Abstract
Objectives:
Body dysmorphic disorder (BDD) is characterized by a preoccupation with a misperceived flaw in appearance, causing significant distress and disability. Neuropsychological research has revealed deficits in executive function and inhibitory control of emotional responses. The few previous structural neuroimaging studies have had inconclusive findings and we aimed to take this field of research forward by contributing high quality structural data.
Methods:
To investigate regional brain volumes we compared 20 BDD participants and 20 matched controls using high-resolution structural T1-weighted magnetic resonance imaging (MRI). The MRI data was subjected to cortical reconstruction and volumetric segmentation using Freesurfer software.
Results:
Results showed the right orbitofrontal cortex, bilateral thalamus, left anterior cingulate cortex, hippocampus and amygdala were significantly smaller in the BDD sample compared to controls. The most pronounced differences were in the right orbitofrontal cortex and left anterior cingulate cortex, as these areas were smaller in BDD participants independent of reduced global brain volumes. Duration of illness significantly negatively correlated with right orbitofrontal cortex volumes.
Conclusions:
This is the largest volumetric neuroimaging study in BDD to date and provides important data on volumetric differences that implicate fronto-limbic circuits.
Introduction
Body dysmorphic disorder (BDD) is a mental disorder characterized by a preoccupation with an imagined defect in physical appearance, or if a slight abnormality is present that the concern for it is excessive. Individuals with BDD have comparable levels of disability to other mental disorders, including social phobia (Coles et al., 2006) and major depression (Phillips et al., 2007). In particular, BDD and obsessive compulsive disorder (OCD) have been linked in terms of symptomatology, familial prevalence and the high degree of comorbidity (Bienvenu et al., 2000; Marazziti et al., 2006). The prevalence of BDD is high, with an Australian study showing 2.3% met diagnostic criteria in a student sample (Bartsch, 2007) and international community samples showing similar rates (Rief et al., 2006).
Neuropsychological studies have provided evidence of cognitive impairments in BDD, including executive function (Dunai et al., 2009; Hanes, 1998), selective attention (Buhlmann, et al., 2002), information processing (Deckersbach et al., 2000), recognition of facial affect (Buhlmann et al., 2004), and a bias towards detailed visual processing (Buhlmann et al., 2004; Feusner et al., 2010a, 2010c). Such research has started to elucidate the cognitive basis of BDD symptoms, and has also provided data to compare BDD against related disorders such as OCD. For example, Hanes’ (1998) executive function results using the Stroop and the Tower of London tasks suggest deficits in prefrontal brain regions. OCD patients show similar difficulties on these tasks, and have had convergent neuroimaging evidence that compellingly confirms prefrontal deficits (Remijnse et al., 2006; Venkatasubramanian et al., 2012). In fact, a pathogenic explanation of pathways involving the orbitofrontal cortex (OFC) and anterior cingulate cortex (ACC) has received consistent support in OCD, while there is a paucity of neurobiological and neuroimaging data in BDD.
Research has begun to provide some characterization of the neurobiology of BDD, with results so far suggesting widespread but varying functional and structural abnormalities (reviewed in Buchanan et al., 2011). Most recently, two diffusion tensor imaging (DTI) studies have investigated white matter integrity in BDD participants. The smaller of these studies (n = 14) found a significant negative correlation between white matter integrity and BDD symptoms (Feusner et al., 2013). The second DTI study found widespread white matter degradation in the BDD participants (Buchanan et al., 2013). Given that performance on cognitive tasks is related to wide distributions throughout the brain, the white matter findings suggesting that there is inefficient communication between distinct grey matter regions may go some way to explain the neuropsychological deficits in BDD.
There have been a small number of structural imaging studies in BDD similar to the current study. Atmaca et al. (2010) investigated brain volumetric parameters in 12 male BDD participants via manually tracing regions of interest (ROIs). They found that OFC and ACC volumes were significantly smaller in BDD than in healthy controls. Length of illness was inversely correlated with OFC volumes in the patient group on both the left and right sides. In addition, this study found increased total white matter volumes and increased thalamic volumes.
Using voxel-based morphometry (VBM), an automated method of group comparisons, Feusner et al. (2009) investigated regional volumes in 12 BDD participants and 12 controls. Analysis revealed no statistically significant volumetric differences; subsequent hand-traced ROI analysis also revealed no regional volumetric differences. However, a trend towards a positive correlation between symptom severity and right amygdala and left inferior frontal gyrus volumes was noted. Given the small sample size, it is likely that the study had inadequate power to detect significance differences.
Perhaps the only consistent property of previous volumetric studies with BDD is that they have all had relatively small sample sizes, from six to 12 participants. In addition, the variety of imaging techniques used, together with the absence of replications, limits the conclusions from neuroimaging studies thus far.
The current study’s aim was to provide robust evidence of volumetric differences in grey matter regions in BDD using automated brain segmentation software: FreeSurfer. We sought to investigate global brain characteristics including gross volumetric differences in the four cortical lobes and, subsequently, across our ROIs. We developed specific hypotheses based on past neuroimaging findings in both BDD and OCD; specifically, that there would be reduced volumes in the OFC, ACC, hippocampus, amygdalae and changes in thalamic volumes between BDD patients and healthy controls. We also conducted exploratory comparisons on 121 other brain regions. In addition, it was hypothesized that symptom severity would correlate to amygdala and OFC volumes, and that OFC volumes would have a negative correlation with duration of illness.
Methods
Participants
Forty individuals were recruited, comprising 20 individuals with BDD and 20 healthy controls, aged between 19 and 64 years. Recruitment for the BDD group was conducted via referrals from St Vincent’s Hospital Body Image clinic in Melbourne, Australia, where clients were identified as having BDD and introduced to the research project. Participants gave their informed consent and diagnosis was then confirmed by the research team using the Body Dysmorphic Disorder Diagnostic Module (BDD-DM) and symptom severity was recorded using the Yale–Brown Obsessive Compulsive Scale Modified for Body Dysmorphic Disorder (BDD-YBOCS) (Phillips et al., 1997). BDD patients were excluded if they had a past or current psychotic disorder, OCD, bulimia nervosa, anorexia nervosa, alcohol or substance abuse history, intellectual/cognitive impairment, metal implants or neurological disturbance. Furthermore, BDD participants were excluded if they had a comorbid mental disorder that was considered to be their primary diagnosis, ensuring that all individuals in the patient sample had BDD as their primary diagnosis.
The control group comprised members of the public and had no personal or family history of a mental disorder. All participants had English as their preferred language and a Wechsler Test of Adult Reading (WTAR) pre-morbid intelligence quotient (IQ) score of >80. Participants were assessed with the Mini-International Neuropsychiatric Interview (MINI; Sheehan et al., 1998), as well as the BDD-DM (Phillips et al., 1997). Handedness was assessed with the Edinburgh Inventory (Oldfield, 1971). A more detailed account of selection criteria and demographic characteristics is described elsewhere (Buchanan et al., 2013).
Magnetic resonance imaging (MRI) acquisition
Participants were scanned using a 3T scanner (Siemens Magnetom TrioTim, Germany) at the Murdoch Children’s Research Institute (Royal Children’s Hospital, Melbourne, Australia). An AC-PC aligned high-resolution structural T1-weighted MPRAGE sequence (512 slices per slab; slice thickness = 1 mm; TE = 2.15 ms; TR = 1900 ms; field of view = 256 mm; in plane resolution 0.5 × 0.5 mm2) was acquired allowing high-quality data for FreeSurfer brain processing.
Data analysis
The MRI data were subjected to cortical reconstruction and volumetric segmentation analysis using the FreeSurfer software package (Version 4.5; www.martinos.org/freesurfer) based on established and largely automated processing steps. These steps included motion correction, removal of non-brain tissue, segmentation of the sub-cortical white matter and deep grey matter volumetric structures, intensity normalization, tessellation of the grey matter – white matter boundary, automated topology correction, and surface deformation following intensity gradients to optimally place the grey/white and grey/cerebrospinal fluid borders at the location where the greatest shift in intensity defines the transition to the other tissue class (Ségonne et al., 2007). Registration to a spherical atlas was employed, which utilized individual cortical folding patterns to match cortical geometry across subjects. The procedures for the measurement of volumes have been validated against manual tracing methods (e.g. Keller et al., 2012).
Raw volumetric data composed 139 brain regions for each participant. Each participant’s brain segmentation was visually inspected and particular attention was paid to regional volumes that fell outside two standard deviations from the group mean. There were 10 regions in total among four participants where there was measurement error due to problems with the automated skull strip, white matter identification or segmentation. For consistency of sample size across regions, these 10 values were replaced with group mean values. Exclusion of these values would have yielded the same overall results. One participant’s scans were reanalyzed after adjusting skull strip tolerances.
Statistical analyses
Data were checked for normality and parametric tests were selected as they were the most suitable. Statistical analyses were performed using the Statistical Package for Social Sciences (SPSS). We restricted the initial analysis to a priori hypothesized brain regions to reduce potential type 1 errors. The p-value for our specific hypotheses was set at 0.05, two tails, and a series of one-way analysis of variance (ANOVA) were conducted to compare the control and BDD group volumes. In addition, an analysis of covariance was conducted to adjust for global brain volume differences and to determine whether volumes for ROIs changed independently from whole brain changes. We used the FreeSurfer output measure of supratentorial volume as a proxy of total brain excluding cerebellum (grey and white matter) and brain stem. It is computed based on everything inside the pial surface, plus any structures that might fall partially or totally outside of the pial (e.g. hippocampus, amygdala and corpus callosum).
Given that FreeSurfer automatically computes many regional brain volumes, an exploratory analysis with a more conservative p-value of 0.01, two tails, was conducted for the remaining 121 regions, some of which were components of the larger ROI already measured. For example, caudal and rostral areas of the ACC combine to create the ACC. For brevity, only areas that were significantly different or showed a trend towards being different across the groups are reported.
Pearson’s correlations were conducted between symptom severity as measured by BDD-YBOCS scores, and the bilateral amygdalae and OFC volumes. Duration of illness was also correlated to our ROIs; for these analyses partial correlations were conducted controlling for the confounding effect of age.
Results
The two participant groups were well matched on age, sex, education, estimated IQ and handedness, as shown in Table 1. The clinical data indicated that the mean BDD severity was in the ‘moderate’ range, which is defined as scores between 16 and 30 on the BDD-YBOCS (Phillips et al., 1997). Areas of aesthetic concern for our sample were generally the face, skin and hair, but included other body areas such as breasts and legs. Duration of illness was included for 17 participants because onset was not determined for three participants.
Demographic and clinical variables of the BDD and control groups.
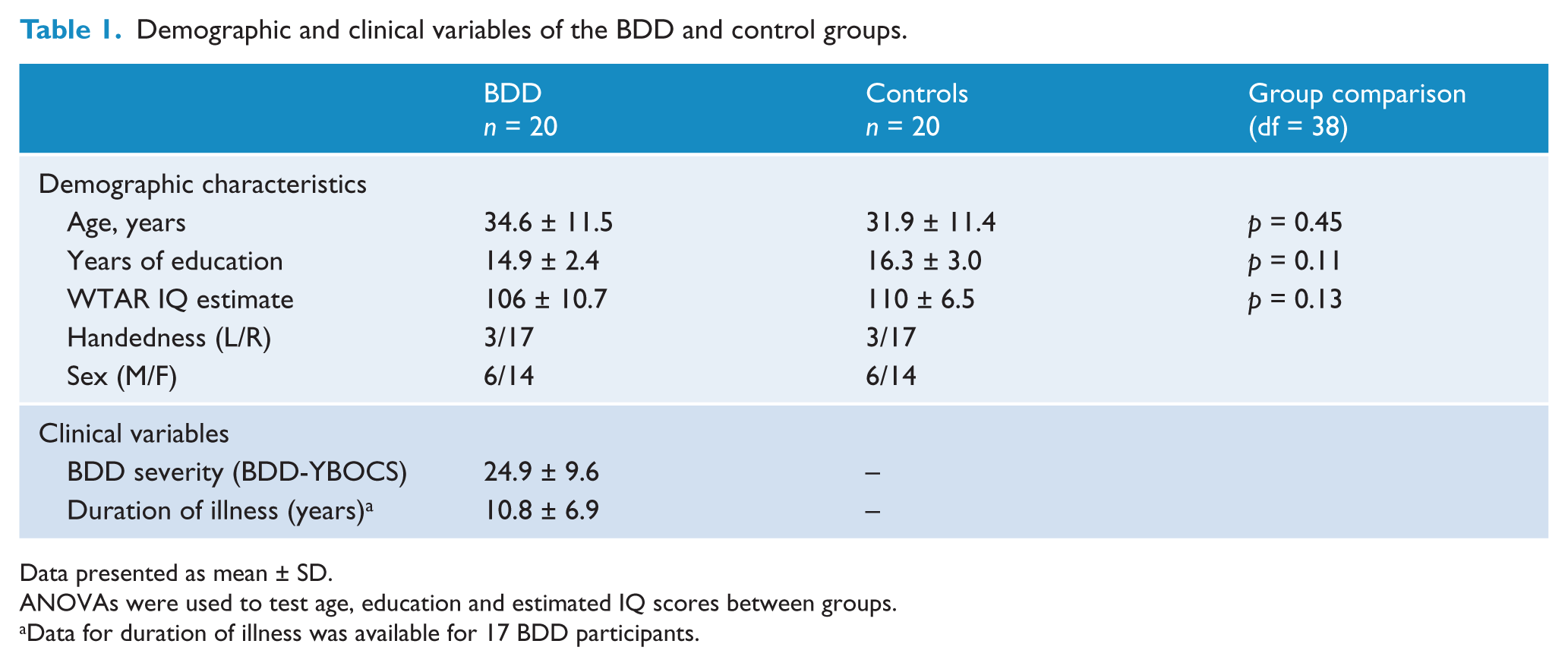
Data presented as mean ± SD.
ANOVAs were used to test age, education and estimated IQ scores between groups.
Data for duration of illness was available for 17 BDD participants.
Whole brain
The mean volumes (cm3) for global brain characteristics are presented in Table 2, as well as the p-value derived from the one-way ANOVA to compare groups. The results indicated that differences between groups in intracranial volume, cerebrospinal fluid and white matter volumes were not statistically significant. Total grey matter volume, however, was significantly smaller among BDD participants.
Whole brain and lobar volumes (cm3) between the BDD and control groups.
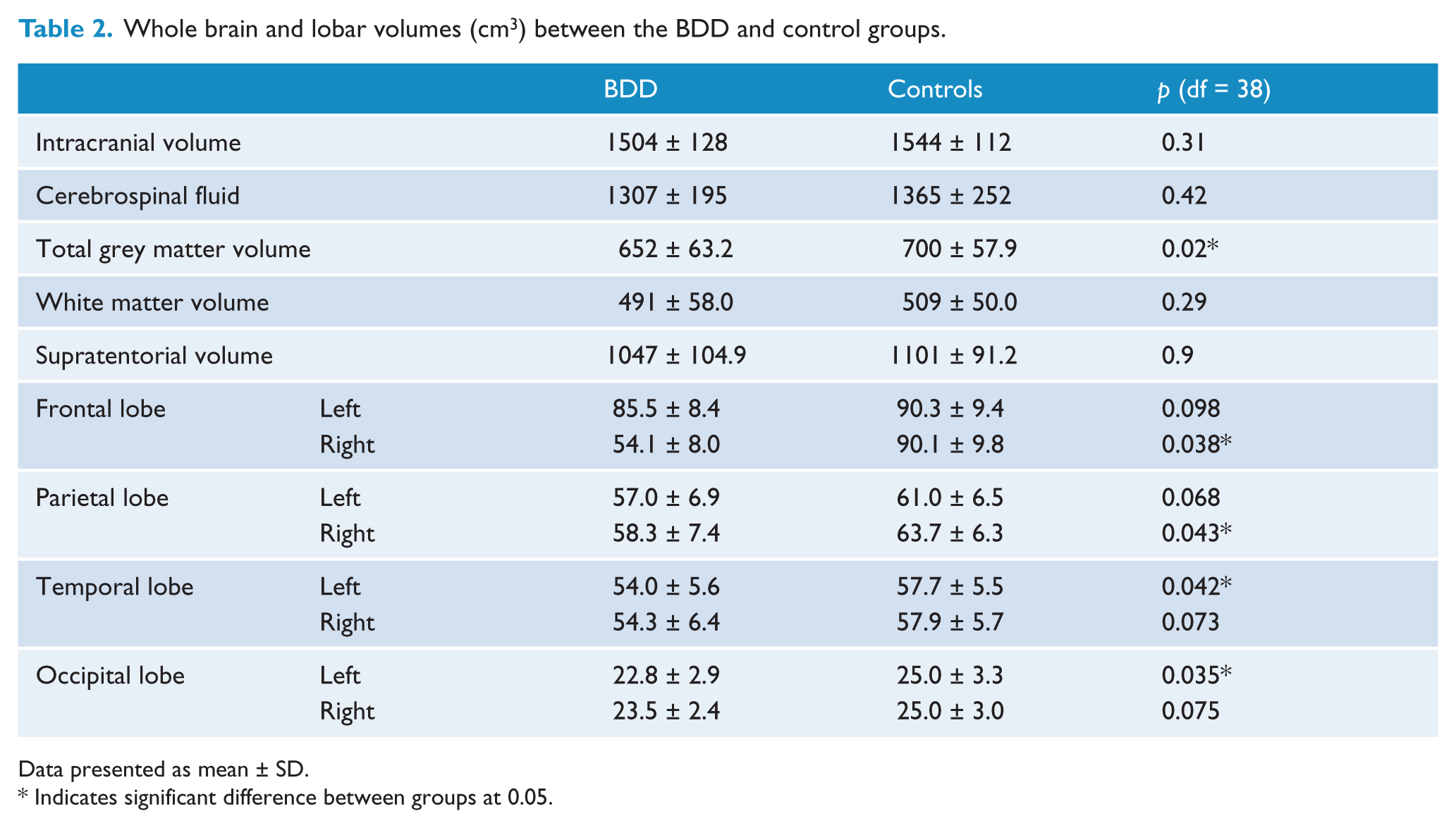
Data presented as mean ± SD.
Indicates significant difference between groups at 0.05.
Our measure for total brain volume supratentorial volume was found to not be significantly different between groups, F = 3.01, p = 0.09, though the trend was sufficient to make it a suitable covariate. In terms of lobar differences, the BDD group had significantly smaller right frontal, right parietal, left temporal and left occipital lobes. However, further investigation co-varying these regions with total brain volume showed that results ceased to have statistical significance, indicating that lobe reductions occurred in the context of total brain reduction rather than a lobe-specific reduction.
Regions of interest
Table 3 shows the mean volumes for our ROIs across groups, as well as the two p-values: the group comparison during an ANOVA and the ANCOVA to co-vary for global brain differences.
Cortical lobes and regions of interest volumes (cm3) across the groups.
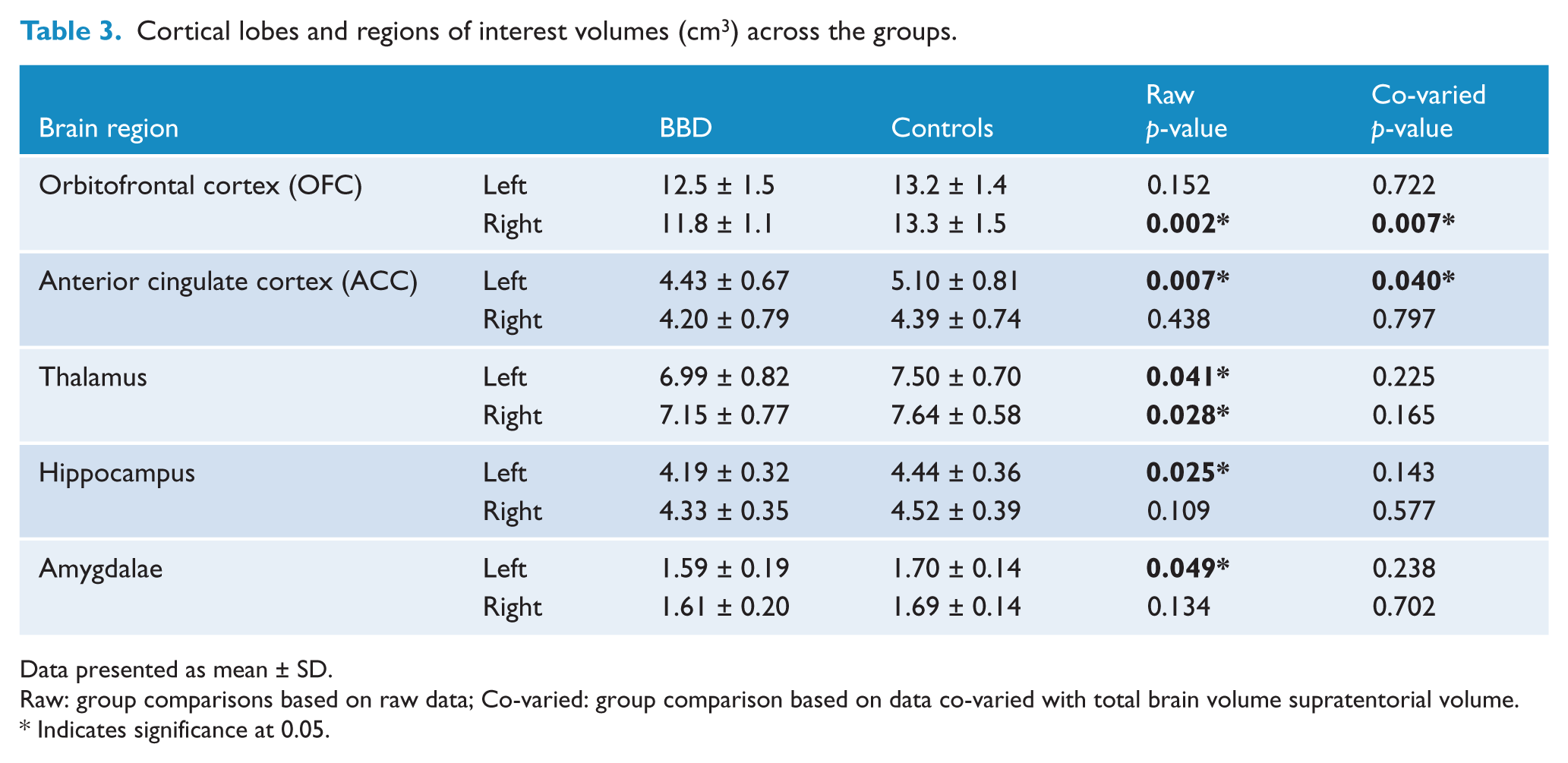
Data presented as mean ± SD.
Raw: group comparisons based on raw data; Co-varied: group comparison based on data co-varied with total brain volume supratentorial volume.
Indicates significance at 0.05.
In terms of our a priori ROIs, the right OFC, left ACC, bilateral thalamus, left hippocampus and left amygdala were significantly reduced in volume in the BDD sample. When co-varying for the total brain difference, the significant differences remained for the right OFC and left ACC, indicating that these areas were smaller in BDD participants independent of smaller global brain volumes. Figure 1 shows the orbitofrontal cortex segmented into medial and lateral regions, and Figure 2 shows rostral and caudal areas of the ACC.
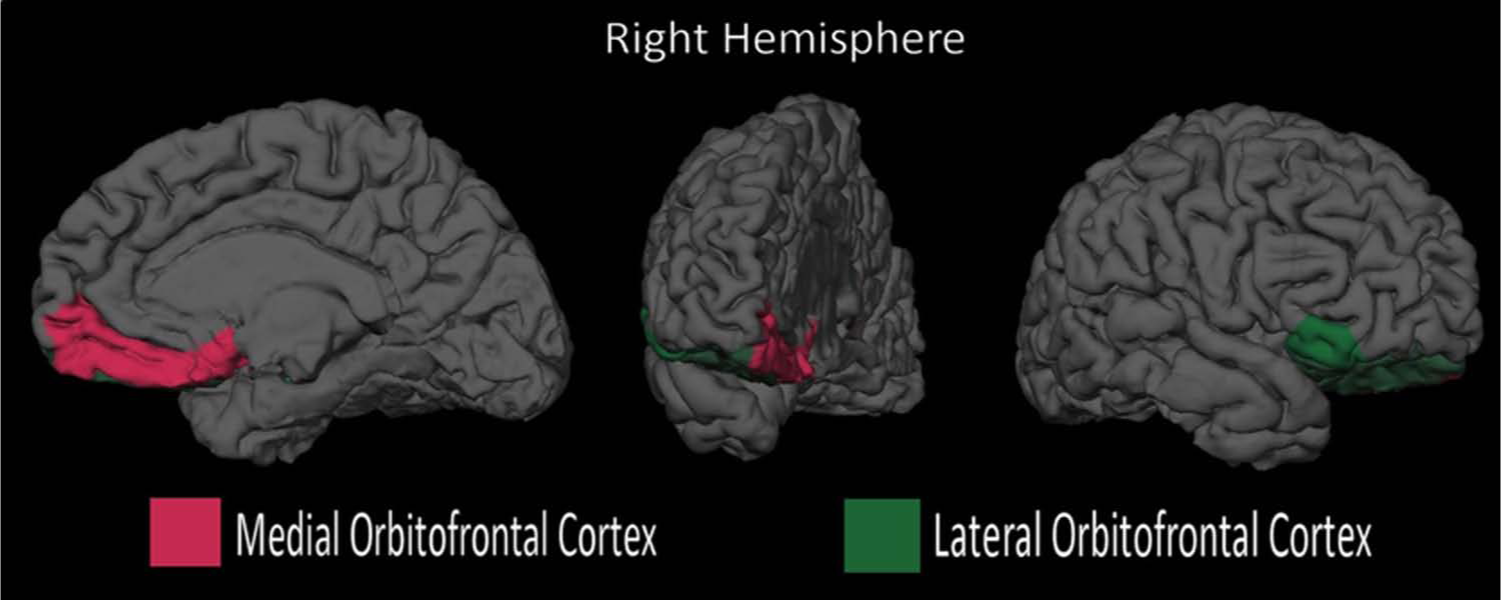
Right orbitofrontal cortex (OFC) in BDD, medial and lateral regions.
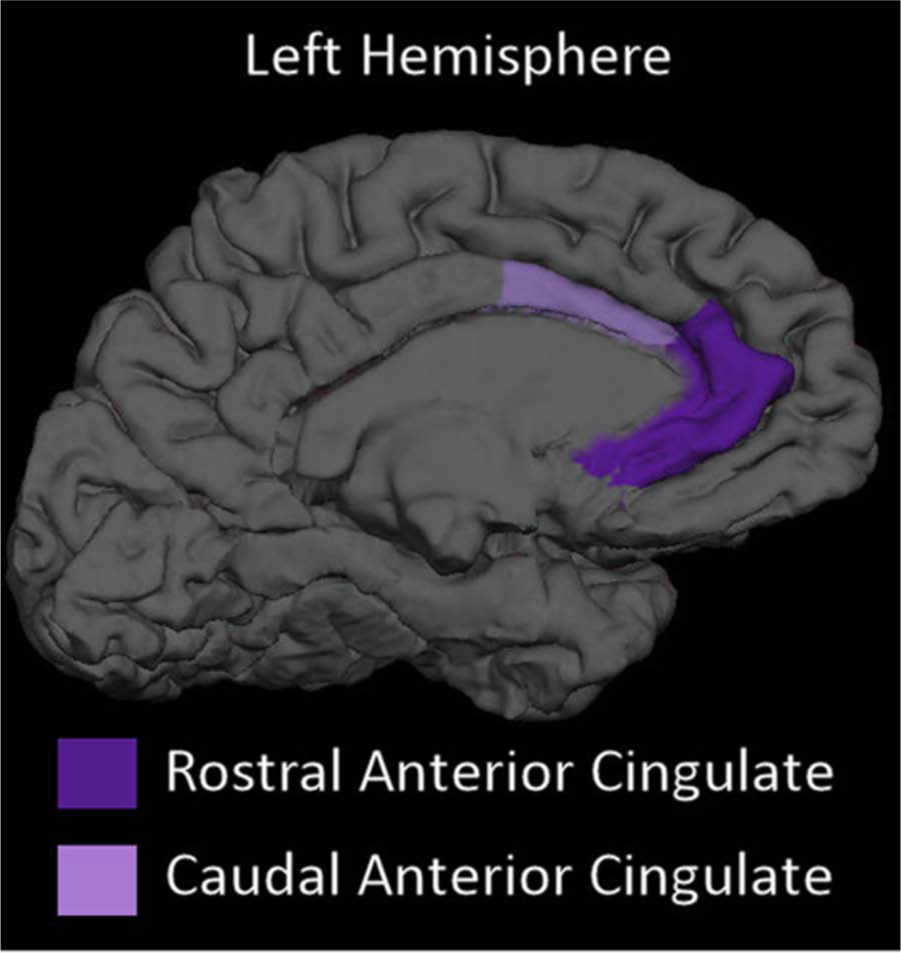
Anterior cingulate cortex (ACC) in BDD, rostral and caudal regions.
The exploratory analysis revealed that four areas were significantly different between groups as defined by a p < 0.01, both for raw volumes and volumes co-varied for total brain volume. The right superior parietal, right precuneus, right lateral OFC and left caudal ACC were significantly smaller in the BDD group compared to controls. The latter two areas were simply smaller components of our a priori ROIs (OFC and ACC) and thus were the predominant contributors to the reduction in the larger areas. The left cerebellum was also significantly smaller in the BDD group for raw volumes but lost its significance when volumes were co-varied for whole brain volume. Other areas that did not reach the required significance value but nevertheless showed a substantive difference (p < 0.05) for raw volumes are also presented in Table 4. For example, the bilateral fusiform gyri were close to, but did not meet, our strict significant test.
Exploratory analysis of regional brain volumes.
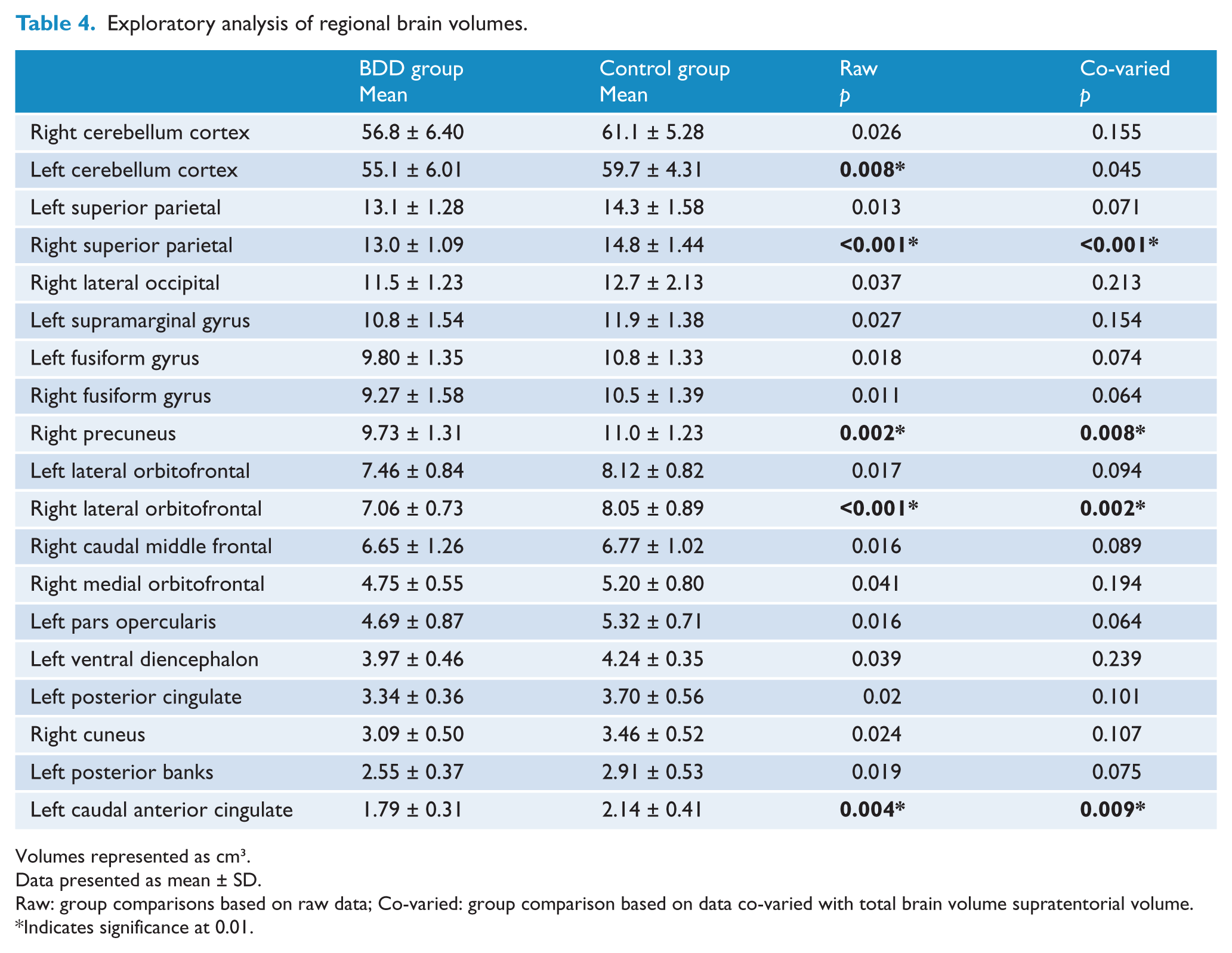
Volumes represented as cm3.
Data presented as mean ± SD.
Raw: group comparisons based on raw data; Co-varied: group comparison based on data co-varied with total brain volume supratentorial volume.
Indicates significance at 0.01.
Correlation analysis revealed a significant negative relationship between symptom severity scores and the left amygdala volumes (r = −0.492, p = 0.027). For the right amygdala there was a non-significant trend towards a negative correlation (r = −0.421, p = 0.065). There was no correlation between symptom severity and OFC volumes on either the left (r = 0.10, p = 0.675) or the right (r = −0.056, p = 0.814).
There was a significant negative correlation between duration of illness and the right OFC (r = −0.520, p = 0.032; Figure 3). The results of the partial correlation between right OFC and illness duration controlling for age revealed a slightly (but not significantly) stronger correlation (r = −0.537, p = 0.032), indicating that duration of illness was related to OFC volumes independently of age. Left OFC volumes were not correlated with illness duration (r = −216, p = 0.405).
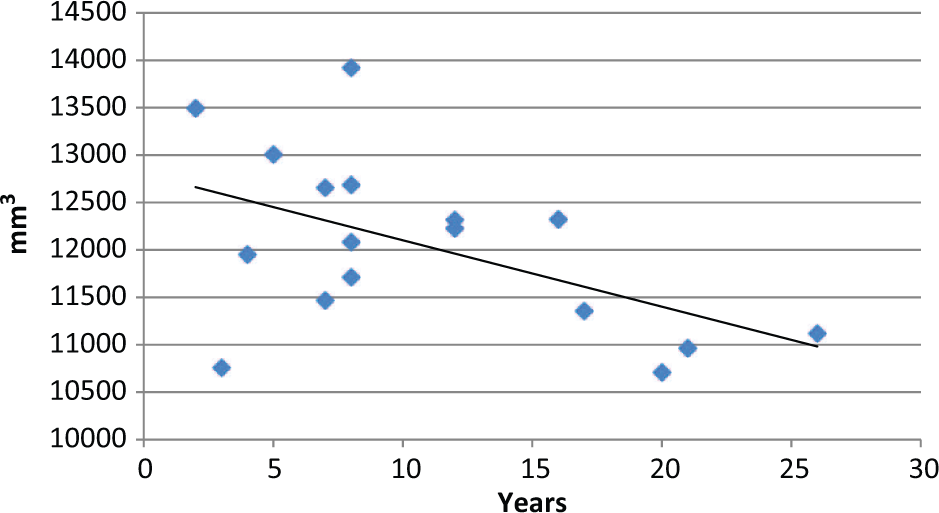
Right orbitofrontal cortex (OFC) volume (mm3) versus illness duration (years).
Discussion
This is the largest volumetric neuroimaging study in BDD patients to date, providing data for hypothesized regions of interest, as well as offering rich exploratory findings. The main finding was that in BDD patients there were reduced volumes in the right OFC and left ACC. The bilateral thalamus, left hippocampus and amygdala were also smaller in the BDD group, although these differences were no longer significant when co-varying for total brain volume.
Our result of reduced left ACC volumes is in part consistent with the findings of Atmaca et al. (2010) who found bilateral volume reductions in this region in BDD patients compared to controls. This finding is particularly meaningful given that the ACC has an important inhibitory effect over emotional responses and is involved with other executive tasks (Albert et al., 2012). Within BDD, dysregulation of emotion is a central affective symptom that has been demonstrated in neuropsychological research using emotion Stroop tasks (Buhlmann et al., 2002). Thus, pathology in the ACC may mediate these symptoms. Reduced inhibitory control mediated by the ACC has also been conceptualized as being of central importance in the pathogenesis of OCD, and is among the most consistent findings in this disorder (Chamberlain et al., 2005; Kühn et al., 2013; Radua et al., 2010; Venkatasubramanian et al., 2012).
Reduced OFC volumes is also noteworthy given it is a common finding in OCD (Rotge et al., 2010) and its involvement in decision-making, emotion regulation and self-focused thinking. It facilitates behavioural flexibility after negative feedback, allowing the unlearning of (emotional) associations (Remijnse et al., 2006). Not only were OFC volumes significantly reduced compared to controls generally, but our data showed that individuals with a longer duration of illness at the time of the scan were likely to have smaller right OFC volumes. The average duration of illness for our BDD participants was almost 11 years (between 2 and 26 years) and the relationship with OFC volumes was confirmed to exist independent of age.
The importance of the OFC in BDD has also been highlighted in functional MRI work by Feusner et al. (2010b), whereby BDD participants showed relative hyperactivity in the left OFC when shown their own face or the faces of others. Furthermore, their study also showed that within the BDD group the level of symptom severity was related to activation in the right OFC. Our results did not indicate that symptom severity was related to right OFC volumes, but, taken together with the past activation data and our own illness duration results, it seems likely that differences in the OFC is a key region in BDD pathophysiology. Indeed, OFC volume reductions due to pathogenic development may lead to poorer outcomes and chronic BDD.
The OFC and ACC are thought to be important to both frontostriatal circuits that mediate inhibitory control, emotional learning and flexibility in responses. In addition, these areas are consistent with frontolimbic dysfunction. Recent DTI results in BDD show that a major white matter connection between frontal areas and amygdalae, the uncinate fasciculus, had reduced directional diffusion, suggesting a weaker neural pathway between these areas (Buchanan et al., 2013). In this context, individuals with BDD may have difficulty with top-down regulation of amygdalae reactivity to control negative affect and mediate threat perception, explaining BDD symptomatology. Such frontolimbic involvement may be related to the frontostriatal explanation that has become well established in OCD (Harrison et al., 2009; Saxena et al., 2001; Venkatasubramanian et al., 2012).
The left amygdala volumes were significantly negatively correlated with symptom severity. The amygdalae are of central importance to many psychiatric and, in particular, anxiety disorders and similar findings have been reported in related disorders such as general anxiety disorder (Stein et al., 2002) and are thought to be a key pathology in emotional dysregulation, eliciting speculation that reduced connection between the amygdala and OFC region could reflect a phenotype that is common among disorders involving emotional dysregulation or impaired social–emotional functioning (Phan et al., 2009).
Our correlation was only found between symptom severity and the left amygdala. On the other hand, our OFC differences were on the right side. Based on previous findings there has been some discussion that laterality effects may be of importance to BDD (Atmaca et al., 2010; Feusner et al., 2011). However, in light of our mixed laterality results (possibly due to insufficient power to detect all differences), there is yet to be a clear picture in terms of left or right brain differences.
Our exploratory analysis revealed significantly smaller volumes in the right superior parietal and right precuneus regions. While these results should not be considered to be as reliable as our a priori hypothesis, due to the multiple comparisons and the chances of type 1 errors, they are presented here so that future research can further investigate their importance. For example, one possibly important exploratory finding was the right superior parietal lobule, which is involved in maintaining internal representations and kinaesthetic attention (Stoeckel et al., 2004). The precuneus is a component of the superior parietal lobule and has specifically been implicated in the recall of imagery, self-reflection and in self-related mental representations (Cavanna and Trimble, 2006). Thus, dysfunction in this area may help explain two important symptoms in BDD: distortions in body awareness and lack of insight.
In terms of whole brain volumes, the BDD group had significantly lower total grey matter. Past BDD studies have shown a similar, yet non-significant, reduction in total grey matter (Atmaca et al., 2010; Feusner et al., 2009). Our data, showing a relatively stable intracranial volume across groups combined with reduced grey matter volume, indicate that it is a true effect and not a sampling error. The findings may be due to either different developmental trajectories or degradation associated with BDD onset. These results are broadly consistent with other anxiety disorders (Koolschijn et al., 2009; Syal et al., 2012), and there is a growing consensus that grey matter changes associated with anxiety-related psychiatric disorders are structural preconditions rather than consequences or side effects of these pathological states (Kühn et al., 2011). Future research could focus on the neurobiological dynamics by investigating developmental pathways for BDD patients at onset and during remission.
In summary, this is the largest volumetric sample of BDD to date, showing a general reduction in grey matter volumes consistent with other psychiatric disorders and highlighting the importance of the OFC and ACC in BDD. The contribution of high-quality evidence implicating frontolimbic circuits is key to creating a coherent neurobiological model of BDD.
Footnotes
Acknowledgements
Ethics approval was granted by The Alfred Hospital’s ethics committee.
Funding
This study was funded by a Monash Strategic Grant.
Declaration of interest
None declared.
