Abstract
Objective:
Metacognitive training is an eight-module, group-based treatment programme for people with schizophrenia that targets the cognitive biases (i.e. problematic thinking styles) thought to contribute to the genesis and maintenance of delusions. The present article is an investigation into the efficacy of a shorter, more targeted, single-module metacognitive training programme, administered individually, which focuses specifically on improving cognitive biases that are thought to be driven by a ‘hypersalience of evidence-hypothesis matches’ mechanism (e.g. jumping to conclusions, belief inflexibility, reasoning heuristics, illusions of control). It was hypothesised that a more targeted metacognitive training module could still improve performance on these bias tasks and reduce delusional ideation, while improving insight and quality of life.
Method:
A sample of 28 patients diagnosed with schizophrenia and mild delusions either participated in the hour-long, single-session, targeted metacognitive training programme (n = 14), or continued treatment as usual (n = 14). All patients were assessed using clinical measures gauging overall positive symptomology, delusional ideation, quality of life and insight, and completed two cognitive bias tasks designed to elucidate the representativeness and illusion of control biases.
Results:
After a 2-week, post-treatment interval, targeted metacognitive training patients exhibited significant decreases in delusional severity and conviction, significantly improved clinical insight, and significant improvements on the cognitive bias tasks, relative to the treatment-as-usual controls. Performance improvements on the cognitive bias tasks significantly correlated with the observed reductions in overall positive symptomology. Patients also evaluated the training positively.
Conclusions:
Although interpretations of these results are limited due to the lack of an optimally designed, randomised controlled trial and a small sample size, the results are promising and warrant further investigation into targeted versions of the metacognitive training programme.
Introduction
Schizophrenia is a severe and disabling psychiatric disorder. It causes considerable suffering for those who have the disorder as well as their families, and creates substantial costs to the community (Landa et al., 2006; Moritz et al., 2011a). Delusions and hallucinations are ‘core symptoms’ of the illness and approximately three-quarters of all people with schizophrenia experience these symptoms (Kaplan and Saddock, 1995). Despite recent advances in antipsychotic treatments, the traditional and dominant psychopharmacological treatment approach is often met with medium effect sizes relative to placebo, high levels of relapse, issues with compliance and illness insight, and serious side effects (Leucht et al., 2009).
Psychotherapies, such as cognitive behavioural therapy for psychosis (CBTp), are now being used alongside the psychopharmacological approach in the treatment of the core symptoms of schizophrenia, such as delusions (Barrowclough et al., 2006; Bechdolf et al., 2010; Garety et al., 1994; Granholm et al., 2006; Landa et al., 2006; Lecomte et al., 2003; Lecomte et al., 2008; Spidel et al., 2006). CBTp, whether administered individually or in group settings, aims to identify and actively modify maladaptive delusional beliefs, attitudes and behaviours that are often associated with schizophrenia. Recent reviews and meta-analyses of its efficacy as an adjunct therapy to psychopharmacological treatments have revealed small-to-moderate effect sizes equal to, and sometimes reportedly better than, the efficacy of antipsychotic medication (Wykes et al., 2008; Zimmermann et al., 2005).
A novel psychotherapeutic approach that builds on the concepts and goals of CBTp is the ‘metacognitive training’ (MCT) programme. Unlike CBTp, which develops an awareness of the implausible content of a patient’s delusion, MCT targets the cognitive biases (i.e. distortions in the collection and processing of information) thought to underlie the formation and maintenance of delusional beliefs (Bell et al., 2006; Freeman, 2007; Garety and Freeman, 1999; Woodward et al., 2006). Developed by Moritz and Woodward (2007b) as a group-based therapy (see Moritz & Woodward, 2007a for a review), MCT aims to bring to the attention of patients the cognitive dysfunctions that may be causing or maintaining their delusional symptoms (i.e. ‘metacognitive’ implies ‘thinking about one’s thinking’). Thus, one of the three fundamental components of the programme is knowledge translation. The second component is a demonstration of the negative consequences of these cognitive biases via exercises that target each bias individually. Finally, patients are offered alternative thinking strategies, which may help them to arrive at more appropriate inferences and thereby avoid the ‘cognitive traps’ that otherwise lead to delusional beliefs (Moritz and Woodward, 2007a). The modules of the programme deal with:
Attributional style. Participants are conveyed the disadvantages of monocausal inferences.
Jumping to conclusions (JTC). Participants are encouraged to avoid hasty decision-making.
Belief inflexibility/bias against disconfirmatory evidence (BADE). Participants are taught to stay open-minded for alternative assumptions as evidence/situations may change over time.
Theory of mind/social cognition. Participants are taught to pay attention to multiple social cues before inferring the state of mind of another person.
Memory/overconfidence. Participants are taught to withhold strong judgements and to prevent false memories.
Mood/self-esteem. Exercises attempt to lift self-esteem and change negative cognitive schemata.
Preliminary research into the efficacy of the MCT programme has thus far yielded promising results. Relative to controls, patients with delusions who were randomised into the MCT intervention group exhibited significant improvements in delusion severity (including distress and conviction), quality of life and illness insight, as well as improved performance on memory tasks and the JTC cognitive bias task (Aghotor et al., 2010; Moritz et al., 2011a; Moritz et al., 2011b). Importantly, patients who received MCT training expressed greater subjective training success and satisfaction with the programme, and greater willingness to recommend it, compared to controls who received other interventions, such as cognitive remediation (Aghotor et al., 2010; Moritz and Woodward, 2007b).
Nevertheless, more research into the efficacy of the MCT programme is required. One of the challenges of conducting such research is the length of time required to conduct the programme in full (i.e. a minimum of 8 weeks, 16 weeks recommended), which can lead to high dropout rates (Aghotor et al., 2010). Moreover, to date, there has been little research conducted on the efficacy of an individually administered MCT programme, entitled metacognitive therapy (MCT+). This variant of the programme combines the process-oriented approach of the group training with elements from individualised cognitive behavioural therapy (i.e. it relates information from the modules to individual experiences, observations and symptoms of the patient). Finally, the individual contribution of each module has not been determined, and the possibility that certain modules may be more effective than others is of clinical importance. For example, there is evidence that the JTC and belief inflexibility/BADE cognitive biases are the most closely related to delusion severity (Sanford et al., 2013; So et al., 2010; Speechley et al., 2012), which suggests that the modules targeting these biases may be the most effective in the treatment of delusions.
The influence and efficacy of a single-module, ‘bias-specific’ programme has recently been investigated (Ross et al., 2011). Using the exercises from the JTC and belief inflexibility/BADE modules, the study found that participants showed a significant increase in post-treatment data processing. The single-module intervention also led to increases in belief flexibility and less conviction in delusions, although these trends were non-significant (Ross et al., 2011). These non-significant trends may be related to the exclusion of delusion-relevant material, a core component of the MCT programme, as they link the cognitive bias to delusional experience. The newly developed Maudsley Review Training Programme substantially extends the adapted MCT module used by Ross et al. (2011), importantly incorporating delusion-relevant material similar to the original MCT modules. Preliminary results of this programme are more encouraging, suggesting improvements in JTC (effect size = 0.30), belief flexibility (effect size = 0.82) and delusional conviction (effect size = 1.06) following treatment (study referenced by Freeman, 2011).
While promising, the study would benefit from replications using larger samples and a randomised control design. Furthermore, there is increasing support that people with delusions exhibit a ‘hypersalience of evidence-hypothesis matches’ mechanism, where even weak matches between the available evidence and existing beliefs or hypotheses are enhanced. It has been argued that this construct may underlie the JTC and BADE effects (Balzan et al., 2012a; Speechley et al., 2010). Other manifestations of this ‘hypersalience’ mechanism have also been reported among people with delusions, including the confirmation bias (Balzan et al., 2013a), illusory correlations and perceptions of control (Balzan et al., 2013b) and reasoning heuristics (Balzan et al., 2012b). Accordingly, MCT replications using the JTC/BADE modules need to determine if performance improvements are limited to the JTC and BADE effects, or whether performance improves on these other cognitive biases as well, as would occur if they were all connected by an underlying ‘hypersalience of evidence-hypothesis matches’ mechanism.
The purpose of the current study was to implement a shorter, more targeted, individually administered MCT module – MCT-T – that combined aspects of the existing JTC and BADE modules. Unlike previous attempts to condense the modules, the current study would stay closer to the original MCT format, incorporating all three fundamental components of the programme (i.e. knowledge translation, demonstration of the negative consequences of cognitive biases and alternative thinking strategies). The study would also include cognitive bias tasks not used in previous MCT studies (i.e. representativeness heuristic and illusion of control tasks), to determine whether these biases could also be improved by the JTC/BADE treatment modules, as would be predicted if they were all driven by the ‘hypersalience’ mechanism. It was hypothesised that this MCT-T programme would still be effective in reducing a patient’s susceptibility to the underlying ‘hypersalience’ construct (as determined by improved performance on the cognitive bias tasks) which, in turn, would reduce delusional severity, distress, conviction and preoccupation, while improving insight into one’s illness and symptoms and one’s subjective quality of life.
Methods
Participants
A total of 28 participants – outpatients with a diagnosis of schizophrenia and at least mild delusional ideation, as determined by a ‘delusions’ (P1) score > 2 on the Positive and Negative Syndrome Scale (PANSS; see the assessments that follow) –were recruited from the Lyell McEwin Health Service Clinical Trials Unit. The diagnosis of schizophrenia was confirmed with the Mini-International Neuropsychiatric Interview (MINI). Exclusion criteria included a history of substance and alcohol abuse and previous brain injury or concussion. All participants were being treated with atypical antipsychotic medications at the time of testing and had been taking this medication for over 12 months.
A subsample of 14 participants (11 men and three women) was initially run in the MCT-T condition, and another 14 participants (nine men and five women) were then allocated to the treatment-as-usual (TAU) control group (i.e. no intervention but continued TAU; after the second assessment session, controls were fully debriefed about the nature of the study and were told that they would be entitled to MCT-T sessions in the near future). Due to the retrospective allocation of the control group, randomisation of group allocation was not possible. All participants who were contacted gave their informed consent and completed all tasks in the trial (i.e. no loss to follow-up); all were fluent in English.
Working memory was assessed with the Wechsler Adult Intelligence Scale–Revised Digits forward and backward subtests (Wechsler, 1997), and premorbid intelligence estimates were made with the National Adult Reading Test (NART; Nelson and Willison, 1991). Socio-economic status was estimated using the Hollingshead Two-Factor Index of Social Position (Hollingshead, 1957), using the highest parental occupation and education level. Scores for these measures and all other demographic information for each group is summarised in Table 1. As indicated in Table 1, the two samples were generally well matched on their age, educational attainment and scores on standardised measures of cognitive and intellectual functioning. The MCT-T group had been diagnosed with schizophrenia for a significantly longer time [t(26) = 2.17, p < 0.05].
Socio-demographic and psychopathological characteristics of study participants (mean; SD).
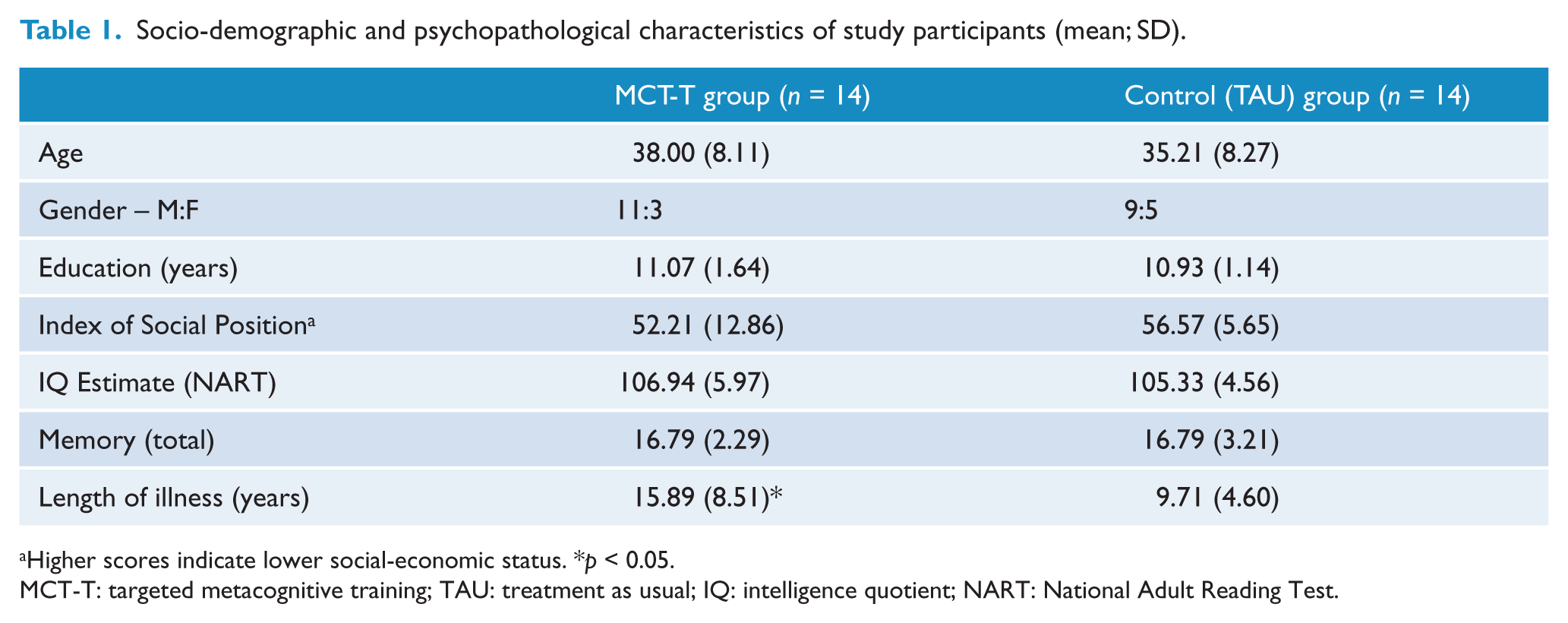
Higher scores indicate lower social-economic status. *p < 0.05.
MCT-T: targeted metacognitive training; TAU: treatment as usual; IQ: intelligence quotient; NART: National Adult Reading Test.
Metacognitive training
The adapted MCT-T module was run with patients individually, which allowed for the personal experiences and symptoms of the patient to be incorporated into the programme, similar to the existing MCT+ programme for patients with psychosis. However, unlike the MCT+ programme, which covers all eight MCT modules, the MCT-T consists of a single 60-minute session that focuses only on the JTC and BADE modules. The session aimed to bring to the attention of the patient the cognitive underpinnings of delusional ideation, and particularly the possibility that delusions may be caused or maintained by a hypersalience of evidence-hypothesis matches (i.e. ‘knowledge translation’). Examples of how hypersalient evidence-hypothesis matches may lead to or maintain delusional beliefs were presented to patients [e.g. a hypersalience between evidence (‘my doctor has a foreign accent’) and a belief (‘my doctor is a spy’) can lead to hasty decisions and resistance to disconfirmatory evidence].
Patients were then given a series of exercises taken from the existing JTC modules (modules 2 and 7) and the belief flexibility/BADE module (module 3). These exercises were designed to point out the negative consequences of making hasty decisions and of inflexible belief systems. For example, visual materials would be presented that would typically lead to false responses if decisions were made prematurely; patients were thus shown that delaying decisions until sufficient evidence had been collected was more optimal. The exercises in modules 2, 3 and 7 are discussed in more detail in Moritz and Woodward (2007a, 2007b) or can be viewed in full at the ‘Metacognitive Training for Psychosis (MCT)’ Web pages of the University Medical Center Hamburg-Eppendorf (Universitätsklinikum Hamburg-Eppendorf, 2013). Patients were also invited to relate these exercises to personal examples. Finally, patients were offered alternative thinking strategies (e.g. delaying decisions, being open to different conclusions from those initially accepted), which were summarised onto a cue card as a ‘take-home’ message. Patients were instructed to go over these strategies whenever they felt insulted or threatened in the time between the training and the follow-up sessions. It is worth noting that although the hypersalience mechanism and the JTC and BADE/belief inflexibility effects were discussed at length during the MCT-T programme, no specific exercises relating to the representativeness heuristic or illusion of control bias (see next section for details) were provided. This was to determine whether any performance improvements post-intervention could be generalised to these biases, which are thought to share the underlying ‘hypersalience’ mechanism with the JTC and BADE effects (Balzan et al., 2012b; Balzan et al., 2013b).
Cognitive bias tasks
Previous MCT efficacy studies have either used the ‘beads task’ (Moritz et al., 2011a; Ross et al., 2011) or the ‘BADE’ task (Aghotor et al., 2010) to assess improvements in cognitive reasoning post MCT. To determine if improvements can be generalised to other biases assumed to share the underlying ‘hypersalience’ mechanism with the JTC and BADE effects, the current study used tasks designed to elucidate the representativeness and illusion of control biases. Prior research indicated that individuals with delusions are particularly susceptible to the ‘lawyer-engineer’ base-rates task used to test the representativeness heuristic and the ‘light-onset’ illusion of control task, compared to non-delusion-prone controls (Balzan et al., 2012a; Balzan et al., 2013b).
Representativeness task
The representativeness task was a modified version of the Kahneman and Tversky (1973) ‘lawyer–engineer’ task. Participants were given short stories that centred on two outcomes, and that initially presented one outcome (the ‘lure’) as more likely than other (the ‘true’ interpretation). Participants were asked to rate the likelihood of each outcome based on this initial information, before being presented with discordant base-rate information (now consistent with the ‘true’ interpretation), and then rated each outcome again (for a full description of this task see Balzan et al., 2012a). Individuals with delusions previously demonstrated less downrating on the representative ‘lure’ after discordant base-rate evidence and less uprating of the ‘true’ interpretation, relative to non-delusion-prone controls (Balzan et al., 2012a). A total of four stories were presented across the two test sessions (two stories at the first session; two stories at the second session) and were randomised across participants.
Illusion of control task
The illusion of control task was a computerised version of the original Alloy and Abramson (1979) light-onset study. In this original task, participants pushed a button to see if they could control the onset of a light. In the current version, participants were presented with images of an illuminated light bulb (‘light on’ stimulus) or an unilluminated bulb (‘light off’ stimulus). They were told that their task was to determine how much control they had over the illumination of the bulb. As in the original study, the onset of light was preprogrammed to occur (i.e. no control over the onset of light) on 30 of the 40 trials (i.e. 75% trials), the order of which was randomised for each participant. At the conclusion of each stimulus set, participants were asked to indicate whether any of their responses lead to bulb illumination, and were also asked to judge their level of control on a 100-point scale, labelled ‘0 = no control’, ‘25 = little control’, ‘50 = intermediate control’, ‘75 = mostly control’ and ‘100 = full control’. Finally, to gauge levels of perceived connection between their responses and the outcome, participants were asked to report on how many of the trials the bulb become illuminated as a result of their response (e.g. 30 trials = ‘full control’). For a full description of this task see Balzan et al. (2013b).
Assessments
Psychopathological symptoms were assessed using the PANSS (Kay et al., 1987), the Scale for the Assessment of Positive Symptoms (SAPS; Andreasen and Olsen, 1982) and the 21-item Peters et al. Delusions Inventory (PDI-21; Peters et al., 2004), which provides a scale for global delusional ideation, and subscales for delusional distress, preoccupation and conviction. Quality of life was determined by the World Health Organization Quality of Life scale (World Health Organization, 2004), which provides estimates for psychological and social well-being. Clinical insight was estimated using the Schedule for Assessment of Insight (SAI) for psychosis patients (adapted from David, 1990), and the Beck Cognitive Insight Scale (BCIS; Beck et at., 2004), which assesses levels of self-reflectiveness and self-certainty (i.e. overconfidence in the interpretation of a person’s experiences). Finally, patients receiving the intervention completed the subjective evaluation of the MCT-T programme, which has been used in other MCT efficacy studies (Aghotor et al., 2010; Moritz and Woodward, 2007b) and has satisfactory internal consistency (Cronbach’s α = 0.73). The evaluation contained six items covering different aspects of training satisfaction (effectiveness, usefulness, applicability to daily life, transparency of the aims, fun, and whether they would recommend the training to others), and two items gauging negative feedback (‘I will not apply the lessons learned to everyday life’; ‘I would rather have spent my time doing something else’). Each item was rated on a 5-point scale (1 = fully disagree; 5 = fully agree), so that higher scores indicated greater satisfaction. Patients were also given a brief, 5-item follow-up evaluation at the next session, which attempted to gauge how often they used the principles of their training and how effective they found these principles to be during the interval.
The PANSS, SAI and SAPS were performed by a trained research nurse. Due to the non-random, sequential allocation of patients to either group, rater blindness to group allocation was not possible. All other measures were completed by self-report. Patients were instructed to complete these self-report assessments based on recent events (i.e. over the last month) for the first session, and over the last fortnight at the second session. All assessments were made prior to and 2 weeks following the intervention (or TAU for controls).
Results
Group differences on the various clinical assessments and cognitive tasks were calculated with 2 (group) × 2 (time) analysis of variance with repeated-measures on time, with a significance level of p < 0.05. Due to significant group differences across the pretreatment clinical assessments (see Table 2), analysis of covariance (ANCOVA) was also conducted for these measures using pretreatment scores as the covariates. Effect sizes were estimated using the partial eta squared (ηp2) statistics. There were no missing data across variables.
Pre- and post-treatment assessments of symptom severity (PANSS; SAPS; PDI-21), quality of life (WHOQoL) and insight (SAI; BCIS) for intervention and control groups (mean; SD).
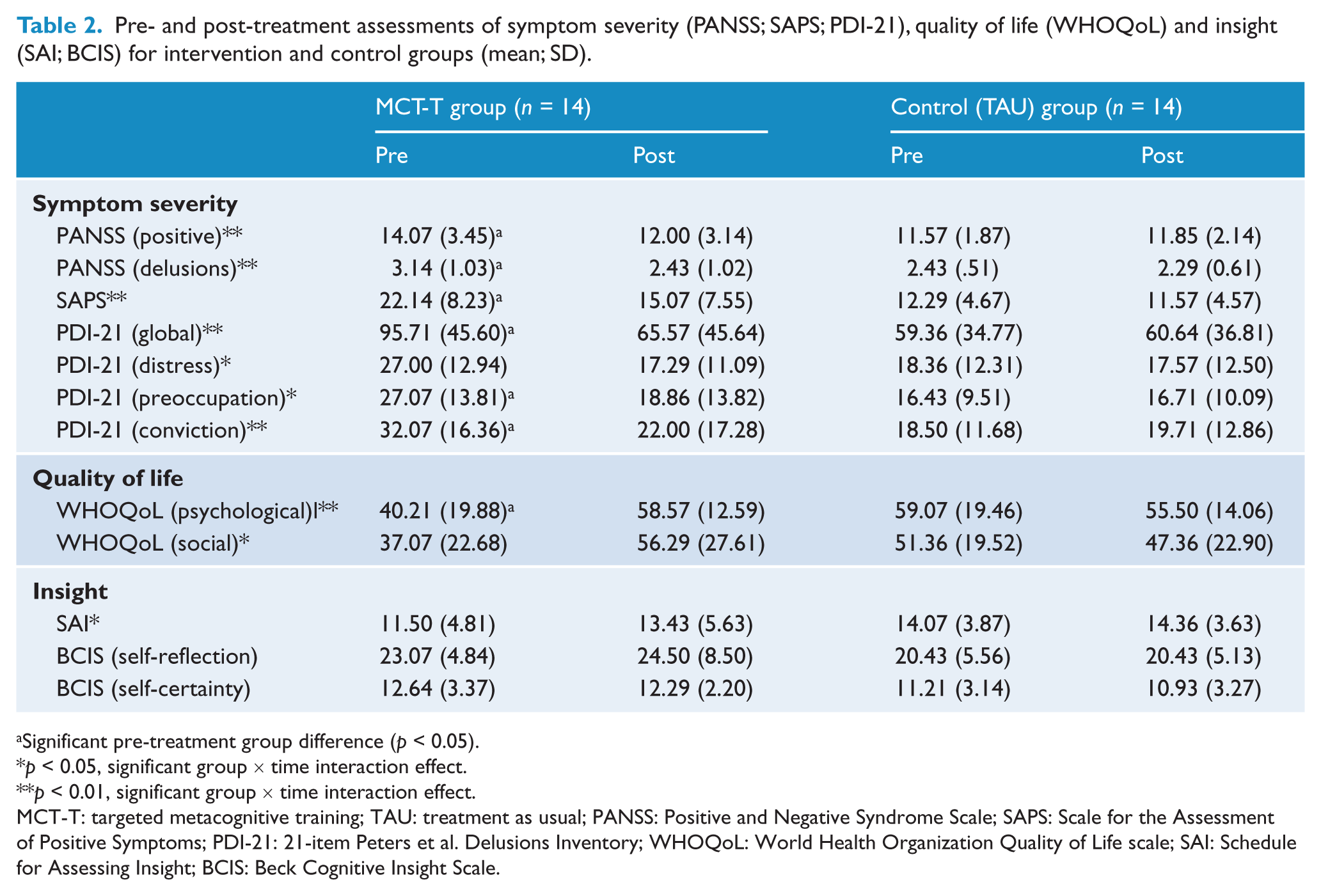
Significant pre-treatment group difference (p < 0.05).
p < 0.05, significant group × time interaction effect.
p < 0.01, significant group × time interaction effect.
MCT-T: targeted metacognitive training; TAU: treatment as usual; PANSS: Positive and Negative Syndrome Scale; SAPS: Scale for the Assessment of Positive Symptoms; PDI-21: 21-item Peters et al. Delusions Inventory; WHOQoL: World Health Organization Quality of Life scale; SAI: Schedule for Assessing Insight; BCIS: Beck Cognitive Insight Scale.
Clinical assessments
Group × time interaction
Table 2 summarises the various assessments used to determine symptom severity, delusional ideation, quality of life and insight at baseline and at the 2-week follow-up for both groups. There were significant interaction effects between group and time for all three measures of symptom severity [PANSS positive scale: F(1, 26) = 18.80, p < 0.001, ηp2 = 0.42; SAPS: F(1, 26) = 17.19, p < 0.001, ηp2 = 0.40; PDI-21 global: F(1, 26) = 9.29, p < 0.01, ηp2 = 0.26], whereby patients in the MCT-T group demonstrated significantly greater decreases in symptom severity post-treatment relative to the TAU controls, whose symptoms remained essentially the same (Table 2). There were similar significant interaction effects for the delusions subscale of the PANSS [F(1, 26) = 9.04, p < 0.01, ηp2 = 0.26], as well as for the distress, preoccupation and conviction subscales of the PDI-21 [F(1, 26) = 7.14, p < 0.05, ηp2 = 0.22; F(1, 26) = 7.51, p < 0.05, ηp2 = 0.22; F(1, 26) = 9.26, p < 0.01, ηp2 = 0.26, respectively].
There were significant interaction effects for both quality of life measures [psychological well-being: F(1, 26) = 8.97, p < 0.01, ηp2 = 0.26; social well-being: F(1, 26) = 6.28, p < 0.05, ηp2 = 0.20], where MCT-T patients rated their psychological and social well-being significantly higher post-treatment, compared to TAU controls, where well-being slightly decreased (Table 2).
The SAI measure of insight also yielded a significant group × time interaction, such that MCT-T patients showed significant improvements in insight into their illness relative to TAU controls post-treatment, where insight remained stable [F(1, 26) = 7.70, p < 0.05, ηp2 = 0.23]. However, neither BCIS self-reported measures of insight (i.e. self-reflection and self-certainty) yielded significant main or interaction effects [i.e. self-reflection: F < 1, main effect for time and time × group interaction, and F(1, 26) = 2.71, p > 0.05, main effect for group; self-certainty: F < 1, main effect for time and time × group interaction, and F(1, 26) = 2.38, p > 0.05, main effect for group].
Analysis of covariance
Using pretreatment scores as the covariate, ANCOVA confirmed that post-treatment results were significant for PANSS [F(1, 25) = 11.23, p < 0.01, ηp2 = 0.23], PANSS delusions [F(1, 25) = 4.95, p < 0.05, ηp2 = 0.17], SAPS [F(1, 25) = 4.73, p < 0.16, ηp2 = 0.16], PDI-21 global [F(1, 25) = 4.73, p < 0.05, ηp2 = 0.16], PDI-21 conviction [F(1, 25) = 4.88, p < 0.05, ηp2 = 0.16] and SAI [F(1, 25) = 7.29, p < 0.05, ηp2 = 0.23]. However, PDI-21 distress [F(1, 25) = 3.72, p > 0.05], PDI-21 preoccupation [F(1, 25) = 3.48, p > 0.05] and both WHOQoL scales [psychological: F(1, 25) = 2.06, p > 0.05; social: F(1, 25) = 3.31, p > 0.05] failed to reach significance under this analysis. This suggested that the initial group differences on these pretreatment measures may better account for the mean group differences post-treatment, rather than the intervention itself.
Cognitive tasks
Base-rates representativeness task
The ratings of ‘lures’ and ‘true interpretations’ before and after the discordant base-rate information are presented in Table 3. Ratings across stories were combined for analysis. There were no significant main or interaction effects on pre-base-rate ratings for ‘true’ interpretations, but there was a significant main effect for time on pre-base-rate ratings for the ‘lures’ [F(1, 26) = 6.30, p < 0.05, ηp2 = 0.20]. This suggested that all participants were slightly less willing to rate the ‘lures’ as highly during the second testing session. However, this was not to suggest that all participants were less hypersalient to evidence-hypothesis matches at the second test session. Rather, the significant group × time interactions for post-base-rate ratings for ‘lures’ [F(1, 26) = 5.38, p < 0.05, ηp2 = 0.17] and ‘true’ interpretations [F(1, 26) = 6.91, p < 0.05, ηp2 = 0.21] suggested that MCT-T patients were significantly more capable than TAU controls of adapting prior beliefs to fit the current evidence post-treatment. It is suggested that this improvement was caused by the intervention, which attempted to abate the hypersalience of evidence-hypothesis matches. Following their training, MCT-T patients were more willing to downrate ‘lure’ ratings and uprate the ‘true’ interpretations following the disconfirmatory base-rate information (Table 3). Conversely, during the second session, TAU controls actually showed slight uprating of ‘lure’ and downrating of ‘true’ interpretation post-base-rates, suggesting that the findings for MCT-T patients were not the result of practice effects.
Mean (SD) prior- and post-base-rate (BR) ratings (maximum = 10) for ‘lures’ and ‘true interpretations’ across groups pre- and post-treatment.
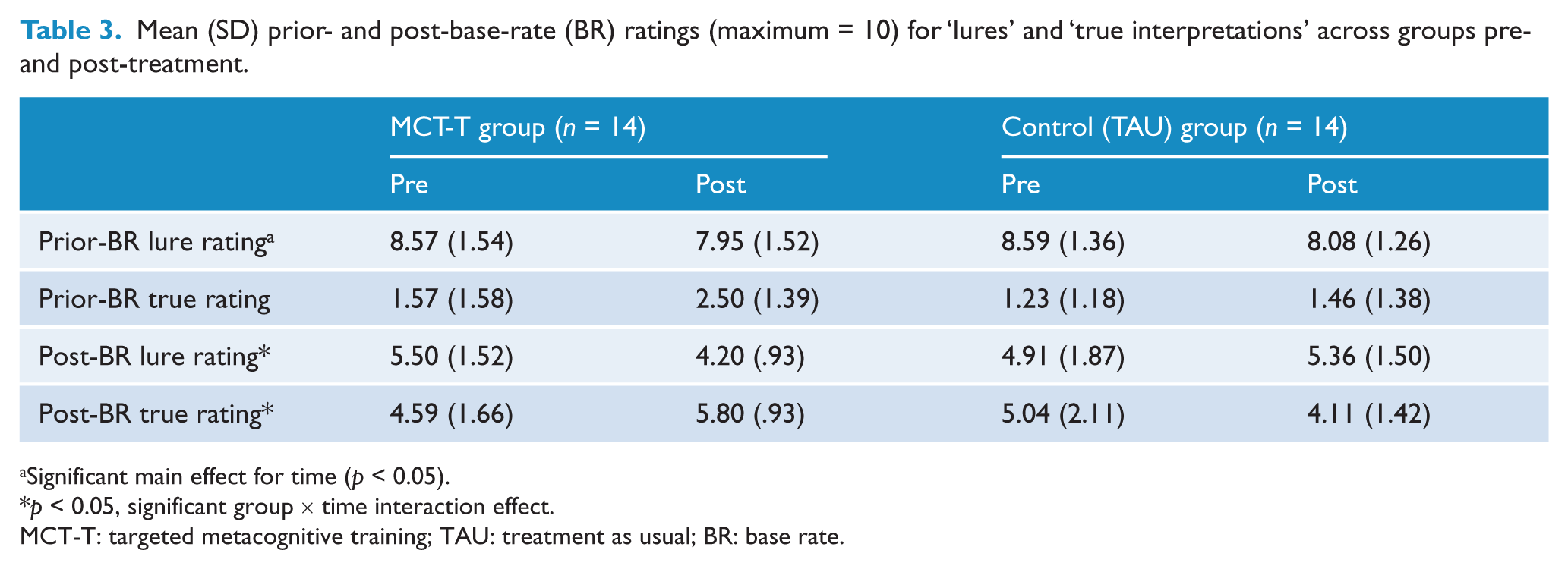
Significant main effect for time (p < 0.05).
p < 0.05, significant group × time interaction effect.
MCT-T: targeted metacognitive training; TAU: treatment as usual; BR: base rate.
Illusion of control task
In the pretreatment session, a majority of participants from both groups (> 70%) indicated that they had control over the outcome [χ2 (1, N = 28) < 1], despite the lack of actual contingency (see Table 4). However, following training, patients within the MCT-T group were significantly less likely to believe they had control over the outcome [χ2 (1, N = 28) = 7.04, p < 0.01], while the illusion of control persisted within the control group (Table 4).
Perceptions of control (n) for MCT-T and control groups over pre- and post-treatment sessions.

p < 0.01 (significant, chi-squared).
MCT-T: targeted metacognitive training; TAU: treatment as usual.
Table 5 shows the patients’ estimated percentage of control and perceived connection both pre- and post-treatment. There were significant group × time interaction effects for both of these measures of control [percentage of control: F(1, 26) = 13.66, p < 0.01, ηp2 = 0.34; perceived connection: F(1, 26) = 9.24, p < 0.01, ηp2 = 0.26]. As shown in Table 5, the MCT-T patient group significantly reduced their estimates of control and perceived connection post-treatment, relative to the TAU controls, where estimates of control were slightly inflated at the second test session. This again demonstrated the lack of potentially confounding practice effects.
Mean (SD) estimated percentages of control and perceived connection across groups over pre- and post-treatment sessions.
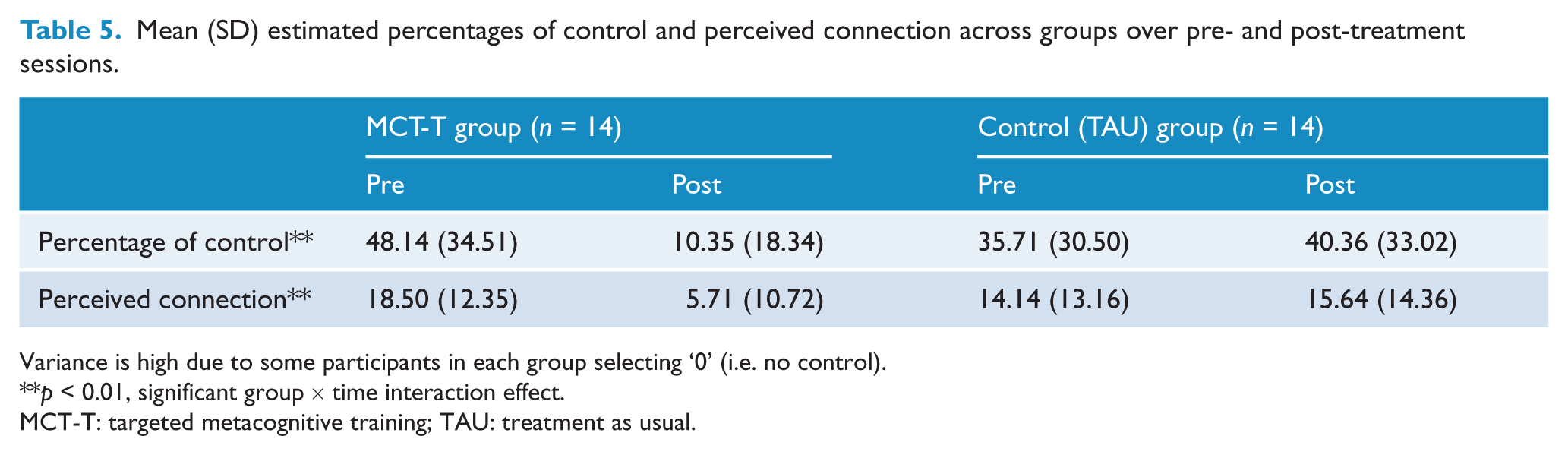
Variance is high due to some participants in each group selecting ‘0’ (i.e. no control).
p < 0.01, significant group × time interaction effect.
MCT-T: targeted metacognitive training; TAU: treatment as usual.
Correlation with post-treatment positive symptom changes
Correlations were conducted between the post-treatment performance improvements for the cognitive bias tasks and the post-treatment improvements on the positive symptomology measures. Difference scores for each measure were calculated to determine the level of change between pre- and post-treatment assessment sessions (i.e. level of improvement); higher scores indicated greater improvement. The only measure of improved positive symptomology to significantly correlate with improvements in cognitive bias performance was the overall PANSS positive measure (correlations: post-base-rate change: r = 0.39, p < 0.05; percentage of control change: r = 0.49, p < 0.01; perceived connection change: r = 0.48, p < 0.01). At first, these results seemed encouraging and suggested that a greater change in positive symptoms (i.e. a reduction in overall severity from pre- to post-treatment) was associated with increased improvements on the representativeness task (i.e. greater downrating of ‘lures’ and uprating of ‘true’ interpretations) and illusion of control task (i.e. reduction in estimates of control and connection). However, no other measure of change in positive symptomology (e.g. SAPS, PDI-21) significantly correlated with these changes in cognitive bias performance, which limited the interpretation that the observed performance improvements were associated with the reduction in delusional ideation.
Evaluation of targeted metacognitive training
Patients in the MCT-T group (N = 14) subjectively evaluated the programme quite favourably on a 5-point evaluation scale (1 = fully disagree; 5 = fully agree). Training was seen as useful (M = 4.00; SD = 1.41), important to their treatment (M = 3.57; SD = 1.16), helpful to their daily routine (M = 3.79; SD = 1.25) and fun (M = 3.50; SD = 1.23). The goals of the training were clear (M = 3.86; SD = 1.23) and participants would recommend the training to others (M = 4.07; SD = 1.30). Negative feedback was generally minimal: ‘I will not apply the lessons learned to everyday life’ (M = 1.64; SD = 0.93) and ‘I would rather have spent my time doing something else’ (M = 2.07; SD = 1.14).
During the follow-up session, participants were asked to complete a second evaluation. Here participants agreed that they had used the principles from training (M = 3.71; SD = 0.99); that training had helped them cope with their special ideas/beliefs (M = 3.86; SD = 0.95); that training had been an important part of their treatment (M = 3.93; SD = 0.92); that what had been learned was useful to their daily routine (M = 3.57; SD = 0.85); and that they would still recommend the training to others (M = 4.50; SD = 0.65).
Discussion
The current study was an investigation into the feasibility of an individually administered, shorter and more targeted MCT programme. The MCT-T module developed for this study combined aspects of the existing JTC and BADE modules, and remained closer to the original MCT format than previous attempts to condense these modules (Ross et al., 2011), incorporating all three fundamental components of the programme (i.e. knowledge translation, demonstration of the negative consequences of cognitive biases and alternative thinking strategies). The study also included cognitive bias tasks that tested for the generalisability of the ‘hypersalience of evidence-hypothesis matches’ construct.
Overall, the results from the clinical assessments and cognitive bias tasks demonstrated the potential benefit of this shorter and more targeted, single-module MCT programme. Delusional severity as assessed by the PANSS, SAPS and PDI-21 had significantly reduced after a 2-week interval for patients who had received the training relative to the TAU controls. Furthermore, as would be predicted by an underlying hypersalience mechanism, MCT-T patients demonstrated significant performance improvements post-treatment on both cognitive tasks (i.e. less susceptible to the representativeness heuristic and illusion of control bias), even though the MCT-T modules only covered JTC and BADE biases specifically, and the hypersalience mechanism more generally. Conversely, no improvements were observed for the TAU controls across the two sessions, which suggested that improvements incurred by patients in the intervention condition were not likely to have been a result of practice effects. This offers further evidence that a hypersalience of evidence-hypothesis matches may underlie many of the cognitive biases reported in schizophrenia, as already suggested in previous studies (Balzan et al., 2012a; Balzan et al., 2013a, 2013b; Speechley et. al., 2010). Moreover, these performance improvements significantly correlated with improvements on overall positive PANSS scores. Finally, MCT continues to be evaluated as a useful, important, helpful and fun treatment programme (for similar evaluations, see Aghotor et al., 2010; Moritz and Woodward, 2007b). Patients generally reported that the goals of training were clear, that they had used the principles from the training, which had helped them cope with their illness, and would recommend the training to others.
However, not all reported findings were consistent. Pretreatment scores on many of the clinical assessment measures significantly differed between the MCT-T and TAU control groups. While ANCOVA confirmed that many of the post-treatment differences on these measures remained significant when the pretreatment scores were analysed as covariates, post-treatment measures of quality of life measures and self-reported delusional distress and preoccupation failed to reach significance. This suggested that any post-treatment differences on these measures were more likely to have been caused by the discrepant pretreatment scores, rather than the intervention itself. Pretreatment scores may have been more balanced between groups had the study used a randomised controlled design, which would have ensured that testing for both groups occurred simultaneously. It remains a possibility that the discrepant pretreatment scores were influenced by the delay between data collection for the MCT-T group (recruited first) and the TAU control group (recruited months later). The lack of a randomised controlled design also ensured that the rater who assessed all patients was not blind to a patient’s group membership, which may have led to a rater bias. Some of the clinical assessment results may be a reflection of this bias. For example, while there was a significant group × time interaction for the rater-assessed SAI scale, no significant differences were found for the self-assessed BCIS. However, this discrepancy may have been due to the different approaches of each scale (i.e. the BCIS gauged overconfidence and self-reflection, while the SAI scale was more concerned with attitudes to diagnosis and medication adherence). Furthermore, the significant group × time interactions found for rater-assessed delusional measures cannot entirely be explained as a rater bias, because the self-assessed PDI-21 also yielded significant group × time interaction effects for delusional severity, and conviction, consistent with the rater scores. Nevertheless, like all self-report measures, there is the possibility that participants were responding in a manner consistent with the experimenter’s expectations. For example, patients in the MCT-T group knew that the purpose of the intervention was to help people with delusions cope with their symptoms. This may have influenced the way they responded to the PDI-21 at the follow-up session. Finally, despite the finding that performance improvements on the cognitive bias tasks (i.e. reduced ‘hypersalience’) significantly correlated with a reduction in overall positive PANSS scores, it cannot be concluded from the present study that a reduction in a patient’s susceptibility to the hypersalience mechanism is associated with a reduction in delusional severity specifically; no other measure of delusional severity (e.g. SAPS, PDI-21) was found to significantly correlate with performance improvements. The initial pretreatment differences on these measures and an overall small sample size may have contributed to this discrepancy.
Ongoing research into the efficacy of this MCT-T module, and the original MCT programme, is clearly required. Future studies should incorporate a randomised controlled trial into the experimental design, and match patient groups on illness duration and baseline delusional severity. An active control condition rather than a TAU control group should be incorporated into the design (e.g. administering controls a cognitive remediation programme aimed at memory, attention and other basic cognitive functions). This would help determine the unique benefits of the MCT programme over other similar, but less targeted, cognitive programmes. Larger samples are also required. Aghotor et al. (2010) conservatively estimated that a sample size of 86 patients in each group would be optimal. Further follow-up sessions (e.g. 6-month; 24-month) are also warranted to accurately determine the long-term effects of any improvements observed. Replications could also improve on the cognitive bias tasks administered to participants (e.g. increase the number of stories/scenarios on the representativeness task).
It would also be beneficial to run MCT and cognitive behavioural therapy (CBT) sessions together within the one study. Both individually and group-administered CBT sessions have also yielded significant decreases in symptom severity with comparable small-to-moderate effect sizes (Garety et al., 1994; Lecomte et al., 2008; Wykes et al., 2008). Hence, the potential benefits of a combined treatment approach, which might optimise psychosocial adjuncts to pharmacotherapy in the treatment of schizophrenia, is worthy of investigation. For example, the treatment outcomes of a combination therapy are more likely to be sustained than either treatment approach used in isolation. These investigations would also benefit from comparing the efficacy of group versus individually administered MCT/CBT interventions. The majority of MCT efficacy studies to date have only investigated group-administered modules; while there are benefits to the group approach, such as higher patient turnarounds (Lecomte et al., 2008), the unique contribution and benefits of an individualised treatment programme are not yet fully determined. For example, group-based interventions may not be as effective as individually administered interventions, which could account for the lack of statistically significant decreases in delusional severity as reported in previous MCT efficacy studies (Aghotor et al., 2010; Moritz et al., 2011a).
In conclusion, despite the methodological limitations, the present investigation into the efficacy of a shorter and more targeted MCT module demonstrated that the MCT-T was a safe and effective intervention. Relative to TAU controls, patients who had received MCT-T experienced significant decreases in delusional severity and conviction, as well as improved insight into their illness. MCT-T patients also demonstrated significant improvements on two cognitive bias tasks, suggesting that the intervention had successfully reduced the hypersalience of the ‘evidence-hypothesis matches’ mechanism. Although a reduction in the hypersalience mechanism was associated with improvements in positive symptoms generally, it remains unclear why this may improve delusional symptoms specifically. Consequently, this line of research should continue and be combined with other psychosocial interventions, so that the current treatment approaches to schizophrenia can more effectively deal with the symptoms of this debilitating disorder.
Footnotes
Acknowledgements
We would like to thank Professor Steffen Moritz for developing the original MCT module and for his permission to adapt modules 2, 3 and 7 into the MCT-T. We would also like to thank Bev Hisee and Imelda Cairney for their tireless efforts at recruiting clinical participants for this study.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
