Abstract
Analysis of the global burden of disease in the year 2000 showed that unipolar depressive disorders were ranked as the fourth highest among all diseases [1]. In the age group 15–44 years they generated the second highest burden. By the year 2020, if current trends for demographic and epidemiological transition continue, depression would become the second leading cause of disability-adjusted life years lost worldwide and the highest ranking cause of disease in the developed regions. A systematic review comparing antidepressants with placebo for treatment of depression in primary care showed that both tricyclic antidepressants and selective serotonin re-uptake inhibitor were effective in treatment of depressive symptoms compared with placebo, and 56–60% of patients responded well to these antidepressants [2]. On the whole, 19–34% of patients with major depressive disorder will become treatment-resistant [3].
Depression is among the 10 most frequent indications for using complementary and alternative medicine [4]. Use of dietary supplements is common among people being treated for depression. Not very much is known about interactions between drugs and supplements, therefore it is particularly important from a clinical point of view to investigate this [5]. Omega-3 fatty acids (ω−3FAs) are among supplements that depressed patients consume. Docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) are the two major long chain ω−3FAs considered important in depression. Epidemiological and treatment studies have suggested that dietary ω−3FA deficiency may be of aetiological importance in depression. It has been reported that depression is inversely related to long-term ω−3FA intake in adults, the elderly and adolescents in the island of Crete [6–8]. An ecologic study has provided a strongly negative correlation between fish consumption and major depressive disorder prevalence in several countries [9]. Edwards et al. reported that severity of depression correlated negatively with the red blood cell membrane levels of ω−3FAs and their dietary intake in depressed subjects [10]. There is evidence suggesting that ω−3FAs have therapeutic effects in unipolar depression but the findings have been inconsistent [11–15]. Furthermore, it has been found that elevated ω−6FA: ω−3FAs ratios could predict suicidal behaviour in major depression [16]. A recent systematic review of published trials about effects of ω−3FAs on depressed mood provided little support for the use of ω−3FAs to improve depressed mood and have suggested more trials to detect clinical effects [17]. In contrast, two other reviews supported protective effect of ω−3FA intake in mood disorders and its benefit in unipolar and bipolar depression, but they have suggested further treatment studies to examine the efficacy of ω−3FA and to determine which ω−3FA is optimal in different disorders [18], [19].
The objective of the present study was to compare therapeutic effects of EPA, fluoxetine and a combination of them in major depression. A placebo-only group could not be included for ethical reasons. To our knowledge this is the first report of EPA monotherapy in major depressive disorder.
Methods
Patients
Patients were referred from Roozbeh Psychiatry Hospital, Tehran, Iran to participate in the study. The study was explained to them and if the patients were eligible and willing to participate, written informed consent was then obtained. The protocol, prepared in accordance with the Helsinki declaration, was approved by the Ethical Committee, Research Department of Tehran University of Medical Sciences.
The patients were 20–59 years of age and met DSM-IV criteria for major depressive disorder without psychotic features based on the semi-structured clinical interview. Their scores on the 17-item Hamilton Depression Rating Scale (HDRS) were ≥15 and they were free of medication for at least 6 weeks. The exclusion criteria were comorbid psychiatric diagnosis other than dysthymia and anxiety, significant medical illness established by medical history, physical examination or laboratory tests, suicidal thoughts, substance abuse, history of hypomanic/manic/mixed episode, pregnancy and lactation, consumption of ω−3FA supplements in the previous year, and dietary intake of more than one serving of fish per week.
Test drugs
Soft gels of 550 mg ethyl-EPA (500 mg pure EPA and 11 mg vitamin E as antioxidant) were supplied by Minami Nutrition, Belgium. The placebo soft gels contained 550 mg rapeseed oil and 11 mg vitamin E and were identical to the ethyl-EPA soft gels in appearance. Fluoxetine was provided as 20 mg capsules and its placebo contained starch and avicel.
Trial design
Patients were randomly allocated into three groups according to pre-arranged balanced block randomization to receive daily either two ethyl-EPA soft gels (1000 mg EPA) plus fluoxetine placebo, or one fluoxetine capsule (20 mg fluoxetine) plus ethyl-EPA placebo, or two ethyl-EPA soft gels (1000 mg EPA) plus one fluoxetine capsule (20 mg fluoxetine) for 8 weeks. The study was double blind and because the fluoxetine capsule and EPA soft gel were not identical we used a double dummy technique to blind patients and the physician who assessed them.
The patients underwent psychiatric assessment by means of HDRS at baseline and at weeks 2, 4, 6 and 8. Adverse events were recorded in a checklist at each visit. Compliance was estimated by counting pills. Patients were considered compliant if they consumed more than 90% of the medication.
Statistical analysis
Given α = 0.05, β = 0.2 and a final difference between the groups of ≥5 on the HDRS, the sample size was calculated to be at least 15 in each group. Patients who remained in the study for at least 4 weeks were included in analysis. Change on HDRS was the primary outcome measure. Analysis of covariance was used to compare HDRS changes among groups. Differences were considered significant for p < 0.05.
Results
Patients characteristics
A total of 60 patients were recruited into the study (20 in each group). Forty-eight patients completed at least 4 weeks of the study, so they were included in the analysis (four patients in each group dropped out). In the fluoxetine group one patient withdrew because of drowsiness after taking the medications, one was lost due to non-compliance and two were lost to follow up. In the EPA group one patient was excluded due to developing suicidal ideation, one due to non-compliance and two were lost to follow up. In the group receiving EPA and fluoxetine combination one patient dropped out due to steatorrhoea, one because of skin rash, one due to non-compliance and one was lost to follow up.
As shown in Table 1 the groups were matched with regard to sex, age, and duration of recent episode, but there was a difference in age of onset and the number of previous episodes, although it was not statistically significant. These variables were included as covariates in the analysis of covariance.
Patient details
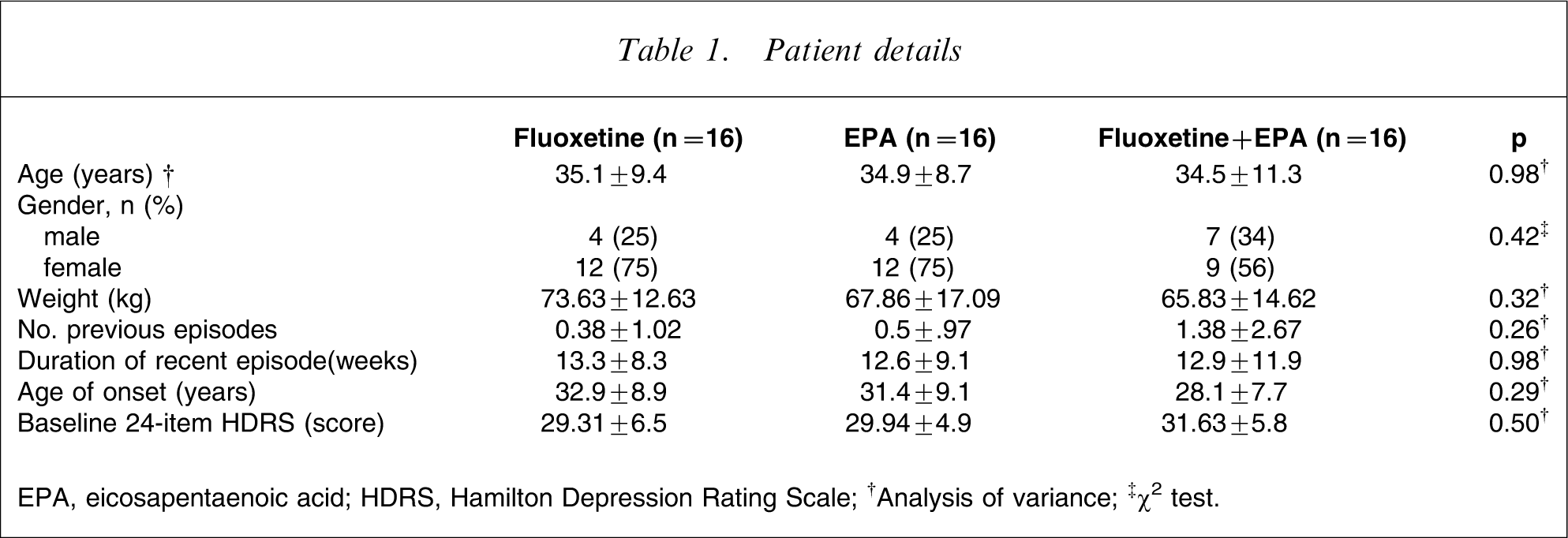
EPA, eicosapentaenoic acid; HDRS, Hamilton Depression Rating Scale.
†Analysis of variance; ‡χ2 test.
Between-group analysis
Figure 1 illustrates HDRS scores in the 48 patients who completed at least 4 weeks of the study, with the last observation carried forward.
Changes in the Hamilton Depression Rating scale (HDRS) over time in the fluoxetine, eicosapentaenoic acid (EPA) and EPA + fluoxetine groups. Analysis of covariance for HDRS adjusted for baseline scores, age of onset and number of previous episodes was performed at weeks 2, 4, 6 and 8. There was a significant effect of treatment at week 4 (F = 4.58, p = 0.016), week 6 (F = 4.28, p = 0.02) and week 8 (F = 6.02, p = 0.005).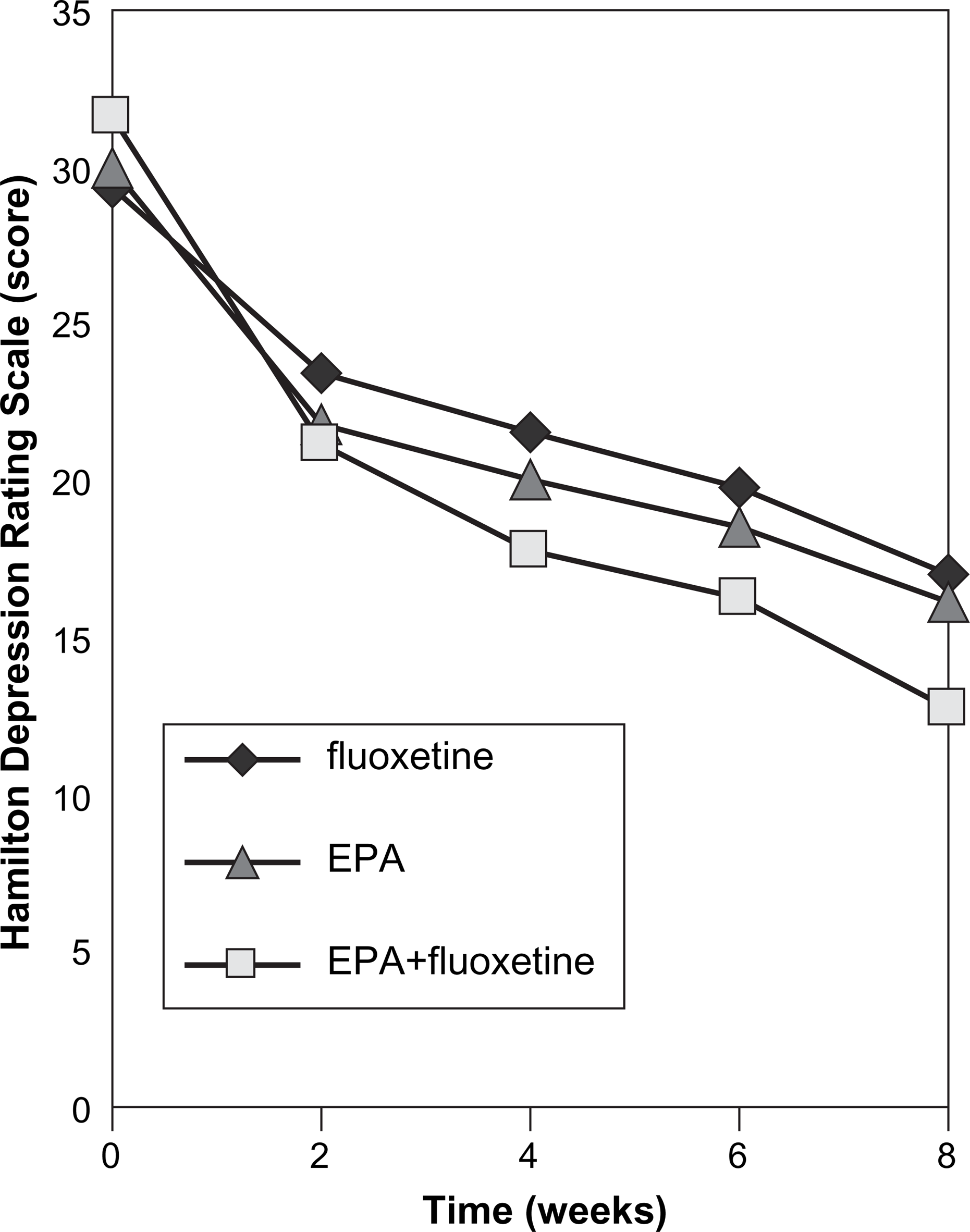
Analysis of covariance (ANCOVA) for HDRS at week 8 across treatment groups was performed. Baseline HDRS, age of onset and number of previous episodes were covariates. Results of ANCOVA are shown in Table 2. Treatment, age of onset and baseline HDRS had a significant effect on HDRS at week 8.
Analysis of covariance for HDRS at week 8 across the three groups
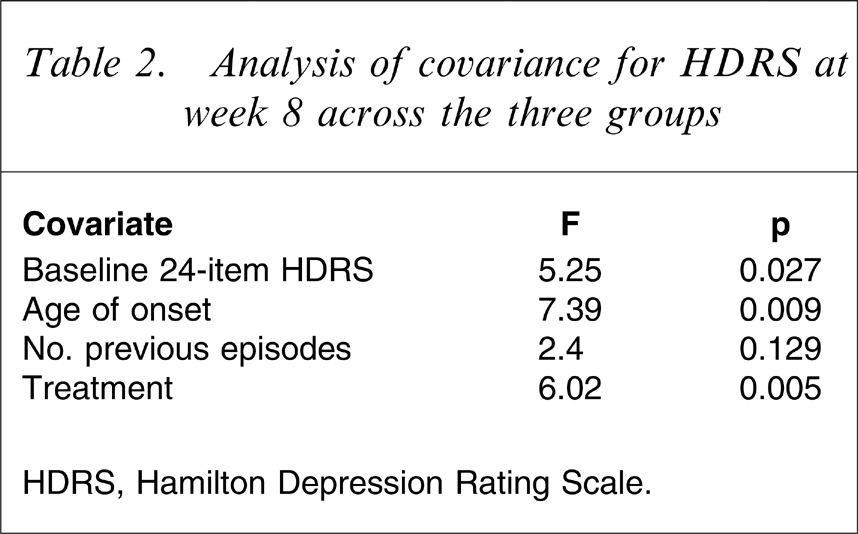
HDRS, Hamilton Depression Rating Scale.
ANCOVA comparing HDRS at week 8 in the fluoxetine group with the other groups and also the EPA group with the EPA + fluoxetine combination is shown in Table 3. The fluoxetine + EPA combination is significantly better than fluoxetine or EPA alone. Fluoxetine and EPA appear to be equally effective in controlling depressive symptoms.
Pairwise analysis of covarience for HDRS at week 8

EPA, eicosapentaenoic acid; HDRS, Hamilton Depression Rating Scale.
∗Treatment is significantly effective.
ANCOVA for HDRS at weeks 2, 4 and 6 showed that treatment had an effect on HDRS at week 4 and 6 (p = 0.016, 0.02 respectively) but not at week 2 (p = 0.113). Response rates (≥50% decrease in baseline HDRS) were 50%, 56% and 81% in the fluoxetine, EPA and combination groups, respectively.
Within-group analysis
Repeated measure analysis of variance was done to test for significant differences in depressive scores over time within each group. There was a significant effect of time within each group and also when all groups were combined, with a noticeable effect starting at week 2 (in all cases p < 0.05).
Adverse events
Sixteen adverse events were reported during the trial (Table 4). Fish aftertaste was reported by two patients in the EPA group and two patients in the combined group. In the EPA group it was limited to the first few days. Anxiety was reported by nine patients in the fluoxetine group, one patient in the EPA group and three patients in the combined group. It was limited to the first weeks for all subjects who reported it. Decreased appetite was reported by six patients in the fluoxetine group. Fisher's exact test showed that the frequency of anxiety and decreased appetite were different among groups (p = 0.009 and 0.002 respectively).
Reported adverse events during the trial
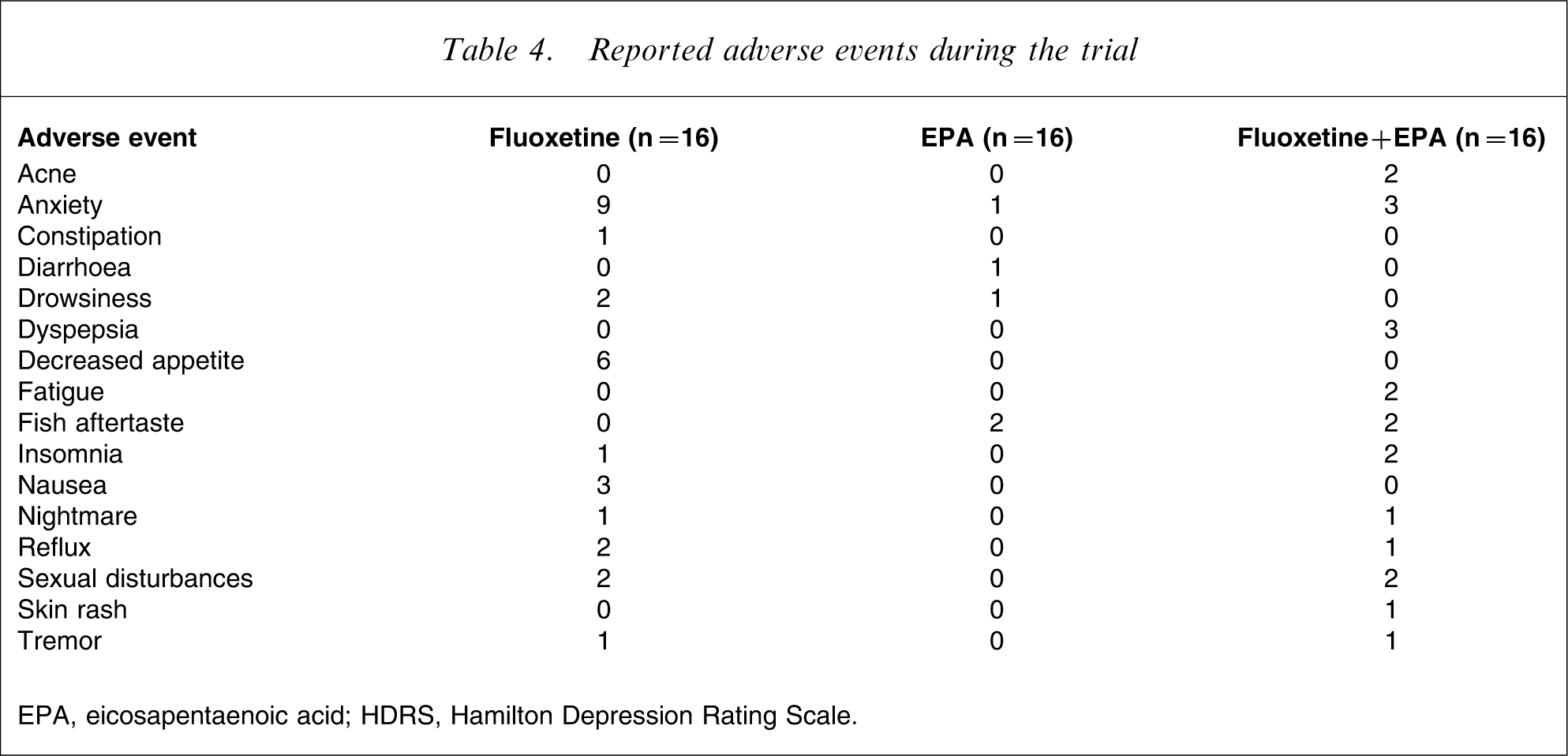
EPA, eicosapentaenoic acid; HDRS, Hamilton Depression Rating Scale.
Discussion
EPA + fluoxetine combination versus fluoxetine
The combination of fluoxetine and EPA was more effective than either of them alone from the fourth week of treatment. It has been reported that EPA or mixture of EPA and DHA at doses ranging from 1 to 9.6 g day–1 were more effective than placebo in three out of four trials assessing therapeutic effects of ω−3FAs as adjunctive therapy for unipolar major depressive episode [11–13], [15]. In contrast, 8 g DHA-enriched fish oil (600 mg EPA), which Silvers et al. used in their study, was not more effective than placebo [15]. In the other studies EPA was provided at a dose of ≥1 g day–1.
Puri et al. reported a case of 21-year old man with a 7 year history of unremitting depressive syndrome. Administration of EPA at a dose of 4 g day–1 as adjunctive treatment led to rapid improvement within 1 month. After 9 months all of the symptoms disappeared [20].
EPA was effective as adjunctive treatment in bipolar depression [21], [22], but 6 g ethyl-EPA was not effective as adjunctive treatment in bipolar and rapid-cycling bipolar disorder [23].
EPA versus fluoxetine
Although EPA alone was as effective as fluoxetine in controlling depressive symptoms, it is possible that neither EPA nor fluoxetine was more effective than placebo. Because their combination was more effective than either alone, the possibility of placebo effect is slight. A few studies have reported the effectiveness of ω−3FA as add-on therapy in childhood, post-partum period, pregnancy and bipolar depression. A randomized controlled trial suggested that ω−3FA (400 mg EPA and 200 mg DHA) monotherapy may have therapeutic benefits in childhood depression [24]. An open-label trial and one dose-ranging (without placebo) study of ω−3FAs suggested that ω−3FA monotherapy may be of therapeutic benefit in the post-partum period and in depression during pregnancy [25], [26]. But the sample size was too small in these studies. In another small open-label trial, ω−3FA monotherapy in bipolar depression had positive results [27]. Chiu et al. reported on a pregnant woman with recurrent depressive episode at 24 weeks of pregnancy who had improvement after ω−3FA monotherapy (4 g EPA and 2 g DHA per day) [28]. ω−3FA supplementation in healthy subjects was associated with improvement of mood status in a randomized controlled trial [29].
In contrast to the aforementioned studies, DHA monotherapy was not more effective than placebo at a dose of 2 g day–1 in unipolar major depressive episode [14]. A small open-label study failed to show any effects of ω−3FAs in preventing post-partum depression [30].
Possible mechanism of action of EPA
The impact of ω−3FAs in depression have been explained by several mechanisms [18]. Verlengia et al. observed marked differences between the effects of EPA and DHA on Jurkat cells. There were opposite effects in some aspects. This indicates that it is not correct to generalize the effects of n-3 fatty acids [31]. EPA and DHA had significant differences in the production of interferon-γ (IFN-γ). EPA caused a higher decrease in the production of IFN-γ [31]. Excessive secretion of cytokines has been proposed as the cause of depression [32], [33]. Cytokines, including IFN-γ, can potentially reduce serotonin levels by lowering the availability of its precursor (tryptophan) through activation of tryptophan-metabolizing enzyme. Increased activity of this enzyme may also contribute to monoaminergic disorders that are observed in depression in an indirect way [34].
Complex reciprocal interactions are observed between the serotonin system and arachidonic acid metabolism. Altered brain arachidonic acid metabolism has been implicated in unipolar disorder [35]. A study showed that EPA had a more pronounced effect on arachidonic acid-dependent processes compared to DHA [36].
In contrast to EPA, DHA is the main ω−3FA in the brain. It is involved in a variety of processes in neural cells [37]. Considering the differential and sometimes potentially opposite effects of EPA and DHA, the discrepancies among the studies assessing ω−3FAs can be explained by the absolute, as well as relative, amounts of EPA and DHA in supplements.
Conclusion
Although this study had some limitation including small sample size and lack of placebo group, the findings suggest that EPA is safe and effective as monotherapy as well as adjunctive treatment for unipolar major depressive episode. Because EPA is a dietary supplement it may be more acceptable to patients than antidepressants.
Considering the health benefits of ω−3FAs especially on cardiovascular disease and the fact that major depression is a risk factor for cardiovascular disease [38], EPA can be prescribed for patients with major depressive disorder both to control depressive symptoms and protect them from cardiovascular disease. Inflammatory cytokines that have an important role in atherogenesis [39] have been reported to increase in major depressive disorder [40] and potentially could be decreased by EPA [41].
Footnotes
Acknowledgements
This work was supported by Vice-Chancellor for Research, Tehran University of Medical Sciences, Tehran, Iran. Thanks are extended to Minami Nutrition, Belgium for supplying the EPA soft gels. We would also like to acknowledge the cooperation and help of Dr. V. Sharifi, Dr. E. Izadian, Dr. M. Arbabi and Mrs. M. Chamari.
