Abstract
Background:
The antipsychotic drug (APD) clozapine (CLZ) is under-prescribed because of concerns about its safety. We evaluated in separate protocols the frequency of cardiomyopathy and hyponatraemia, which are adverse drug effects, where few comparative studies are available.
Methods:
Cross-sectional studies in subjects treated for at least 3 consecutive months with the same drug were conducted. Cardiomyopathy: Patients undergoing treatment either with CLZ (n = 125) or with other typical or atypical APDs (n = 59) were examined by a cardiologist who also recorded echocardiograms and electrocardiograms in order to diagnose cardiomyopathy. Hyponatraemia: Fasting sodium levels were assessed in patients receiving any of the following treatments: CLZ (n = 88), other atypical APDs (n = 61), typical APDs (n = 23), typical + atypical APDs (n = 11), and other drugs/drug-free (n = 36).
Results:
Cardiomyopathy: No case of cardiomyopathy was detected. The frequency of abnormal ventricular ejection fraction (< 55%) was similar in both treatment groups (p = 1). Hyponatraemia: The frequency of hyponatraemia (percentage; 95% CI) was: CLZ (3.4%; –0.7, 7.1); other atypical APDs (4.9%; –0.5, 10.3); typical APDs (26.1%; 8.2, 44.0); typical + atypical APDs (9.1%; –7.8, 26.0); other drugs/drug-free (0%). None of the CLZ hyponatraemia subjects were on monotherapy.
Conclusions:
Our results are at odds with previous studies of CLZ-associated cardiomyopathy. However, they must be compared to further cross-sectional or prospective studies because most published data come from either case reports or pharmacovigilance systems. The frequency of hyponatraemia during CLZ administration was similar to that observed with other atypical APDs, and it was significantly lower than that recorded with typical agents. These results, along with numerous case reports on the effects of CLZ in patients with polydipsia and water intoxication, point to a safe or even positive profile of CLZ on electrolytic regulation.
Introduction
Even though clozapine (CLZ) is at the top of effectiveness among the antipsychotic drugs (APD) in the treatment of refractory schizophrenia (Kane and Correll, 2010; Meltzer, 2012), it is currently under-prescribed because of concerns about its safety (Agid et al., 2010) and the need for haematological monitoring. In fact, in the Unites States, it is used only in about 14% of potential candidates (Stroup et al., 2009).
The most studied adverse drug reactions (ADRs) of CLZ are neutropaenia, agranulocytosis, gastrointestinal hypomotility, excessive weight gain, carbohydrate and lipid metabolism dysregulation (Agid et al., 2010). Other CLZ ADRs such as myocarditis, cardiomyopathy, a procoagulant state and a complex effect on water and electrolyte balance, mainly sodium regulation (De Berardis et al., 2012; de Leon et al., 1995), have been less studied.
Numerous pharmacovigilance and case report studies have been published about the association between CLZ, myocarditis, pericarditis and cardiomyopathy (Committee on Safety of Medicines, 1993; Coulter et al., 2001; Haas et al., 2007; Hagg et al., 2001; Kilian et al., 1999; La Grenade et al., 2001; Merrill et al., 2005; Montastruc et al., 2010; Reinders et al., 2004; Vesterby et al., 1980; Walker et al., 1997).
The term cardiomyopathy refers to a chronic myocardium disease which is grouped into the following specific morphological and functional phenotypes: dilated, hypertrophic, restrictive, and arrhythmogenic cardiomyopathies. Each phenotype is then sub-classified into familial and non-familial forms (Elliot et al., 2008).
The first case dealing with the possible association between CLZ and cardiomyopathy was reported by Juul Povlsen et al. (1985). Thereafter, a small number of case series (Kilian et al., 1999) and pharmacovigilance studies (La Grenade et al., 2001) have been published. Merrill et al. (2005) reviewed the available information about 54 suspected cases. The ages ranged between 20 and 59 years (median 34 years), 78% were males, treatment duration ranged between 2 weeks and 7 years (median 9 months) and the dose range was 200–500 mg/day. Eleven deaths were reported.
Novartis Pharmaceuticals (Clozaril® 2011; downloaded 17 November 2012) reported a rate for cardiomyopathy of 8.9 per 100,000 person-years in CLZ-treated patients in the US. This figure was similar to an estimate of the cardiomyopathy incidence in the US general population derived from the 1999 National Hospital Discharge Survey data (Popovic, 2001) (9.7 per 100,000 person-years (0.01%)). About 80% of CLZ-treated patients in whom cardiomyopathy was reported were less than 50 years of age. The duration of treatment prior to cardiomyopathy diagnosis varied, but was > 6 months in 65% of the reports. Dilated cardiomyopathy was the most frequent type.
Hyponatraemia (low sodium levels) is detected in about 4–10% of schizophrenia patients (de Leon 2003; Mannesse et al., 2010, Meulendijks et al., 2010). Hyponatraemic symptoms may be either neurological (nausea, vomiting, delirium, ataxia, seizures and coma) or psychiatric in nature (worsening of psychosis, agitation and irritability) (de Leon et al., 1994). Administration of APDs has been directly or indirectly (through polydipsia) linked to the development of hyponatraemia, mainly by the typical APD (Bersani, 2007). However, the causal evidence, time course and the dose–effect relationship are more controversial and weaker than those observed with the use of carbamazepine, oxcarbazepina and serotonin selective reuptake inhibitors (SSRIs) (de Leon et al., 1994; Fabian et al., 2004; Jessani et al., 2006; Mannesse et al., 2010; Meulendijks et al., 2010; Wilkinson et al., 1999).
In a recent case–control study based on 3,881,518 ADR reports, Mannesse et al. (2010) calculated an adjusted odds ratio (OR) for hyponatraemia of 1.66 (95% CI 1.45, 1.91) for typical APD and of 1.55 (95% CI 1.41, 1.69) for atypical APD (p > 0.05). The typical agents associated with the highest risk (OR > 2.0) were fluphenazine (6.91), flupenthixol (3.65), trifluoperazine (2.24) and zuclopenthixol (2.0). For the atypical APD the highest figures were 2.81 for ziprasidone, 2.27 for risperidone and 2.14 for olanzapine. The adjusted OR for CLZ was 1.22 (95% CI 0.35, 1.73), and the figures were lower for loxapine (0.78) and cyamemazine (0.77) only (Mannesse et al., 2010).
The risk of hyponatraemia during CLZ administration thus appears to be low among all the APDs. In fact, numerous case reports suggest that CLZ rather may improve the electrolytic balance in some subjects with polydipsia, water intoxication, syndrome of innapropriate antidiuretic hormone secretion (SIADH) and hyponatraemia (de Leon et al., 1995; Leadbetter and Shutty, 1994). However, only one case–control study involving CLZ has been carried out (Spigset and Hedenmalm, 1996). This study reported hyponatraemia in 25.9% and 13.6% of the patients receiving perphenazine or CLZ, respectively. To the best of our knowledge, no randomised controlled study on the association between CLZ or hyponatraemia has been published so far (Meulendijks et al., 2010).
Given the important role of CLZ in clinical psychiatry, more studies are needed to compare its safety with that of other APDs. We report here two cross-sectional, naturalistic studies that quantified the frequency of cardiomyopathy and hyponatraemia in patients undergoing prolonged treatment with CLZ or other typical or atypical APDs. Besides, we explored the association between metabolic syndrome, sodium levels and heart function.
Methods
Subjects
Both studies were conducted in Venezuela between 2007 and 2011 and comprised the CLZ-treated outpatients attending a clinic for individuals with schizophrenia and their families (CATESFAM, Maracaibo), inpatients from Pampero Hospital (Barquisimeto) and Peribeca Hospital (San Cristóbal), and two outpatient private clinics in Mérida, where consecutively admitted outpatients were evaluated. The comparison groups comprised patients receiving other atypical or typical APDs. In the hyponatraemia study, an additional group included subjects receiving other psychotropic agents or drug-free patients.
The inclusion criteria were: (1) the patients must have been under pharmacological treatment for 3 or more consecutive months; and (2) they must have signed an informed consent for voluntary participation. The study was approved by the ethics committee from each institution. Several APD-treated subjects participated in both studies, but no attempt was made to conduct any between-study data crossing.
The following information was recorded: present and past drug treatments, demographic features, and psychiatric diagnosis using the Structured Clinical Interview for DSM-IV Axis I Disorders (First et al., 1997). We also assessed, in fasting conditions, body weight (kg), height (m), body mass index (weight/height2), waist circumference (cm), blood pressure, and obtained a cubital venous blood sample for immediate analysis. Liver and kidney function was assessed in all subjects. Glucose and lipid levels were quantified with enzymatic methods from Human (Wiesbaden, Germany). Sodium and potassium levels were assessed with a selective ion electrode method (Easylite Medica Corporation, Bedford, MA, USA). The inter-site variability was < 10% for glucose and lipid levels, and 1.8% for sodium levels. Metabolic syndrome was diagnosed according to the Adult Treatment Panel III (Third Report of the National Cholesterol Education Program, 2002).
Procedure
Cardiomyopathy study
Each subject was evaluated at a university setting by a clinical cardiologist who explored cardiovascular-respiratory symptoms (chest pain, oedema, breathlessness, abdominal discomfort, palpitations, fatigue and tachycardia) and conducted a M-B mode 2D echocardiogram with Doppler, an electrocardiogram and a general clinical evaluation. The primary echocardiographic variables were chamber morphology, ventricular ejection fraction (biplane Simpson’s method) (normal values: either ≥ 50% or ≥ 55%) and the degree of diastolic dysfunction (I–III according to Nagueh et al., 2009). With all these clinical and instrumental data, the cardiologist reached a morphological and functional diagnosis of (a) cardiomyopathy, that is, myocardial disorders in which the heart muscle is structurally and functionally abnormal in the absence of coronary artery disease, hypertension, and valve and congenital heart disease sufficient to cause the observed myocardial abnormality (Elliot et al., 2008); (b) any other specific heart disease; or (c) no heart disease.
Patients were divided into two treatment groups: CLZ or any other APD. Continuous variables were analysed with the two-tailed t-test for independent samples (when normally distributed) or otherwise with the Mann–Whitney U-test.
The analysis was conducted with the SPSS version 17.0 (SPSS Inc., Chicago, IL, USA). The frequency of dichotomous variables (normal or abnormal systolic and diastolic function, specific heart disorder, metabolic syndrome) was compared using the chi-squared test, Fisher’s exact test and binary logistic regression analysis, with age, sex, ethnicity and metabolic syndrome as covariates. The CLZ and other APD doses were expressed as olanzapine equivalents (Gardner et al., 2010) in order to conduct bivariate correlation analyses with the ventricular ejection fraction. Results were considered significant when p < 0.05.
Hyponatraemia study
Symptoms of hyponatraemia were explored, including nausea, general malaise, headache, muscle cramps, lethargy, confusion, delirium and agitation (Meulendijks et al., 2010). These symptoms, however, were difficult to separate from those of the specific mental disorder or from other ADRs.
Hyponatraemia severity was categorised according to serum sodium levels as follows: mild (130–135 mmol/L), moderate (125–129.9 mmol/L) and severe (< 125 mmol/L) (Ellison and Berl, 2007).
The frequency of hyponatraemia for each cut-off point (dependent variable) was compared among the groups with binary logistic regression analysis, with treatment, age and sex as covariates.
A Pearson bivariate correlation analysis was conducted between the serum sodium levels and potassium, glucose, triglycerides and total cholesterol levels, APD dose (in olanzapine equivalents; Gardner et al., 2010), treatment duration (in months) and age. Results were considered significant when p ≤ 0.05.
Results
Cardiomyopathy
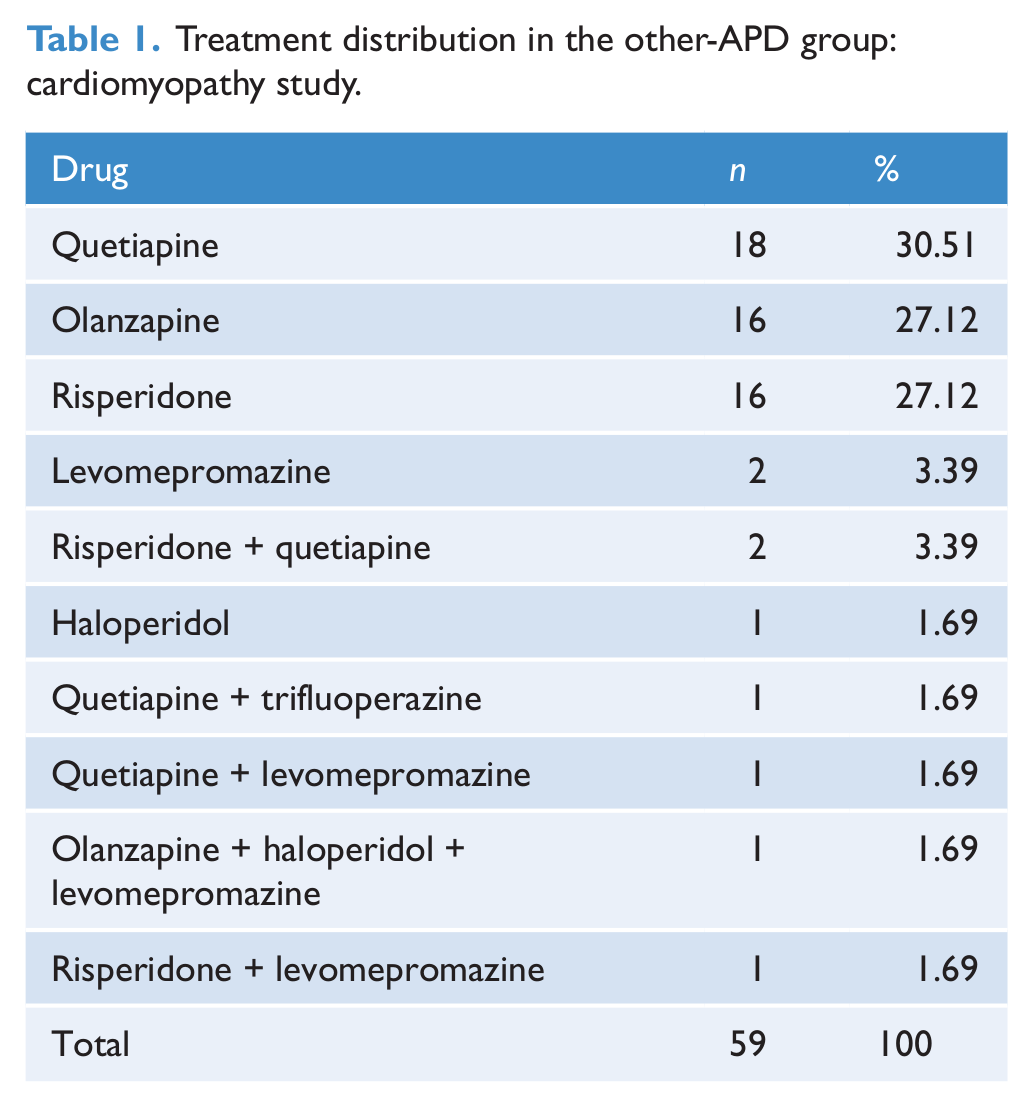
The study comprised 125 CLZ and 59 other APD-treated patients. Most subjects in the latter group received olanzapine, risperidone or quetiapine (Table 1). The CLZ group was younger and included more males and schizophrenia diagnosis than the other-APD group (Table 2). One hundred and eighteen out of 125 CLZ-treated patients (94.4%) only received that agent as APD therapy, but most subjects received intermittent benzodiazepine or antidepressant treatment. Because of the disease chronicity, the magnitude and frequency of such a polypharmacy was not properly quantified.
Treatment distribution in the other-APD group: cardiomyopathy study.
Demographic and clinical features: cardiomyopathy study.
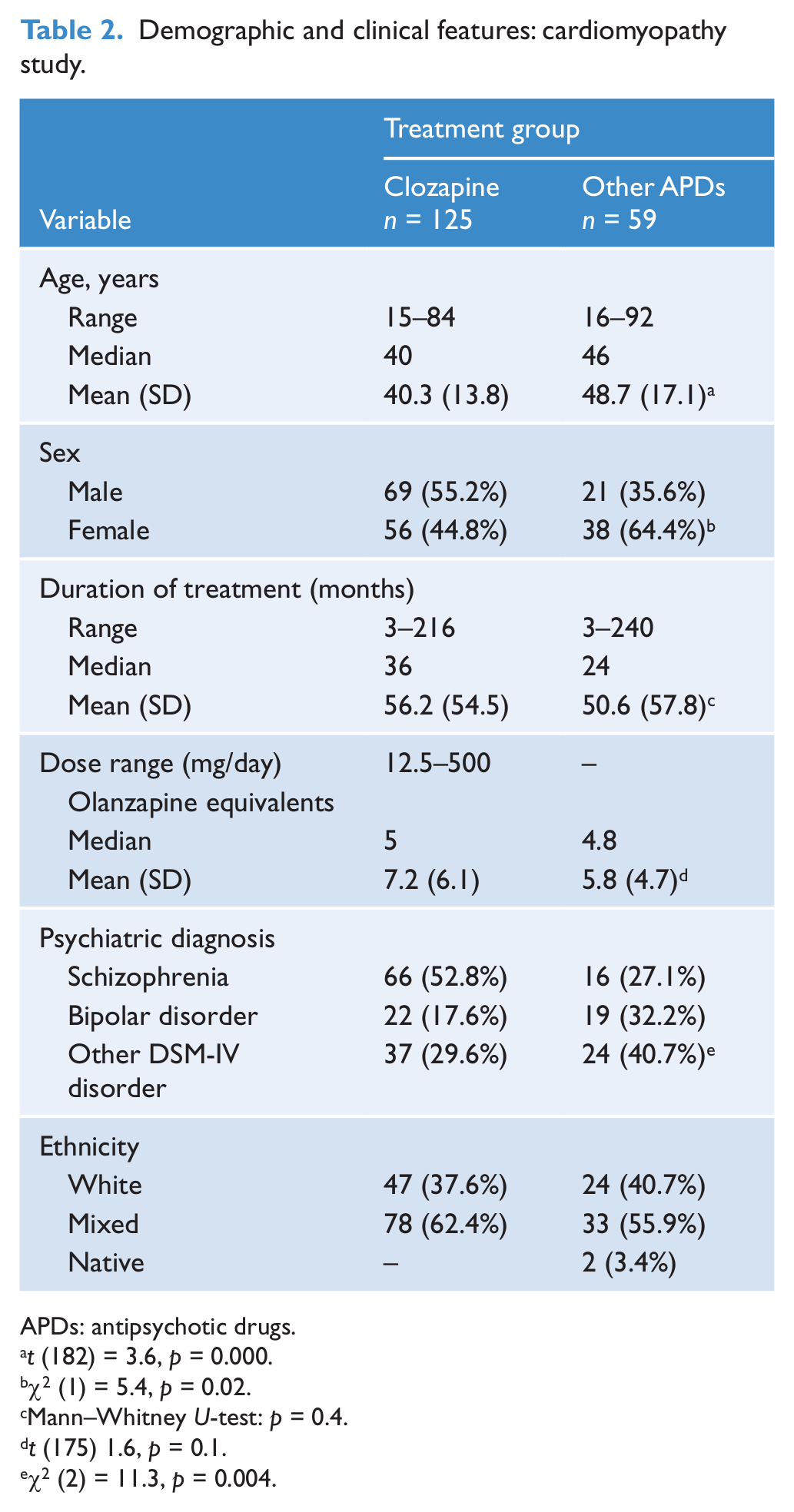
APDs: antipsychotic drugs.
t (182) = 3.6, p = 0.000.
χ2 (1) = 5.4, p = 0.02.
Mann–Whitney U-test: p = 0.4.
t (175) 1.6, p = 0.1.
χ2 (2) = 11.3, p = 0.004.
No case of cardiomyopathy was detected. One subject in each group was diagnosed with coronary heart disease. In fact, these were the only patients with grade II diastolic dysfunction (Table 3).
Cardiovascular and metabolic evaluation.
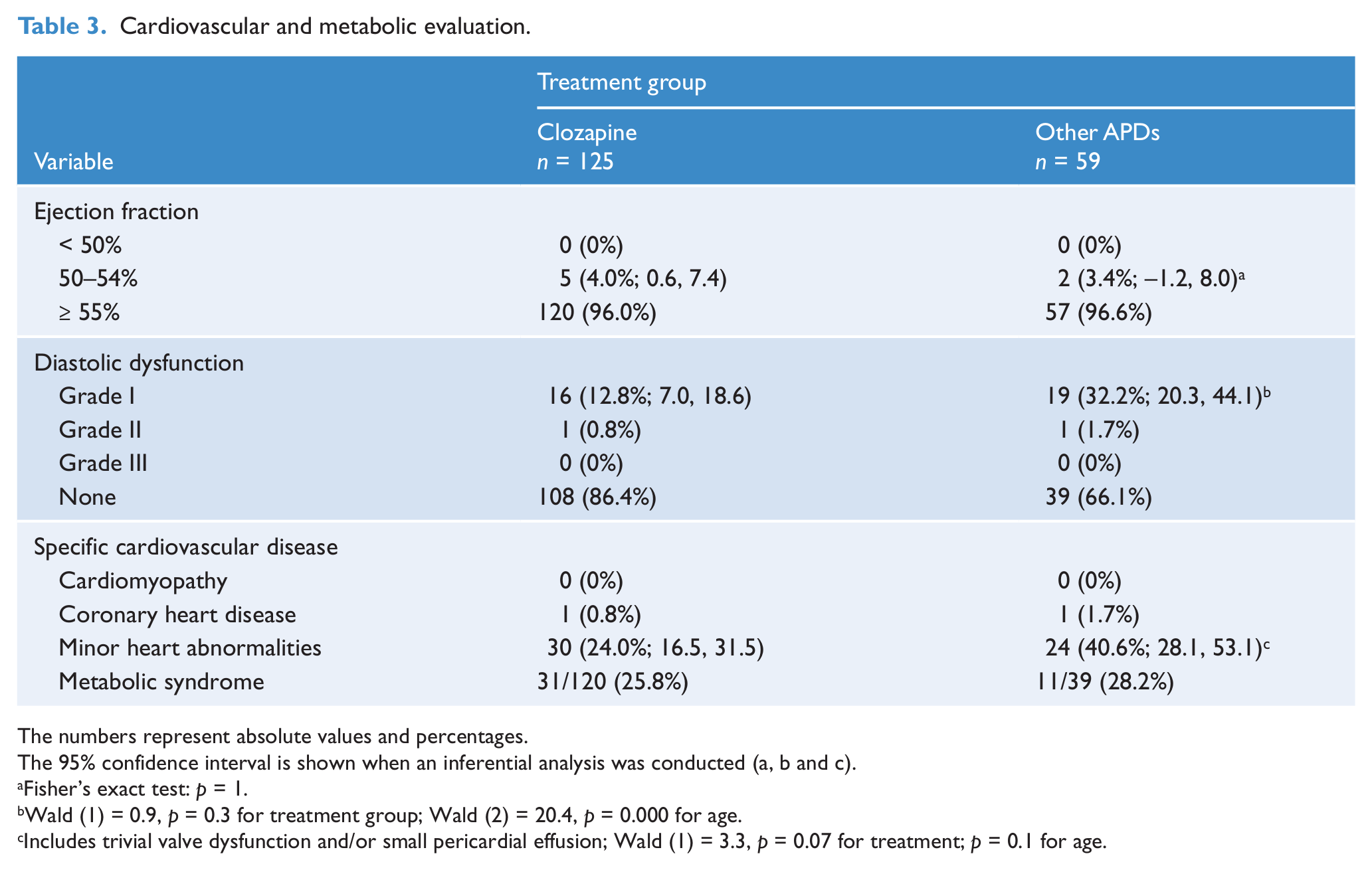
The numbers represent absolute values and percentages.
The 95% confidence interval is shown when an inferential analysis was conducted (a, b and c).
Fisher’s exact test: p = 1.
Wald (1) = 0.9, p = 0.3 for treatment group; Wald (2) = 20.4, p = 0.000 for age.
Includes trivial valve dysfunction and/or small pericardial effusion; Wald (1) = 3.3, p = 0.07 for treatment; p = 0.1 for age.
The frequency of grade I diastolic dysfunction was higher in the other-APD group, but the difference was accounted for by these subjects’ older age (Table 3).
No subject displayed an ejection fraction below 50%. The proportion of subjects with marginal ejection fraction levels (50–54%) was similar in both treatment groups (Table 3). No significant correlation was observed between the APD dose (in olanzapine equivalents) and the ejection fraction in any treatment group (p = 0.4 in both groups).
Minor heart abnormalities (trivial valve dysfunction and/or small pericardial effusion) were observed in a higher proportion in the other-APD-treated group (p = 0.07; Table 3). Interestingly, this marginal significance was not accounted for by age in the logistic regression analysis (p = 0.1 for age; Table 3).
Hyponatraemia
The study comprised 219 subjects (Table 4). Patients receiving other psychotropic drugs (OPD, n = 27) or who were drug-free (n = 9) were collapsed into one single group because of a small sample size. The CLZ group comprised a higher proportion of males than the other groups (p = 0.000; Table 4). The percentage of schizophrenia patients in the treatment groups were: CLZ: 59.8%; other atypical APDs: 16.9%; typical APDs: 56.5%; typical + atypical APDs: 58.3%; other drugs/drug-free: 0%.
Demographic and clinical characteristics in the hyponatraemia study.
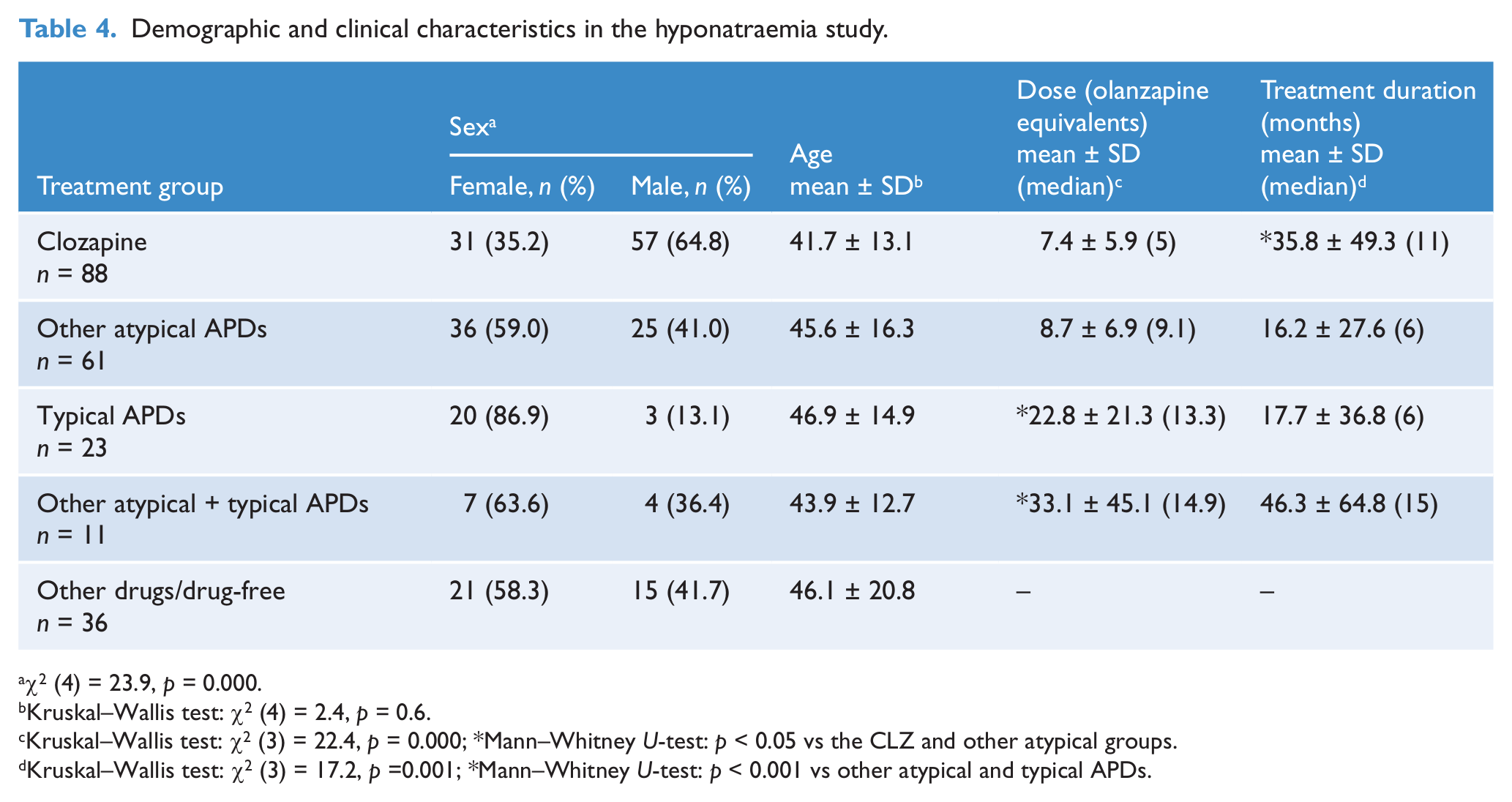
χ2 (4) = 23.9, p = 0.000.
Kruskal–Wallis test: χ2 (4) = 2.4, p = 0.6.
Kruskal–Wallis test: χ2 (3) = 22.4, p = 0.000; *Mann–Whitney U-test: p < 0.05 vs the CLZ and other atypical groups.
Kruskal–Wallis test: χ2 (3) = 17.2, p =0.001; *Mann–Whitney U-test: p < 0.001 vs other atypical and typical APDs.
Drug treatment distribution
The CLZ dose (in olanzapine equivalents) was the lowest among all the APD treatment groups (Table 4). The daily dose distribution in the CLZ group (number and percentage) was: < 100 mg: n = 30, 34.1%; 100–200 mg: n = 27, 30.7%; > 200 mg: n = 31, 35.2%. Thirty-four subjects (38.6%) only received CLZ and 54 subjects (61.4%) received CLZ + other drug treatments (anticonvulsants, antidepressants or benzodiazepines).
The drug type in the atypical APD group was: olanzapine (n = 13, 21.4%), risperidone (n = 14, 22.9%), quetiapine (n = 14, 22.9%), aripirazole (n = 5, 8.2%), APD + other drugs (n = 15, 24.6%). Nineteen patients (82.6%) in the typical APD group received one of the following agents: haloperidol, trifluoperazine, levomepromazine, or a combination of them.
Thirty percent of the subjects in the other drug group received serotonin selective reuptake inhibitors (SSRIs) alone or in combination with benzodiazepines. However, the dose distribution was not calculated due to the heterogeneity of the prescribed agents.
Frequency of hyponatraemia
Since no subject presented hyponatraemia in the drug-free/other-drug group, that group was excluded from the logistic regression analysis.
We detected 13 subjects with hyponatraemia: 10 with mild hyponatraemia, one with moderate hyponatraemia and two with the severe type. Three CLZ-treated subjects (3.4%), one in every severity group, displayed hyponatraemia, and all of them were receiving other psychotropic drugs (Tables 5 and 6). This figure was significantly lower than that observed in the typical APD group (26.1%), and similar to that of the atypical (4.9%) and combined (typical + atypical APD) (9.1%) groups (Table 5).
Frequency of hyponatraemia.
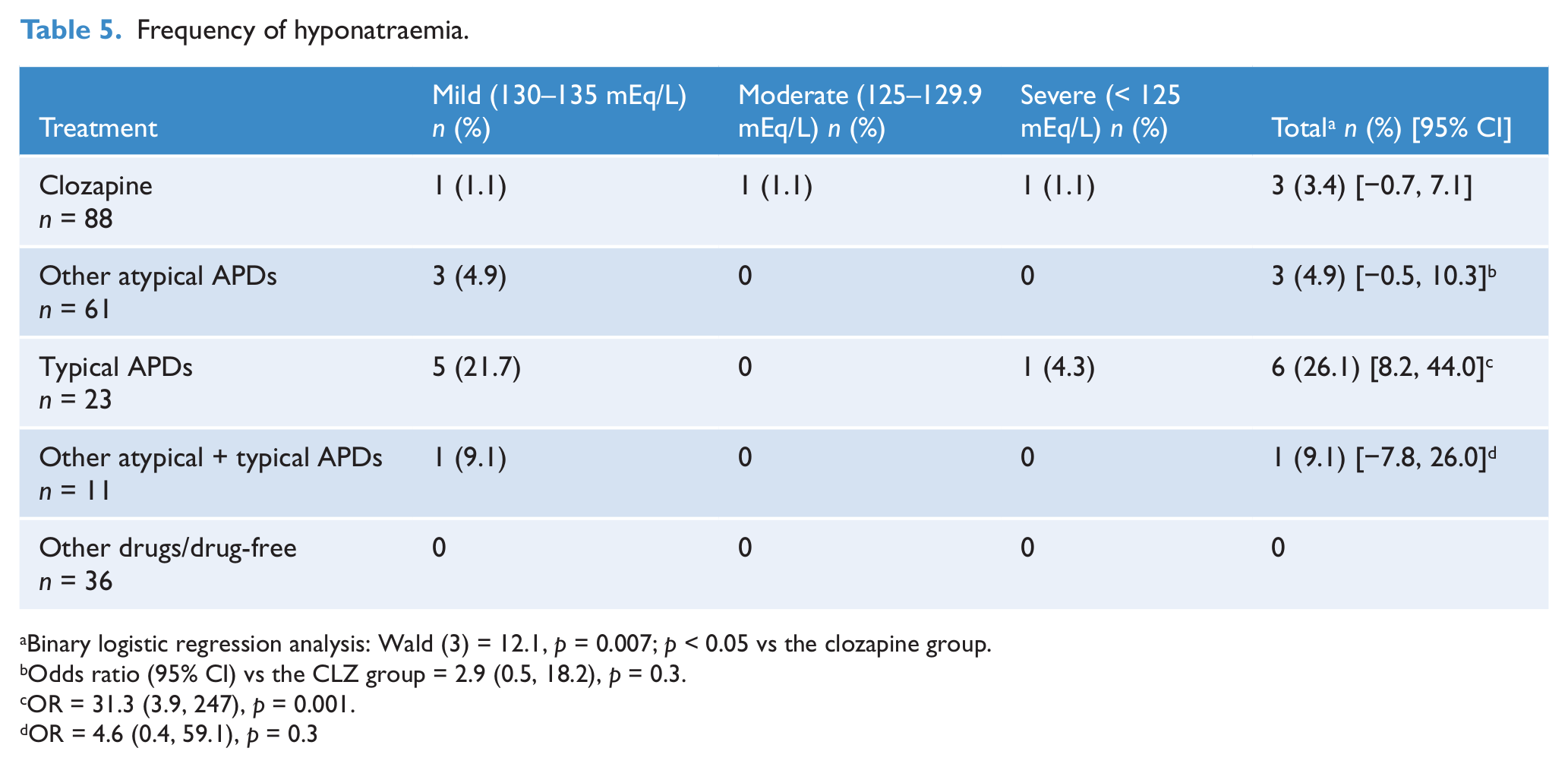
Binary logistic regression analysis: Wald (3) = 12.1, p = 0.007; p < 0.05 vs the clozapine group.
Odds ratio (95% CI) vs the CLZ group = 2.9 (0.5, 18.2), p = 0.3.
OR = 31.3 (3.9, 247), p = 0.001.
OR = 4.6 (0.4, 59.1), p = 0.3
Clinical, demographic and treatment features of the subjects with hyponatraemia.
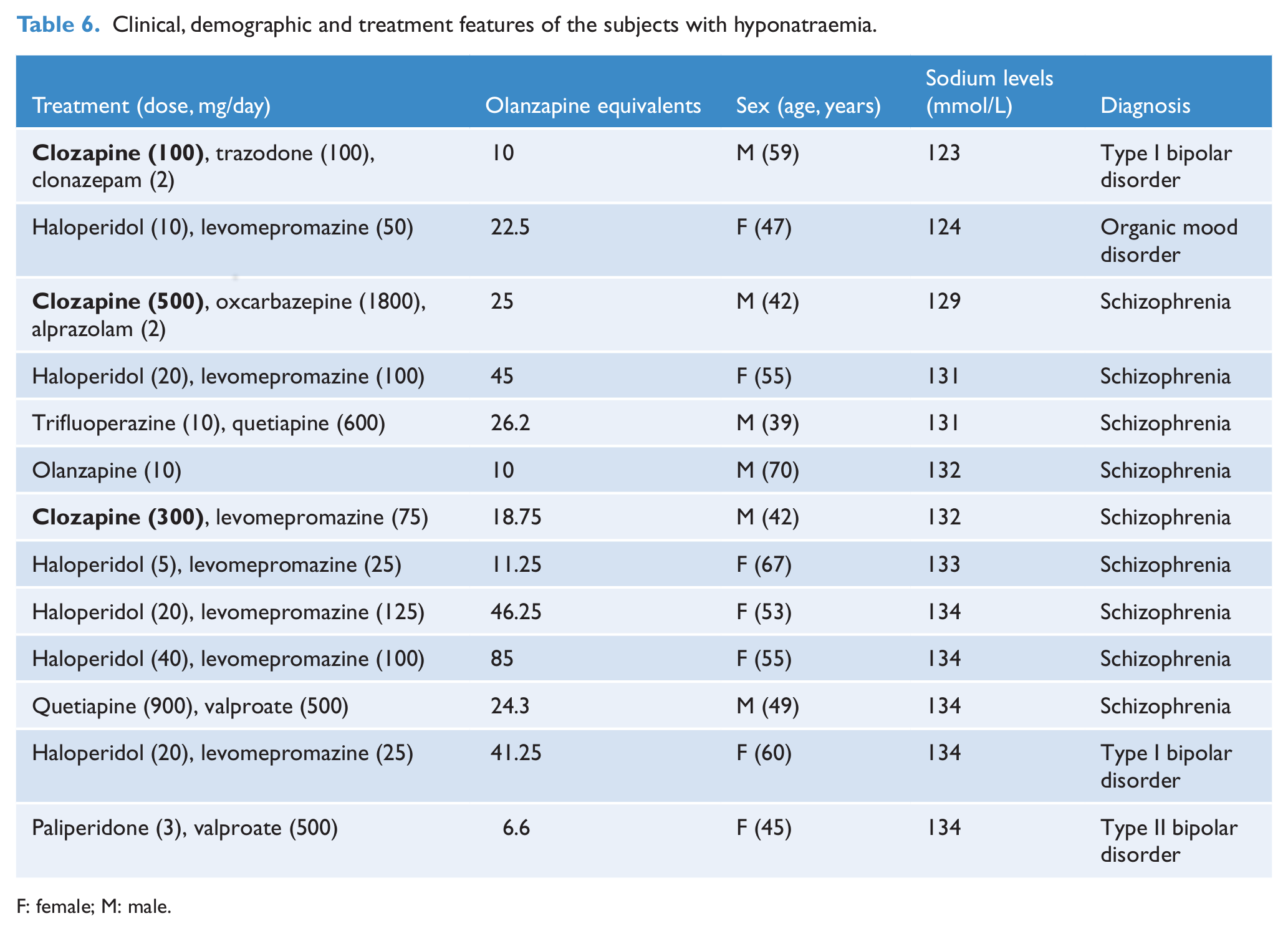
F: female; M: male.
Table 6 describes the demographic, clinical and treatment features of the 13 patients with hyponatraemia. One subject (# 6 in the table) was in treatment with a single psychotropic agent. Only subject # 3 consistently reported dizziness which lessened when oxcarbazepine was substituted by lamotrigine and sodium levels gradually increased up to 133 mmol/L.
Correlation analysis
For this analysis, the CLZ group was subdivided into subjects receiving CLZ alone or combined with other drugs.
A significant, negative correlation was observed between the serum sodium levels and the APD dose in the whole CLZ group (r [80] = −0.33, p = 0.002) and in the typical APD group (r [21] = −0.45, p = 0.03). Treatment duration and sodium levels positively correlated in the CLZ-alone group (r [30] = 0.53, p = 0.003).
Sodium and glucose levels positively correlated in the CLZ-alone (r [30] = 0.4, p = 0.024) and in the other atypical APD groups (r [49] = 0.29, p = 0.035).
Discussion
Cardiomyopathy
We did not find a single case of cardiomyopathy in our sample. Besides, the frequency of marginally abnormal ventricular ejection fraction (50–54%) and grade I diastolic dysfunction, which could be pointers of mild myocardial dysfunction, was similar in both treatment groups. As a matter of fact, minor valve abnormalities and small pericardial effusion were rather more frequent in the other-APD group.
Among the studies conducted about the cardiotoxicity of CLZ, this is one of the few that reports negative results. However, we did not find in the literature any cross-sectional or between-group studies with which to compare our findings. Kelly et al. (2009) did not find any significant difference in any of the cardiac findings at autopsy in people with severe mental illness who had received either CLZ or risperidone. Given the limited number of publications, these negative results must be cautiously evaluated.
Novel research protocols are needed to clarify the relationship between CLZ and cardiomyopathy. For example, longitudinal studies may be conducted that evaluate personal and familial risk factors and include additional tests for the early recognition of myocardial dysfunction, such as the serum levels of N-terminal pro-B-type natriuretic peptide (Rostagno et al., 2011) and heart magnetic nuclear resonance (Ariyarajah et al., 2010).
The application of some of the proposed protocols for the early detection of myocarditis may help clarify the relationship between this early inflammatory insult and cardiomyopathy (Guglin and Nallamshetty, 2012). For example, Ronaldson et al. (2011) recommend obtaining baseline troponin I/T, C-reactive protein and echocardiography, and monitoring troponin and C-reactive protein on days 7, 14, 21 and 28 after starting CLZ, along with monitoring for high heart rate or signs or symptoms of infective illness. Cessation of CLZ is recommended when troponin levels are more than twice the normal upper limit or when C-reactive protein levels are over 100 mg/L. From this set of studies, conducting a slow CLZ-dose titration is also recommended when starting the treatment to prevent myocarditis (Ronaldson et al., 2012).
The present study has the strengths of including a comparison group and a direct cardiovascular examination. In fact, we found that a significant proportion of patients in both treatment groups had grade I diastolic dysfunction. Whereas such an abnormality is usually asymptomatic and unspecific, in a large epidemiological study it was associated with a fivefold increase in 3–5-year mortality (Redfield et al., 2003).
Hyponatraemia
The frequency of hyponatraemia during CLZ administration (3.4%) was similar to, or significantly lower than the values observed in other atypical APD-treated patients (4.9%) and in the typical APD group (26.1%), respectively. All subjects were male outpatients, receiving CLZ + other drugs that may induce hyponatraemia, and two of them had schizophrenia.
Serum sodium levels showed a negative correlation with the CLZ dose. Interestingly, a significant negative correlation was also observed in the typical APD group. In agreement with our findings, Spigset and Hedenmalm (1996) reported a trend for a negative correlation (p = 0.12) in 112 subjects treated with the typical APD perphenazine. However, in contrast with our results, these authors found a trend for a positive correlation (p = 0.07) in 59 CLZ-treated patients. Meulendijks et al. (2010) reviewed 91 previously published case reports or case series and found a trend for a positive correlation for typical APDs (r [n = 56] = 0.25, p = 0.061) and a negative one for atypical APDs (r [n = 22] = −0.16, NS). While these last results agree with ours, a strict comparison is not possible because Meulendijks et al. used a daily dose-equivalent dose calculation for the APDs, whereas we used olanzapine equivalents (Gardner et al., 2010).
These correlation results may provide clues into the pathogenesis of hyponatraemia; for example, the relationship between dopamine and sigma receptor blockade and increase in antidiuretic hormone levels (Meulendijks et al., 2010). On the other hand, it is of therapeutic value because an inverse relationship suggests that the APD dose reduction should improve sodium levels. Studies about this association are scarce, and the only existing one discussed by Meulendijks et al. (2010) found no effect in lowering the dose of typical APD down to 33% on sodium levels in five schizophrenia subjects with hyponatraemia (Canuso and Goldman, 1996). However, in our study, the severity of hyponatraemia was not significantly associated with APD dose (n = 13, r = 0.4, p = 0.2).
Considering the APD treatment length, a positive significant association with sodium levels in the CLZ-alone group (p = 0.003) and a trend in the whole-CLZ (p = 0.058) and in the typical-APD (p = 0.065) groups were observed. Hence, low sodium levels were not associated with prolonged APD treatment. This finding indirectly supports the claim that hyponatraemia tends to be observed early in APD treatment (Meulendijks et al., 2010).
Since elevated serum glucose or lipid levels may induce an artificial hyponatraemia (Ellison and Berl, 2007), we conducted an additional correlation analysis. Unexpectedly, sodium and glucose levels showed a significant positive correlation in the CLZ-alone (p = 0.024) and in the other atypical APD groups (p = 0.029). No significant correlations were observed between sodium and triglyceride levels in any treatment group. When the analysis was conducted in subjects with hyponatraemia only, no significant correlation was observed either (data not shown).
Older age has been invoked as a predisposing factor for APD-induced hyponatraemia (Meulendijks et al., 2010). In fact, our 13 positive patients were significantly older than the combined group of all the other APD-treated subjects: 52.8 ± 9.5 years (median 53) versus 43.1 ± 14.6 years (median 41): t (180) = 2.4, p = 0.019. The same results were obtained when including the other-drug/drug-free group (data not shown). These results agree with those of Mannesse et al. (2010) in a case–control study, but disagree with Meulendijks et al. (2010) who reported that the age of hyponatraemia subjects in their secondary analysis was similar to that observed in patients with acute hyponatraemia of various aetiologies as controls. Again, both results are not comparable because we used APD-treated subjects without hyponatraemia as a comparison group. Lastly, serum sodium levels did not correlate significantly with age in the whole APD-treated group or in APD-treated subjects with or without hyponatraemia (data not shown).
It has been proposed that males are at a higher risk than females for polydipsia and water intoxication (de Leon et al., 1994), whereas hyponatraemia associated with antidepressants is more frequently observed in women (Meulendijks et al., 2010). In the present study, among the subjects with hyponatraemia, 46.2% were males (Table 6). This agrees with Meulendijks et al.’s (2010) study that reports a lack of association between hyponatraemia and sex during APD administration.
The specific mental disorder, particularly schizophrenia, and inpatient status, have also been suggested as predisposing factors for polydipsia and water intoxication (de Leon et al., 1994). Indeed, nine out of 13 subjects in our study had schizophrenia, which supports the role this disorder plays in the hydro-electrolytic metabolism regulation. Fifty percent of the hyponatraemia subjects were inpatients, and among the three subjects with severe hyponatraemia (sodium < 130 mmol/L) two were outpatients (subjects 1 and 3). Hence, the inpatient/outpatient status was not a determinant factor in our study. However, the number of positive subjects was too small to reach a definitive conclusion.
Limitations and general conclusions
Both studies could be underestimating the rate of prevalence due to single measurement but at the same time overestimating effects due to the naturalistic setting of design (e.g. incompliance, no drug accountability). Besides, the CLZ and control group were not strictly comparable.
Cardiomyopathy limitations
The sample size is relatively small, particularly in the other-APD group, given the low prevalence of cardiomyopathy.
Most CLZ studies have been conducted in schizophrenia patients. Nonetheless, in our study, this diagnosis only accounted for 52.8% of the sample, and a significant proportion received CLZ for off-label indications, such as bipolar and severe anxiety disorders. Unfortunately, the sample size was not powered enough to study the impact of the specific mental disorder.
Thirty-seven percent of the sample received less than 100 mg/day of CLZ, which can be considered a low dose. However, myocarditis has been observed with CLZ doses ranging from 12.5 to 500 mg/day (Hill et al., 2008).
It was not possible to obtain reliable information about the symptoms of myocarditis early in the CLZ treatment.
The cardiologist was not blinded to the treatment group. Future studies should also control for current tobacco abuse and drugs used before starting the CLZ treatment because some of them may also have a cardiotoxic effect (Montastruc et al., 2010).
Hyponatraemia limitations
Sodium levels were assessed only once – in fasting conditions only. Since hyponatraemia may be intermittent (Meulendijks et al., 2010), sodium should be repeatedly measured, including during the late afternoon (Verghese et al., 1996).
We could not assess serum and urine osmolality and sodium in urine to discard pseudohyponatraemia.
The small sample sizes in the comparison groups precluded the proper assessment of individual agents.
The CLZ dose (in olanzapine equivalents) was the lowest among all the APD-treated subjects.
The use of polypharmacy in most subjects is an impediment for attributing a causal role to a particular drug. Hence, the safety of CLZ should be tested in subjects undergoing monotherapy.
In our two naturalistic studies we found no higher risk for cardiomyopathy or hyponatraemia in patients under CLZ treatment compared to patients under other antipsychotic treatment. Owing to the above-mentioned limitations of our study design and for achieving a more thorough evaluation of the safety profile of CLZ, further studies with improved methodologies should be undertaken. This will assist clinicians in the adequate use of this APD in schizophrenia (Meltzer, 2012) and in other mental disorders where its use is currently off-label.
Footnotes
Acknowledgements
The authors thank Françoise Meyer for her editorial assistance and Abdel Fuenmayor Arocha for his critical reading of the manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
