% of patients improved considerably and 23% had a chronic protracted course and a crude mortality rate of 0.32%. There was a 10–32% mean rate of crossover to other eating disorder diagnoses, most commonly to OSFED, followed by anorexia nervosa. A low rate of conversion to binge eating disorder may have been partially because the term had not been introduced when many of the older outcome measures were performed. Childhood obesity, substance use disorder and having a personality disturbance have most consistently been poor predictors of outcomes in bulimia nervosa, although it has been difficult to establish such predictors across studies (NICE, 2004a; Steinhausen and Weber, 2009). The long delays from illness onset to presentation likely contribute to poorer outcomes but there is, to our knowledge, no direct evidence for this.
Summary of recommendations
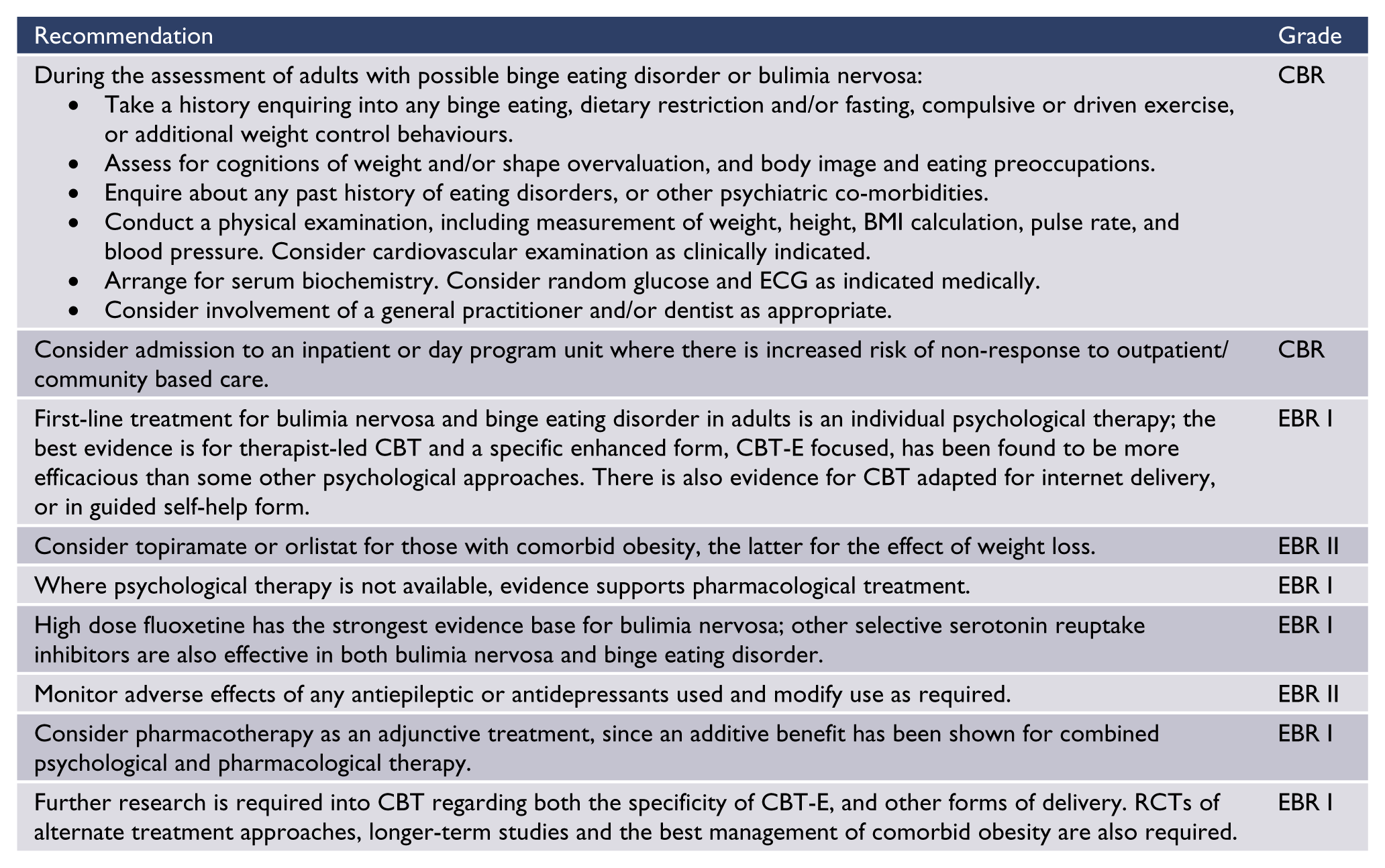
Section three: avoidant/restrictive food intake disorder (ARFID)
Introduction
ARFID (APA, 2013) is a new disorder to DSM-5. It replaces and extends the DSM-IV diagnosis of feeding disorder of infancy and early childhood as well as DSM-IV somatoform disorders that were characterised by phobic food avoidance. Due to general paucity of data and absence of published data concerning this condition in older adolescents or adults, consideration of ARFID in this guideline is confined to the following brief overview.
Overview
The key diagnostic features of ARFID are restriction of food intake accompanied by one of the following: significant weight loss; significant nutritional deficiency; marked interference with social functioning; or dependence on enteral feeds or oral supplements, in the absence of body image concerns. It may occur at any age. Data from three recently published studies on early onset eating disorder has shown that between 21.2% and 35.2% of children aged 12 years and under presenting with weight loss and deliberate food avoidance do not report abnormal body image or fear of weight gain. These children presented with similar physical complications of their malnutrition and similar rates of psychiatric comorbidity to children meeting diagnostic criteria for anorexia nervosa. To date there have been no published studies to guide appropriate treatment interventions or inform prognosis for this group (Madden et al., 2009; Nicholls et al., 2011a; Pinhas et al., 2011).
Section four: future research in treating eating disorders
The most important challenge for future research is the elucidation of assessment and treatment of the newly introduced disorders, especially ARFID. Second, although CBT is a first-line therapy for people with bulimia nervosa, further evaluation of CBT and alternate therapies is needed. This is particularly needed for anorexia nervosa (where in adults there is no clear first-line psychological therapy) and those with additional complex problems such as borderline personality disorder for which broader treatment approaches have been found to be associated with improved outcomes (Fairburn et al., 2009). More randomised controlled trials of approaches with an emerging evidence base, for example acceptance and commitment therapy (ACT) and psychodynamic therapy, and novel biological treatments such as neuromodulation and deep brain stimulation, are also needed.
From a psychopharmacological perspective, greater clarification of the neurobiological basis of eating disorders and mechanisms of action of treatments may help elucidate other treatment options, for example in bulimia nervosa antidepressants appear to have actions on satiety separate to their effects on mood modulation. In addition, data on the combination or augmentation of psychological and pharmacological approaches may guide further management of those who do not respond to first-line treatments.
More information on the long-term efficacy of treatments would be gained by longer-term trials and post-treatment follow-up, since many studies last less than six months. Inclusion and subgroup analyses of more heterogeneous and clinically complex patients (e.g. those with severe co-morbidities, as seen clinically) would expand the data on effectiveness of what treatments work and for whom.
In bulimia nervosa even after treatment is successful in reducing behaviours such as binge eating and purging, abstinence rates may remain low. Since the long-term outcome is likely better if abstinence is achieved, improvement of such rates is important. Further identification of features differentiating those who achieve remission from those who remain symptomatic may clarify factors moderating outcome.
Further research is also needed into non-specialist therapist-guided self-help, given that in comparison to therapist-led CBT, adherence to therapy and outcomes may not be as good, especially in treatment of bulimia nervosa; more specificity about the provision of such guidance would also be helpful. In binge eating disorder associated with obesity, energy restriction has, in the short-term, been associated with weight loss; however, longer-term studies of weight reduction management in this population are required.
Future studies should also address specific treatment needs and approaches as they may apply to groups underrepresented in current research. This includes, but is not limited to males and to Aboriginal and Torres Strait Islander peoples and Māori.
Conclusion
Assessment and management of people with anorexia nervosa should be multidisciplinary and include specific specialist psychological therapies and family based treatments in younger people. Recommendations for treatment in this CPG were based on evidence (see Table 7) of variable levels. The evidence base for therapies is stronger in bulimia nervosa and binge eating disorder where a specific transdiagnostic CBT has a high level of evidence. CBT can also be provided in less intensive guided self-help and online forms for less severe eating disorders. The majority of people can be treated as an outpatient with inpatient or day patient care needed for more severe illness, and particularly low weight people with anorexia nervosa. In the absence of evidence, trials evaluating treatments for ARFID are urgently needed to guide clinical practice.
Role and evidence base support for specific treatments in eating disorders.
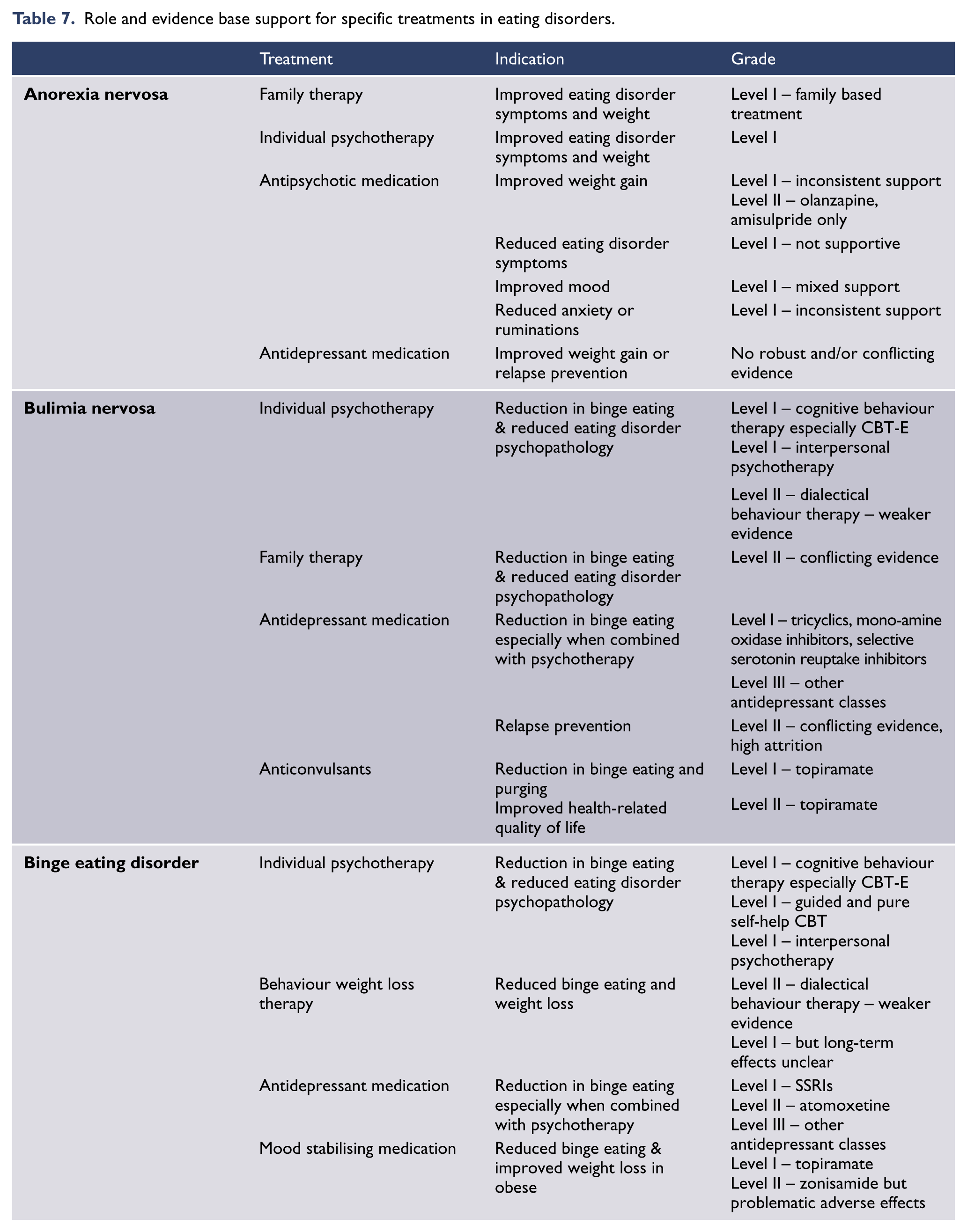
Footnotes
Acknowledgements
The following people and organisations contributed information that was used in the RANZCP Clinical Practice Guideline for Eating Disorders.
Feedback received during Public and Stakeholder consultations
Dr Richard Benjamin
Ms Claire Diffey
Dr Alby Elias
Dr Susanna Every-Palmer
Dr Brad Hayhow
Ms Amy Lewis
Dr Andrew Neilsen
Associate Professor Thinh Nguyen
Associate Professor Elizabeth Rieger
Dr Maugan Rimmer
Dr Beth Shelton
Associate Professor Victor Storm
Ms Emma Sutich
Dr Frances Wilson
Dietitians Association of Australia
Special Acknowledgements
Professor Mark Oakley Browne
Dr Roger Mysliwiec
Adjunct Research Fellow Hunna Watson
RANZCP Project Team
Ms Joyce Goh, Project Manager
Ms Helen Walters, Project Officer
Disclaimer
Compiled for the Royal Australian and New Zealand College of Psychiatrists (RANZCP), this information and advice is based on current medical knowledge and practice as at the date of publication. It is intended as a general guide only, not as a substitute for individual medical advice. The RANZCP and its employees accept no responsibility for any consequences arising from relying upon the information contained in this publication.
Funding
The Clinical Practice Guideline for Eating Disorders was funded by the Royal Australian and New Zealand College of Psychiatry.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
