Abstract
Objective:
To compare the therapeutic effects of vitamin D3 plus fluoxetine and fluoxetine alone in patients with major depressive disorder.
Methods:
In the present double-blind, randomized, placebo-controlled trial, 42 patients with a diagnosis of major depressive disorder based on DSM-IV criteria were randomly assigned into two groups to receive daily either 1500 IU vitamin D3 plus 20 mg fluoxetine or fluoxetine alone for 8 weeks. Depression severity was assessed at 2-week intervals using the 24-item Hamilton Depression Rating Scale (HDRS) as a primary outcome measure and the 21-item Beck Depression Inventory (BDI) as a secondary outcome measure. Serum 25(OH) vitamin D was measured at baseline and after intervention.
Results:
Forty patients completed the trial. A two-way repeated-measures analysis of variance showed that depression severity based on HDRS and BDI decreased significantly after intervention, with a significant difference between the two groups. The vitamin D + fluoxetine combination was significantly better than fluoxetine alone from the fourth week of treatment.
Conclusions:
In the present 8-week trial, the vitamin D + fluoxetine combination was superior to fluoxetine alone in controlling depressive symptoms.
Introduction
Unipolar depressive disorders were the third cause of disability-adjusted life years lost in all age groups based on a World Health Organization report of the year 2004 and, if the current trend continues, it is anticipated to be the leading cause of morbidity by 2030 (WHO, 2008).
It is reported that about 19–34% of major depressive disorder cases resist good response to routine antidepressants (Arroll et al., 2005; Fava and Davidson, 1996).
Considering the wide use of complementary and alternative medications such as dietary supplements in depressed patients and our insufficient knowledge about the interaction between drugs and supplements, investigating the subject from a clinical point of view seems important (Kessler et al., 2001).
Vitamin D receptors exist in many different tissues; thus, hypovitaminosis D theoretically can cause various adverse effects on health (Holick, 2007). Recently, growing evidence has linked this vitamin to brain function (McGrath et al., 2001). Active target sites have been identified for this nutrient in different parts of the brain, such as the cortex and hippocampus (Kalueff and Tuohimaa, 2007; Langub et al., 2001). Moreover, the main enzyme responsible for changing vitamin D to its active form exists in the brain (Eyles et al., 2005; Neveu et al., 1994). Finally, animal studies have confirmed the fundamental role of vitamin D in the development of the brain (Eyles et al., 2009).
Some information about the relationship of this vitamin with psychiatric diseases in humans was derived from studies showing worse cognitive performance in age-related hypovitaminosis D (Buell and Dawson-Hughes, 2008; Cherniack et al., 2009) and accelerated osteoporosis or less bone mineral density in psychiatric patients (Eskandari et al., 2007; Halbreich and Palter, 1996; Michelson et al., 1996). Furthermore, cross-sectional studies have shown that there is an inverse association between vitamin D status and depressive symptoms (Armstrong et al., 2007; Hoogendijk et al., 2008; Jorde et al., 2008; Nanri et al., 2009; Oren et al., 1994; Schneider et al., 2000; Stewart and Hirani, 2010; Wilkins et al., 2006). Also, a large cross-sectional and prospective study in women aged 50–79 years has shown that higher intakes of vitamin D were associated with lower depressive symptoms and a lower risk of depressive symptoms after 3 years (Bertone-Johnson et al., 2011). Few interventional studies have assessed the effects of vitamin D supplementation on mood and depression symptoms in healthy (Lansdowne and Provost, 1998), overweight and obese subjects (Jorde et al., 2008), or in patients with seasonal affective disorder (Gloth et al., 1999) or normal women (Harris and Dawson-Hughes, 1993). The results have been inconsistent, which may be due to the doses of vitamin D given, different rates of prevalence of vitamin D deficiency, or clinical depression in the participants. We could not find any published randomized, placebo-controlled clinical trial about vitamin D supplementation in major depressive disorder. A recent open trial by Zanetidou and colleagues (2011) has shown that a single high dose of vitamin D had beneficial effects in elderly patients with major depressive disorder.
Taking into account the potential therapeutic role of vitamin D in mood disorders, we designed the present study to determine the therapeutic effects of vitamin D as an adjunctive therapy to fluoxetine in patients with major depressive disorder. As low doses of vitamin D had not been effective in previous studies (Dumville et al., 2006; Harris and Dawson-Hughes, 1993), we chose a dose of 1500 IU/day, which was greater than the recommended dietary allowance (RDA) and lower than the upper tolerable intake level (UL) for this vitamin (Institute of Medicine, 2010).
Materials and methods
Patients
The patients were 42 outpatients aged between 18 and 65 years old with a diagnosis of major depressive disorder without psychotic features based on DSM-IV criteria and with a score of ≥ 15 on the 17-item Hamilton Depression Rating Scale (HDRS) who were referred from Roozbeh Psychiatry Hospital, Tehran University of Medical Sciences, Tehran. The patients had not taken any antidepressant or dietary supplements during the previous 2 months, were free from other psychiatric diagnoses, significant medical illnesses or suicidal thoughts and were willing to participate in the study. Substance abuse, pregnancy and lactation, and the occurrence of important adverse effects from medications were exclusion criteria.
Test drugs
Tablets of 1000 IU vitamin D3 were supplied by Merk (Germany). The placebo tablets contained starch, were identical to the vitamin D3 tablets in appearance and were supplied by SOHA Pharmaceutical Company (Iran). Fluoxetine was provided as 20 mg capsules.
Trial design
After signing written informed consent, eligible patients were randomly assigned to two groups to receive daily either 1.5 tablets (1500 IU) of vitamin D3 plus one capsule (20 mg) fluoxetine or vitamin D placebo plus 20 mg fluoxetine for 8 weeks in a double-blind, placebo-controlled manner. Depression severity was assessed at 2-week intervals using the 24-item HDRS as a primary outcome measure and the 21-item Beck Depression Inventory (BDI) as a secondary outcome measure.
Blood samples were collected after 8–10 hours of fasting at baseline and serums were frozen at −80°C to measure 25(OH)D levels by enzyme-linked immunosorbent assay (ELISA).
The protocol of the study was compliant with the declaration of Helsinki and was approved by the Ethical Committee of Tehran University of Medical Sciences. The trial was registered in Iran Registry of Clinical Trials (IRCT ID: IRCT201201072394N6).
Compliance was measured by counting fluoxetine pills and measuring serum 25(OH)D.
Results
Sampling was started in November 2010 and the last subject entered the study in October 2011. Analysis was performed on the 40 patients who completed the study. One patient was lost to follow-up from the fourth week of treatment in the fluoxetine group and one was excluded from the fluoxetine + vitamin D group because of severe anxiety at week 2. No other important adverse effect was reported. The baseline characteristics of patients are shown in Table 1. There were no significant differences between the two groups with regard to age, weight, body mass index (BMI), serum 25(OH)D, sex, use of sunscreen, smoking status or depression severity. Moreover, the BMI did not change significantly during the intervention period (p > 0.05).
General characteristics of the patients.
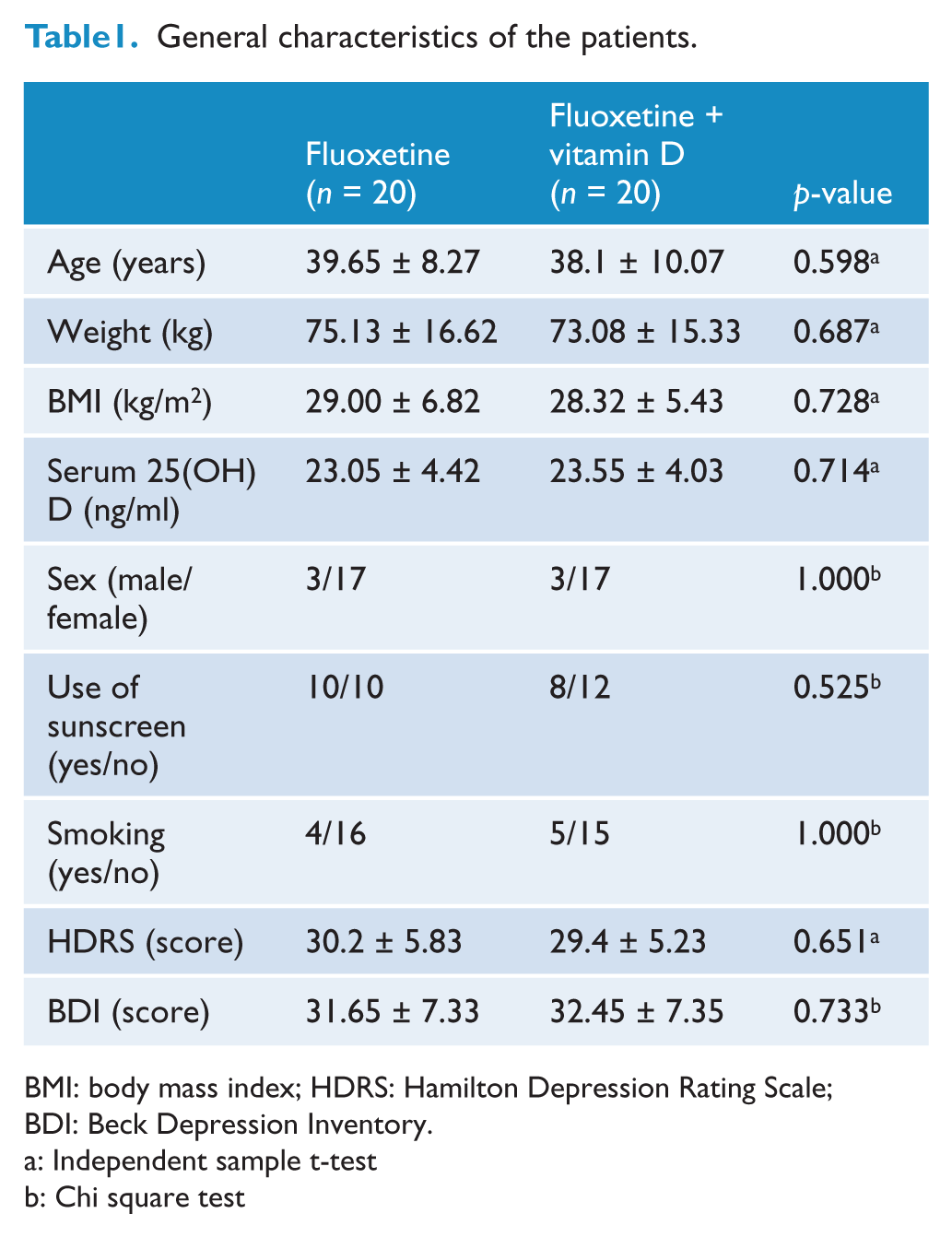
BMI: body mass index; HDRS: Hamilton Depression Rating Scale; BDI: Beck Depression Inventory.
Independent sample t-test
Chi square test
With regard to compliance, all patients consumed more than 90% of the fluoxetine tablets and the serum 25(OH)D increased significantly in the fluoxetine + vitamin D group from 23.55 ± 4.03 to 46.85 ± 14.14 (p < 0.001). There was a negative correlation between baseline serum 25(OH)D and depression severity (Figure 1).
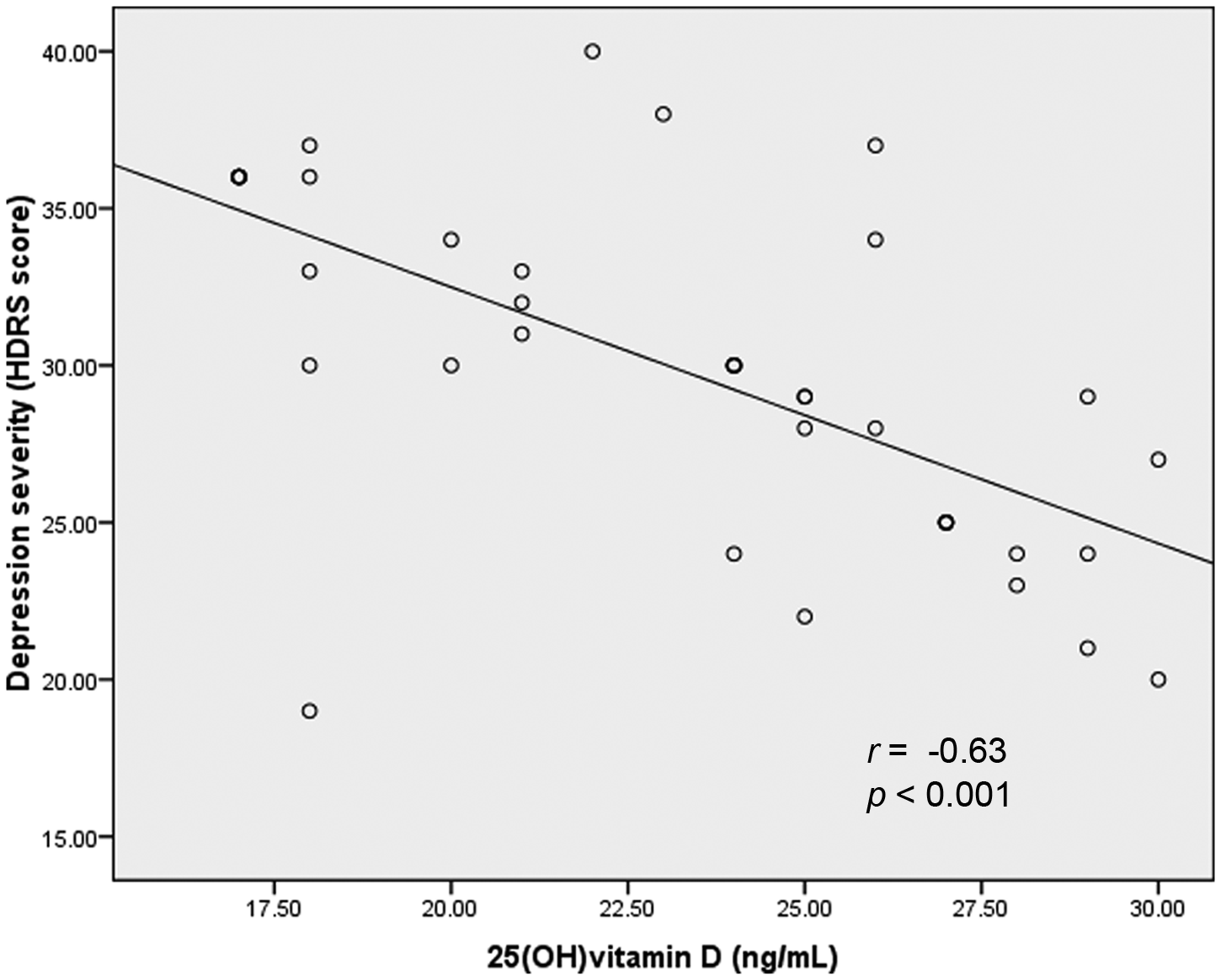
Correlation between vitamin D status and depression severity (HDRS: Hamilton Depression Rating Scale).
As Table 2 shows, the two-way repeated-measures analysis of variance showed that depression severity decreased significantly after intervention with a significant difference between the two groups. Analysis of covariance for depression severity adjusted for baseline values at weeks 2, 4, 6 and 8 showed that the vitamin D–fluoxetine combination was significantly better than fluoxetine alone from the fourth week of treatment.
Depression severity during 8 weeks of intervention.
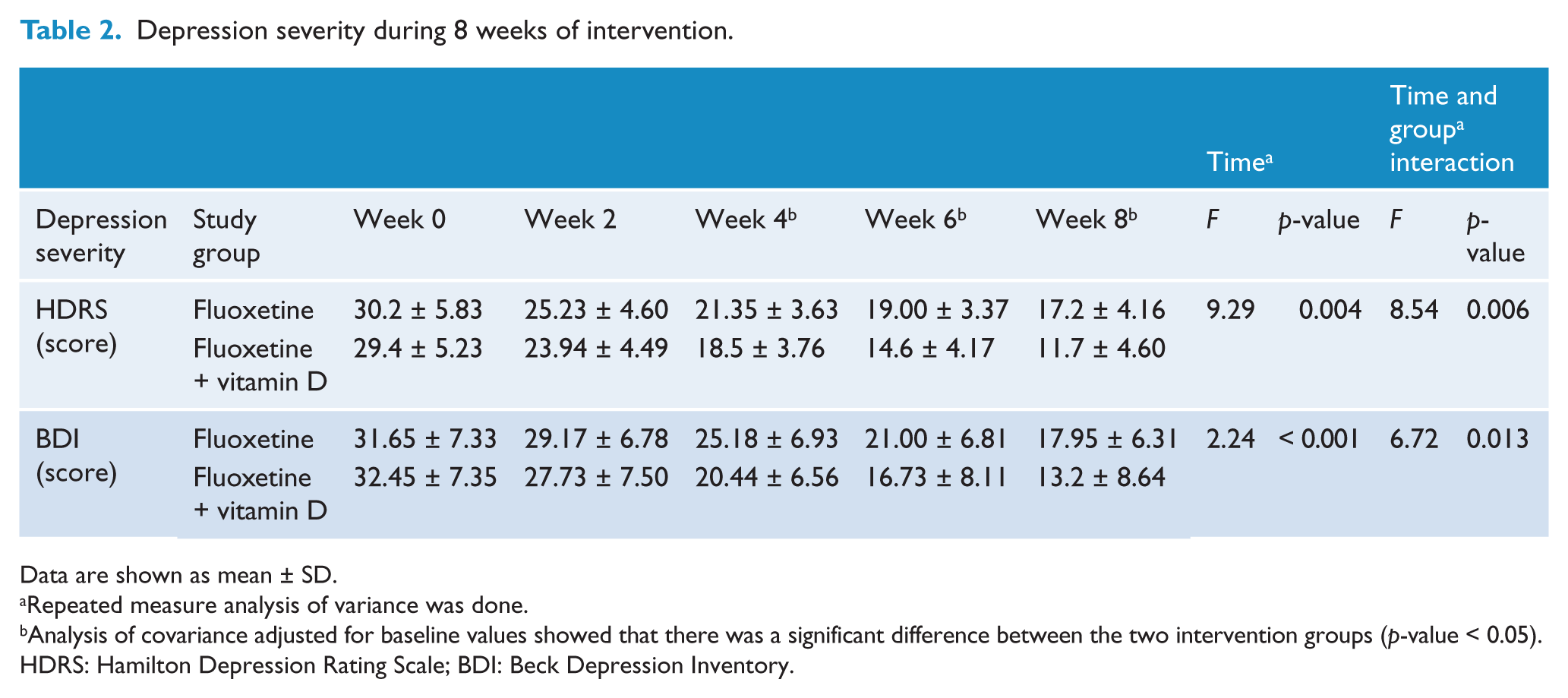
Data are shown as mean ± SD.
Repeated measure analysis of variance was done.
Analysis of covariance adjusted for baseline values showed that there was a significant difference between the two intervention groups (p-value < 0.05).
HDRS: Hamilton Depression Rating Scale; BDI: Beck Depression Inventory.
Discussion
To our knowledge, the present study is the first randomized, placebo-controlled trial specifically designed to determine the effects of vitamin D supplementation in patients with major depressive disorder, and the results showed that vitamin D is safe and effective as an adjunctive treatment to fluoxetine in major depressive disorder. It should be considered that 95% of patients participating in the study were vitamin D-deficient based on serum 25(OH)D concentrations < 30 ng/ml and were moderately or severely depressed.
The few previous studies assessing the effects of vitamin D supplementation on mood and depression symptoms have shown inconsistent results. Some of them have reported positive effects of supplementation in overweight and obese people (Jorde et al., 2008), patients with seasonal affective disorder (Gloth et al., 1999) and healthy subjects (Lansdowne and Provost, 1998), but other results were not consistent to ours, which may be due to inadequate doses of vitamin D (Harris and Dawson-Hughes, 1993), a low prevalence of vitamin D deficiency (Dean et al., 2011) and a low prevalence of clinical depression, or seasonal affective disorder (Dumville et al., 2006) in the participants. Moreover, in a recent open trial, it was shown that a single 300,000 IU dose of vitamin D could decrease depression severity in elderly patients with major depression (Zanetidou et al., 2011). However, a large randomized trial in older women has shown that a single annual dose of 500,000 IU of vitamin D3 for 3–5 years had no benefit on mood, although there was a non-significant improvement in those taking medications for anxiety and depression (Sanders et al., 2011). In another study, 40,000 IU per week of vitamin D for 6 months in people with low levels of serum 25(OH)D had no significant effect on depressive symptom scores compared to placebo (Kjaergaard et al., 2012). The daily doses of vitamin D used in these studies were too high compared to the UL and probably the relationship between serum vitamin D and depression is not linear as it is between neonatal blood vitamin D and risk of schizophrenia (McGrath et al., 2010).
An inverse linear correlation was also seen between depression severity and serum 25(OH)D levels in our study. Some previous studies have also reported an association between depression status and lower levels of serum vitamin D metabolites (Hoogendijk et al., 2008; Jorde et al., 2006; Schneider et al., 2000; Wilkins et al., 2006).
The mechanism of action of vitamin D in the brain and its effects on mood is not completely understood. A possible mechanism is an increased expression of the tyrosine hydroxylase gene (the rate-limiting enzyme of the catecholamine synthesis pathway) and enhancement of the bioavailability of some neurotransmitters such as dopamine, noradrenalin and adrenalin by vitamin D. This vitamin may also affect the cholinergic system by activating choline acetyltransferase, which is considered to play a role in the pathophysiology of mood disorders. Moreover, it was shown that calcitriol potentially can enhance the level of neurotrophic factors involved in depression such as nerve growth factor (NGF), glial-derived neurotrophic factor (GDNF) and neurotrophin 3 (NT-3) (Humble, 2010).
In conclusion, it seems that vitamin D deficiency has a role in the pathophysiology of depression and that its dietary supplementation is safe and effective as an adjunctive treatment in major depressive disorder, at least in vitamin D-deficient subjects.
Footnotes
Acknowledgements
This work was supported by the Vice-Chancellor for Research, Tehran University of Medical Sciences, Tehran, Iran. We would also like to acknowledge the cooperation and help of Dr Farhoud, Dr Nejatisafa and Dr Arbabi. Thanks are also extended to SOHA Pharmaceutical Company for supplying placebo tablets.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
