Abstract
Objective:
To examine characteristics that predict the progression from mild cognitive impairment to dementia.
Methods:
Of 970 patients recruited from nine memory clinics around Australia, 185 had mild cognitive impairment diagnosed. Measures of cognitive ability, functional ability, and neuropsychiatric symptoms were completed at baseline and over 3 years of follow up.
Results:
Over 3 years, 52 (28%) patients with mild cognitive impairment developed dementia. Older age, lower cognitive ability at baseline, and faster decline in cognitive ability over the first 6 months of follow up, but not depression, predicted progression to dementia.
Conclusions:
The findings confirm that simple clinical data such as age, cognitive ability at baseline, and rate of cognitive decline are important predictors of progression from mild cognitive impairment to dementia over 3 years.
Introduction
Mild cognitive impairment (MCI), often conceptualised as an intermediate state between normal ageing and dementia, is characterised by cognitive deficits that are noticeable to the patients or their families, yet do not interfere significantly with the patients’ ability to function in everyday life. While patients with MCI are at a higher risk of developing dementia then the general population (Petersen et al., 2009; Winblad et al., 2004), not all patients with MCI develop dementia (Xie et al., 2011). There is considerable controversy over the validity of MCI criteria and the prognostic significance of various proposed subtypes of MCI (Allegri et al., 2008; Visser and Brodaty, 2006). More accurate identification of which patients with MCI will develop dementia would be helpful to clinicians to advise their patients. Of particular importance is whether easily measured variables such as older age (Bruscoli and Lovestone, 2004), lower cognitive ability (Bruscoli and Lovestone, 2004), rates of cognitive and functional decline (Hensel et al., 2009), and neuropsychiatric symptoms (Teng et al., 2007), especially depression (Modrego and Ferrández, 2004; Palmer et al., 2007; Teng et al., 2007), increase the risk of progression.
Most research on predictors of progression from MCI has emanated from tertiary referral centres or population studies (Mitchell and Shiri-Feshki, 2009; Petersen et al., 2009), especially from the USA. This is significant given that rates of progression to dementia in patients with MCI vary depending on location and setting (Bruscoli and Lovestone, 2004; Brodaty et al., 2013). We examined predictors of dementia in patients with MCI who were attending memory clinics across Australia. Measures of cognitive ability, functional ability, and neuropsychiatric symptoms were completed at baseline and patients were followed for 3 years. Following previous research, we hypothesised that older age, lower cognitive ability at baseline, more neuropsychiatric symptoms (especially greater depression at baseline), and steeper trajectory of cognitive decline would predict progression to dementia.
Materials and methods
Design
The participants were drawn from the PRIME study in Australia (Brodaty et al., 2011). This study was a 3-year, nonprescriptive, observational study examining relationships between predictors and outcome variables in patients with dementia and MCI. All participants were receiving specialist assessment or treatment at one of nine memory clinics in Australia. The memory clinics were in four of the eight states and territories of Australia and included both regional and capital centres. Patients were followed up at 3, 6, 12, 24, and 36 months by a research nurse or their specialist clinician. Patients were also followed up after their regular clinical appointments if their specialist clinician indicated a change in diagnosis. Ethics approval was obtained from institutional ethics committees associated with individual referring centres (National Institute of Health clinical trials registry number: NCT00297271).
Participants
Of the 970 patients in the PRIME study, 189 had MCI at baseline and 781 had dementia. All patients were living in the community, were fluent in English, and provided written informed consent. Four patients with MCI were excluded from analyses because they were taking medication for Alzheimer’s disease or were concurrently participating in a clinical trial of an investigational drug. This paper specifically focused on the remaining 185 patients with MCI at baseline. Diagnoses of MCI were made according to the Petersen criteria by a specialist psychogeriatrician, geriatrician or neurologist (Winblad et al., 2004); subsequent diagnoses of dementia and types were made by specialist clinicians using DSM-IV criteria (American Psychiatric Association, 2000). More specifically, criteria for MCI required that: (i) patients or an informant report signs of cognitive decline; (ii) patients show objective evidence of cognitive decline as measured by the Mini-Mental State Examination (MMSE; Folstein et al., 1975); (iii) patients retain generally preserved functioning in activities of daily living; and (iv) patients do not meet DSM-IV criteria for dementia. Data relating to the subtypes of MCI (Winblad et al., 2004) were not collected. The number of patients at each time point and the reasons that patients were lost to follow up are shown in Figure 1. Patients who were lost to follow up did not significantly differ from those who completed the study on any of the measured variables (all p>0.06)
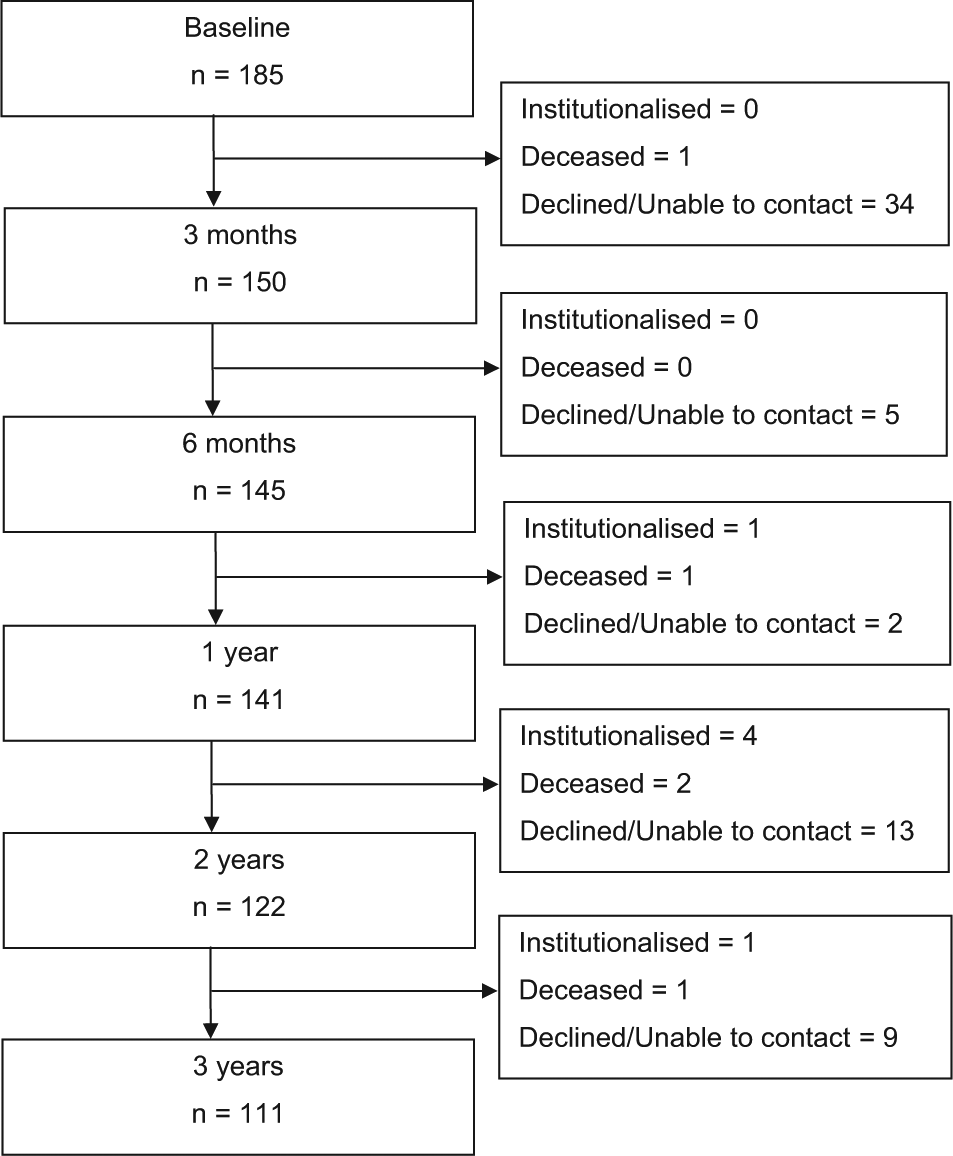
Flow diagram indicating sample sizes across time points and the reasons that participants were lost to follow up.
Measures and procedure
At baseline and at each subsequent visit, a research nurse or specialist clinician administered measures of cognitive ability, functional ability, and neuropsychiatric symptoms. Cognitive ability was assessed using the MMSE (Folstein et al., 1975). Functional ability was assessed using the Functional Autonomy Measurement System (SMAF; Hébert et al., 1988). Neuropsychiatric symptoms, including depression, were assessed using the 12-item Neuropsychiatric Inventory (NPI; Cummings, 1997) with an informant.
Statistical analyses
Patients with MCI who developed dementia were compared to patients who did not on demographic variables and measures of cognition (MMSE), decline in cognitive ability over time (decline in MMSE), functional ability (SMAF), decline in functional ability over time (decline in SMAF), neuropsychiatric symptoms (NPI), change in neuropsychiatric symptoms over time (change in NPI), and individual neuropsychiatric symptoms – namely delusions, hallucinations, agitation, depression, anxiety, euphoria, apathy, disinhibition, irritability, aberrant motor behaviour, night-time disturbances, and appetite disturbances. Categorical variables – namely sex and education – were compared using chi-squared analyses. All other variables were compared using independent-samples t-tests. The relationship between neuropsychiatric symptoms and MMSE score was examined using Pearson correlations.
A Cox’s proportional hazard model was used to examine which variables predicted a diagnosis of dementia within the 3-year period. Patients who withdrew from the study or died before being institutionalised were classed as censored. Given the statistical constraint of one predictor per 10 events (Peduzzi et al., 1995), analyses focused on age, sex, MMSE), decline in MMSE at 6 months, and depression as predictors. Analyses were also repeated with total NPI score and change in total NPI at 6 months included in the model instead of depression. All statistical procedures were conducted using SPSS version 21.
Results
Patient characteristics
The baseline characteristics of the patients who developed dementia and those who did not are shown in Table 1. Patients who developed dementia in the 3-year period had a lower MMSE score at baseline, a greater decrease in MMSE score over time, and greater depression at baseline.
Characteristics of patients who did and did not develop dementia.
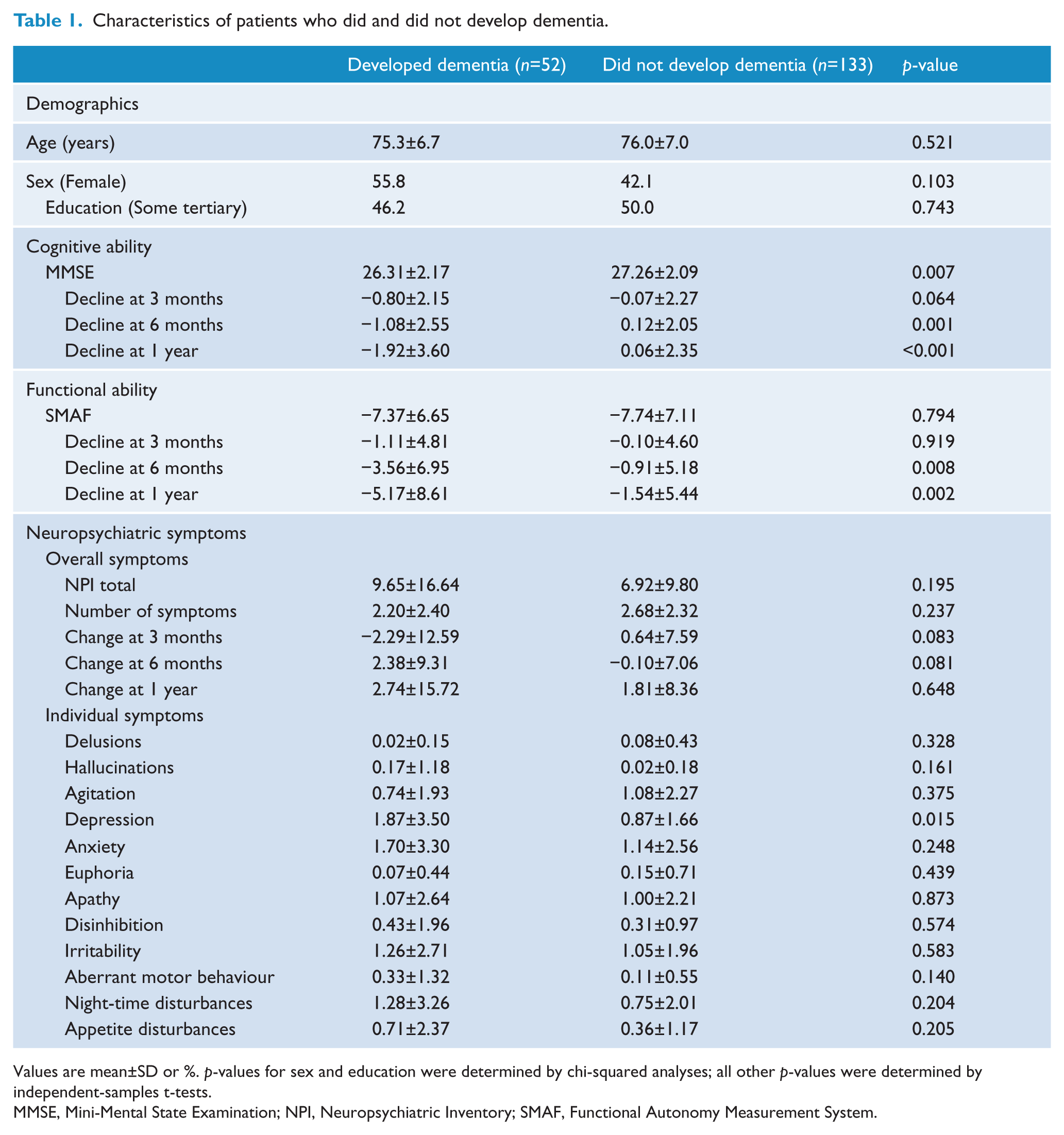
Values are mean±SD or %. p-values for sex and education were determined by chi-squared analyses; all other p-values were determined by independent-samples t-tests.
MMSE, Mini-Mental State Examination; NPI, Neuropsychiatric Inventory; SMAF, Functional Autonomy Measurement System.
Across all patients, 118 (63.8%) were reported to have at least one neuropsychiatric symptom at baseline. Overall, eight (4.3%) patients were reported to have delusions, two (1.1%) hallucinations, 54 (29.2%) agitation, 61 (33%) depression, 59 (31.9%) anxiety, 10 (5.4%) euphoria, 59 (31.9%) apathy, 25 (13.5%) disinhibition, 67 (36.2%) irritability, 12 (6.5%) aberrant motor behaviour, 34 (18.4%) night-time disturbances, and 26 (14.1%) appetite disturbances. Chi-squared analysis indicated no significant difference in the distribution of symptoms at baseline between patients who developed dementia and those who did not (all chi-squared<3.62, all p>0.07). Baseline depression and MMSE score were negatively correlated, r=−0.17, p=0.03. There were no statistically significant correlations between other neuropsychiatric symptoms and MMSE score (all r<0.14, all p>0.09).
Progression to dementia
Of the 185 patients with MCI at baseline, 52 (28.1%) were diagnosed with dementia over the 3-year period. They comprised 38 (73.1%) patients with Alzheimer’s disease, two (3.8%) with vascular dementia, five (9.6%) with mixed Alzheimer’s and vascular dementia, one (1.9%) with dementia with Lewy Bodies, four (7.7%) with frontotemporal dementia, one (1.9%) with semantic dementia, and one (1.9%) with atypical dementia. The duration until progression to dementia was (mean±SD) 98.1±61.1 weeks.
Predictors of progression to dementia
A Cox’s proportional hazard model that included age, sex, baseline MMSE, decline in MMSE at 6 months, and baseline depression as predictors found that only age, MMSE, and decline in MMSE at 6 months predicted the progression from MCI to dementia. Sex and depression were not statistically significant predictors. Results are shown in Table 2.
Predictors of time to progression from MCI to dementia.
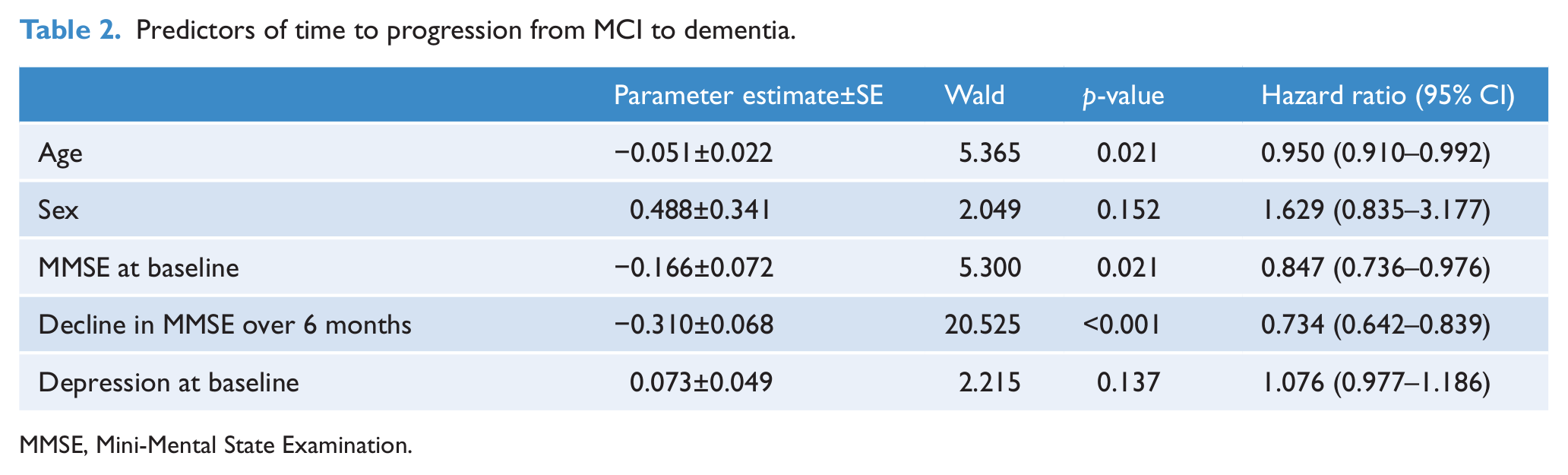
MMSE, Mini-Mental State Examination.
When analyses were repeated with the total NPI score or the change in the total NPI score over 6 months included in the model instead of depression, results were unaltered and neither of these variables emerged as significant predictors.
Discussion
The current study found that approximately 30% of patients with MCI defined by the Petersen criteria progressed to dementia within 3 years. This is consistent with previous research which found annual conversion rates from Petersen-defined MCI to dementia of just under 10% (Mitchell and Shiri-Feshki, 2009). As in previous research (Bruscoli and Lovestone, 2004), older age and lower cognitive ability predicted progression from MCI to dementia. Rate of decline in cognitive ability over 6 months was also a significant predictor. While decline rather than impairment has not been as widely examined in previous research (Hensel et al., 2009), early decline in MMSE scores in patients with MCI is associated with steeper longer-term trajectories of cognitive decline (Xie et al., 2011). Although the comparison of scores over time is influenced by other factors, such as measurement error, practice effects, and regression to the mean, the current findings underscore the clinical importance of assessing decline over time, particularly given the ease with which it can be calculated.
Depression, however, did not predict progression to dementia. It is unlikely that the issue could be simply attributed to statistical power: the current study, for example, involved a larger sample size and longer follow-up period than other studies that found depression was a significant predictor (Modrego and Ferrández, 2004; Palmer et al., 2007; Teng et al., 2007). The findings from the current study are nevertheless consistent with other research which found that depression does not predict progression to dementia (Brodaty et al., 2012; Rozzini et al., 2005). One possible explanation is that depression may reflect patients’ levels of cognitive decline, which in turn is a robust predictor of progression to dementia. Consistent with this possibility, there was a significant negative correlation between depression and cognitive ability at baseline in this sample.
A limitation of the current study was that we did not establish the reliability of raters across different testing sites. In addition, we did not have data on subtypes of MCI to determine whether these would assist in predicting outcome or how MCI subtype matched dementia type. Among MCI subtypes, multidomain amnaestic MCI has been reported to have the highest rate of progression to dementia (Brodaty et al., 2013). These limitations are relevant given the heterogeneity of patients with MCI. Rates and prevalences of neuropsychiatric symptoms in patients with MCI vary widely across different studies (Apostolova and Cummings, 2008; Monastero et al., 2009), possibly due to sample and methodological differences. Another limitation was the lack of data on other variables associated with MCI progression, such as loss of smell (Sun et al., 2012), biomarker information (e.g. apolipoprotein E genotype), and neuroimaging (Cui et al., 2011).
The implications from this study are particularly relevant in primary care. A diagnosis of MCI raises great concern to patients themselves and their families. Our findings indicate that longitudinal assessment with simple clinical tools can be helpful in determining which patients will develop dementia.
Footnotes
Acknowledgements
The Dementia Collaborative Research Centre is funded by the National Health and Medical Research Council. We thank the staff at nine sites who collected data and the patients and caregivers who supported the study. Janssen Pharmaceuticals sponsored the collection of data in the nine memory clinics and data management.
Funding
This work was supported in part by Janssen-Cilag.
Declaration of interest
HB and MW have worked on drug trials for patients with MCI and Alzheimer’s disease sponsored by major pharmaceutical companies including Eisai Pharmaceuticals, Eli Lilly, GlaxoSmithKline, H Lundbeck, Janssen-Cilag, Medivation, Merck Sharp and Dohme, Novartis Pharmaceuticals, Nutricia, Pfizer, Prana Biotechnology, Sanofi-Aventis, Servier, Tau Rx, Voyager Pharmaceutical Corporation, and Wyeth. MW also works on trials sponsored by Forest, Takeda, and the Buck Institute. HB has also been a consultant, advisory board member, and sponsored speaker for Baxter, H Lundbeck, Janssen-Cilag, Medivation, Novartis Pharmaceuticals, Pfizer, Prana Biotechnology, Voyager Pharmaceutical Corporation, and Wyeth. MW has also been a consultant and speaker for these companies as well as bioCSL, Prana Biotechnology, Eli Lilly, and Merck Sharp and Dohme. DA has served as a paid consultant to Janssen-Cilag and has also received payment for consultancies from Baxter, Eli Lilly, Lundbeck, Prana, Pfizer, and Novartis. MHC reports no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
