Abstract
Objective:
To determine the association between insomnia, obstructive sleep apnoea (OSA), and comorbid insomnia-OSA and depression, while controlling for relevant lifestyle and health factors, among a large population-based sample of US adults.
Method:
We examined a sample of 11,329 adults (≥18 years) who participated in the National Health and Nutrition Examination Survey (NHANES) during the years 2005–2008. Insomnia was classified via a combination of self-reported positive physician diagnosis and high-frequency ‘trouble falling asleep’, ‘waking during the night’, ‘waking too early’, and ‘feeling unrested during the day’. OSA was classified as a combination of a positive response to a physician-diagnosed condition, in addition to a high frequency of self-reported nocturnal ‘snoring’, ‘snorting/stopping breathing’ and ‘feeling overly sleepy during the day’. Comorbid insomnia-OSA was further assessed by combining a positive response to either insomnia (all), or sleep apnoea (all), as classified above. Depressive symptomology was assessed by the Patient Health Questionnaire-9 (PHQ-9), with scores of >9 used to indicate depression. Odds ratios (ORs) and 95% confidence intervals (CIs) for sleep disorders and depression were attained from logistic regression modelling adjusted for sex, age, poverty level, smoking status and body mass index (BMI).
Results:
Those who reported insomnia, OSA or comorbid insomnia-OSA symptoms reported higher rates of depression (33.6%, 22.2%, 27.1%, respectively), and consistently reported poorer physical health outcomes than those who did not report sleep disorders. After adjusting for sex, age, poverty level, smoking status and BMI (kg/m2), insomnia (OR 6.57, 95% CI 3.89-11.11), OSA (OR 5.14, 95% CI 3.14–8.41) and comorbid insomnia-OSA (OR 6.67, 95% CI 4.44–10.00) were associated with an increased likelihood of reporting depression.
Conclusions:
Insomnia, OSA and comorbid insomnia-OSA are associated with significant depressive symptomology among this large population-based sample of adults.
Keywords
Introduction
Sleep disorders represent a significant social and personal burden, with research suggesting that as many as 10% of the population experience these problems during their lifetime (Ram et al., 2010). It is estimated that as many as 50–60 million Americans have insomnia symptoms annually (Hossain, 2002). Similarly, population-based research has suggested that as many as 5% of adults in western countries meet classification for clinically significant obstructive sleep apnoea (OSA) (Davies and Stradling, 1996). The implications for these disorders are substantial, both in terms of direct and indirect costs for health care systems and individuals. Both insomnia and OSA are associated with poorer occupational and interpersonal functioning, increased medical expenditure and increased instances of work absenteeism and reduced productivity in the workplace (Hossain, 2002). In addition, both disorders have previously been linked to increased medical and psychiatric comorbidity (such as cardiovascular risk) and all-cause mortality via a combination of these factors (Vgontzas et al., 2010; Yaggi et al., 2005).
OSA is a disorder characterised by repeated occlusions to the upper airway during periods of nocturnal sleep (Punjabi, 2008), resulting in snoring, nocturnal hypoxemia, fragmented sleep and daytime symptoms such as irritability, reduced affect, and reduced quality of life (Engleman and Douglas, 2004). Insomnia is defined as the experience of subjective and objective poor or inadequate sleep despite ample time in bed (Summers et al., 2006). Patients may also report daytime symptoms (e.g. daytime fatigue and confusion), as well as deficits in interpersonal functioning (e.g. malaise, reduced motivation) (Summers et al., 2006). Research conducted among individuals attending sleep clinics has shown that as many as 26% of patients describe themselves as suffering from depression, and up to 67% of those patients reported an episode of major depression in the previous 5-year period (Mosko et al., 1989). Concordant research conducted by Vandeputte and de Weerd (2003) assessed the association between depression and sleep disorders among a sample of patients presenting at a sleep clinic. It was demonstrated that the overall prevalence of depressive symptoms among those with a sleep disorder was high, with 60.5% of those with insomnia and 41% of those with OSA indicating significant levels of depression (Vandeputte and de Weerd, 2003).
Although both insomnia and OSA are often considered to represent mutually exclusive disorders, particularly in terms of clinical presentation, new evidence suggests that a high level of comorbidity exists between these two conditions (Smith et al., 2004). Indeed, some studies cite a concordance rate of as high as 50% (Krakow et al., 2001); however, others report more conservative numbers (Smith et al., 2004). Although speculative, it has been suggested that the sustained physiological arousal characteristic of insomnia may act to increase nocturnal heart rate and sympathetic processes, thereby exacerbating existing symptoms of OSA. Concordantly, periods of nocturnal hypoventilation and hypoxemia experienced by OSA patients may act to increase nocturnal sympathetic processes, thereby mimicking and promoting a negative feedback loop which acts to develop and sustain insomnia pathology (Luyster et al., 2010).
Despite the apparent relationship between sleep disorders and depression among clinical samples, it is unclear if these findings are represented at a population level when assessing a non-clinical sample, or whether these findings are comparable among differing diagnoses using a heterogeneous sample. Moreover, there is little indication of the impact of OSA and insomnia on depression among a representative sample, as the disorders are often assessed in isolation (Schroder and O’Hara, 2005), or as a function of the other (Smith et al., 2004). Moreover, despite increasing evidence suggesting the high rates of concordance between these disorders, and given the established individual associations with depression, there is a paucity of information pertaining to the association between these combined associations and depressive symptomology. Indeed, of the population-based research available for analysis, generalisability of findings is often limited due to specific patient groups or samples used, and by use of individual assessment of these disorders. For example, research conducted by Ohayon (2003) aimed to assess the association between depression and sleep-related breathing disorders in the general population of Europe (spanning five countries). Assessment of 18,940 adults revealed that 18% of individuals who met criteria for major depressive disorder were also identified as having a Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) breathing-related sleep disorder. Moreover, multivariate modelling demonstrated that those with major depressive disorder were five times more likely to have a DSM-IV diagnosis of a sleep-related breathing disorder (Ohayon, 2003). Utilising the National Health and Nutrition Examination Survey (NHANES), Wheaton and colleagues (2012) explored the association between OSA-specific symptom items and depressive disorders. The findings indicated that men with physician-diagnosed OSA were two times more likely to report depressive symptoms, while for women the likelihood of having depression was increased by fivefold for those with OSA (Wheaton et al., 2012). Of the available literature, however, comparable community-based research has indicated that a significant proportion (20%) of those with insomnia also reported concordant depressive symptomology (Taylor et al., 2005).
Both OSA and insomnia are implicated in the expression of depression both among clinical and non-clinical samples, and represent a significant mediating factor in successful treatment outcomes. However, at present, only a small number of studies exist which investigate these combined associations among a single large population-based sample pool. In order to assess this relationship in light of a number of health and lifestyle factors, we utilised data from the NHANES dataset across two points: 2005–2006 and 2007–2008.
Method
Study population
NHANES is a stratified, cross-sectional, population-based sample of non-institutionalised individuals, spanning all age groups. Approximately 5000 individual assessments are performed each year, aimed at assessing aspects of health (disease burden) and nutrition. A more detailed description of the NHANES assessment protocol is available elsewhere. NHANES 2005–2006 was the first time during which questionnaire data were used to assess the presence of both physician-diagnosed sleep disorders and sleep-related subjective questionnaire items. All health interviews were conducted in respondents’ homes, and health measurements were performed in specially designed and equipped mobile centres located throughout the country. Interview teams conducting these assessments consisted of physicians, medical and health technicians, and trained dietary and health interviewers. These items were also included for the 2007–2008 testing period. For the purpose of this study, adults (aged ≥18 years) who participated in NHANES at two sampling time points (2005–2006 and 2007–2008) were included for analysis, resulting in a total sample of 11,329 individuals. The overall response rate for these surveys was 76.4% (Centers for Disease Control and Prevention, 2014). NHANES received approval from the National Center for Health Statistics Research Ethics Review Board and written informed consent was obtained for all participants.
Insomnia
The presence of insomnia was first determined by a positive response to a questionnaire item referring to physician-diagnosed insomnia (n=185). Insomnia was also separately classified as a combination of frequency questionnaire data referring to subjective difficulties with sleep. For the purpose of this study, a response to items of ‘often’ or ‘almost always’ for all four questions was classified to indicate the presence of insomnia symptoms (n=764). The use of frequency indicators as markers of the disorder has previously been employed in population samples (Karacan et al., 1976). Insomnia was then finally classified as the sum of a positive indication of diagnoses, and the sum of individuals classified as having insomnia via the application of subjective symptoms. Those individuals who met criteria for both objective and subjective insomnia classification were counted only once in the original files and thus removed from further analysis (n=58). Removing these cases avoided duplication in the subsequent analysis. After merging both the depression and sleep databases, those for whom depression information were not available (n=56) were excluded from further analysis. Using this criteria, a total of n=835 cases were identified.
Sleep apnoea
The presence of sleep apnoea was first determined by a positive response to a questionnaire item referring to physician-diagnosed sleep apnoea (n=481). The presence of sleep apnoea was further separately indicated via frequency questionnaire items referring to common symptoms, such as ‘how often do you snore’. For the purpose of this study, a positive response to variable items ‘occasionally’ and ‘frequently’ (nocturnal symptoms), and ‘often’ and ‘almost always’ (daytime sleepiness) on all three questions was considered to be indicative of sleep apnoea symptoms (n=290). The presence of sleep apnoea was then finally classified as the sum of positive response to disease diagnosis and positive response to subjective symptom items. Assessment of symptoms utilising frequency data has previously been used and validated among clinical (Netzer et al., 2003; Rowley et al., 2000) and population samples (Kapur et al., 2002), and is considered an effective and reliable screening method for identifying those at risk for the disorder (Maislin et al., 1995). Those individuals who met criteria for both objective and subjective sleep apnoea classification in the original files were counted once (n=83). Removing these cases avoided duplication in the subsequent analysis. After merging both the depression and sleep databases, those for whom depression information was not available (n=24) were excluded from analysis. Using this classification, a total of n=664 cases were identified.
Comorbid insomnia and OSA
The presence of comorbid insomnia and OSA was assessed by combining a positive response to either insomnia (all), or sleep apnoea (all), as classified above. Those who met criteria for both insomnia and sleep apnoea (n=136) were counted only once for analysis. Using this criteria, a total of n=1363 cases were identified. Of those who met the insomnia criteria (835), 136 (16.3%) also met criteria for comorbid OSA. Similarly, of those meeting the OSA criteria (664), 136 (20.5%) cases were comorbid with insomnia (Figure 1).
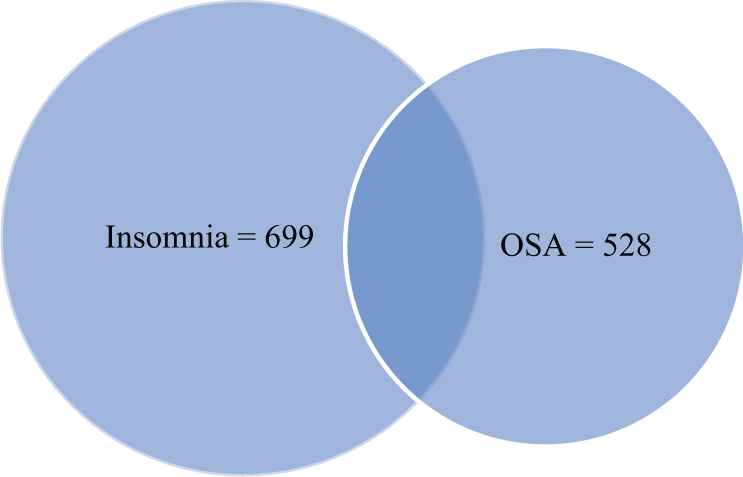
Proportional Venn diagram of the unweighted sample showing respondents with insomnia, OSA or both (comorbid).
Depression assessment
Depressive symptoms were assessed via the Patient Health Questionnaire-9 (PHQ-9). The PHQ-9 is a self-administered questionnaire aimed at assessing the presence of common mental disorders, as derived from the DSM-IV classification system. The assessment tool includes nine frequency questionnaire items (0 = not at all, 3 = nearly every day) pertaining to aspects of anhedonia, depressed mood, difficulty concentrating, trouble sleeping, and guilt or worthlessness, among others (Sidebottom et al., 2012). The PHQ-9 is considered an effective tool for assessing depressive symptomology and is considered as a reliable tool for assessing instances of both major and sub-threshold depression among population samples (Kroenke and Spitzer, 2002), A total PHQ-9 score of >9 (out of possible 27) has been indicated for use in the event of single screening assessments (Kroenke and Spitzer, 2002). For the current study, a PHQ-9 score of >9 was used to indicate the presence of depression.
Lifestyle and health information
Information regarding health and lifestyle factors was collected via home interview at the time of assessments. Demographic variables included: sex, race/ethnicity (e.g. Hispanic, non-Hispanic, white), age group (18–24, 25–34, 35–44, 45–54, 55–64, ≥65 years), education level (student, less than 9th grade, 9–11th grade, high school graduate, some college, college graduate and above), marital status (married, widowed, divorced, separated, never married, living with partner), poverty level (below 100%, 100% to below 200%, 200% or higher), and smoking status (current or former). Sedentary behaviour was indicated by self-reported daily average TV time (hours) and computer time (hours).
Information regarding health factors was obtained via self-report and by laboratory data. The presence of diabetes, cancer and asthma was assessed utilising a self-report question asking the participant whether they have ever been told by a doctor or other health professional that they have diabetes or sugar diabetes, with responses categorised into yes, no and borderline or pre-diabetes. The presence of cancer and asthma was similarly assessed by asking the participant if they had been previously diagnosed with these conditions. The presence of cardiovascular disease (CVD) was classified as a positive response to the questionnaire item asking if participants had ever been told by a doctor or other health professional that they have coronary heart disease, congestive heart failure, angina/angina pectoris, heart attack or stroke. The presence of high blood pressure was similarly classified as a positive response to the question ‘have you ever been told by a doctor or other health professional that they have high blood pressure?’.
Height and weight measurements were obtained at the time of the assessments via a mobile examination centre, and body mass index (BMI) was calculated as weight/height squared (kg/m2). BMI categories were defined as underweight (BMI ≤18.5), normal (BMI >18.5 to <25), overweight (BMI >25 to <30), and obese (BMI ≥30) for all participants.
C-reactive protein (CRP) levels (ml/dL) were quantified using latex-enhanced nephelometry; analyses were conducted in the Department of Laboratory Medicine, Immunology Division at the University of Washington Medical Center.
Dietary intake in the NHANES 99–00 is based on recollection of foods eaten the previous day by the respondent, coupled with known nutritional content of each of these foods (24-hour recall). The total daily dietary intakes for magnesium were derived from the 24-hour recall information about foods eaten the previous day. These interviews were conducted by trained personnel, and food intake information was coded using the USDA Food and Nutrient Database for Dietary Studies 3.0 (FNDDS 3.0). This database was then used to produce nutrient intake values for magnesium (other nutrients not used).
Statistical analysis
Demographics variables and sleep data were merged into a master file and additional variables were created for analysis. Weighting was applied to account for the stratified multistage survey design, oversampling and non-response and were constructed according to the CDC guidelines (Centers for Disease Control, 2013) . Differences in characteristics between those with and without sleep disorders (insomnia or OSA) were analysed using appropriate parametric tests where data were normally distributed, and non-parametric statistical tests for categorical or non-normal data. Odds ratios (ORs) with 95% confidence intervals (CIs) were determined using binary logistic regression models for categorical outcomes to investigate the association between the presence of sleep disorders and depression. In all models, sleep disorder (insomnia, OSA or comorbid insomnia/OSA) was the exposure variable. Sex, age, poverty level, smoking status, BMI category, sedentary behaviours (expressed as average daily TV time and computer use) and CRP levels were tested sequentially, and potential confounders and effect modifiers were checked in all statistical models. Covariates that significantly influenced the outcome variable were retained and applied for all final statistical models (significance level set at p=0.05). Stata 12 (survey procedures) (StataCorp., College Station, TX, USA) was used for all statistical analyses.
Results
Insomnia
Unweighted population characteristics for those with insomnia and OSA are presented in Table 1. Overall, 835 individuals were classified as having insomnia, and women were almost twice as likely to report insomnia as men (66% vs 34%, respectively). Those who fell within the 45–54 years age bracket were most likely to report insomnia, with those in the 18–24 age group least likely to report these symptoms. In regard to ethnicity, non-Hispanic white adults were more than twice as likely to report insomnia as those who identified themselves as non-Hispanic black (50.2% vs 20.2%), and were more than five times more likely to report insomnia than Mexican Americans (50.2% vs 15%). Those individuals who identified themselves as ‘married’ were most likely to report insomnia, with those who were ‘separated’ reporting the least symptoms. Moreover, those regarded in the highest income bracket (above 200%) reported more insomnia than those in the 100–200% or <100% brackets. Those who reported never smoking were more likely to report insomnia than those who identified as either a ‘former or current’ smoker. In regard to body composition, those who were overweight or obese reported higher rates of insomnia than those who were classified as within the normal BMI range, or those who were considered underweight. Finally, although a greater proportion of those with insomnia reported ‘normal’ CRP levels (0–1 mg/dL), almost one in five reported clinically high levels (>1 mg/dL) (16.9%) (Table 1).
Demographic characteristics by type of sleep disorder, 2005–2008, aged 18 years and above (unweighted).
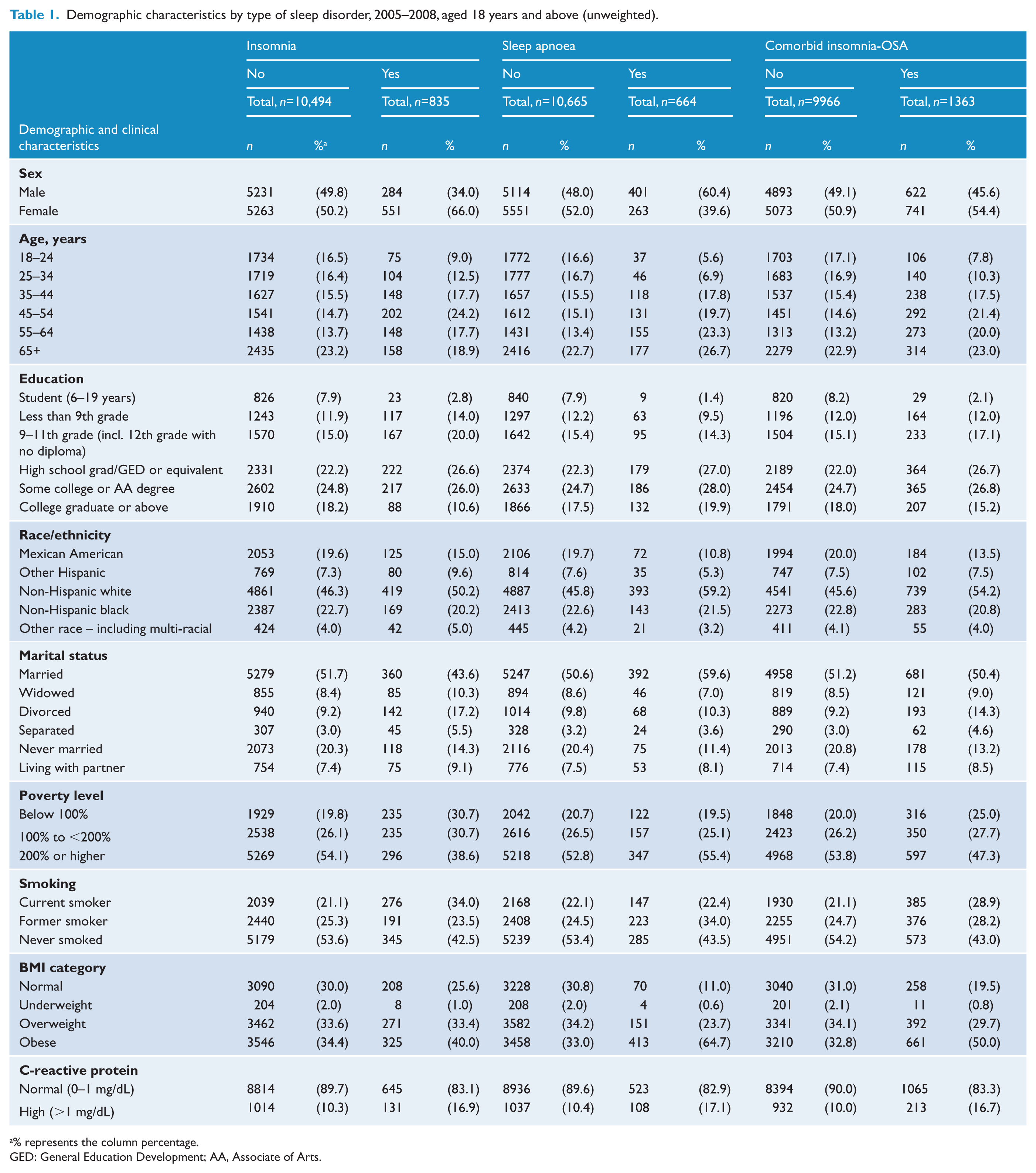
% represents the column percentage.
GED: General Education Development; AA, Associate of Arts.
Univariate analysis of weighted data revealed that those with insomnia were significantly older at the time of assessment, had a greater overall weight and BMI, and were more likely to report a greater time spent in sedentary activities than those without insomnia (all p<0.001) (Table 2). Similarly, individuals with insomnia were more likely to report depression than those without insomnia, and were more likely to report negative health status, such as instances of asthma, diabetes, high blood pressure and cancer (all p<0.001) (Table 3).
Clinical characteristics and sedentary behaviour by sleep disorder, aged 18 years and above (weighted) a .
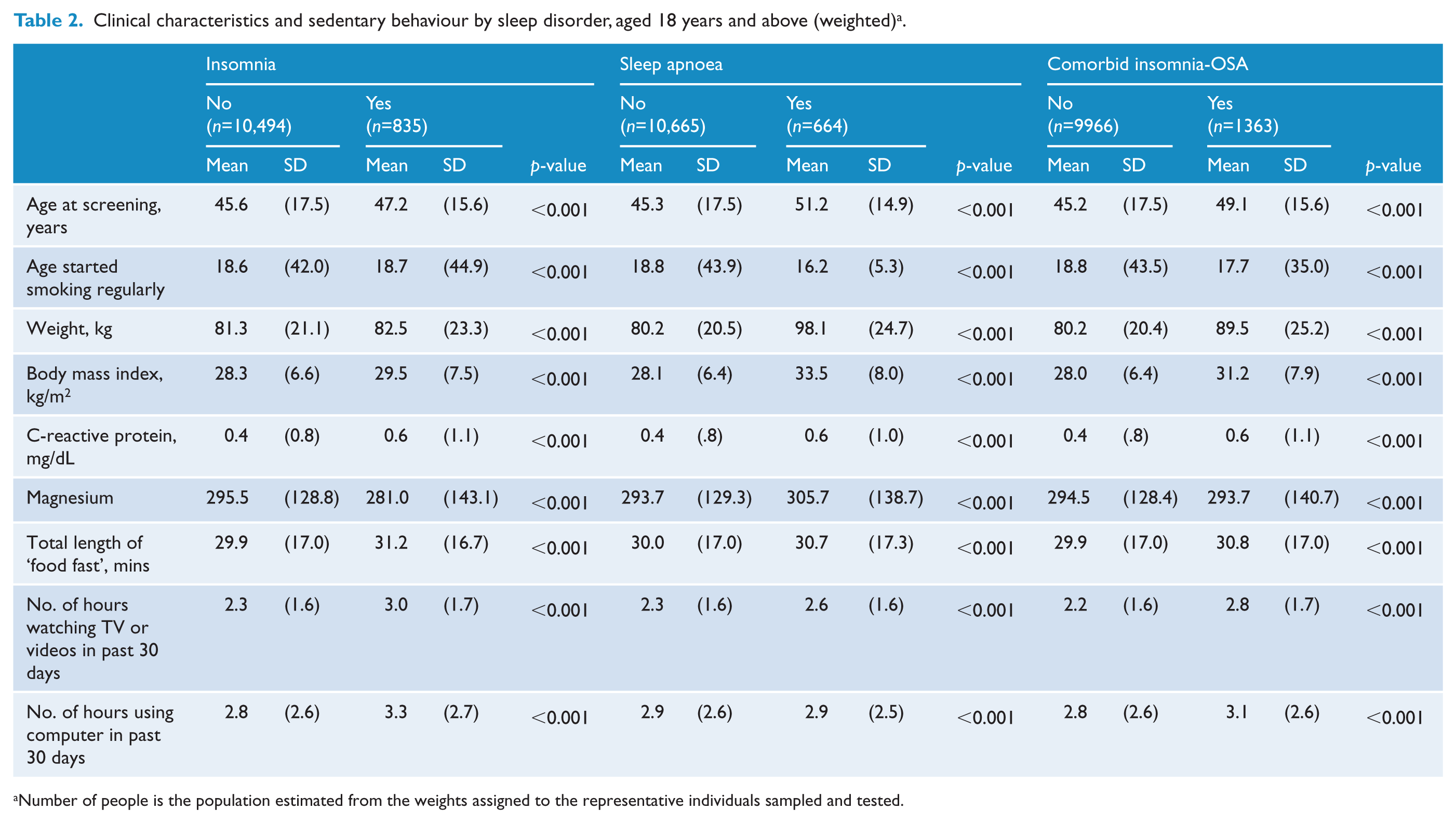
Number of people is the population estimated from the weights assigned to the representative individuals sampled and tested.
Medical conditions by sleep disorder, aged 18 years and above (unweighted).
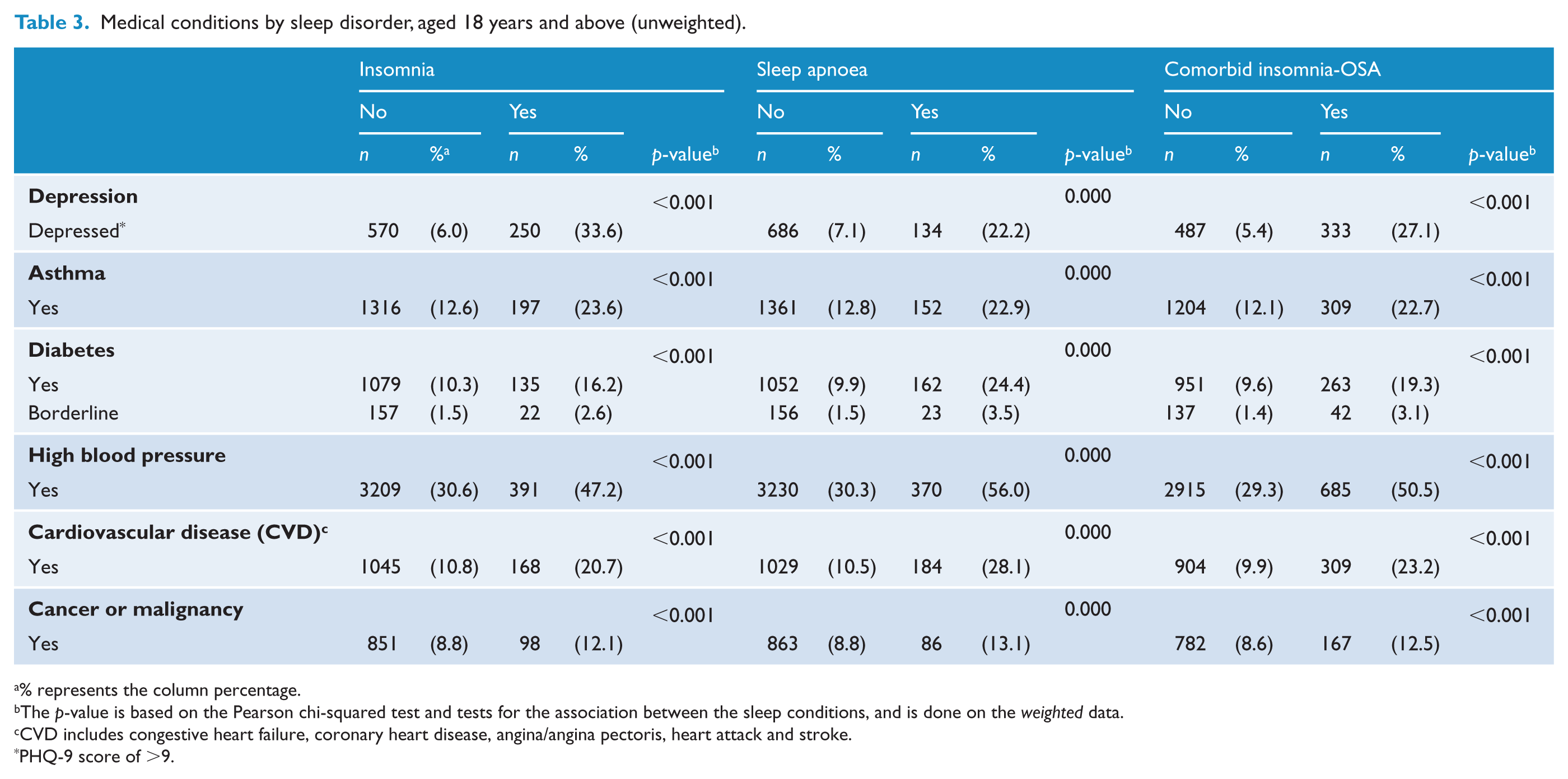
% represents the column percentage.
The p-value is based on the Pearson chi-squared test and tests for the association between the sleep conditions, and is done on the weighted data.
CVD includes congestive heart failure, coronary heart disease, angina/angina pectoris, heart attack and stroke.
PHQ-9 score of >9.
After adjusting for age, sex, poverty level, smoking status and BMI, insomnia was associated with a 6.57-fold increased likelihood of reporting depression (OR 6.57, 95% CI 3.89-11.11). These findings were not explained by CRP levels or sedentary behaviour (Table 4).
Results of multivariable logistic regression analyses assessing predictors of depression including insomnia.
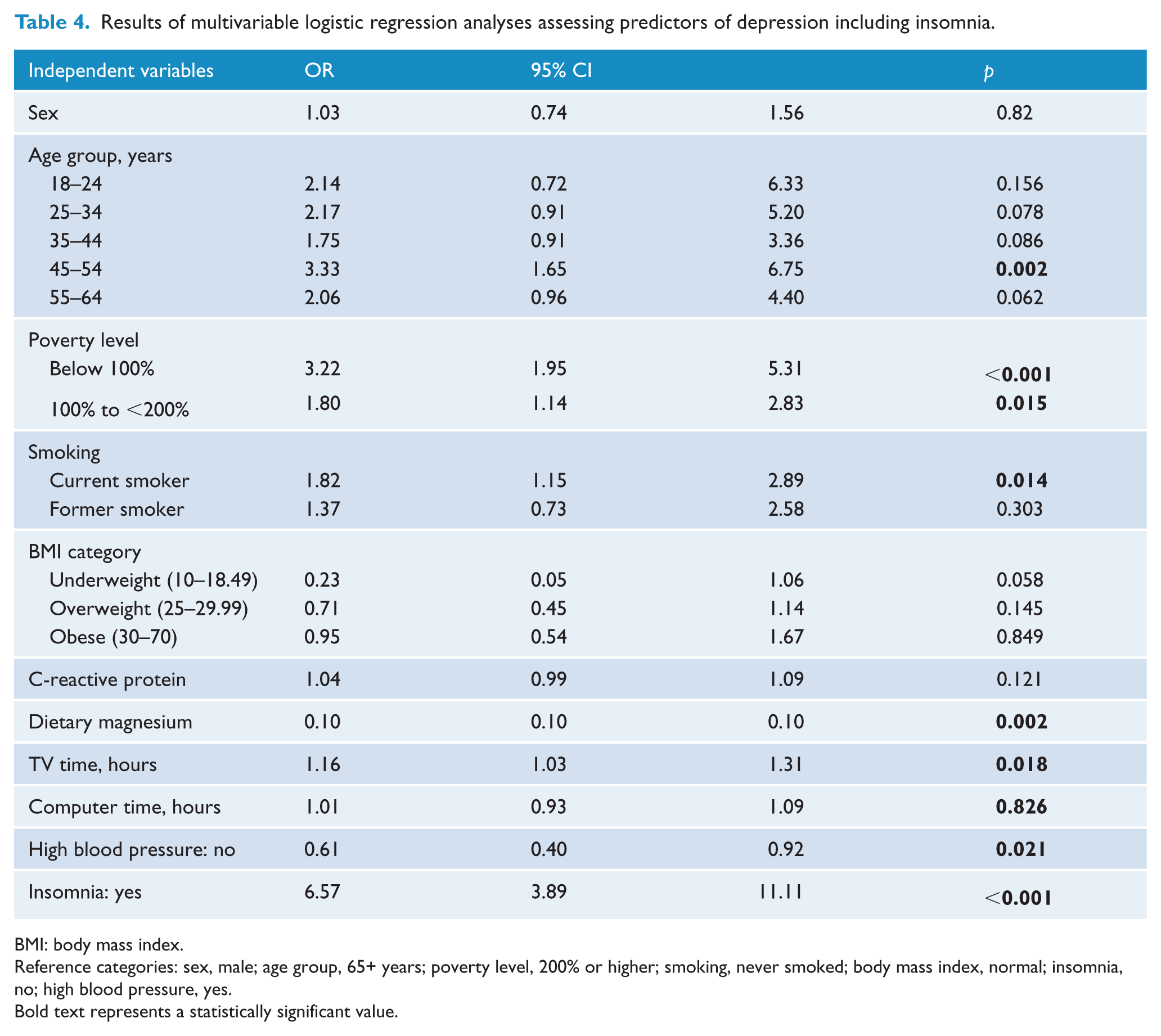
BMI: body mass index.
Reference categories: sex, male; age group, 65+ years; poverty level, 200% or higher; smoking, never smoked; body mass index, normal; insomnia, no; high blood pressure, yes.
Bold text represents a statistically significant value.
Sleep apnoea
Overall, 664 individuals were classified as having OSA, with these symptoms more common among men than women (60.4% vs 39.6%, respectively). There was a general trend of increased prevalence among increasing age brackets, and those aged 65+ years reported the highest instances of the disorder (Table 1). The highest proportion of OSA was seen among those who identified their ethnicity as either non-Hispanic white or non-Hispanic black, with the lowest proportion seen among those who identified as ‘other Hispanic’ or ‘other race – including multi-racial’ (5.3% and 3.2%, respectively). The largest proportion of those who reported OSA also identified themselves as ‘married’, with those who are ‘separated’ or ‘widowed’ reporting the lowest prevalence. Similarly, those individuals who fell into the highest income bracket reported the highest proportion of OSA, with those in the lowest bracket concordantly reporting the lowest prevalence of the disorder. Those who reported having ‘never smoked’ had nearly a twofold increased proportion of OSA compared to those who identified themselves as a ‘current smoker’. Individuals who were classified as ‘overweight’ or ‘obese’ had the highest proportion of OSA, compared to those who were considered ‘underweight’ or within the ‘normal’ weight range, who reported the lowest (0.6% and 11%, respectively). Lastly, those with OSA similarly reported the highest proportion of ‘normal’ CRP levels (Table 1).
Univariate analysis of weighted data revealed that those individuals who reported OSA were significantly older and had greater overall weight and BMI than those without OSA. Similarly, these individuals reported spending more time participating in sedentary activities than those who did not report the disorder (Table 2). Assessment of disease comorbidity revealed that those with OSA were significantly more likely to report depression than those without OSA. Similarly, they reported significantly more medical comorbidity, such as diabetes, high blood pressure and CVD than those without the disorder (all p<0.001) (Table 3).
After adjusting for age, sex, poverty level, smoking status and BMI, OSA was independently associated with a 5.14-fold increased likelihood of reporting depression (OR 5.14, 95% CI 3.14–8.41). These findings were not explained by sedentary behaviour (Table 5).
Results of multivariable logistic regression analyses assessing predictors of depression, including sleep apnoea.
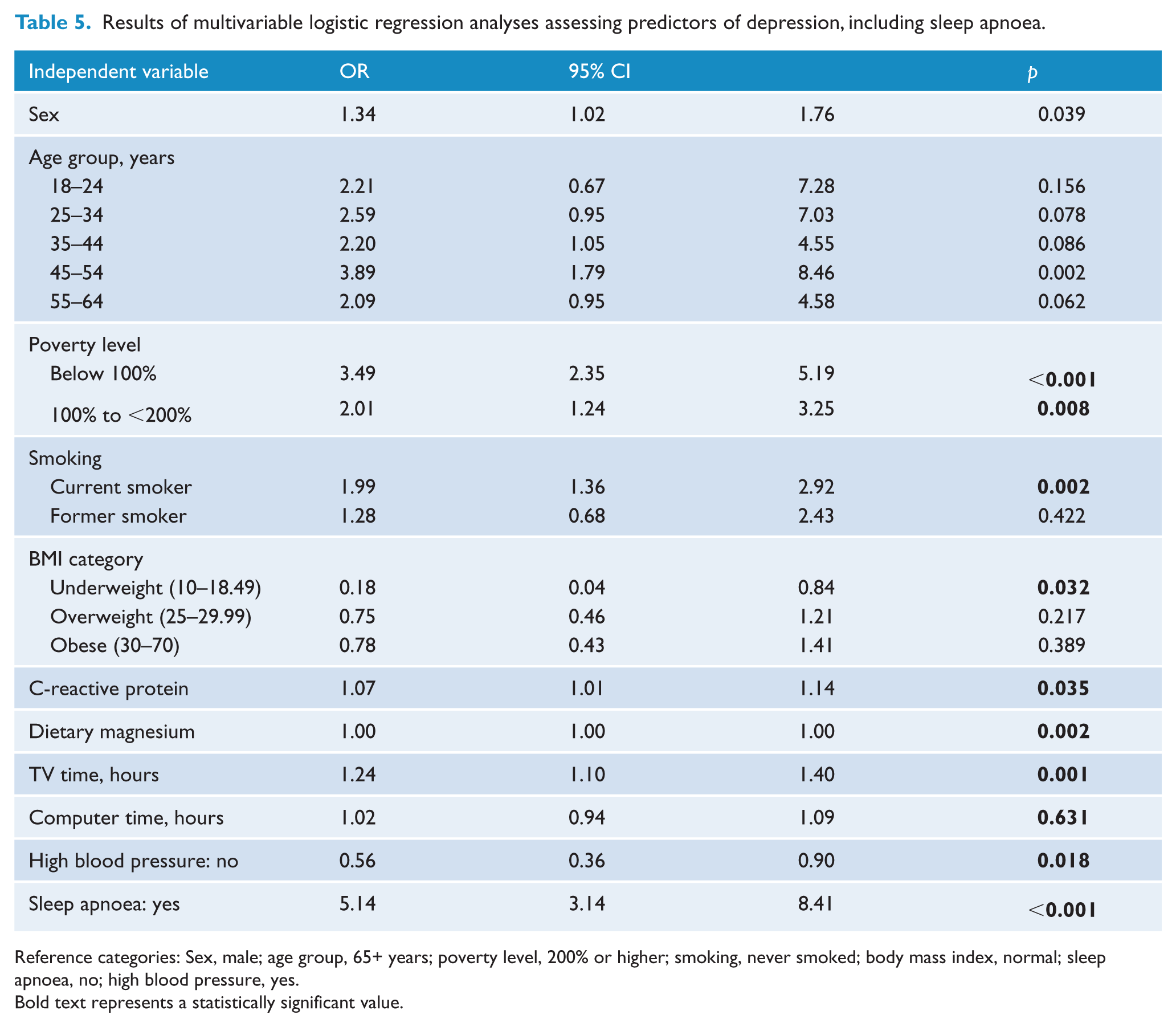
Reference categories: Sex, male; age group, 65+ years; poverty level, 200% or higher; smoking, never smoked; body mass index, normal; sleep apnoea, no; high blood pressure, yes.
Bold text represents a statistically significant value.
Comorbid insomnia and OSA
In total, 1363 individuals were classified as meeting criteria for comorbid insomnia-OSA, with higher rates of comorbidity seen among women compared to men (54.4% vs 45.6%, respectively). A general trend of increased prevalence was noted among increasing age groups, with the highest incidence rate noted among those aged 65+ years (Table 1). The proportion of individuals of non-Hispanic white ethnicity reported nearly double the prevalence of comorbidity compared to non-Hispanic black individuals (54.2% vs 20.8%), and nearly triple that of the prevalence compared to Mexican American individuals (54.2% vs13.5%). In regard to marital status, those who identified as ‘married’ also reported the highest rate of comorbidity (50.4%), with individuals who identified as ‘separated’ reporting the lowest incidence (4.6%). Those who identified as belonging to the highest income bracket similarly reported the highest incidence of comorbidity (47.3%), almost double that of the mid-range and lowest income brackets (25.0% and 27.7%, respectively). Those individuals who identified as having ‘never smoked’ were most likely to report comorbid insomnia and OSA, with almost double prevalence seen among these individuals compared to those who were classified as ‘current smokers’ (28.9%) or ‘former smokers’ (28.2%). Individuals who were classified as obese or overweight had the highest proportion of comorbidity, compared to those who identified as being of ‘normal weight’ or ‘underweight’. Those who reported comorbidity similarly reported a higher rate of ‘normal’ CRP levels (Table 1).
Following the application of univariate analysis, those with comorbid insomnia-OSA were seen to be significantly older, and reported a higher overall weight and BMI than those who reported no comorbidities. Serum levels of CRP were also elevated among those individuals reporting comorbid insomnia and OSA. Assessment of the association between comorbid insomnia and OSA showed significantly higher rates of depression, asthma, CVD, high blood pressure, and cancer or malignancy among these individuals compared to those who reported no comorbidity (all p=<0.001).
After adjusting for sex, age, poverty level, smoking status and BMI (kg/m2), the presence of comorbid insomnia and OSA was independently associated with more than a sixfold increased likelihood of reporting depression (OR 6.67, 95% CI 4.44 - 10.00). These findings were not explained by CRP levels or sedentary behaviour (Table 6).
Results of multivariable logistic regression analyses assessing predictors of depression, including comorbid insomnia-OSA.
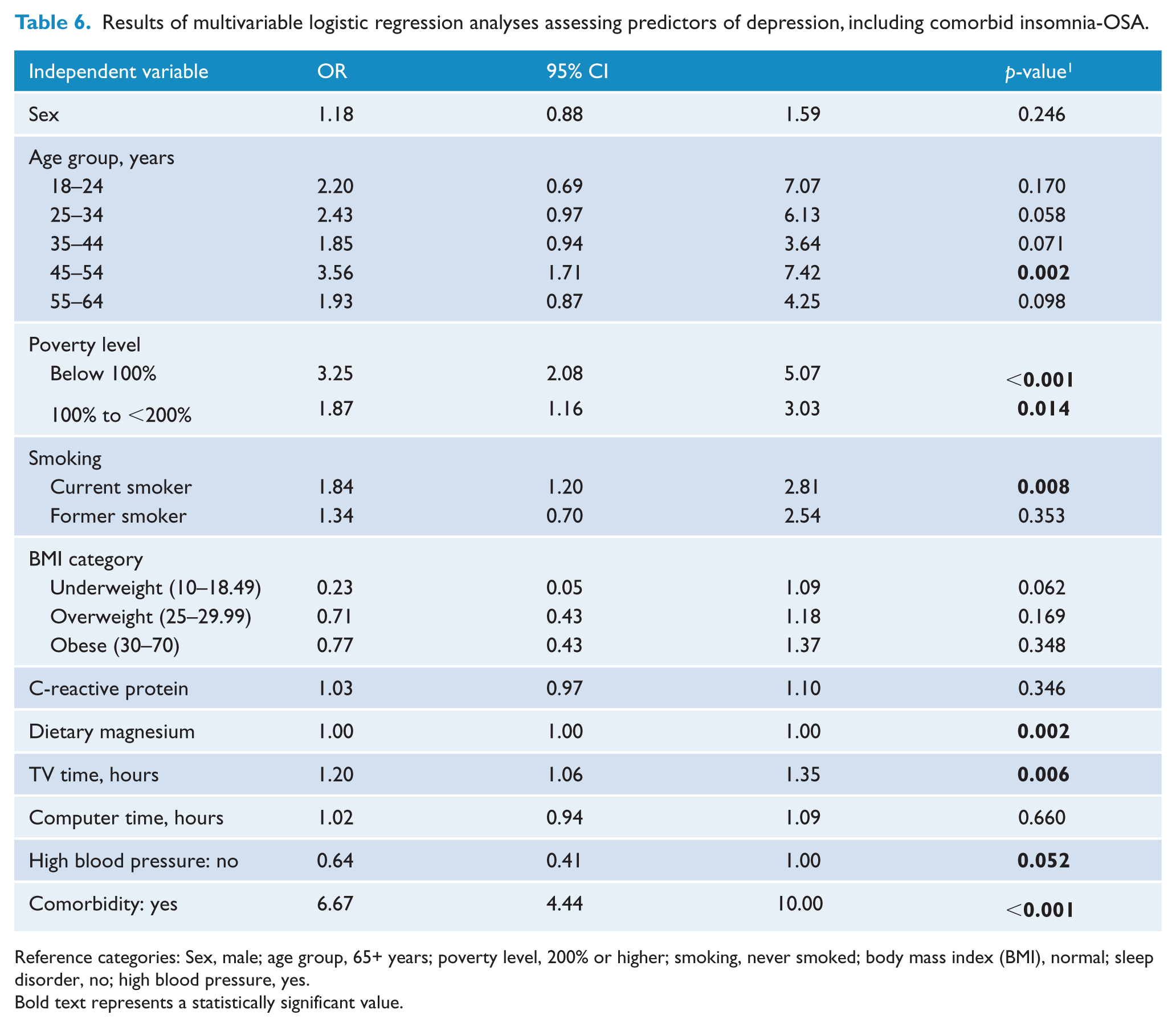
Reference categories: Sex, male; age group, 65+ years; poverty level, 200% or higher; smoking, never smoked; body mass index (BMI), normal; sleep disorder, no; high blood pressure, yes.
Bold text represents a statistically significant value.
Discussion
Within this large, cross-sectional population study of American adults, we identified a robust association between independent insomnia and OSA, as well as comorbid insomnia and OSA, and the experience of depressive symptoms. Moreover, after adjusting for relevant lifestyle and health factors, both insomnia and OSA (independent), and comorbid insomnia and OSA were associated with greater than a fivefold increased likelihood of also reporting depression. These findings were not related to CRP levels, dietary magnesium or sedentary behaviour.
The strong relationship between sleep disorders and depression has been demonstrated among a number of clinical (Vandeputte and de Weerd, 2003) and intervention studies (Manber et al., 2008), with some studies suggesting that this relationship may also be considered bidirectional. Longitudinal research conducted by Morphy and colleagues (2007) demonstrated that individuals who reported insomnia at the time of baseline assessment were more likely to report instances of depression at the time of the 12-month follow-up. These findings have also been supported by a number of review studies, with an almost unambiguous link between self-reported insomnia and the later development of depressive symptoms (Baglioni et al., 2011). Analysis of individuals who participated in the Wisconsin Sleep Cohort Study revealed a dose–response relationship between the severity of sleep disordered breathing (SDB) symptoms and the subsequent risk for developing depressive symptoms (Peppard et al., 2006). The strength of this association has also been demonstrated among a number of treatment studies, with documented improvements in depressive symptoms following the application of Continuous Positive Air Pressure (CPAP) therapy (Kawahara et al., 2005).
Possible mechanisms for the association between sleep disorders and depression have been cited as shared deficits in the quality or continuity of nocturnal sleep, albeit derived from different pathological origins, for both insomnia and OSA. Indeed, depressive symptoms in individuals with OSA are often attributed to periods of nocturnal hypoxemia and sleep fragmentation, which are characteristic of the disorder (Akashiba et al., 2002; Pochat et al., 1993; Schroder and O’Hara, 2005; Yue et al., 2003). Research conducted by Wheaton and colleagues (2012) demonstrated that individual symptoms characteristic of OSA, such as pauses in breathing, but not snoring, are strongly associated with the experience of depression, supporting this view. Within the current study, individual symptoms were not assessed. Rather, the presence of the disorder was assessed via the cumulative response to relevant questionnaire items, in addition to a positive identification of a physician-diagnosed disorder. Such methods have been previously applied among similar samples (Kapur et al., 2002) and have demonstrated a higher overall prevalence of OSA than previous research. Within our study, utilising this method, we report a high overall prevalence of individuals reporting OSA, which may be due to the methodological characterisation of the syndrome. However, these figures are not standardised to the wider population, and thus need to be interpreted with some caution. Despite this, characterising the disorder in this manner allows for identification of possible sub-threshold cases of the disorder, of which has significant clinical usefulness in regard to indications of the burden of disease.
To our knowledge, this is the first research of its kind to demonstrate a strong association between comorbid insomnia and OSA and depression. As outlined previously, recent research suggests a high concordance rate between insomnia and OSA, with some authors suggesting this rate could be higher than 50% (Krell and Kapur, 2005). Given that the possible mechanisms of action supporting the association between both insomnia and OSA have been well-described individually, it is surprising, therefore, that there is such little research investigating these conditions simultaneously. We postulate that the strength of the reported relationship may, in part, be attributed to the complementary symptomatic characteristics associated with the disorder (i.e. systematic and chronic disrupted sleep), shared daytime impairments, and manifest physiological effects of one or both of the disorders remaining untreated. Indeed, other studies have shown that treatment of OSA in insomnia patients improves subjective symptom ratings (Krakow et al., 2004), depressive symptoms improve among insomnia patients treated with cognitive behavioural therapy for insomnia (CBT-I) (Taylor et al., 2007), and amelioration of insomnia among patients with concordant mild OSA using CBT-I has been shown to improve objective measured sleep architecture (Guilleminault et al., 2008).
Depressive symptomology among insomnia patients has previously been ascribed to shared biological dysregulation of circadian clock genes (Serretti et al., 2003), as well as deviations from optimal neuroendocrine or neurological system functioning, such as that induced by reduced immune functioning or increased oxidative stress (Berk et al., 2013). Indeed, as both insomnia and OSA were found to be associated with a number of negative health outcomes in the current research (such as diabetes, obesity and hypertension), which are both known to share these neurological pathways (Kahn and Flier, 2000), and often feature in the clinical presentation of these disorders, such biological mechanisms may provide some explanation for these observed relationships. Shared neuroinflammatory processes may also provide a possible explanation for these associations, as reduced cardiovascular and metabolic functioning has been shown to be impaired among insomnia and OSA patients (Kahn and Flier, 2000). Indeed, we demonstrated that the relationship between OSA and depression was somewhat attenuated by CRP. Elevation in these inflammatory biomarkers has also been attributed to increased risk for depressive symptomology (Berk et al., 2013); however, this relationship may also be considered bidirectional (Motivala et al., 2005). As effective treatment of OSA has been shown to improve cardiac outcomes (Milleron et al., 2004), and untreated insomnia has been linked to reduced cardiac outcomes in specific patient groups (Chien et al., 2010), amelioration of these conditions may therefore provide a possible therapeutic target among these sleep-disordered patients, and thus improve treatment outcomes, particularly among patients with concordant depressive symptoms.
The design of the current study does not allow for differentiation between possible sub-types of insomnia (i.e. sleep maintenance, early morning awakening). However, similar methods have been applied among other studies (Ancoli-Israel and Roth, 1999) and classification grouping of symptoms does give some indication of disease burden among the sample. Future research may benefit from focusing on the strength of the association among sub-types of insomnia and depression among this sample in order to investigate possible underlying mechanisms.
The primary strength of the current study includes the use of a large, population-based, representative cohort in the assessment of the relationship between sleep disorders and depression. Similarly, to our knowledge, this is the first study to assess the relationship between comorbid insomnia and OSA. Importantly, the use of the NHANES dataset addresses many of the limitations presented by previous research; it provides information on a large sample of the general public, as opposed to a specific patient group, and thus provides information regarding the degree of disease burden at this level. Similarly, the current study actively addressed the limitations of previous NHANES data which has assessed the association between sleep and depression by controlling for, and accounting for, a large number of associated health and lifestyle covariates during analysis. Such techniques provide more detailed assessment of the strength of these associations.
As inherent in any population-based survey that employs self-report symptom classification, the reported sleep disorders may be under or over-reported, and thus may not be accurately representative of population prevalence. Specifically, those with insomnia may overestimate the degree of impairment, a bias which has been previously noted (Edinger and Means, 2005). Similarly, no report was obtained from the bed partners or third parties of those who were identified as having OSA. As these symptoms occur primarily when the patient is sleeping, symptom frequency may be under-reported. Despite this, methods of self-report for both insomnia and OSA have been found to have good reliability (Bliwise et al., 1991). Although a number of health and lifestyle factors were accounted for during analysis, information regarding medication use was not included. Treatment of underlying depressive illness has been shown to improve OSA symptom outcomes as a function of improved adherence to continuous positive air pressure (CPAP) therapy (Harris et al., 2009), and antidepressant use among those with comorbid depression-insomnia can result in improvement in subjective symptoms (Serretti et al., 2005). As these factors were not assessed specifically, it is possible that reported associations may be under or over-reported. Lastly, the cross-sectional nature of the analysis does not allow for interpretation regarding the direction of the relationship, and thus it is recognised that the associations between insomnia, OSA and depression may be considered bidirectional.
In summary, this study provides population-relevant information regarding the association of the global assessment of the two most common sleep disorders, insomnia and OSA, in addition to comorbid insomnia-OSA, and their robust relationship to depressive symptomology. The strength of these associations highlights the need for appropriate clinical screening for people reporting such symptoms, in order to optimise patient treatment and outcomes.
Footnotes
Funding
Michael Berk is supported by a NHMRC Senior Principal Research Fellowship 1059660. Lana Williams is supported by a NHMRC of Australia Career Development Fellowship (GNT1064272).
Declaration of interest
Amie Hayley, Gerard Kennedy, Kamalesh Venugopal and Julie Pasco have no conflicts of interest, including specific financial interests and relationships and affiliations relevant to the subject matter or materials discussed in the manuscript. Michael Berk has received grant/research support from the NIH, Simons Foundation, CRC for Mental Health, Stanley Medical Research Institute, MBF, NHMRC, Beyond Blue, Geelong Medical Research Foundation, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Organon, Novartis, Mayne Pharma, Servier and AstraZeneca. He has been a paid consultant for AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Lundbeck and Pfizer and a paid speaker for AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Lundbeck, Organon, Pfizer, Sanofi Synthelabo, Solvay and Wyeth. Lana Williams has received grant/research support from Eli Lilly, Pfizer, The University of Melbourne, Deakin University and the NHMRC.
