Abstract
Rational pharmacotherapy involves weighing the relative risks and benefits of drug treatment. Usually this means balancing the therapeutic effect of a given agent against its profile of adverse effects. Many medications used in psychiatry have adverse effects, such as sedation, constipation or weight gain, which cause discomfort to the patient, and may jeopardize compliance, but are not in themselves life-threatening. Other adverse effects, such as agranulocytosis, neuroleptic malignant syndrome or cardiac arrhythmias, are less common but have the potential to be life-threatening.
A variety of psychotropic and other drugs affect the electrical activity of the heart, generally producing minor and clinically insignificant delays in cardiac conduction [1]. Serious arrhythmias rarely occur at usual doses, but are more common with overdosage of, for example, antipsychotics or tricyclic antidepressants [2].
The potential cardiotoxicity of thioridazine (a tricyclic phenothiazine) has been recognized since the 1960s [3–6], but this has not, until recently, generated much concern. In the last decade, larger, more systematic studies have found that thioridazine may be significantly more likely than other antipsychotics to produce both benign and serious ECG effects, including sudden cardiac death [7, 8].
With around 10 000 prescriptions presented quarterly, thioridazine has traditionally been the most widely prescribed antipsychotic agent in New Zealand, having only in the year 2000 been overtaken by risperidone [see Table 1. In general psychiatry, thioridazine has largely been employed as a low-potency, sedating antipsychotic agent, similar to chlorpromazine, used for the symptomatic control of acute psychotic states. Thioridazine is also used for the non-specific control of agitation, insomnia, anxiety and disturbed behaviour, particularly in children, the elderly, and the intellectually disabled. These additional indications probably account for its relatively low average daily dose, compared to the equipotent chlorpromazine (see Table 2). The great majority of prescriptions for thioridazine are written by GPs (see Table 3).
Prescription volumes for antipsychotics in New Zealand
Average daily doses (mg) for classical antipsychotics in New Zealand
Prescription of thioridazine in New Zealand
Recent concern about the cardiotoxicity of thioridazine [8] has led to marked restrictions on its use in the UK, North America and Australia. In 2001 New Zealand considered similar restrictions, prompting a focused review of ‘thioridazine’, ‘antipsychotic’, ‘arrhythmia’ and ‘cardiotoxicity’ in the scientific literature (MedLine via Ovid) and around the websites of pharmaceutical regulatory authorities in the UK, USA, New Zealand and Australia. Particularly pertinent results were obtained from online searches of the Lancet (www.lancet.com) and websites dedicated to the study of cardiac arrhythmias (see list before the References). A contribution is also noted from the website of the Royal Australian and New Zealand College of Psychiatrists (www.ranzcp.org) and from free text searches in major search engines, especially http://www.alltheweb.com.
Based on our review, we consider whether thioridazine, at normal therapeutic doses, causes unacceptably high rates of cardiac arrhythmia compared to other antipsychotics, particularly those with sedative actions and comparable non-specific clinical use. We also consider whether thioridazine has particular therapeutic advantages when used for its various indications, both specific and non-specific. We conclude with recommendations about its availability, and what should be done for patients presently receiving thioridazine, including those with and without other risk factors for cardiotoxicity.
Current indications and use
The data sheet for ‘Melleril’ (thioridazine hydrochloride, Novartis, Avondale, Auckland, New Zealand) lists a broad range of indications, from psychosis, through anxiety and depression, to agitation and insomnia. Thioridazine is fully subsidized in New Zealand by PHARMAC, although the ‘Melleril’ brand attracts a minor part charge in some presentations (25, 50 and 100 mg tablets) for which a cheaper generic equivalent (‘Aldazine’) is available [9]. Thioridazine is listed as an antipsychotic, but it is likely that substantial non-specific use contributes to the large volumes and modest average daily doses prescribed in this country (see above, and Tables 1–3).
Background considerations: cardiac electrophysiology
Drugs that alter ventricular repolarization are sometimes recognized by prolongations of the cardiac output (QT) interval in the surface electrocardiogram, and QT prolongation has been associated with malignant ventricular arrhythmias (especially the distinctive polymorphic ventricular tachycardia torsade de pointes) and death [2, 10].
With any given repolarization-inhibiting drug, the risk of malignant arrhythmia seems to multiply with increasing QT interval, but there is no well-established threshold duration, below which a prolonged QT interval is known to be harmless. The extent of QT prolongation seen with a given drug and patient may be non-linearly related to serum levels of the drug and/or its metabolites. Moreover, patient factors play a major role; for example female sex, old age, hypokalaemia and hypomagnesemia, and congestive heart failure all predispose to QT prolongation and its sequelae. The actual incidence of malignant arrhythmias, even in association with the drugs most known to induce them, is relatively low, so failure to observe malignant arrhythmias during clinical trials of ordinary size and duration does not provide substantial reassurance. This has made postmarketing reporting and surveillance of adverse drug reactions (ADRs) essential for malignant arrhythmias, as with other rare but serious ADRs [11].
On the surface electrocardiogram, the QT interval is the time from the beginning of the Q wave (or of the R wave, if there is no Q wave) to the return to baseline at the end of the T wave. The QT interval is normally shorter when the heart rate is rapid, although changes in heart rate may precede the changes in QT interval by several beats. QT intervals described as ‘prolonged’ typically have been explicitly or implicitly normalized for heart rate, using one or another formula. The common formulae result from curve-fitting of QT intervals plotted against heart rates in healthy subjects. In each of these formulae, the heart rate conventionally appears in the form of its reciprocal, the length of the cardiac cycle (the RR interval). When the normalization is explicit, for example using the Bazett formula, the normalized (‘corrected’) QT interval is called the QTc, in this case defined as QT divided by the square root of the RR interval [10].
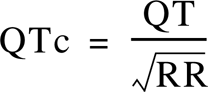
QT prolongation and associated arrhythmias result ultimately from alterations in channelled ionic flux through cardiac cell membranes. Some cells (e.g. those of the Purkinje system) seem especially susceptible to these changes. Altered ion fluxes affect the time-course of the action potential and its propagation within a given cell and from cell to cell within syncitia and from tissue to tissue within the heart. At a higher level of aggregation, one sees ‘afterdepolarizations’ in the terminal portion of the action potential: spontaneous beats triggered by afterdepolarizations; propagation of these beats to other cells; and re-entrant excitation [1]. IKs (‘s’ denotes slow) and IKr (‘r’ denotes rapid) are the delayedrectifying potassium currents responsible for phase 3 repolarization in the heart. Most or all drugs associated with torsade de pointes block the rapidly activating delayed-rectifier current, IKr. This current plays a key role in repolarizing the excitable membranes of the heart, and preparing for the next action potential. Using voltage-clamp techniques, IKr can be studied in myocyte and other physiological systems and provides a powerful preclinical model with which to study repolarization and its interference by drugs. Genetic abnormalities in this channel appear to arise from mutations of the humanether-a-go-go related gene (HERG), and cause one form of inherited long QT syndrome (LQTS type 2) [12]. Although this syndrome is very rare (affecting 1 in 10 000–15 000 individuals) it has contributed to the understanding of, as well as greatly facilitating, drug induced LQTS. The role of HERG in normal cardiac physiology appears to be suppression of depolarizations that lead to premature firing, protecting against arrhythmogenic afterbeats.
Thioridazine, quinidine and other drugs appear to prolong the QT interval by blocking the IKr channel, coded for by HERG [13], and thereby can cause serious ventricular arrhythmias, particularly during overdoses or in combination with other risk factors that lengthen QTc. In vitro testing of drugs for effects on HERG channels may predict risk of QTc prolongation, ventricular arrhythmias, and sudden death. An impressive collection of these data is available electronically [10], but a systematic study has yet to be published in the conventional press. Limitations of such an approach include the uncertain connection between in vitro pharmacology and clinical effects, the effect of drug dose and pharmacokinetics [13], and the effect of concomitant medication [10].
The Ir/HERG current depends on extracellular potassium concentration, and thus repolarization abnormalities, both in patients with inherited LQTS and with quinidine-induced acquired LQTS, can be corrected by raising serum potassium. It seems likely that the effect of psychotropics as Ir/HERG blockers can be similarly mitigated by high extracellular potassium, but this has thus far only been observed in vitro with amitriptyline [14].
Background considerations: patient vulnerability
Thioridazine appears to have the potential to lengthen the QT interval, and ultimately induce torsade de pointes, in any subject, but what differs from one subject to another is the dose at which cardiotoxicity is manifest. Clearly, there are some patients with pre-existing heart disease (such as congenital LQTS), who are at much higher risk of developing arrhythmias [12]. As mentioned above, other more common risk factors include female sex, concomitant QT prolongation by other medications, and hypokalemia or hypomagnesemia. Old age is of particular clinical importance, as it constitutes both a common independent risk factor and also covaries with the prevalence of other risk factors including cardiac disease, electrolyte disturbances, and frequent concomitant medications.
Among potential high risk patients, one group of particular interest are slow metabolizers. Thioridazine, along with a variety of other antipsychotic and antidepressant drugs, is metabolized by the cytochrome P450 2D6 iso-enzyme (CYP2D6) [15]. It has been found that 5–10% of European populations inherit the CYP2D6 poor metabolizer phenotype, which appears to confer increased risk of various concentration-dependent adverse drug reactions. This may be particularly true of cardiac effects of these drugs, since CYP2D6 is also expressed in heart tissue [16]. Screening patients for the poor metabolizer phenotype prior to antipsychotic or antidepressant drug treatment has been advocated, but has yet to be adopted in practice [17]. Genotype determination has become technically feasible, and may offer further advantages [18].
Cardiotoxicity of thioridazine
The literature which describes the cardiotoxic effects of various drugs ranges from in vitro studies of electrophysiological aspects of toxicity in tissue preparations, through to large scale clinical trials in which cardiotoxic adverse effects of drugs are of particular interest. Current knowledge about the risk inherent in prescribing drugs such as thioridazine is thus a synthesis of evidence gathered at a variety of levels of investigation.
The potential cardiotoxicity of thioridazine has been recognized since the 1960s, but this recognition does not seem, until recent times, to have gained a very high profile. One of the studies that can be credited with raising this profile was published in the Lancet in March 2000 [8]. In this study, ECG recordings from 495 psychiatric patients treated in a variety of inpatient and outpatient settings were compared with 101 healthy controls. ECG recordings from the reference group were used to generate a normal range of values for the QT interval. A ratecorrected QT interval of 456 ms was considered the threshold beyond which the QT interval was classified as abnormally long − 40 (8%) of the 495 patients in the study fell into this group. Several statistically robust predictors of an abnormally long QT interval were found, notably:
– Age over 65
– Use of tricyclic antidepressants
– Use of thioridazine
– Use of droperidol
– High antipsychotic dose
Other antipsychotic and antidepressant drugs failed to prolong QT significantly, but the modest numbers in each treatment group limits interpretation of this negative finding, particularly as drugs such as chlorpromazine are nearly as active as thioridazine in preclinical models of Ir/HERG antagonism [13].
QT prolongation by thioridazine has been related to the plasma concentration of the drug and principal metabolites. Interestingly, this effect is dose-dependent and occurs at quite modest doses of the drug (10 mg or 50 mg), even in rapid metabolisers [19] taking no other medication. Other studies have examined the impact of drug interactions on the QTc interval. This is particularly important given the large number of drugs used in psychiatric practice which are metabolized by, or inhibit, CYP isozymes [20].
Decisions of regulatory bodies
The Federal Drug Administration (FDA) in the USA, and the Medicines Advisory Committee (MAC) in the United Kingdom have both acted in 2000 to markedly restrict the approved indications for thioridazine, in the wake of concern regarding its potential cardiotoxicity. In addition, the Royal Australian and New Zealand College of Psychiatrists has recently advised its members that the use of thioridazine should be drastically restricted. In essence, these bodies have recommended that thioridazine use be closely monitored and restricted to patients with schizophrenia unresponsive to other therapy. A typical set of stringent restrictions is described in the FDA website [21].
Alternatives to thioridazine
Restriction of the historically broad indications for thioridazine treatment to the single indication of treatment-refractory schizophrenia would essentially remove the drug from the market. In New Zealand, a range of antipsychotic medications is currently available, including the ‘atypical’ agents clozapine, olanzapine and risperidone [9]. To the best of our knowledge, thioridazine does not have any greater efficacy in the treatment of schizophrenia than other drugs available and is hardly likely to be chosen ahead of the other available drugs as a second line treatment for schizophrenia. There is, as yet, little evidence to support our impression that thioridazine's non-specific indications can be met comparably well by other sedative antipsychotics, such as methotrimeprazine or pericyazine. These drugs can be tried as alternatives to thioridazine, and appear to pose distinctly less cardiac risk (as noted above, the same may not be true of chlorpromazine).
Conclusion and recommendations
Regulatory authorities in the United Kingdom, Ireland, USA and Australia have all responded to concerns regarding the cardiotoxic potential of thioridazine by markedly restricting the indications for this drug to include only the second (or third) line treatment of schizophrenia. It is likely that the number of new prescriptions for thioridazine in these countries will be negligible, and that patients already stabilized on thioridazine for nonapproved indications are being switched to alternative drugs. Withdrawal syndromes and psychosocial disruption are likely to follow unless this is done gradually with careful follow up.
Fatal cardiac arrhythmia is a rare complication of treatment with thioridazine, but this agent is by no means the only antipsychotic demonstrated to lengthen the QT interval. For example, droperidol, which had been regarded as a comparatively safe antipsychotic, has been shown to lengthen the QT interval in a dose-dependant manner, in much the same way as thioridazine. Janssen Cilag announced in December 2000 that droperidol would be withdrawn by the end of 2001, ostensibly because of its effect on cardiac conduction. As noted above, other antipsychotics are also known to interfere with Ir/HERG currents, and are also likely to prolong QT and, in susceptible patients, promote torsade de pointes [10, 13].
The close restriction on the indications for thioridazine now in place in Australia, the United Kingdom and North America could be seen as something of an overreaction by the regulatory bodies concerned. Fortunately, New Zealand's Ministry of Health has taken a more measured, realistic approach to the problem in its communication posted to prescribers in June 2001 [22] This document recommends:
– thioridazine should be initiated only by a specialist and only as third line therapy and after careful review of cardiac risk factors. (we note that it is scarcely conceivable that new patients with schizophrenia would receive this agent after unsuccessful trials of multiple other agents) – patients established on thioridazine should be carefully assessed for arrhythmia risk factors, and ECG (in the morning after a nocte dose) and serum potassium should be monitored. Patients with a QTc over 500 ms should have their thioridazine dose reduced (we suggest those with a QTc between 460 and 500 should be withdrawn where possible, and other risk factors addressed) – Children should continue thioridazine only after specialist review – Drugs which inhibit CYP2D6 or otherwise impair metabolism of thioridazine (cimetidine, antidepressants, pindolol, propranolol) should not be co-prescribed with thioridazine – Drugs which can also prolong QT (antiarrhythmics, antipsychotics, cisapride) should not be co-prescribed with thioridazine – Doses over 200 mg/day should be avoided, and patients appearing to require such doses should be reviewed by a specialist (N.B. 800–900 patients in New Zealand receive prescriptions at this level, see Table 3).
Wherever possible or convenient, alternatives to thioridazine should be considered and tried, after or during its gradual withdrawal (see Alternatives to Thioridazine, above). The decision to require withdrawal of successful treatment with thioridazine has been adopted in a number of countries. New Zealand authorities have avoided such an extreme measure, and indeed have wisely decided against requiring specialist review of such patients, except for children and those receiving doses above 200 mg/day. There would be, in any case, enormous practical difficulty in arranging review of so many patients, the great majority of whom receive their prescription from a GP [Table 3. It would be disruptive for many patients to transfer to alternative medications, and such requirement is predicted to increase rates of decompensation and hospital admission. This is likely to occur more frequently in countries, such as UK, USA and Australia, requiring the great majority of thioridazinetreated patients to be withdrawn. Patients treated for long periods with thioridazine may experience marked behavioural and physiological reactions when the drug is withdrawn [23], and the month-long withdrawal recommended by the New Zealand Ministry of Health may turn out to be insufficient in many cases.
We endorse the New Zealand government's position that all patients taking thioridazine be monitored, and only those judged high risk (according to the above criteria) be necessarily withdrawn and transferred to alternative treatments. It should be emphasized that the overwhelming majority of patients prescribed thioridazine in New Zealand appear to tolerate the drug well, and particularly those with modest dose ranges (see Table 3) and average daily doses (Table 2) would appear to be at low risk of cardiotoxicity.
The present recommendations will accelerate the already diminishing use of thioridazine (see Table 1), but allow its continuation in a population of patients demonstrably benefiting from treatment. Many medications have rare but life-threatening adverse effects, yet they continue to be prescribed because the therapeutic benefit conferred by these medications is judged to outweigh potential risks associated with their use.
