Abstract
Objective:
To date, insufficient studies have focused on the relationship between childhood trauma and white matter integrity changes in healthy subjects. The aim of the present study was to explore the potential effects of childhood trauma on white matter microstructural changes by using voxel-based diffusion tensor imaging (DTI) to examine alterations in fractional anisotropy (FA) values in a group of young healthy adults.
Methods:
A total of 21 healthy adults with a history of childhood trauma exposures and 21 age- and sex-matched individuals without childhood trauma were recruited in the present study. The Childhood Trauma Questionnaire was used to assess five aspects of childhood trauma exposures. DTI data were obtained on a Philips 3.0-Tesla scanner. Voxel-based analysis was conducted to compare white matter FA values between groups.
Results:
Adults with self-reported childhood trauma experiences showed decreased white matter FA values in the genu and body of the corpus callosum and the left occipital fusiform gyrus (p < 0.001 uncorrected, voxel > 100). There was no significant difference in FA values between individuals with single and multiple childhood trauma exposures at the defined threshold.
Conclusion:
Our findings suggest that childhood trauma is associated with reduced microstructural integrity of the white matter in adulthood. These effects are still evident even in the absence of current psychiatric or medical symptoms, which may represent the vulnerability for developing mental disorders after childhood trauma experiences.
Introduction
Childhood trauma, including a spectrum of sexual, physical and emotional forms of abuse, as well as physical or emotional neglect, is highly prevalent both in western countries (Scher et al., 2004) and in China (Zhang et al., 2013a). It has been reported that childhood trauma is associated with the development of multiple psychiatric disorders and unfavourable behaviour patterns in later life (e.g. mood and anxiety disorders, personality disorders and substance dependence) (Kendler et al., 2000; MacMillan et al., 2001; Nelson et al., 2002). Although this relationship has been demonstrated in plenty of epidemiologic studies, less is known about the mechanisms underlying this association. Recently, biological findings such as reduced grey matter volumes of specific brain areas (van Harmelen et al., 2010), dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis (Heim et al., 2008a) and activation of the inflammation system (Danese et al., 2007) among individuals with childhood trauma, even without psychiatric diagnoses, have been revealed, which may contribute to possible pathways from childhood trauma to mental disorders.
In the last two decades, neuroimaging has emerged as an important approach to discover brain structural and functional alterations and subsequently has been used to investigate the pathophysiology of mental diseases since its origin. In particular, structural magnetic resonance imaging (MRI) studies have documented a range of regional grey matter volume changes in subjects suffering from childhood trauma, which have been highly corroborated by a systematic review (Heim and Binder, 2012). Moreover, in two recent studies, childhood trauma has been found to be associated with remarkable corticostriatal-limbic grey matter volume reductions both in healthy adolescents (Edmiston et al., 2011) and in adults (Dannlowski et al., 2012). The neurotoxic impact of childhood trauma may also inhibit white matter myelination (Kikusui and Mori, 2009); however, to the best of our knowledge, less research has focused on the relationship between childhood trauma and white matter integrity changes. Diffusion tensor imaging (DTI), which can better reflect the integrity of white matter by analysing the anisotropic diffusion of water molecules in brain tissues in vivo, enables us to detect the microstructural changes in white matter (Bandettini, 2009). A most common parameter used in DTI measurements is the fractional anisotropy (FA) value, which is considered as a quantitative marker of white matter integrity, reflecting fibre density, axonal diameter and myelination (Daniels et al., 2013). To date, limited DTI studies have provided findings showing decreased FA values in the corpus callosum, left uncinate fasciculus, left inferior longitudinal fasciculus (left lateral occipital lobe), arcuate fasciculus (left superior temporal gyrus), cingulum bundle (left fusiform gyrus) and the left body of the fornix in subjects with early life stress (Choi et al., 2009, 2012; Eluvathingal et al., 2006; Jackowski et al., 2008; Seckfort et al., 2008; Teicher et al., 2010). However, the majority of these DTI studies did not perform whole brain analyses, merely regions of interest-based analyses, and most of these studies recruited subjects without specific exclusion of patients with post-traumatic stress disorder and other diagnoses. Therefore, the current outcomes require verification in further studies that well overcome these limitations in order to exactly describe the effects of childhood trauma on white matter integrity.
Owing to these limitations, we present here a study trying to detect microstructural abnormalities in young healthy adults with self-reported childhood trauma exposures by using voxel-based DTI to examine alterations in FA values as a mean to evaluate whole brain white matter integrity. Voxel-based analysis (VBA) is a highly reproducible, reliable and user-independent method which can assess comprehensive whole brain structure changes without the restrictions imposed by the prior selection of regions of interest (Zhang et al., 2013b).
Method
Participants
A total of 42 subjects (male/female, 18/24), aged 18–33 years, including 21 subjects with childhood trauma experiences (CT group) and 21 age- and sex-matched subjects without childhood trauma exposures (non-CT group), were recruited in the present DTI study from a survey that we had carried out to investigate the occurrence of childhood trauma in local communities and universities. For assignment to the CT group, individuals must have had experienced chronic moderate-severe trauma exposures (abuse and/or neglect) before the age of 16 years, which were assessed by the Childhood Trauma Questionnaire (CTQ). Subjects responded with no direct reference to childhood trauma as a key variable in the study. All subjects were thoroughly interviewed by two professional psychologists and were free from any current or lifetime history of psychiatric disorders according to the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria, as screened with the Structured Clinical Interview for DSM-IV interview (SCID). The general exclusions were as follows: (a) left-handedness, (b) standard scores > 50 on Zung’s self-rating depression scale (SDS) (Zung et al., 1965) or > 40 on Zung’s self-rating anxiety scale (SAS) (Zung, 1971), (c) significant medical illness, (d) presence of major sensorimotor handicaps, (e) history of seizures, head trauma or unconsciousness, (f) intake of any psychotropic medication or hormone, (g) alcohol or substance abuse, (h) women who were pregnant/lactating or in their menstrual period, (i) contraindications to the MRI scan, including metallic implants, retractors or braces and claustrophobia, and (j) an inability to keep still during MRI scanning. The demographic data were collected from all the participants using a self-designed questionnaire. This study was approved by the ethic committee of the Second Xiangya Hospital of Central South University. A complete description of the study was provided to every subject, after which written informed consent was obtained from each participant.
Assessment of childhood trauma
The existence or absence of childhood trauma was determined by the CTQ in all subjects. The CTQ is a reliable and valid self-reporting questionnaire with 28 items. It can yield five subscales which evaluate five aspects of childhood trauma exposures: emotional abuse, emotional neglect, sexual abuse, physical abuse, and physical neglect. Every subscale has five items and each item scores as 1 = never true, 2 = rarely true, 3 = sometimes true, 4 = often true, and 5 = very often true. Subjects who score higher than the threshold (moderate-severe) of a subscale are treated as existence of corresponding childhood trauma experiences. The cut-offs of each subscale for moderate-severe exposure are as follows: (a) emotional abuse ≥ 13, (b) emotional neglect ≥ 15, (c) sexual abuse ≥ 8, (d) physical abuse ≥ 10, and (e) physical neglect ≥ 10 (Bernstein and Fink, 1998). The Chinese version of the CTQ was introduced in our study. It was translated into Chinese by Zhao et al. (2005). The CTQ has good internal consistency: Cronbach’s alpha for the CTQ total score = 0.77 and the five subscales range from 0.41 to 0.68 in a Chinese sample of 794 individuals. It has been widely used in many Chinese studies, although many researchers would like to revise the CTQ so its subscales are more suitable for the Chinese population (Lu et al., 2013; Pu et al., 2013; Zou et al., 2013). Based on our survey, the internal consistency coefficients (Cronbach’s alpha) for the five subscales range from 0.46 to 0.78.
MRI acquisition
Imaging data were acquired using a Philips 3.0-Tesla scanner (Philips, Best, The Netherlands) in the Magnetic Resonance Center belonging to the Second Xiangya Hospital of the Central South University. Subjects were asked to lie on the scanner and keep their eyes closed. A standard birdcage head coil was used and the restraining foam pads were placed on two sides of the head to minimize head motion while cotton plugs were used to diminish the noise. DTI data were acquired using a single-shot spin-echo echo planar imaging (SE-EPI) sequence. The integral parallel acquisition technique (iPAT) was used with an acceleration factor of 2. The diffusion sensitizing gradients consisted of acquisition of the baseline image without diffusion weighting (b = 0 s/mm2) and the diffusion-weighted images (b = 1000 s/mm2) along 33 nonlinear directions. Images of the whole brain were acquired in an axial orientation with the following parameters: repetition time (TR) = 6500 ms, echo time (TE) = 70 ms, slice thickness = 2.5 mm, gap = 0 mm, field of view (FOV) = 240 × 240 mm2, acquisition matrix size = 128 × 128, slices = 60, scan time = 8’27’’.
Volumetric T1-weighted anatomic reference images were acquired using a three-dimensional magnetization-prepared rapid-acquisition gradient echo (3D-MPRAGE) sequence with the following parameters: slice thickness = 1 mm, gap = 0 mm, TR = 7.6 ms, TE = 3.7 ms, FOV = 256 × 256 mm2, flip angle = 8°, matrix size = 256 × 256, slices = 180, scan time = 2’58’’.
Voxel-based analysis of DTI data
Diffusion Toolkit software (trackvis.org/dtk/) was used to calculate the b0 map and fractional anisotropy map for each subject. The resulting fractional anisotropy images were normalized into Montreal Neurological Institute standard space with Statistical Parametric Mapping (SPM) 8; the steps were as follows: (a) the b = 0 image for each subject was co-registered with the T1 image of the same person, (b) the co-registration parameters were applied to the fractional anisotropy maps, (c) the T1 image of every subject was normalized to the SPM T1 template (in Montreal Neurological Institute standard space), (d) the normalization parameters were applied to the co-registered fractional anisotropy images, (e) all images were resampled with a voxel size of 2 mm × 2 mm × 2 mm, and (f) all the resulting fractional anisotropy images were spatially smoothed by an 8-mm full width at half maximum (FWHM) Gaussian kernel. The masking threshold for fractional anisotropy values was set at 0.20 for excluding voxels containing partial volume of cerebrospinal fluid and grey matter when comparisons were made.
Statistical analysis
Demographic and clinical data were analysed using the Statistical Package for the Social Sciences, version 16.0 (SPSS Inc., Chicago, IL, USA). Independent two-sample t-tests and chi-squared tests (χ2) were respectively used to test for the continuous variables and categorical variables between the two groups. Values are given as mean ± standard deviation. The level of two-tailed statistical significance was set at p < 0.05 for all tests.
Between-group differences of white matter FA values were analysed using two-sample t-tests on a voxel-by-voxel basis with SPM8 software. The potential confounders, such as education level, body mass index (BMI), scores of SDS and SAS, were included as covariates of no interest in the comparison. Clusters of 100 voxels or greater, surviving an uncorrected threshold of p < 0.001, were considered significant. Furthermore, the mean FA values of the clusters that had shown differences between groups in VBA analysis were extracted using region of interest (ROI) analyses. A two-sample t-test (SPSS 16.0) was used to compare these FA values.
Results
Demographics and clinical measures
As indicated in Table 1, the two groups did not significantly differ in age, sex ratio, years of education, BMI, SDS score, and smoking status. Individuals with childhood trauma showed higher levels in the SAS and CTQ scores (except sexual abuse) as compared with those without childhood adverse experiences. In maltreated subjects, the most common type of childhood trauma was emotional neglect (71.4%); a proportion of 61.9% (13/21) experienced at least two forms of childhood trauma exposures.
Demographic and clinical characteristics of all subjects (n = 42).
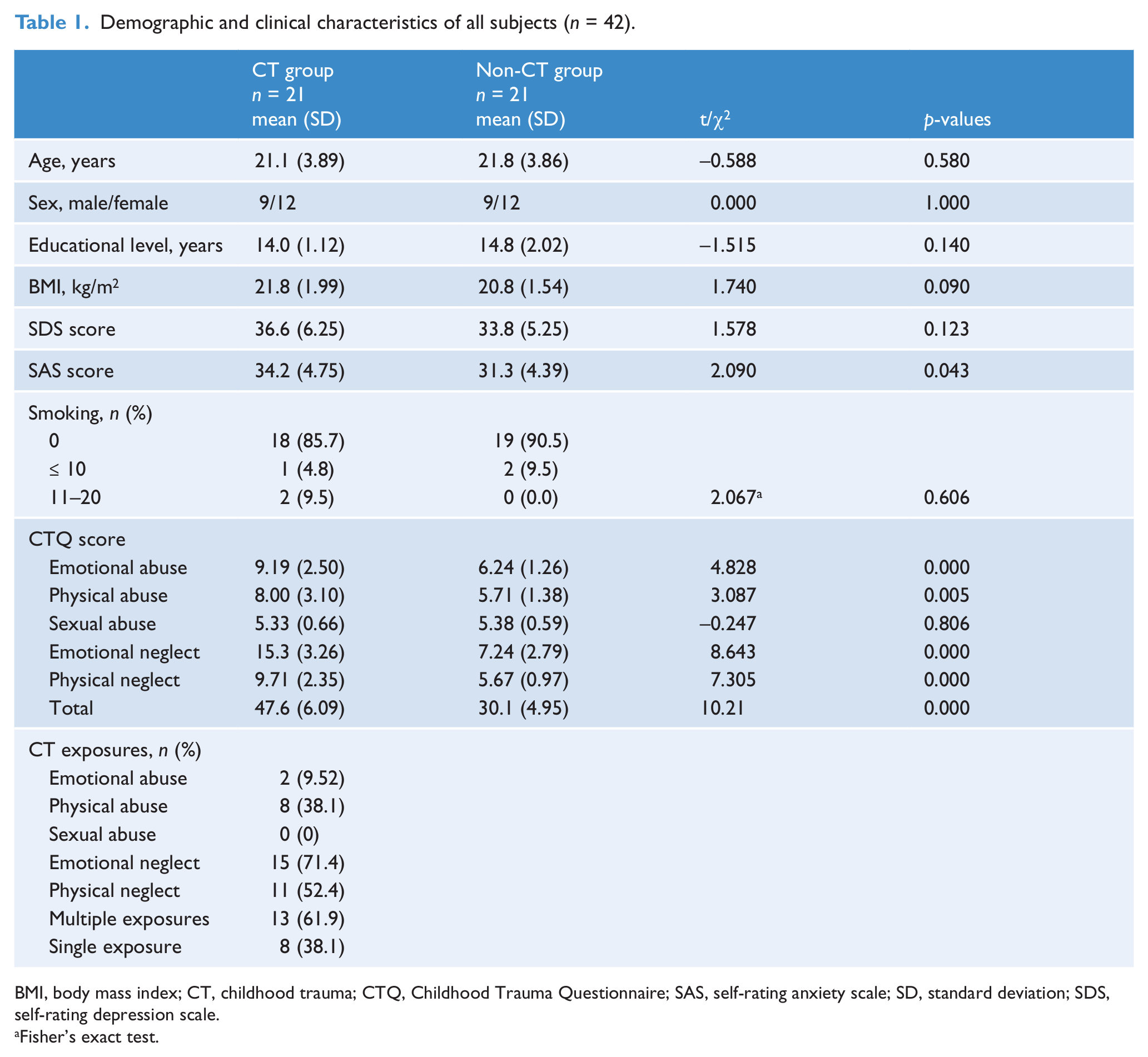
BMI, body mass index; CT, childhood trauma; CTQ, Childhood Trauma Questionnaire; SAS, self-rating anxiety scale; SD, standard deviation; SDS, self-rating depression scale.
Fisher’s exact test.
In subjects with childhood trauma, there was no statistical difference in demographics and scores of SAS and SDS between individuals with single and multiple childhood trauma exposures (all p > 0.35); however, subjects with multiple exposures had higher levels in CTQ physical neglect (10.2 ± 2.45 vs 7.50 ± 1.51, p < 0.05), emotional neglect (17.0 ± 1.68 vs 12.6 ± 3.46, p < 0.01) and total (51.8 ± 3.06 vs 40.8 ± 1.98, p < 0.01) scores.
Neuroimaging measures
The voxel-wise analysis revealed two regions where the white matter FA values were significantly lower in individuals with childhood trauma experiences. These two regions were mainly located in the genu and body of the corpus callosum and the left occipital fusiform gyrus (Figure 1). However, no region with increased FA values was found in maltreated adults. We also compared the FA values between individuals with single and multiple childhood trauma exposures, but no significant difference was found at the defined threshold.
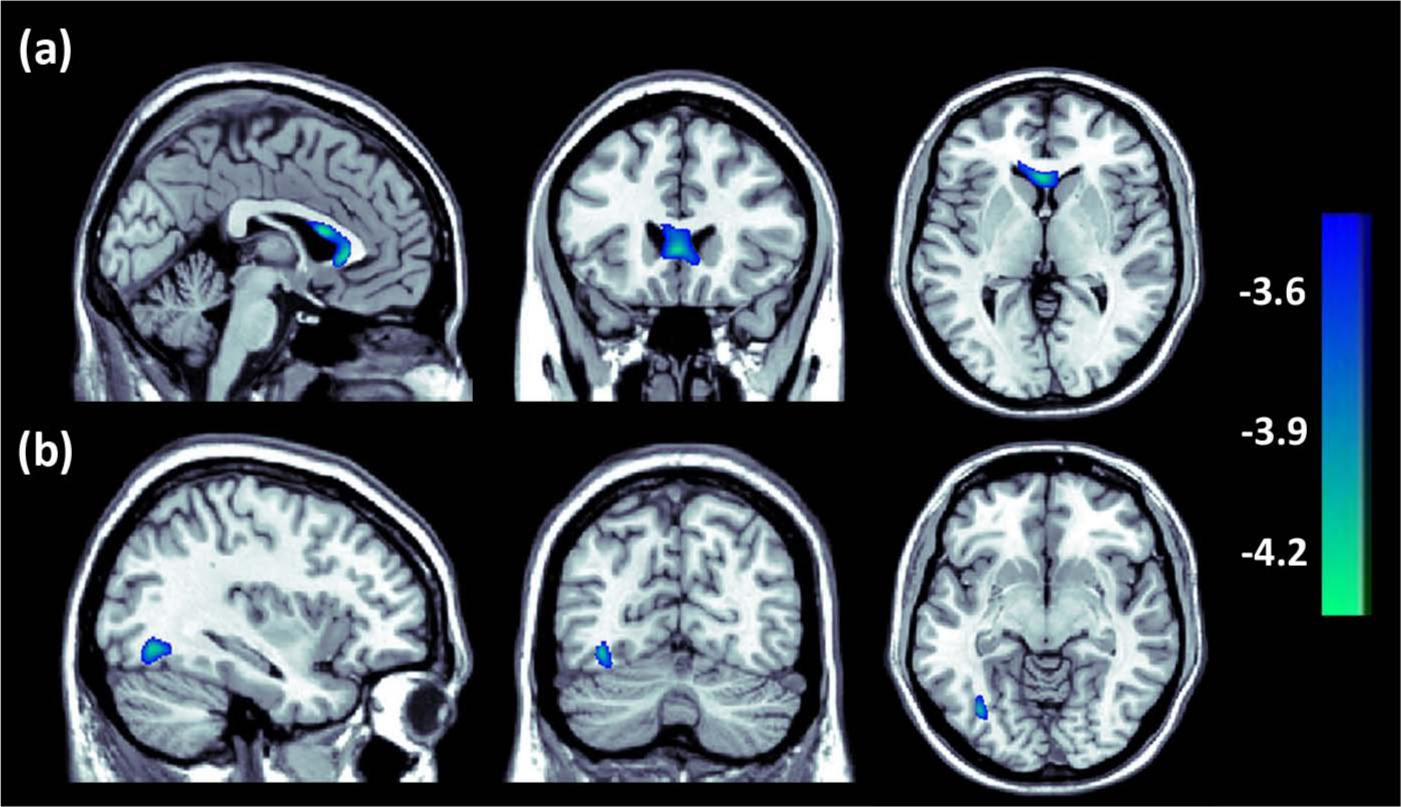
Regions of different FA values between individuals with and without childhood trauma (p < 0.001 uncorrected, voxel > 100). Decreased white matter FA values were detected in (a) the genu and body of the corpus callosum (x = −2, y = 24, z = 2, cluster size = 322, T = 4.34) and (b) the left occipital fusiform gyrus (x = −36, y = −68, z = −8, cluster size = 112, T = 4.23) in subjects with childhood trauma.
The mean FA values of the regions that showed significant differences in the above between-group analysis were extracted from each subject. As compared with subjects without childhood trauma, maltreated individuals exhibited 15.6% and 16.4% reductions of FA values in the corpus callosum and left occipital fusiform gyrus, respectively (Figure 2).
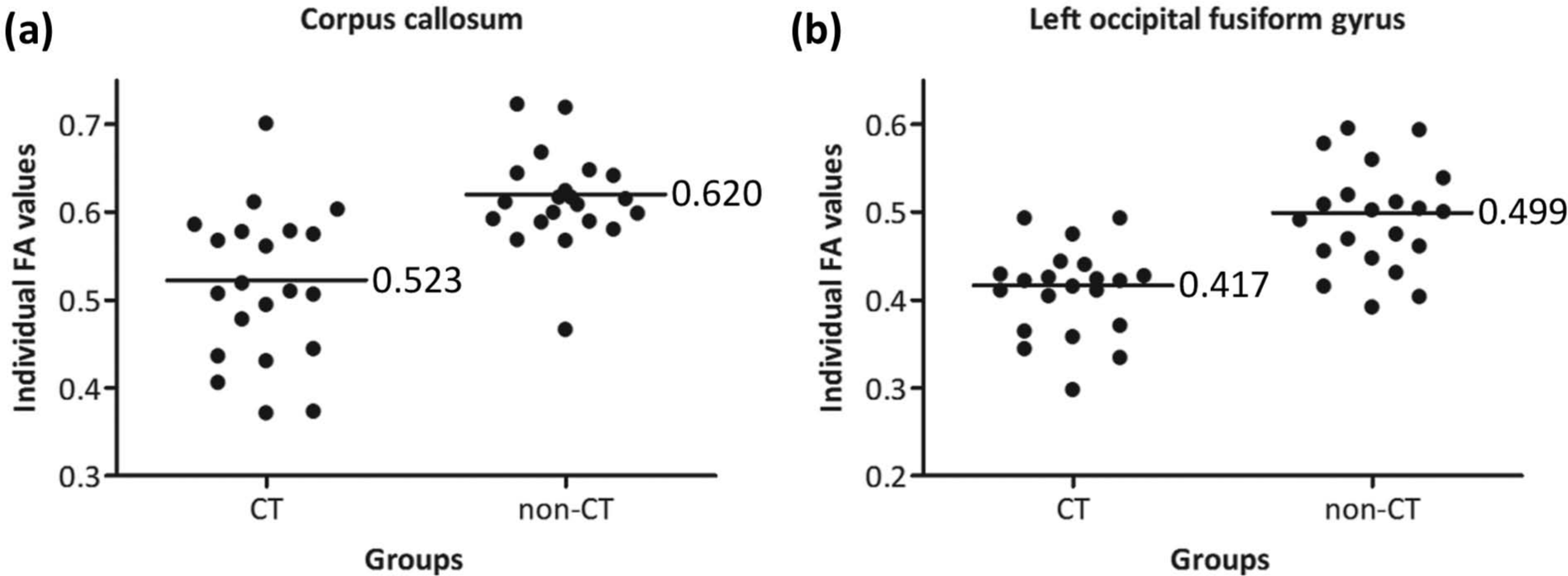
Comparisons of regional FA values between subjects with and without childhood trauma (p < 0.001). (a) Scatter plot of FA values in the corpus callosum. (b) Scatter plot of FA values in the left occipital fusiform gyrus.
Discussion
In this study, individuals with a history of childhood trauma exposures showed reduced white matter FA values in the corpus callosum and the left occipital fusiform gyrus. However, the FA values in subjects with single and multiple childhood trauma exposures did not differ significantly, which might suggest that there was not a dose–response relationship between extent of childhood trauma exposures and white matter integrity alterations in these maltreated adults. The current results may be outstanding since our sample is rigorously screened for both medical and psychiatric diagnoses, and therefore representative of a very healthy group of subjects; we have also analysed the white matter integrity across the whole brain volume. Taken together, our findings may have potential significance for contributing to understanding the brain mechanism of early life stress.
In the present study, FA values were significantly reduced in the genu and body of the corpus callosum among healthy individuals exposed to childhood trauma. The corpus callosum, which is involved in integrating sensory-motor functions, attention, language and memory-executive functions, is a largest fibre tract interconnecting the left and right hemispheres (Bellani et al., 2009). Our finding is consistent with previous observations that have identified the corpus callosum as most vulnerable to the adverse effects of early life stress. Our results are congruent with findings in a group of children with post-traumatic stress disorder secondary to intrafamilial abuse (Jackowski et al., 2008) as well as findings in healthy children with a high level of early life events (Seckfort et al., 2008), both demonstrating decreased FA values in the corpus callosum after childhood trauma exposures. The current finding is also generally comparable to a prior report in healthy non-clinical participants showing significant reductions in FA values within the genu of the corpus callosum among those exposed to the greatest number of early life stressors, suggesting reduced microstructural integrity of the corpus callosum associated with increased early life stress (Paul et al., 2008). In addition, our outcome is probably in parallel with a volumetric study revealing an association between macrostructural reductions of the corpus callosum and a history of childhood neglect (Teicher et al., 2004). From the evidence given above, we speculate that white matter integrity changes in the corpus callosum may exist early after exposure to childhood trauma and always persist to adulthood.
Another noteworthy finding in our study is decreased white matter FA values in the left fusiform gyrus (located in the occipital lobe). The fusiform gyrus, usually considered as the visual association cortex, is implicated in processing colour information, face and body recognition, word recognition and within-category identification (Liao et al., 2013). The fusiform appears to contain fibres from the cingulum bundle which is the most vital tract of the limbic lobe and connects the limbic lobe with the neocortex, especially the cingulate gyrus (Niedtfeld et al., 2013). The visual cortex is a plastic structure with early myelination which may be extensively modified by early life experiences (Sowell et al., 2003; Teicher et al., 2010). In a structural MRI study, Tomoda et al. (2009) have demonstrated that exposure to childhood sexual abuse is associated with decreased grey matter volume in the visual cortex bilaterally (including an 18.0% reduction in the left fusiform gyrus). Meanwhile, in a recent DTI study, similar alterations were found as well, revealing a strong relationship between witnessing domestic violence in childhood and reduced FA values in the visual limbic pathway in young adults (Choi et al., 2012). The current result also stands in line with a preliminary finding by Choi et al. (2009) who have reported decreased FA values in (a) arcuate fasciculus in the left superior temporal gyrus, (b) cingulum bundle in the left fusiform gyrus, and (c) the body of the fornix in young adults exposed to parental verbal abuse, although the mean FA values of the cingulum bundle in the left fusiform were generally lower than our finding. Finally, in functional MRI studies, altered responsiveness of the fusiform gyrus to negative emotional cues has been revealed to be associated with early life stress as well (Dannlowski et al., 2013; Edmiston and Blackford, 2013; Tottenham et al., 2011). It is quite interesting that childhood trauma is associated with structural and functional abnormalities in the visual cortex in many studies, including ours. In this context, we proposed that childhood trauma, especially being an eye-witness to adverse experiences, might exert a strong influence on the development of the visual cortex, which subsequently contributes to white matter integrity changes and grey matter atrophy in the visual cortex.
The origin of white matter FA changes in the corpus callosum and the left occipital fusiform in our study is a history of childhood trauma exposures. However, structural abnormalities in these two brain regions are frequently observed in many mental disorders; for example, major depressive disorder (Alexopoulos et al., 2008; Liao et al., 2013), bipolar disorder (Benedetti et al., 2011; James et al., 2011), post-traumatic stress disorder (Hedges et al., 2007; Jackowski et al., 2009), personality disorder (Carrasco et al., 2012; Niedtfeld et al., 2013) and schizophrenia (Ehrlich et al., 2013; Henze et al., 2012), which are closely correlated with childhood trauma. Therefore, the current findings of white matter integrity alterations in the corpus callosum and the left occipital fusiform in a group of young healthy adults may represent the biological vulnerability for developing mental disorders after childhood trauma experiences. Owing to these findings, it is also crucial to consider the neurobiological effects of childhood trauma in future studies in order to clearly define which of psychiatric diagnosis or childhood trauma induces abnormalities relative to normal controls.
Limitations
Several limitations should be considered when interpreting the present results. First of all, in our study, we did not find any difference in FA values between individuals with single and multiple childhood trauma exposures; however, the sample size in the present study is modest, which may give rise to the problem that some subtle changes in the brain cannot be observed. Second, we did not investigate the effects of different types of childhood trauma on white matter integrity changes. Third, childhood trauma was assessed by a retrospective questionnaire, which may lead to information bias (Heim et al., 2008b). In that case, we carried out a face-to-face interview with subjects to confirm the childhood adverse experience and reduce information biases as much as possible. Finally, our study was a cross-sectional design, which precluded causal inferences. Altogether, the findings of the present study need to be confirmed in further studies that can overcome these limitations.
In conclusion, our findings suggest that childhood trauma is associated with reduced microstructural integrity of the white matter in adulthood. These effects are still evident even in the absence of current psychiatric or medical symptoms, which may represent the vulnerability for developing mental disorders after childhood trauma experiences.
Footnotes
Acknowledgements
The authors would like to thank all the participants of this study.
Funding
The authors sincerely thank the support of funds from the Major Project of Chinese National Programs for Fundamental Research and Development (973 program, 2009CB918303 to LJL), the National Natural Science Foundation of China (30830046, 81171286 & 91232714 to LJL), and the Innovation Project of Hunan Graduate Education (CX2012B100 to SJL).
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
