Abstract
Objective:
Dementia is a major public health issue, with notably high rates in persons with mood illnesses. Lithium has been shown to have considerable neuroprotective effects, even in trace or low doses. The aim of this review is to summarize the current understanding of lithium benefits in trace or low doses in dementia prevention and for other behavioral or medical benefits.
Methods:
A systematic review identified 24 clinical, epidemiological, and biological reports that met inclusion criteria of assessing lithium in standard or low doses for dementia or other behavioral or medical benefits.
Results:
Five out of seven epidemiological studies found an association between standard-dose lithium and low dementia rates. Nine out of 11 epidemiological studies, usually of drinking water sources, found an association between trace-dose lithium and low suicide/homicide/mortality and crime rates. All four small randomized clinical trials of lithium for Alzheimer’s dementia have found at least some clinical or biological benefits versus placebo. Only one small randomized clinical trial (RCT) of trace lithium has been conducted, assessing mood symptoms in former substance abusers, and found benefit with lithium versus placebo.
Conclusions:
Lithium, in both standard and trace doses, appears to have biological benefits for dementia, suicide, and other behavioral outcomes. Further RCT research of trace lithium in dementia is warranted.
Introduction
Dementia is a major public health problem, rising in prevalence from 1% of the population at age 60 years to over 30% of the population by age 90 (Nunes et al., 2007), and affecting an estimated 35.6 million people worldwide, a number projected to nearly double every 20 years. Nearly 7.7 million new cases occur yearly, or about one new case every 4 seconds. Prevalence rates in Australia (9% above age 65) and Europe (6.4%) are similar, with an expected increase by one-third in the next decade (Australian Institute of Health and Welfare, 2012; Lobo et al., 2000). The estimated worldwide cost of dementia is US$604 billion (World Health Organization, 2012). There are no known preventive, or even notably ameliorative, treatments for most dementias – the most common type of which is Alzheimer’s disease (AD) (Broberg et al., 2011).
A prominent risk factor of dementia is depression, which doubles the risk of dementia (da Silva et al., 2013; Kessing et al., 2008). Subjects with depression or bipolar disorder have been found to experience long-term cognitive impairment, even when in a euthymic state (Forlenza et al., 2011). Depression itself is a common illness, occurring in about 5–10% of the population in its unipolar variety, and about 2–5% of the population in its bipolar variety (Ohgami et al., 2009).
Lithium, a very effective agent for the prevention of mania and depression, has also been shown to have considerable neuroprotective effects, far more in extent and human relevance than any other psychotropic agent (Broberg et al., 2011; Hampel et al., 2009; Helbich et al., 2012; Nunes et al., 2013; Zarse et al., 2011). Clinical studies have also demonstrated that lithium can modulate in vivo cellular cascades related to neuronal resilience and neuroprotection. Lithium treatment has been linked to increased phosphor-glycogen synthase kinase-3 beta (GSK-3B) levels and, consequently, reduced enzymatic activity in leukocytes of patients with bipolar disorder (Forlenza et al., 2011). It has also been found to induce autophagy (enhanced clearance of toxic cellular substrates) via inositol monophosphatase (IMPase) inhibition, which leads to free inositol depletion and reduced myo-inositol-1,4,5-triphosphate (IP3) levels. The induction of cellular autophagy by lithium may be a mechanism to prevent neurodegeneration (Forlenza et al., 2012; Sarkar and Rubinsztein, 2006; Sarkar et al., 2005). Phosphorylated tau (p-tau) levels are reduced with long-term lithium in mild cognitive impairment (MCI) (Forlenza et al., 2011), and the concentration of brain-derived neurotrophic factor (BDNF) is increased after only 10 weeks of lithium treatment in mild AD (Leyhe et al., 2009).
In this paper, we review the clinical, epidemiological, and biological literature related to whether or not lithium might have any benefit in the prevention or treatment of dementia, including AD.
Methods
This systematic review was conducted between September 2012 and August 2013 following PRISMA guidelines (Moher et al., 2009), searching the PubMed database, augmented by bibliographic cross-referencing, and use of any other sources available, including conference proceedings or abstracts from 1969 to 2013 (Figure 1).
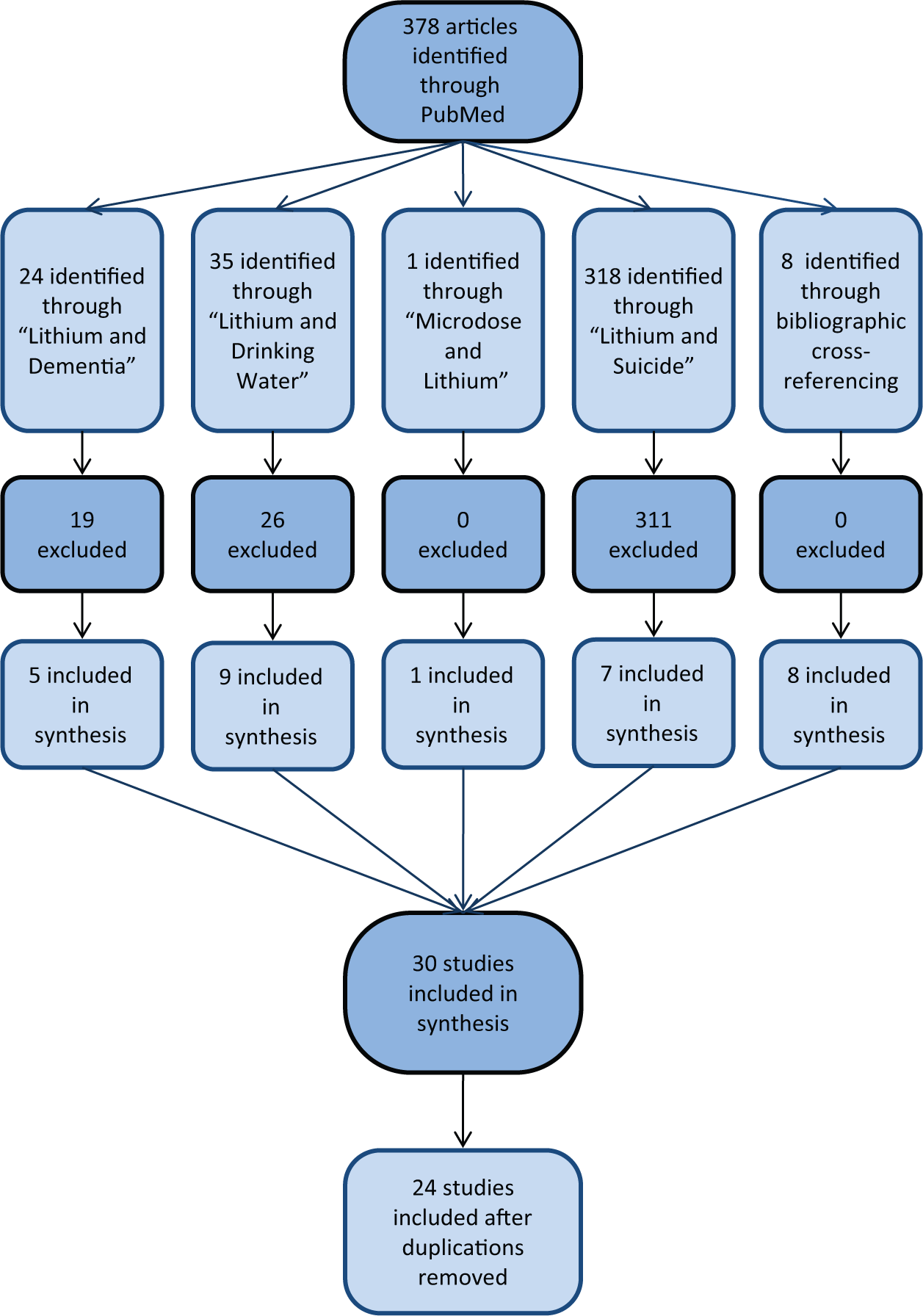
Flow chart depicting steps in the systematic review of articles.
Inclusion criteria were as follows: studies had to use the English language, include human subjects, and represent original research data. When using standard doses of lithium, studies had to examine lithium in relation to a cognitive outcome, such as minimal cognitive impairment, dementia, or other cognitive impairment. Since there are no specific studies of cognitive outcomes or dementia risk with trace lithium levels, we included any outcomes in studies of trace lithium levels to obtain a sense of whether trace lithium levels can have detectable biological or neurocognitive effects. Exclusion criteria included reviews, commentaries, case reports and studies limited to animal populations only.
In sum, we included clinical studies of lithium in humans with cognitive outcomes, and studies of trace lithium levels in humans assessing any outcome.
Key words and application of inclusion/exclusion criteria produced the following results: “Lithium AND dementia”, filtered by clinical trials and human subjects, yielded 24 papers, of which 19 were excluded because six related to Huntington’s chorea, four were reviews, seven were letters or case reports, and two were irrelevant (one was related to the antipsychotic risperidone, and another to depression, not dementia). “Microdose AND lithium”, filtered by clinical trial, yielded one article, included in this review. “Lithium AND drinking water” yielded 35 references, of which 26 were excluded because eight were letters, seven were reviews, six involved diseases irrelevant to the focus of this review (porphyria, vascular disease, agranulocytosis, anencephaly, dental caries and thyroid levels), two were not in English, one did not assess any outcomes, one involved sodium not lithium, and one used an animal sample only. “Lithium AND suicide” yielded 318 references, which, after applying the inclusion criterion of including only trace lithium studies, led to exclusion of 311 studies because 176 did not involve trace lithium, 125 were reviews, seven were not in English, and three were letters. Using bibliographic cross-referencing from the above studies, we identified eight more studies for inclusion in this review: seven human clinical trials of lithium for cognition, and one more clinical study of trace lithium for any outcome.
Results
Clinical and epidemiological studies of standard lithium doses
As seen in Table 1, three case–control studies, three retrospective cohort studies, and one prospective cohort study, ranging in sample size from 22 to almost 20,000 subjects and a follow-up of 1–10 years, have been published. Five of seven studies found an association between lithium use and low dementia rates.
Clinical and epidemiological studies of standard doses of lithium for cognition or dementia.
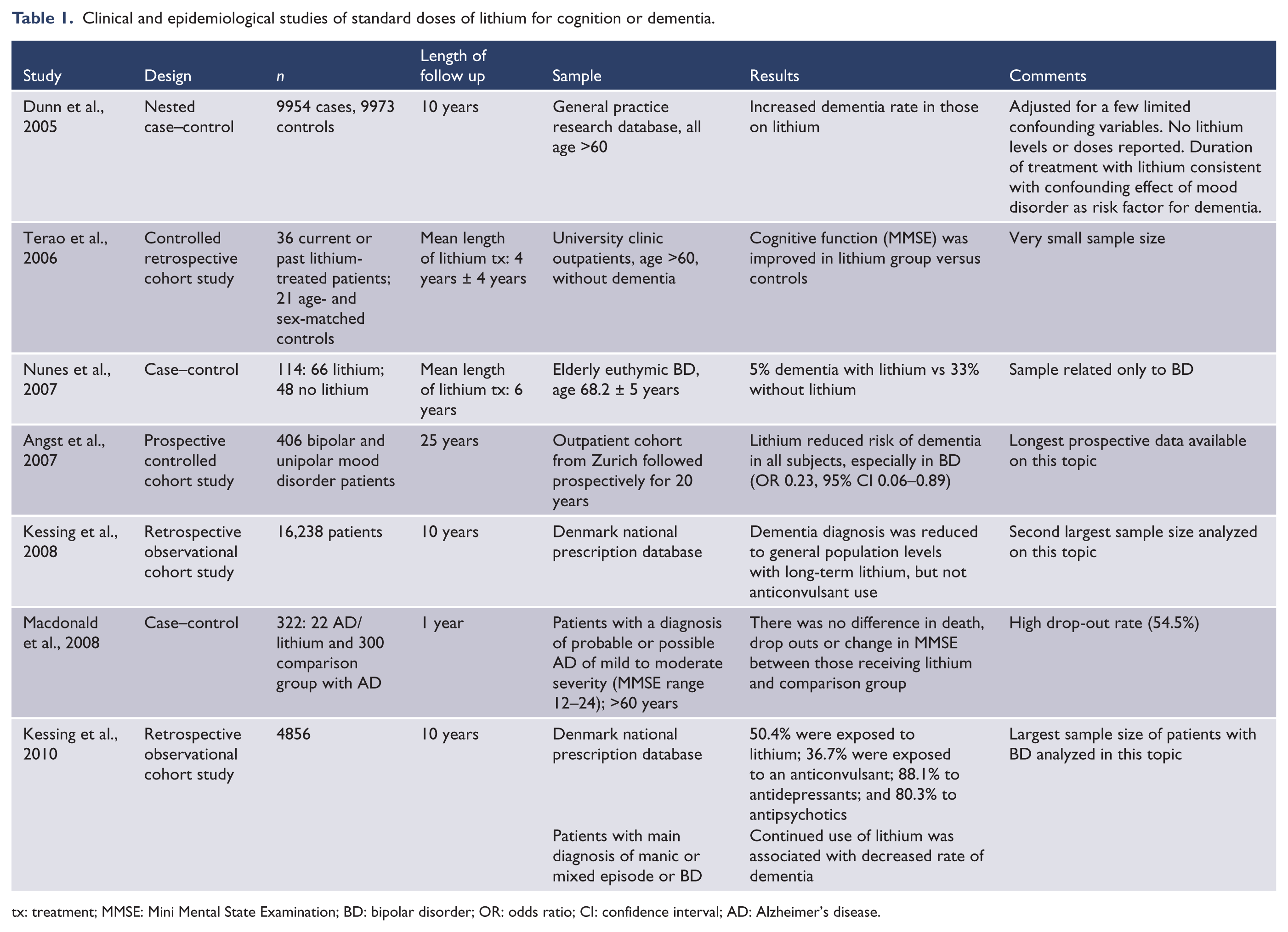
tx: treatment; MMSE: Mini Mental State Examination; BD: bipolar disorder; OR: odds ratio; CI: confidence interval; AD: Alzheimer’s disease.
The only prospective study, with up to 26 years’ outcome data in a mood disorder population from Zurich, found a major decrease in dementia with lithium treatment (77% reduction in odds ratio) (Angst et al., 2007). Of the three retrospective studies (Kessing et al., 2008, 2010; Terao et al., 2006), all of them found benefit. The notable benefit in the Danish bipolar samples (Kessing et al., 2008, 2010) may suggest, as with the Zurich study, special effectiveness for dementia prevention in mood disorders, as opposed to the general population.
Two out of three case–control studies showed no benefit with lithium (Dunn et al., 2005; Macdonald et al., 2008); both studies were in non-mood disorder samples. One case–control study found notable benefit with lithium (Nunes et al., 2007); unlike the others, it was limited to a bipolar sample.
Epidemiological studies of trace lithium doses
For trace lithium levels with any outcome, we identified 11 epidemiological studies, many conducted by geology specialists (Table 2). Sample sizes were often large (5 million in one study, 8 million in another), including regions such as over two dozen Texas counties (Dawson et al., 1970; Schrauzer and Shrestha, 1990), the entire state of North Carolina (Voors, 1972), the 100 largest American cities, or 99 districts in Austria. In all cases, water levels were tested for lithium availability. Outcomes often involved major behavioral abnormalities (such as suicide, homicide, psychiatric admissions, crimes) or some medical illnesses (such as cardiovascular disease), or overall mortality. Suicide was the most commonly measured outcome.
Epidemiological studies of trace amounts of lithium.
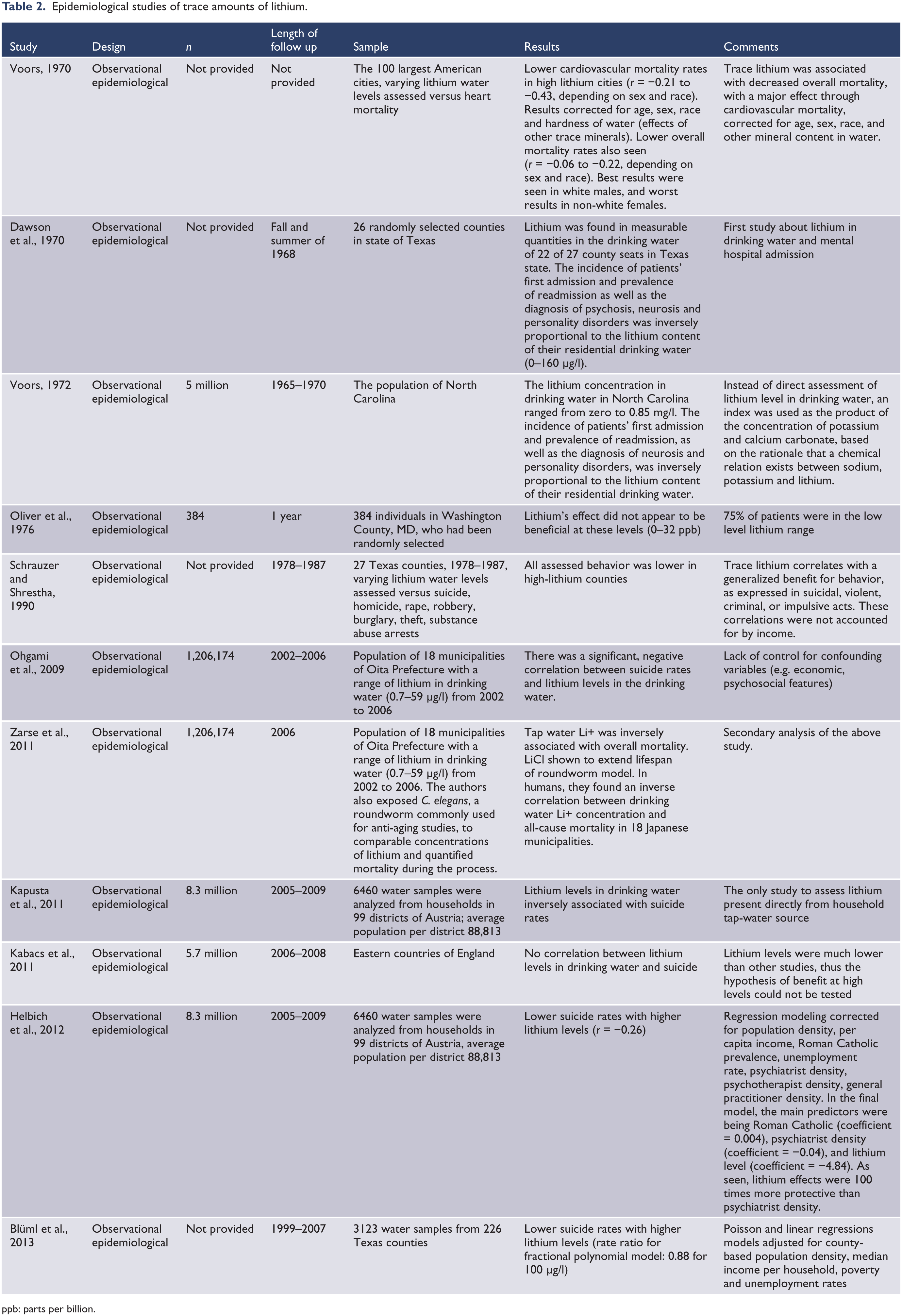
ppb: parts per billion.
Nine of 11 studies found an association between higher levels of trace lithium in drinking water and beneficial clinical, behavioral, legal, and medical outcomes. The most commonly assessed outcome – suicide – was reduced in four of five studies.
The major risk in epidemiological studies is confounding bias (Agabegi and Stern, 2008; Ely, 1992; Ghaemi, 2009), which is best addressed by regression modeling and other forms of matching for confounding factors (Ghaemi, 2009). The two most valid studies in this group (Blüml et al., 2013; Helbich et al., 2012) conducted regression models adjusting for potential confounding factors, such as economic factors like poverty, unemployment, and social factors (e.g. religious denomination prevalence and psychiatrist density). A third study (Voors, 1970) matched for age, sex, race, and mineral content in water. All of these studies, which corrected for at least some statistical confounding factors, found notable benefit with lithium for suicide prevention and mortality. Another report which found a suicide benefit with lithium did not correct for other confounding factors but is the only study to assess lithium presence directly in household water-tap sources (Kapusta et al., 2011).
In both negative studies (Kabacs et al., 2011; Oliver et al., 1976), most water samples had low levels of lithium; thus, benefits of high trace lithium levels may not have been detectable.
In the other positive studies, two, which assessed psychiatric admission rates, found consistent association with lower hospitalization in areas with high lithium water levels (Dawson et al., 1970; Voors, 1972), two found decreased overall mortality (with specific benefit for cardiovascular mortality in one study) (Voors, 1970; Zarse et al., 2011), and another study found a range of behavioral benefits including lowered crime and homicide rates (Schrauzer and Shrestha, 1990).
Randomized clinical trials (RCTs) of dementia or cognitive symptoms
Four RCTs, ranging from 6 to 15 months in 27–113 subjects, have been conducted to assess lithium’s effects on cognitive impairment or dementia. Benefits were seen on at least some clinical or biological parameters in all four studies. The impression has been given that this literature is negative (Tariot and Aisen, 2009) because the first published RCT (Hampel et al., 2009) reported a negative result for its primary outcome (GSK-3 biological activity at 6-month follow-up). However, the same study reported benefit with another biological outcome (BDNF levels) (Leyhe et al., 2009) and there was a decrease in worsening of clinical cognitive symptoms in that elderly MCI population: using the Alzheimer’s Disease Assessment Scale – cognitive subscale (ADAS-Cog), exactly twice as many lithium- than placebo-treated subjects, 28.6% vs 14.3%, had notable cognitive improvement (ADAS-Cog improved >4 points). This benefit is not statistically significant, but given the small sample size, type II error is not improbable. The effect size is notable: using confidence intervals (CIs), we recalculated these data to find a twofold benefit with lithium (risk ratio = 2.0, lower CI 0.64, upper CI 6.20), with probability of positive benefit being more likely above than below the null value.
The other three positive studies included a 6-month study of glial cell line-derived neurotrophic factor (GDNF) expression in Alzheimer’s dementia (Straten et al., 2011), and two studies of about 1 year in duration finding prevention of worsening of cognitive functions compared to placebo (Nunes et al., 2013), as well as other biological benefits (reduction in p-tau in lithium-treated subjects) (Forlenza et al., 2011).
Although unrelated to cognitive outcomes, we have included the only RCT of trace lithium in humans in Table 3, since it confirms most of the epidemiological data in Table 2 indicating that, indeed, trace lithium levels can have important biological and neuropsychiatric effects. In the only RCT on the topic (Schrauzer and De Vroey, 1994), trace lithium levels improved mood substantially more than placebo in former substance abusers.
Randomized clinical trials (RCTs) of lithium for dementia or cognitive symptoms. a
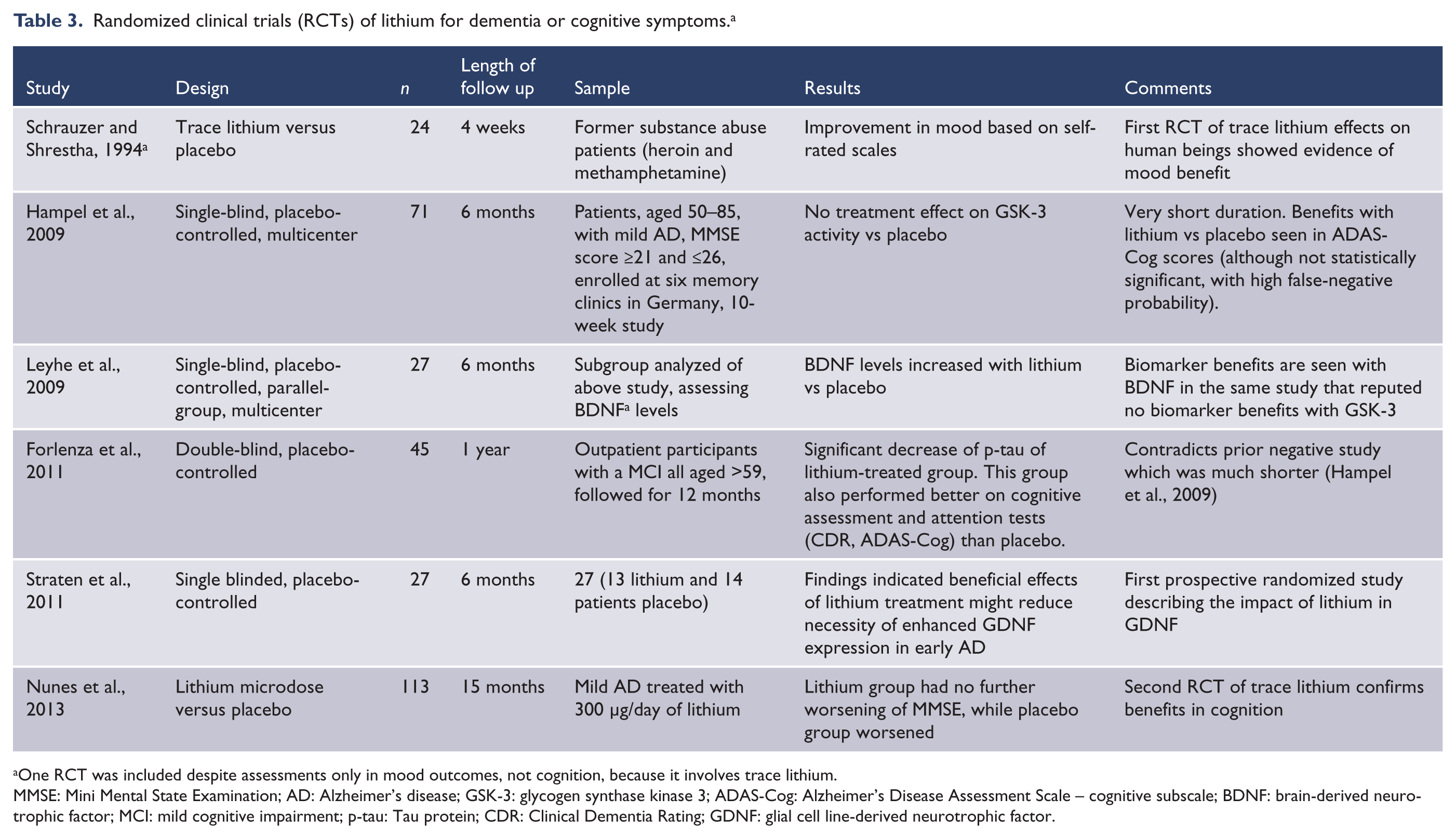
One RCT was included despite assessments only in mood outcomes, not cognition, because it involves trace lithium.
MMSE: Mini Mental State Examination; AD: Alzheimer’s disease; GSK-3: glycogen synthase kinase 3; ADAS-Cog: Alzheimer’s Disease Assessment Scale – cognitive subscale; BDNF: brain-derived neurotrophic factor; MCI: mild cognitive impairment; p-tau: Tau protein; CDR: Clinical Dementia Rating; GDNF: glial cell line-derived neurotrophic factor.
Discussion
This systematic review finds that most studies on lithium in standard doses showed notable cognitive benefits or effectiveness in dementia prevention. Epidemiological studies are numerous, large, and replicated. Most RCTs show clear biological or clinical benefits as well. Further, trace lithium levels are associated rather consistently in epidemiological studies with a range of medical and behavioral benefits, especially decreased mortality and suicide; these trace behavioral effects have been confirmed in the only RCT using trace lithium levels.
In sum, standard doses of lithium may have cognitive benefits for dementia prevention, and, although trace levels have not been studied for that outcome, trace lithium levels may have a range of other medical/behavioral benefits.
This systematic review of the literature supports the need for more attention to lithium, in standard and trace doses, for potential benefit in dementia prevention. This literature represents a formidable amount of pilot data supportive of possible, or even probable, benefit with lithium for dementia prevention. Extensive research, in both time and funding, has been conducted with putative dementia prevention interventions involving complex mechanisms, often related to apoE4 gene mechanisms or effects on neurofibrillary tangles and plaques. The simple intervention of lithium, even at low or trace doses, has been mostly ignored in dementia clinical trial research. This literature review would suggest that renewed attention should be given to the potential of lithium as a preventive intervention for dementias.
Based on initial signals of prediction of response in these epidemiological and randomized studies, it may be that lithium would yield preferential benefits in dementia prevention in persons with mood disorders, at standard or possibly trace levels, and that such benefit may not involve improvement in cognitive function but rather prevention of further decline.
In this respect, it may also be relevant that recent research suggests that the dementia process likely begins biologically at least a decade before the first clinical signs, with the largest biological changes occurring within the fifth decade of life. During this age threshold about 40% of individuals at high risk for AD (e.g. apoE4 carriers) have senile plaques, despite the absence of clinical signs of dementia (Schrauzer and De Vroey, 1994). Thus, true biological prevention may need to begin in individuals before even the earliest clinical phase of dementia.
The potential benefit of trace lithium for dementia can be connected to the existence of basic science evidence on lithium’s neuroprotective effects. Both animal and human studies show that lithium has a range of potent neuroprotective and neuroplastic effects (Bearden et al., 2007; Chen et al., 2000; Gould and Manji, 2005; Manji et al., 1999; Moore et al., 2000; Rowe and Chuang, 2004). In animal studies, when lithium has been compared at lower versus higher concentrations, neuronal viability seems to be enhanced with any concentration of lithium. Although standard lithium concentrations of 0.6–0.8 have the most benefit for enhancing neuronal viability, even ‘low’ levels of 0.2–0.4 have that benefit, compared to placebo (Hashimoto et al., 2002). Trace levels of lithium would be undetectable in standard blood tests, which generally do not measure lithium levels below 0.2. But, in some of the available animal research, even concentrations below 0.2 lead to enhanced neuronal viability (Hashimoto et al., 2002).
This basic neuroscience evidence, combined with the many clinical studies on the biological activity of trace lithium, would support clinical research on trace lithium prevention of dementia. If effective, such trace dosing would likely be much more tolerable than standard lithium levels, especially in aging populations, thus making this intervention more feasible from a public health standpoint. In summary, this review of the literature suggests that this promising avenue of intervention for perhaps the most devastating neuropsychiatric condition, dementia, should be explored more intensively.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of interest
In the past 12 months, Dr Ghaemi has received a research grant from Takeda Pharmaceuticals. Neither he nor his family hold equity positions in pharmaceutical corporations. Dr Sivan Mauer and Dr Derick Vergne declare no conflicts of interest.
