Abstract
Objective:
This paper aims to provide an overview on the nocebo effect, focusing on recognition — its phenomenology, at-risk demographic profiles, clinical situations and personality factors, as well as discriminating somatic symptoms in the general population from treatment-related adverse effects. Lastly, the paper addresses available evidence-based strategies for management and minimisation of the nocebo effect.
Method:
Data for this paper were identified by searching PubMed using the search terms "nocebo" and “nocebo effect”, augmented by a manual search of the references of the key papers and the related literature.
Results:
The nocebo effect refers to non-pharmacodynamic, harmful or undesirable effects occurring after inactive treatment, a phenomenon that also occurs in the context of active therapy. Known drivers include classical conditioning and negative expectations concerning treatment. Recent meta-analyses have reported a considerable prevalence, ranging from 18% in the symptomatic treatment of migraine, to more than 74% in multiple sclerosis. Recognition of the nocebo-driven adverse effects presents a challenge, especially because of its non-specific nature and the similarity to the active medication’s expected profile. Traits such as neuroticism, pessimism and type A personalities may predispose individuals to this phenomenon. Clinical management of the nocebo effect includes awareness and recognition, changing the manner of disclosure of potential drug-related adverse effects, shaping patients’ expectations and enhancing the treatment alliance.
Conclusion:
The nocebo effect is a common, clinically significant, yet covert driver of clinical outcomes. Increased awareness of its features, as well as knowledge of strategies on how to manage it, are fundamental so that clinicians can mitigate its impact on clinical practice.
Introduction
The placebo effect (from Latin “I will please”) has attracted much attention, and many studies have been conducted to elicit its prevalence, physiology, impact and management. There is, however, scant data and awareness of its “dark side” — the nocebo effect (from Latin “I will harm”), despite the fact that it may be a significant driver of clinical outcomes. The first description of the nocebo effect was by Kennedy in 1961, after noticing non-specific adverse reactions to drugs, which he hypothesised to have parallels to the placebo effect (Kennedy, 1961). A classic example of the nocebo phenomenon is illustrated by the experiment of Schweiger and Parducci (1981). They recruited a group of students and informed them that they would be subjected to a procedure — administration of electric current to their heads — that could induce headaches. Even though no electricity was administered, the majority of participants reported headache.
The nocebo effect refers to non-pharmacodynamic, harmful, unpleasant, or undesirable effects a person experiences after receiving an inactive treatment. Like the placebo effect, this additionally has the potential to impact on active therapy. Hahn (1997) expanded this concept, noting that the generation of negative symptoms follows their expectation and associated emotions, and may lead to undesirable effects. Expectation may in some situations be a more potent mediator of clinical response than the treatment itself (Chen et al., 2011). The nocebo effect may be a cause of adverse reactions and treatment discontinuation. For example, the nocebo effect has been linked to non-adherence to oral contraception. It is not always clear if non-specific side effects of many drugs are more common than with placebo, and such reactions are considered to be one of the main causes of treatment discontinuation (Grimes and Schulz, 2011). The aim of this paper is to synthesise the latest data on the nocebo effect, focusing on its prevalence and biological and psychological explanations, and to provide information that will help clinicians to recognise and manage this covert mediator of clinical outcomes.
Material and methods
Data for this paper were identified by searching PubMed using the search terms “nocebo” and “nocebo effect”, augmented by a manual search of the references of the key papers and the related literature.
Results
Prevalence
Data on the prevalence of the nocebo side effects are largely derived from the adverse reactions in the placebo arm of clinical trials. This provides only an estimate, since natural history comparison is not available. In phase one studies of healthy volunteers, the rate of adverse reactions on placebo was 7.9% (Sibille et al., 1998). A review of 109 double-blind, placebo-controlled studies indicated that around 19% of 1228 healthy participants reported adverse reactions to an inert substance (Rosenzweig et al., 1993). The clinical population and background symptomatology have a major influence on prevalence; in the placebo arms of two trials of methylphenidate and donepezil for cancer-related fatigue, 71% of the patients reported more than two side effects (De la Cruz et al., 2010). Recently, a series of meta-analysis has been conducted to estimate the frequency of nocebo effects in several disorders, using adverse effects and drop-out rate in the placebo arm of drug trials (Table 1). Nocebo responses ranged from 18% in the symptomatic treatment of migraine to more than 74% in multiple sclerosis. Treatment discontinuation due to side effects was up to 9.5% in fibromyalgia.
Meta-analyses of nocebo effects in the placebo arms of clinical trials.
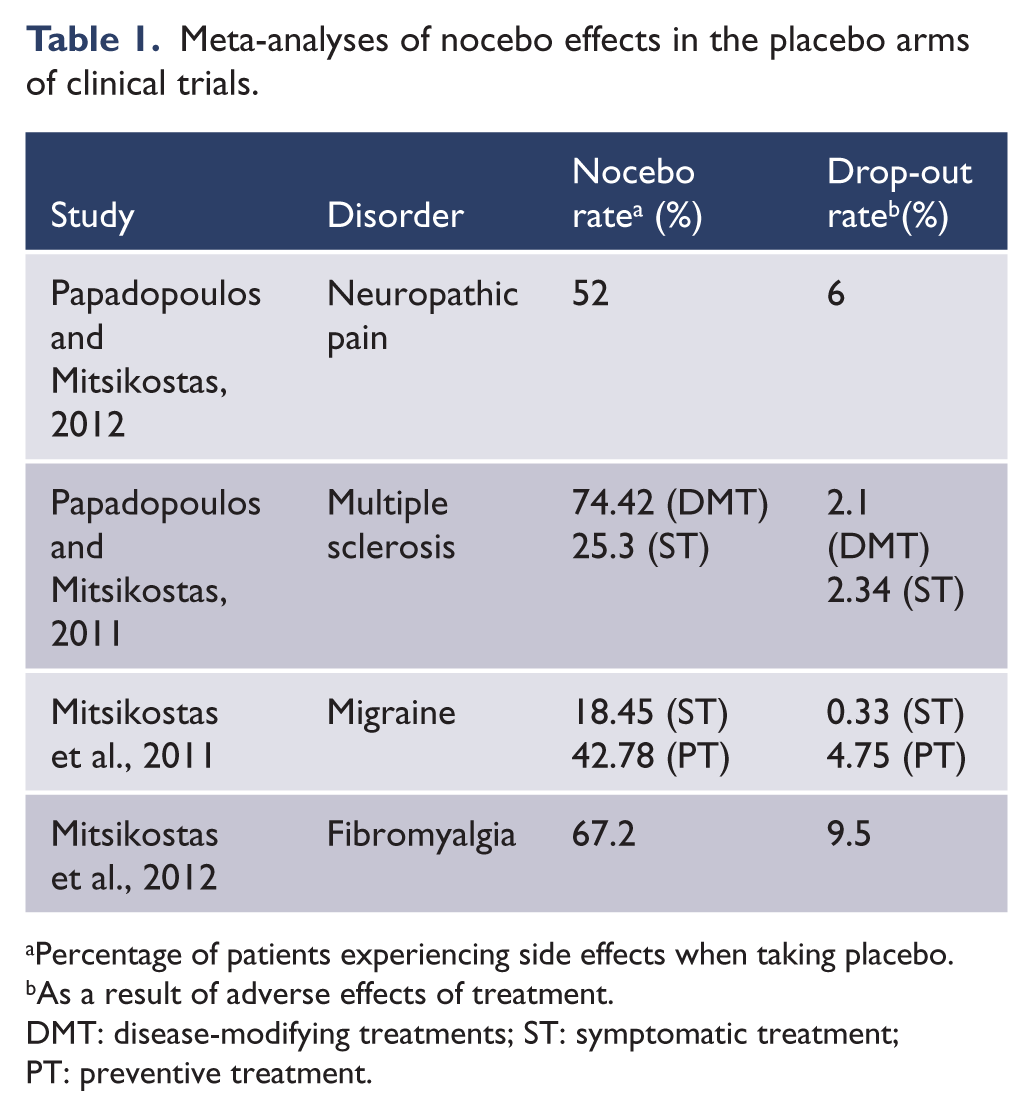
Percentage of patients experiencing side effects when taking placebo.
As a result of adverse effects of treatment.
DMT: disease-modifying treatments; ST: symptomatic treatment; PT: preventive treatment.
A study of 600 patients with a history of adverse effects to different medications was conducted (Liccardi et al., 2004). Patients were first challenged with an inert substance, and then with two different concentrations of an active substance that was different from the drug that allegedly induced side effects. The induction of symptoms by the inert agent ranged between 24 and 30% of patients. Male subjects reported fewer side effects (19%) than females (30%). However, a more recent study, using a similar method, reported only a 3% nocebo rate (Lombardi et al., 2008). The pattern of gender effects has been replicated, with females suffering from panic disorder being more likely than males to develop increased anxiety following administration of an inert substance, a finding also seen in depression (Casper et al., 2001; Strohle, 2000). Similarly, medication side effects may be more frequent amongst females (Zopf et al., 2008).
Aetiology
In part because of a lack of awareness of the nocebo effect, its covert nature, as well as ethical issues in studying unnecessary worsening of symptoms, there is less knowledge about the mechanisms of the nocebo response compared to the placebo effect. Most studies have been carried out in the field of pain. Two dominant psychological mechanisms are thought to be involved in the nocebo response — expectation and classical conditioning (Benedetti et al., 2003).
Expectation
A paradigmatic example of how expectations are involved in a nocebo effect is the experiment of sham current causing headaches conducted by Schweiger and Parducci (1981) mentioned above. In another informative study, participants were told that they would receive either an inert or herbal substance, even though all participants ingested an inactive compound. Fully 89% reported it induced at least one symptom; 11% reported only nocebo symptoms, while 28% reported both placebo and nocebo effects (Link et al., 2006).
In a study using the tourniquet technique to induce ischaemic pain, providing suggestions that the pain would increase led to diminished pain tolerance. Similarly, expectation of nausea in cancer patients undergoing chemotherapy treatment predicted its development (Colagiuri and Zachariae, 2010). In a study examining the impact of verbal suggestions on the induction of pruritus and pain, both were promoted by the person’s expectation (Van Laarhoven et al., 2011).
Treatment outcomes are influenced by whether a therapy is overt or hidden. Among people treated with morphine to reduce pain after surgery, delayed rises in pain intensity were reported if they were unaware that the treatment had been stopped, while unconcealed treatment suspension triggered symptom recurrence. Similarly, with diazepam treatment for anxiety, open interruption of diazepam resulted in worsening of anxiety, while concealed suspension did not (Colloca et al., 2004). In aggregate, these studies support expectancy as a key operative pathway of the nocebo effect.
Classical conditioning
Classical conditioning is another pathway to the nocebo effect. For example, patients who received a lemon-lime beverage (conditioned stimulus) concurrent with administration of chemotherapy reported significantly more nausea than control subjects when the conditioned stimulus was presented again without the chemotherapy agents (Bovbjerg et al., 1992). The finding that both expectations and prior conditioning are relevant in the development of nocebo-induced hyperalgaesia has been replicated, and it appears that the duration of the conditioning positively influences the perpetuation of the nocebo effect (Colloca et al., 2008; Colloca et al., 2010). Clinically, awareness of past adverse experiences should warn the clinician of the probability of nocebo effects intruding into treatment. The genders may differ in terms of the relative role of expectation and conditioning. In men, the nocebo response may be driven more by expectations, while conditioning appears more salient in women (Klosterhalfen et al., 2009).
Neurobiology of the nocebo phenomenon
Changes in the hypothalamic-pituitary-adrenal axis, including rises in adrenocorticotrophic hormone and cortisol, have been linked to pain perception and expectation (Benedetti et al., 2006; Johansen et al., 2003). Cholecystokinin pathways may additionally mediate the link between anxiety and pain in nocebo-induced hyperalgaesia (Benedetti et al., 1997; Benedetti et al., 2006). Neuroimaging studies have examined this phenomenon. A positron emission tomography study reported changes in μ-opioid and dopamine D2/D3 neurotransmission with the nocebo effect, and functional magnetic resonance imaging studies have suggested the involvement of specific brain structures, such as anterior cingulate, insula and the prefrontal cortex (Kong et al., 2008; Koyama et al., 2005; Scott et al., 2008).
Recognition
Before management can commence, recognition of the at-risk profile is necessary. Individuals who have high levels of anxiety, somatisation or hypochondriasis, histories of adverse events, trauma or abuse or unsatisfactory experiences with medical care, signs of personality disorder and conflictual relationships with authority figures or systems of care need to be identified. These profiles will be examined in this section.
Recognising the nocebo response
Characteristically, nocebo-driven adverse events are non-specific (i.e. not necessarily directly linked to the drugs’ pharmacological profile, although knowledge of the potential side effects may drive expectation-driven events), with drowsiness, asthenia, dizziness, nausea and vomiting being the most frequent (Rosenzweig et al., 1993). One consequence of expectation is that nocebo-driven side effects tend to resemble the ones known to be part of a medication’s profile or the expected symptoms of the disorder, adding opacity to interpretation (Mitsikostas et al., 2012; Rief et al., 2009; Amanzio et al., 2009).
Somatic symptoms
In order to contextualise adverse events, it needs to be noted that physical symptoms not attributable to any disease (somatisation) are frequent in the general population, with prevalence rates of up to 81.6%. The most frequent symptoms are headache, back and joint pain, intolerance to food, and sexual dysfunction. Female gender, old age, lower levels of education and socio-economic status, as well as living in rural regions, are all linked to such symptoms (Hiller et al., 2006; Rief et al., 2001). Some adverse effects like headache, fatigue or back pain may even be more frequent in the general population than in trial reports (Rief et al., 2006). Moreover, subjects may mislabel their symptoms according to prior experiences and expectations, environmental influences, knowledge about the bodily sensations, and different emotional states (Mechanic, 1972). The proneness to interpret sensations as more distressing, known as “amplification”, is associated with anxiety, depressive and dysphoric mood (Barsky et al., 1988; Uhlenhuth et al., 1998). However, not all studies have found a clear link between the nocebo effects and anxiety (Link et al., 2006). It is plausible that the nocebo effect with an active treatment might overlap with unexpected and unusually prominent side effects reported by patients with a genuine sensitivity to a particular drug, for example, those with significant genetically determined slow metabolism.
Personality
Some studies have addressed the role of personality as a mediator of the nocebo response and adverse event reporting. Traits such as neuroticism, pessimism and type A personalities may increase the risks for such phenomena (Table 2). Linked to personality, one’s prior experiences profoundly mould expectation and outcomes. People who have had prior adverse events or dissatisfaction with medical services are more likely to be guarded, distrustful and expectant of pattern repetition. More broadly, those people who have had adverse early life experiences or trauma histories tend to be more vulnerable to distrust in general and to experience feelings of rejection or abandonment of care. When placed in a situation of need, they may manifest a hostile-dependent pattern of interaction with caregivers, asking for care, but at some level expecting a repeat of prior patterns of interaction. As an exemplar, haemodialysis patients who manifested conflictual relationships with care had developmental histories in which there were major issues with dependency/independency conflicts and more adverse current life circumstances (Procci, 1981).
The role of personality in the nocebo effect.
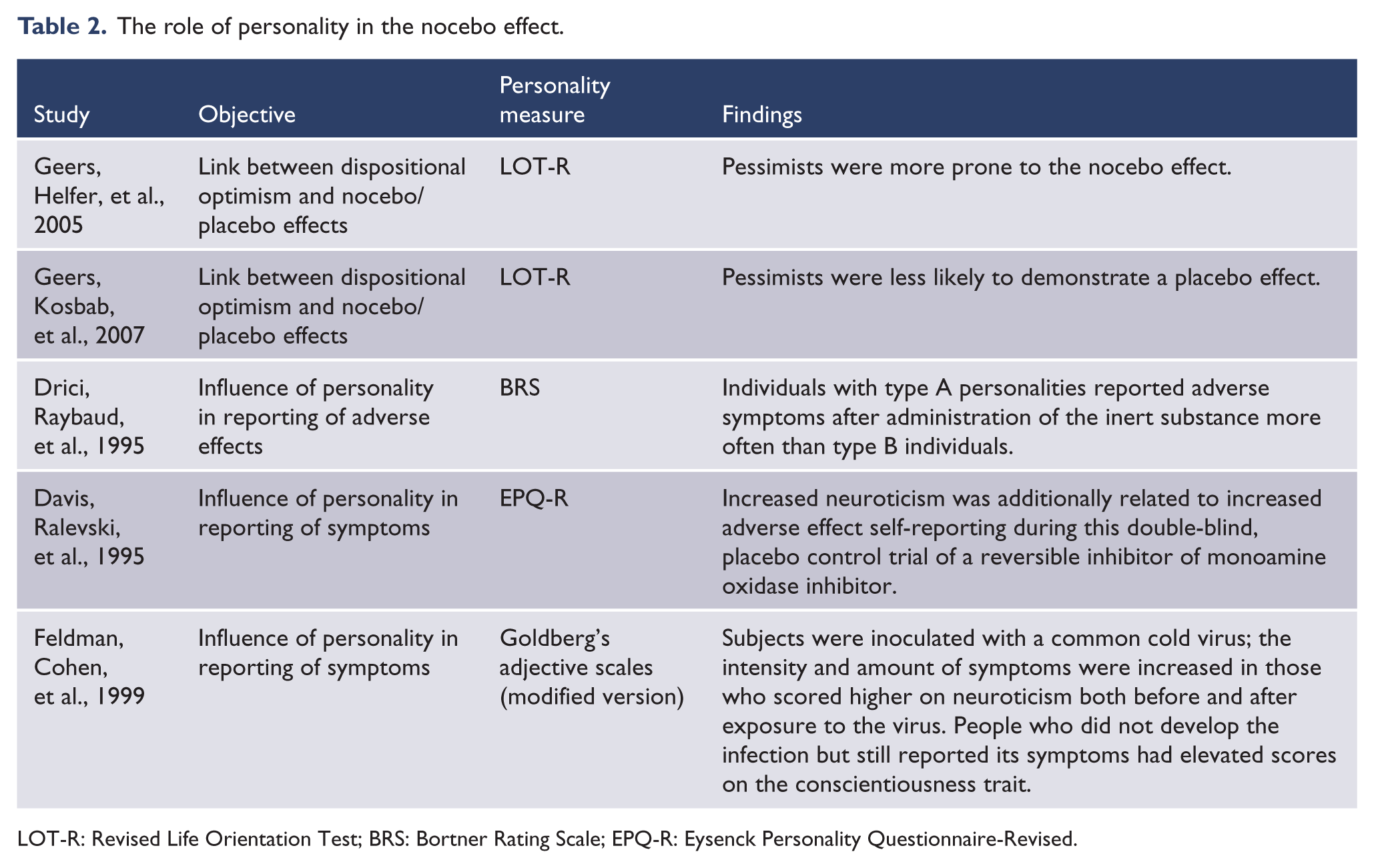
LOT-R: Revised Life Orientation Test; BRS: Bortner Rating Scale; EPQ-R: Eysenck Personality Questionnaire-Revised.
Involuntary treatment and perceived coercive treatment settings are linked to a lower probability of placebo effects and an increased likelihood of nocebo effects, resulting in a decreased effectiveness of treatment (Meynen and Swaab, 2011).
Clinical management
Informed consent
A major factor in the development of the nocebo effect is the manner of disclosure of potential medication adverse effects. In a study on the sexual adverse reactions of finasteride — a drug used to treat benign prostate hyperplasia and androgenetic alopecia — 44% of people who were briefed on the potential occurrence of sexual adverse effects reported them, compared to only 15% of the subjects who were not told (Mondaini et al., 2007). Similar results using different drugs for different disorders are reported (Myers et al., 1987; Silvestri et al., 2003). Thus, the clinician needs to consider how to present information to the patient and that this information can shape the person’s expectations. In a litigious environment, doctors may over-emphasise the risks of treatment for pre-emptive medicolegal reasons, which can increase the risk of inadvertently driving the nocebo effect. In clinical care, this can compromise therapy, and in the context of informed consent for clinical trials, can drive the rate of adverse event reporting.
Presenting information to patients
Physicians as well as patients undertake risk-benefit assessments in decision making. How clinicians present information about risks and benefits of therapies therefore markedly affects the perceptions of the patient, and thereby adherence, engagement and treatment choices. Physicians can transmit their messages to patients in different ways. While not refraining from delivering the necessary information, it may be preferable to bring attention to benefits rather than losses (Thaler and Sunstein, 2008). This method is called “framing” and it has been suggested to guide patient’s approach and choices regarding treatment and prevention of disease (for a review on ‘framing’, see Edwards et al., 2001). When discussing side effects, there is some evidence that if doctors make explicit the difference between the probabilities of patients feeling adverse symptoms and their actual prevalence in a general population, they may become less worried about their occurrence (Zikmund-Fisher et al., 2008). One study of influenza vaccination reported lower absentee rates and adverse effects if the clinical explanation focused on the advantages (i.e. framed as the percentage of people who will remain well and have no side effects) instead of the disadvantages (i.e. patients who will get influenza and develop side effects) (O’Connor et al., 1996).
Shaping expectations
As an exemplar, it is common for physicians to warn patients of the painful nature of an impending procedure, such as the administration of a local anaesthesia. However, this may have counterproductive effects, as one study reported lower scores for pain if the message was focused on the injection’s beneficial effects, rather than it being painful (Varelmann et al., 2010).
The treatment alliance and way the medication is prescribed are also factors that may assist the clinician to reduce the likelihood of the nocebo effect. Engaging in a collaborative model of shared decision making is helpful, so that the person has a sense of a degree of both control and ownership and of the decision-making process and the therapeutic decisions. This enhancement of autonomy increases perceived ownership of the conjoint plan, enhances the therapeutic alliance and makes adherence and engagement with treatment more likely (Berk et al., 2004; Berk et al., 2010).
Attitude
Di Blasi conducted a systematic review on the effects of context in the effectiveness of treatment. Doctors who were more encouraging, kind and affectionate and provided a clear diagnosis seemed to be more effective in, for instance, reducing perceived pain levels and time to improvement than physicians who adopted a more rigid attitude and did not offer any consolation (Di Blasi et al., 2001).
Mitigating reaction to side effects
A “two-step strategy” has been proposed to minimise the nocebo phenomenon. Especially in situations where long-term medication is envisaged, the person has a clinical profile that puts him or her at risk for the nocebo effect, where there are few alternatives to the chosen therapy, and where time pressure is not acute (e.g. lithium for maintenance of bipolar disorder), the person should first be started on a dosage of medication that is overtly under what is considered to be therapeutic. The use of a pill cutter to shave chips off a tablet may be a more explicit demonstration of safety than use of a lower dose formulation. Following this, small increases should follow until the desirable dose is achieved. If nonspecific adverse effects do occur, they should be thoroughly discussed with the patient, and the attending physician should take time to explain the benign or time-limited nature of such reactions (Barsky, 2002; Rogers, 2003). However, in some cases, the development of adverse reactions will end up being inevitable. When it happens, it may be more helpful and pragmatic to assist patients to cope, rather than attempting to suppress them. A discussion of acceptance of adverse events as a price of health can enhance a sense of acceptance of the treatment choice. Rogers also suggested that telling patients these reactions are benign and mean that the medicine is “in their system” may be helpful (Rogers, 2003).
Conclusions
The nocebo effect is a significant driver of clinical outcomes. Clinicians need to be aware of its presence and influence, and be able to recognise the phenomenon. There are strategies on how to manage and minimise the nocebo effect, including how to optimally present information to patients, shape their expectations and manage emergent side effects. Only by being aware of potholes can one steer around them.
Footnotes
Acknowledgements
All the participating authors have been listed and both authors contributed equally to this review.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of interest
MB has received grant/research support from the National Institutes of Health (NIH), Simons Autism Foundation, Cancer Council of Victoria, Stanley Medical Research Foundation, MBF, National Health and Medical Research Council (NHMRC), Beyond Blue, Geelong Medical Research Foundation, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Organon, Novartis, Mayne Pharma and Servier, has been a speaker for Astra Zeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck, Merck, Pfizer, Sanofi Synthelabo, Servier, Solvay and Wyeth, and served as a consultant to Astra Zeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck and Servier. JDF has no potential conflicts of interest.
