Abstract
Objective:
Many patients with depression respond or remit with current treatments, but often experience persistent distress, in part because they perceive that they have not returned to their normal or premorbid state. Some continue to have a lack of subjective psychological well-being and positive affect following treatment. It would be useful to measure these deficits and explore whether interventions can improve them. Currently, no clinically useful scale has been developed to measure positive affect. To fill this gap, we developed the Clinical Positive Affect Scale (CPAS).
Method:
The purpose of this study is to describe the development and validation of the CPAS, a 16-item self-report measurement of self-perceived affective and cognitive correlates of positive affect, in a sample of 300 college students.
Results:
A principal component analysis with varimax rotation showed one major factor of positive affect, with all items revealing high loadings (≥ 0.65) on the single factor. The CPAS also demonstrated good internal consistency (α = 0.97) and strong part-whole correlations. Finally, the CPAS revealed some degree of divergent validity through moderately strong negative correlations with validated measures of depression, anxiety and drug abuse.
Conclusions:
This study supports the validity of the CPAS, which may help clinicians and researchers to assess patients’ current self-perceived levels of hedonic capacity and enthusiasm for life.
Introduction
While antidepressant and psychosocial treatments are effective for major depressive disorder (MDD), the absence of depressive symptoms is not necessarily the presence of psychological well-being (Keyes, 2002; Ryff et al., 2006; Singer et al., 1998). Specifically, when patients respond or remit with treatments, they often continue to experience residual symptoms (Nierenberg et al., 2010), such as lack of motivation, low energy or anhedonia, associated with a lack of subjective well-being.
Psychiatry has tended to focus on symptoms and syndromes. In contrast, the psychological literature has focused on an expanded multidimensional perspective of mood with two dominant factors: positive affect, which functions as the primary characteristic of well-being, and negative affect (Ryff et al., 2004; Watson and Tellegen, 1985; Watson et al., 1988) that can be directly related to distress. These two dimensions are not simply opposites, as the everyday use of the terms imply, but, instead, are independent constructs (Watson and Tellegen, 1985). While positive affect refers to enthusiasm, motivation, hedonic capacity and excitement, negative affect describes distress, dysphoria and fear (Watson and Tellegen, 1985).
According to structural models, negative affect correlates with both anxiety and depressive symptoms, while low positive affect significantly contributes to depression only (Brown, 2007; Brown and Barlow, 2009; Jolly et al., 1994). Despite such evidence that positive affect is strongly related to depression, the literature supports the notion that positive affect and depression are indeed distinct constructs. For instance, low positive affect is not the sole predictor of depression; integrative models suggest that a combination of negative affect, depressive cognitions and low positive affect tend to predict depressive symptoms (Jolly et al., 1994). Further, the depressive domain is conceptualized as including aspects other than low positive affect, such as low self-esteem and hopelessness (Brown et al., 1997; Lovibund and Lovibund, 1995). Clinical observation also illustrates that treatment of depression can lead to a decrease in negative affect without a concomitant increase in positive affect. Clinically, patients frequently report that their depression (negative affect) is improved but that they don’t feel that they are back to 100% of their pre-depressed self (lack of positive affect). In sum, both empirical evidence and clinical observations suggest that positive affect is a distinct, albeit related, construct from depression.
Well-Being Therapy (WBT) addresses the gap between positive and negative affect by inverting the usual focus of cognitive behavioral therapy (CBT) for depression (Fava et al., 1998a, 1998b). CBT focuses on correcting dysfunctional thoughts associated with negative events and dysphoria. In contrast, WBT addresses cognitive interruptions of hedonic experience to enable people to experience pleasure. WBT has been found to be equally effective as traditional CBT in symptom reduction for those with residual depressive symptoms, and led to greater improvement in depressive symptoms (Fava et al., 1998a). For patients with recurrent depression, WBT resulted in significantly fewer symptoms and lower rates of relapse compared to CBT (Fava et al., 1998b). Remarkably, WBT was given for ten 30-minute sessions every other week and the protective effect of WBT lasted for up to 6 years.
To the best of our knowledge, no scales that measure perceived well-being or positive affect have been developed for use in a clinical setting. The Psychological Well-Being (PWB) scale comprises six specific subscales that measure positive relations with others, autonomy, environmental mastery, self-acceptance, purpose in life, and personal growth (Ryff, 1989). However, the PWB scale primarily measures eudaimonic well-being (i.e. human potential and functioning in life; Ryff et al., 2003), not the more hedonic components of well-being, such as happiness, hope, and confidence. In addition, the PWB does not use a definitive metric or time frame, which may lead to ambiguous answers. Finally, the PWB scales are lengthy and therefore burdensome for patients and clinicians alike. The WHO (Five) Well-Being Index (WHO-5), a five-item multidimensional measure of psychological well-being, protective factors, and subjective quality of life, may be too limited in scope to adequately assess an individual’s well-being and has been validated predominantly as a screening measure for depression (Bech et al., 2003; Bonsignore et al., 2001; Primack, 2003).
The Positive and Negative Affect Scales (PANAS), with uncorrelated measures of positive and negative affect (Watson et al., 1988), is used widely as a research tool. While suggestions have been made to use the PANAS clinically (Crawford and Henry, 2004), we are not aware of any migration of the PANAS into the clinic. To the best of our knowledge, there is also no normative PANAS data available from large non-clinical populations in the United States beyond that of the initial validation study (Watson et al., 1988). Administration of the PANAS permits clinicians to change time frames (Watson et al., 1988), a format that increases flexibility but also introduces variability and may potentially confound comparisons across individual patients and settings. Further, interpreting changes in the positive affect subscale of the PANAS requires additional subscale calculations (i.e. joy, activation and interest; Egloff et al., 2003) that may be too burdensome for routine clinical care. By using single-word items, the PANAS also may lack sufficient detail and specificity necessary to accurately assess levels of positive affect (Nemanick and Munz, 1994). In sum, while the PANAS is a well-validated indicator of negative and positive affect, its variable time frames, required subscale calculations and single-word format leave ample room for a brief, easily administered, accurate measure of positive affect for standardized use in clinical settings.
To fill the need for a brief measure of hedonic positive affect that helps distinguish positive affect from depression for clinicians and researchers, we developed the Clinical Positive Affect Scale (CPAS), a self-report scale that assesses the self-perceived affective and cognitive hedonic correlates of positive affect in the past week. Items of the CPAS capture the enthusiasm (e.g. ‘Feel enthusiastic’), excitement (‘Get excited about something pleasurable’), and hedonic capacity (‘Enjoy activities’, ‘Have fun’) characteristic of positive affect, rather than core symptoms of depression. The CPAS was developed out of clinical observations that many depressed patients who had responded to treatment continued to experience distress because treatment had failed to bring them back to what they would consider their normal level of hedonic capacity and enthusiasm for life. In contrast to the PANAS, the ‘past week’ time frame of the CPAS is standardized and does not vary across administrations.
The CPAS was administered in a recent trial of mindfulness-based cognitive therapy (MBCT) for bipolar disorder (Deckersbach et al., 2012). Analyses of pre- and post-treatment CPAS scores revealed a large effect size and a trend towards significant increase in CPAS scores over the course of treatment. These findings suggest that the CPAS effectively captured participants’ increased positive affect that resulted from mindfulness-based exercises.
Given prior administration of the CPAS to a clinical sample, the purpose of the present study was to provide data from a non-clinical population and validate the CPAS for face and construct validity in a large student sample.
Materials and methods
Participants
The present study was part of a larger ongoing study investigating a suicide prevention screening program for college students at multiple Boston-area universities. The data included in the present study were collected at one mid-size college located in Boston, and encompasses a total of 300 (190 female) college students (age in years: mean 19.5, SD 1.7). These participants were recruited through flyers and word of mouth. Out of these students, 32.4% were freshmen, 26.1% were sophomores, 26.1% were juniors and 11.4% were seniors, and 4.0% either identified as ‘other’ or chose not to respond. Ethnicity was self-identified as white/not of Hispanic origin (77.3%), Asian or Pacific Islander (7.7%), Hispanic (5.3%), black, not of Hispanic origin (2.7%), American Indian or Alaskan native (0.7%) and other (5.7%).
Assessments
The assessments analyzed for the present study are a subset of the standard packet of questionnaires used for the aforementioned college suicide prevention program. These instruments include the following items.
Clinical Positive Scale (CPAS)
The CPAS is a 16-item self-rated measure of positive affect. For the preceding week, respondents are asked to rate to which degree they were getting excited about something pleasurable, were looking forward to things, anticipated having a good time or feel good about the things they were able to get done (Table 1). Each item is rated on a 0 (not at all; completely absent) to 4 (same as best or normal self) scale, with total scores ranging from 0 to 64. Higher scores represent more well-being/positive affect. The CPAS was originally developed with 16 items, all of which were administered in the present study.
Items and factor loadings for the Clinical Positive Affect Scale.

Depression
The Beck Depression Inventory (BDI) is a 21-item self-rated assessment of depression that measures both psychological and physical symptoms (Beck, 1961). Prior research has supported the reliability and validity of the BDI for use in various populations (Beck et al., 1961; Bumberry et al., 1978; Jegede, 1976; Lester and Beck, 1977). Previous research using the BDI proposes a cut-off score of 16 to screen for depression (Beck et al., 1961).
Anxiety
The Beck Anxiety Inventory (BAI) is a 21-item self-report measure of anxiety (Beck et al., 1988). Scores of all items are summed to yield a single score of anxiety. Previous research using the BAI recommends that scores of 0–9 represent normal anxiety; 10–18 indicate mild to moderate anxiety; 19–29 suggest moderate to severe anxiety; and 30–63 should be interpreted as severe anxiety (Osman et al., 1993), with a cut-off score of 16 to screen for anxiety (Kunik et al., 2007).
Drug abuse
The Drug Abuse Screening Test (DAST) is a 28-item self-report instrument for clinical screening and treatment evaluation research (Skinner, 1982). Each item is answered by endorsing ‘Yes’ or ‘No’, and is combined in a total score that yields a quantitative index of problems related to drug misuse. A higher total score is indicative of more problems of drug abuse. The DAST is correlated with the frequency of drug use during the past 12 months (Gavin et al., 1989; Staley and el-Guebaly, 1990).
Procedures
All study procedures were approved by the Partners Institutional Review Board. Study participants received a description of the study aims and screening procedures and provided written informed consent prior to participating in the research. Study participants completed a self-report questionnaire packet that contained various measures assessing mental health symptoms, including the BDI, BAI, DAST and CPAS. The CPAS was included for validation purposes. Upon the return of completed packets, a research assistant scored the BDI while the student was present to determine whether a further diagnostic evaluation with the mood module of the Structured Clinical Interview for DSM-IV (SCID) was indicated (Spitzer et al., 2002). The SCID was administered if BDI total score ≥ 13 and/or BDI suicide item #9 score ≥ 1. Subjects with a BDI score ≥ 13 (
Results
Factor structure and reliability
First, we explored the factor structure of the CPAS (rather than conducting a confirmatory analyses), given that this was the first evaluation of this measure, and we sought to consider the most appropriate model that fits the data. We conducted a principal component analysis (PCA) with varimax rotation, which yielded one major factor that explained 68.4% of the total variance with each of the additional factors explaining less than 5.5% of the variance. The one-factor structure was determined in accordance with the Kaiser criterion, with the PCA resulting in a single eigenvalue greater than one (10.85). A scree plot also suggested retaining one factor, with remaining factors failing to explain additional variance. The revealed dominant single factor structure rendered employing additional methods (e.g. principal axis factoring) to further explore this one-factor structure unnecessary. Replications of the PCA among subgroups (e.g. men, women, Caucasian, non- Caucasian) suggested the same single-factor structure as evidenced in the overall sample. Among the total sample, all 16 items had loadings of 0.65 or higher on the first factor (Table 1), with 15 items exhibiting a loading above 0.77.
Second, we examined the internal consistency of the CPAS. Internal consistency and part-whole correlations showed that the 16-item CPAS demonstrated good internal consistency (Cronbach’s α = 0.97). Part-whole correlations (i.e. correlations of each item with the CPAS sum score without that particular item) ranged from
Construct validity
If the CPAS is a valid measure of positive affect, scores should be negatively correlated with measures of divergent validity. We tested the divergent validity of the CPAS by examining the correlations with the BDI, BAI and DAST. Specifically, the CPAS had moderately strong negative correlations with the BDI (
Criterion-related validity
Previous findings suggest that low positive affect should, to some extent, distinguish between depressed and non-depressed individuals (Dyck et al., 1994). Participants with BDI ≥ 16 (
Distribution of CPAS scores
Table 2 shows the descriptive statistics for all total scale scores included in the present analyses (i.e. CPAS, BDI, BAI, DAST) in the entire sample, and as a function of sex and race. There were no significant differences in mean total CPAS scores according to sex (males:
Descriptive statistics of CPAS, BDI, BAI and DAST.
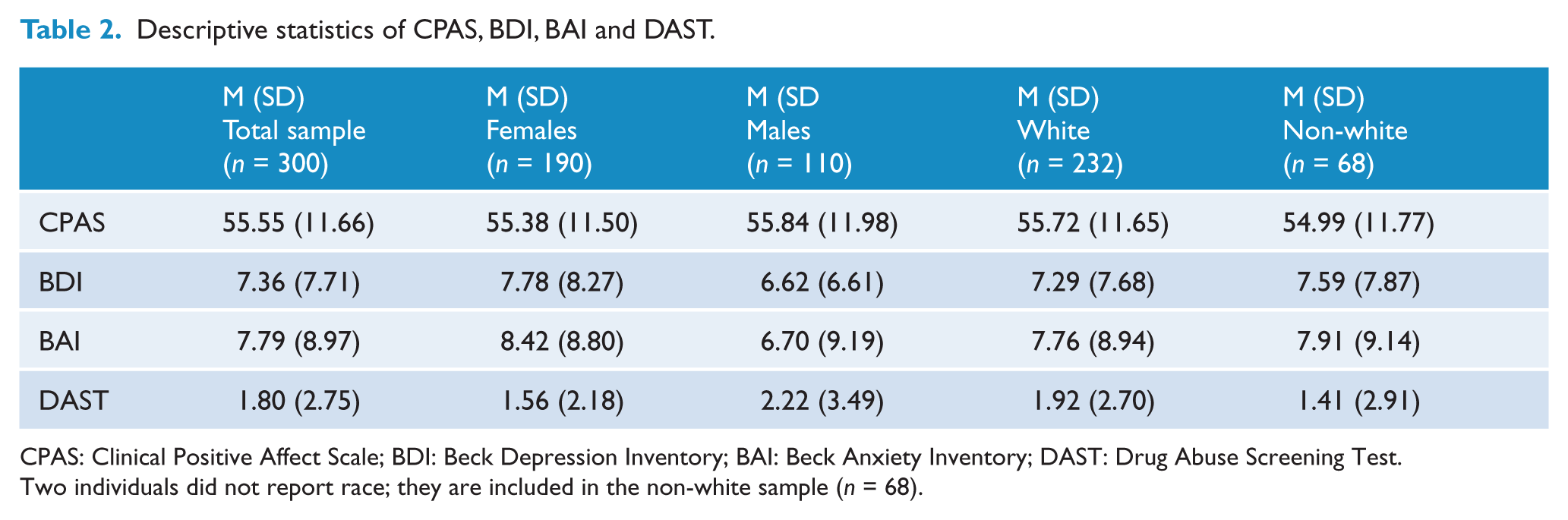
CPAS: Clinical Positive Affect Scale; BDI: Beck Depression Inventory; BAI: Beck Anxiety Inventory; DAST: Drug Abuse Screening Test.
Two individuals did not report race; they are included in the non-white sample (
Table 3 illustrates the distribution of CPAS total scores. Overall, the frequency distribution of the CPAS was left-skewed with a tendency for most students being close to a maximum score (i.e. close to their normal or best self).
Frequency distribution of CPAS total scores.
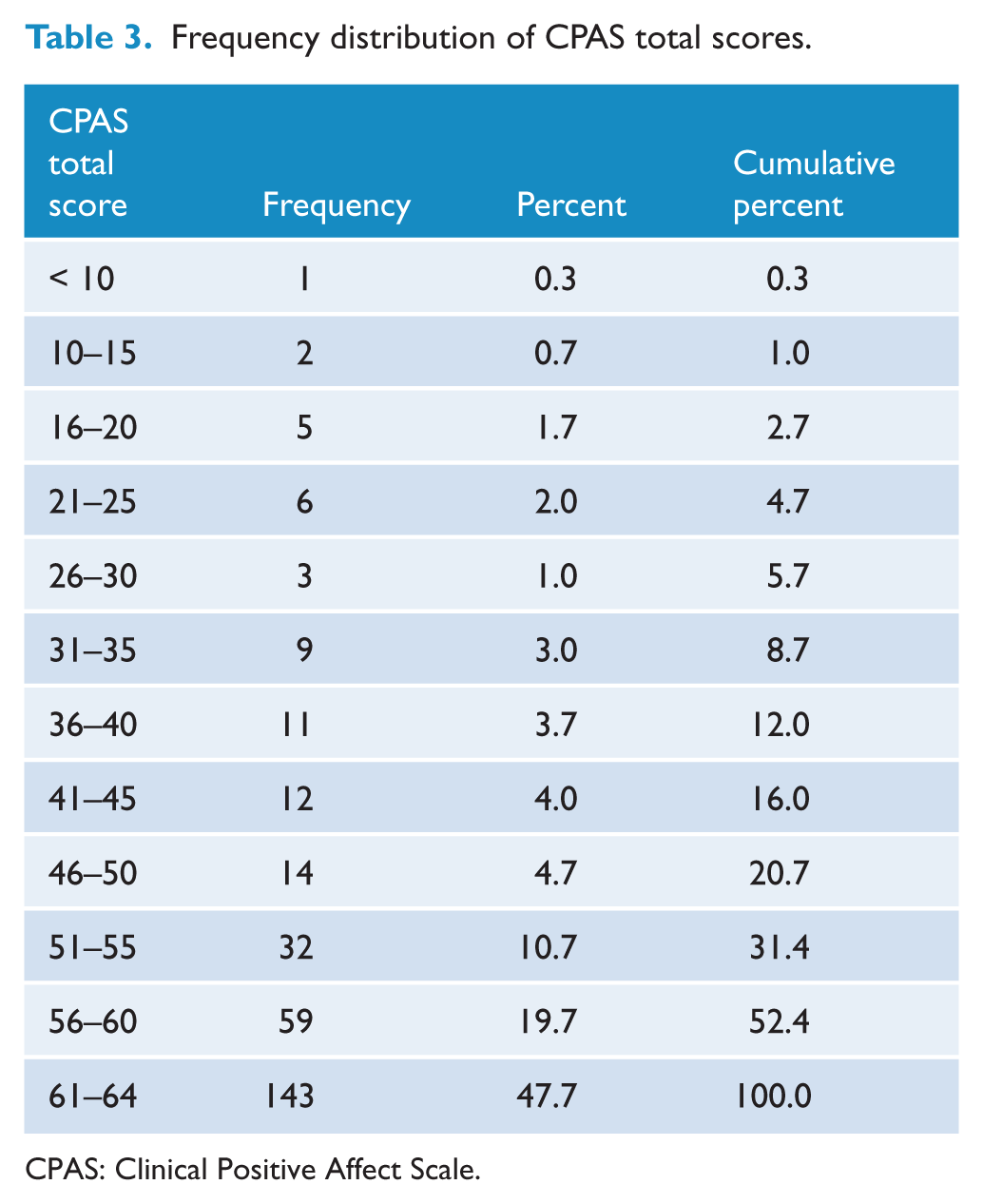
CPAS: Clinical Positive Affect Scale.
Discussion
The purpose of this study was to assess the psychometric properties of the CPAS, which measures positive affect over the past week. Results indicated a homogeneous factor structure of the CPAS, with each of the scale’s 16 items demonstrating high loadings in this single-factor structure. The discrepancy between the ‘Want to get things don’ item loading (0.65) and other item loadings may potentially be informed by contemporary well-being models. According to an ESS Well-Being Module (for a review see: Huppert et al., 2009), two dimensions of well-being exist: positive feeling (i.e. more transient state of well-being) and positive functioning (i.e. more sustainable well-being). It could be argued that the majority of CPAS items primarily assess the feeling dimension (e.g. ‘Feel optimistic’, ‘Anticipate having a good time’, ‘Feel confident’), while the ‘Want to get things done’ item reflects the functioning dimension of well-being. It is possible that, if additional items targeting the functioning dimension of well-being were included in the CPAS, a correlated two-dimensional factor structure may have emerged. Conversely, it is also possible that wanting to get things done may differ inherently from the positive well-being items, and better reflect stress or negativity associated with demanding tasks. However, given that this item may indeed reflect a feeling, rather than functionality (e.g. ‘Get things done’), as well as the acceptably high loading of the ‘Want to get things done’ item, we chose to preserve all 16 items of the original scale. Future psychometric work with the CPAS should continue to examine the ‘Want to get things done’ item in order to evaluate its appropriateness for inclusion in later iterations of the scale.
The good internal consistency of the CPAS (Cronbach’s α = 0.97) was demonstrated by large positive part-whole correlations, which revealed strong associations between each individual item and the total CPAS score. These results, strongly suggestive of reliability, provided further support for upholding all items and this one-factor structure.
Present findings indicate that non-depressed individuals tend to score higher on the CPAS than those with depression. Given evidence that low positive affect contributes significantly to depression (Brown, 2007; Brown and Barlow, 2009; Jolly et al., 1994), it is expected that within a non-clinical population, most individuals would obtain high scores; in line with such expectations, 47.7% of participants in the present study scored in the 61–64 (a maximum score) range (see Table 3). However, sizeable variation (e.g. 19.7% in the 56–60 range, 10.7% in the 51–55 range) within the approximately 80% of individuals who scored toward the upper end of the CPAS score distribution (51–64; see Table 3) was present. In other words, despite both a homogeneous sample and a skewed distribution within a homogeneous sample, ample variation exists at the upper end of the distribution. These findings, in conjunction with evidence of high reliability, indicate that the CPAS discriminates between high and extremely high levels of positive affect. Results suggest that while non-depressed individuals tend to score highly on the CPAS, there is some variation in positive affect among a non-clinical sample. However, the psychometric properties of the CPAS require further investigation in (potentially) more heterogeneous normal and psychiatric populations.
The present investigation also supported the construct validity of the CPAS. Given evidence that high positive affect reliably indicates the absence of distress, we expected that total scores on the CPAS would display a negative relationship with scores on measures of depression (BDI). We also expected total scores on the CPAS to negatively correlate with measures of constructs related to anxiety (BAI) and drug abuse (DAST). Specifically, we saw a moderately strong negative correlation with the depression and anxiety measures, and a low-to-moderate negative correlation with the drug abuse measure.
With respect to the moderately strong negative correlations with the BDI and BAI, it should be noted that CPAS scores explained approximately 50% of the variance, indicating that about half of the variance in BDI and BAI total scores cannot be accounted for in differences in scores on the CPAS. The high internal consistency demonstrated by the CPAS (Cronbach’s α = 0.97), in addition to the correlations of the CPAS with the BDI and BAI, suggests that the remaining variance in positive affect unexplained by depressive and anxiety symptoms is not necessarily due to reliability related issues. While we acknowledge that alternate explanations for the moderately strong correlations and unexplained variance exist (e.g. high reliability of the BDI and/or BAI, measurement error, statistical issues related to the distribution), these results may indicate that the CPAS does not simply measure absence of depression, but captures a distinct, yet related, construct (i.e. positive affect).
Despite the strength of the findings, the study is limited by several factors. First, all participants in our sample population completed the BDI and therefore were assessed for depression via this non-stringent cut-off for depression (BDI total score ≥ 16). However, only a subsample completed the mood module of the SCID, and as a result was eligible to receive a possible diagnosis according to this more conservative measure. In addition, the number of depressed individuals in our college student population is likely lower than in other populations. Therefore, studies of the psychometric properties of the CPAS should use diagnostic instruments such as the SCID in patients with MDD before and after treatment.
Second, while our overall sample size was rather large, generalizability may be limited because all participants were college students and the majority of participants were female. In addition, a college student sample is inherently more homogeneous than other non-clinical populations (e.g. limited age range, higher education, higher socioeconomic status (SES)), which may limit the generalizability of our results with other groups. It is possible that these students who participated on their school campus were unwilling to provide honest responses because of concerns regarding confidentiality.
Third, it may be that the set-up of the study, in which the majority of participants completed the questionnaire packet with their peers present at the screening site, as opposed to a more private setting, impaired their willingness to provide truthful responses. The current results may not replicate in other non-clinical cohorts, especially among individuals unwilling to participate in research, or in different settings.
Future studies may choose to explore the psychometric qualities of the CPAS in depressed or other clinical samples, as the present data may not generalize across groups. Specifically, nearly half of the participants in the present study responded with a maximum score on most, if not all, items. Given the relatively low variability in the present dataset, we cannot conclude that the suggested factor structure will generalize to other samples; it may be that exploring the CPAS factor structure in data with heightened variability (i.e. from a clinical sample) would suggest an alternate factor structure. Furthermore, the present study relied on self-report measures with one objective assessment of diagnosis in a subgroup.
Finally, we were unable to assess for convergent validity given that no validated scale of positive affect was included in the survey battery. Future studies exploring the psychometric properties of the CPAS would benefit from including an established positive affect measure (e.g. PANAS) to adequately assess convergent validity. While we expect that the CPAS would demonstrate higher correlations with measures of positive affect than measures of related constructs (e.g. BDI, BAI), we are unable to confirm this speculation in the present study.
Despite the limitations of the study, the results provide preliminary support for the validity of the CPAS. Given the brevity of the scale, it may be useful in the clinic and in clinical trials to assess the important metric of positive affect.
Footnotes
Acknowledgements
We thank the professional staff and research coordinators of the Depression Clinical and Research Program who organized the parent study and conducted the interviews.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of interest
AAN is a full-time employee of the Massachusetts General Hospital (MGH).
As of 26 May 2011, he has served as a consultant to: American Psychiatric Association (only travel expenses paid), Appliance Computing, Inc. (Mindsite), Basliea, BrainCells, Inc., Brandeis University, Bristol Myers Squibb, Dainippon Sumitomo, Eli Lilly and Company, EpiQ, Novartis, PGxHealth, Shire, Schering-Plough, Takeda Pharmaceuticals, and Targacept. He has consulted for the following through the MGH Clinical Trials Network and Institute (CTNI): Astra Zeneca, BrainCells, Inc., Dianippon Sumitomo/Sepracor, Johnson and Johnson, Labopharm, Merck, Methylation Science, Novartis, PGxHealth, Shire, Schering-Plough, Targacept, and Takeda/Lundbeck Pharmaceuticals.
AAN has received grant/research support through MGH from: Cephalon, NIMH, PamLabs, Pfizer Pharmaceuticals, and Shire. He has received honoraria from: American Society for Clinical Psychopharmacology, APSARD, Belvoir Publishing, Hillside Hospital, IMEDEX, ISBD, MBL Publishing, and Physicians Postgraduate Press.
AAN is a presenter for the Massachusetts General Hospital Psychiatry Academy (MGHPA). The education programs conducted by the MGHPA were supported through Independent Medical Education (IME) grants from the following pharmaceutical companies in 2008: Astra Zeneca, Eli Lilly, and Janssen Pharmaceuticals; in 2009: Astra Zeneca, Bristol-Myers Squibb, and Eli Lilly.
AAN is (was) on the advisory boards of: Appliance Computing, Inc., BrainCells, Inc., Eli Lilly and Company, Forest Pharmaceuticals, Johnson and Johnson, Sunovion, Takeda/Lundbeck, and Targacept. He has had no speaker bureaus or boards since 2003.
AAN owns stock options in Appliance Computing, Inc. and BrainCells, Inc.
Through MGH, AAN is named for copyrights to: the Clinical Positive Affect Scale and the MGH Structured Clinical Interview for the Montgomery Asberg Depression Scale exclusively licensed to the MGH Clinical Trials Network and Institute (CTNI).
KHB has no support to report.
AHF’s research has been funded by NARSAD, HMS, and NIMH. She has received honoraria from the MGH Psychiatry Academy. She has equity holdings in Pfizer, GlaxoSmithKlein, and Wyeth.
MF has received research support from the following: Abbott Laboratories, Alkermes, Inc., Aspect Medical Systems, AstraZeneca, BioResearch, BrainCells, Inc., Bristol-Myers Squibb, Cephalon, Inc., Clinical Trials Solutions LLC, Clintara LLC, Covidien, Eli Lilly and Company, EnVivo Pharmaceuticals, Inc., Forest Pharmaceuticals, Inc., Ganeden Biotech, Inc., GlaxoSmithKline, Icon Clinical Research, Johnson & Johnson Pharmaceutical Research & Development, Lichtwer Pharma GmbH, Lorex Pharmaceuticals, NARSAD, NCCAM, NIDA, NIMH, Novartis AG, Organon Pharmaceuticals, PamLab LLC, Pfizer, Inc., Pharmavite® LLC, Roche, RCT Logic LLC, Sanofi-Aventis US LLC, Shire, Solvay Pharmaceuticals, Inc., Synthelabo, and Wyeth-Ayerst Laboratories.
MF has provided advisory/consulting to the following: Abbott Laboratories, Affectis Pharmaceuticals AG, Amarin Pharma, Inc., Aspect Medical Systems, AstraZeneca, Auspex Pharmaceuticals, Bayer AG, Best Practice Project Management, Inc., BioMarin Pharmaceuticals, Inc., Biovail Corporation, BrainCells, Inc., Bristol-Myers Squibb, CeNeRx BioPharma, Cephalon, Inc., Clinical Trials Solutions LLC, CNS Response, Inc., Compellis Pharmaceuticals, Cypress Pharmaceutical, Inc., DiagnoSearch Life Sciences (P) Ltd, Dov Pharmaceuticals, Inc., Eisai, Inc., Eli Lilly and Company, EPIX Pharmaceuticals, Inc., Euthymics Bioscience, Inc., Fabre-Kramer Pharmaceuticals, Inc., Forest Pharmaceuticals, Inc., GenOmind LLC, GlaxoSmithKline, Gruenthal GmbH, Janssen Pharmaceutica, Jazz Pharmaceuticals, Inc., Johnson & Johnson Pharmaceutical Research & Development LLC, Knoll Pharmaceuticals Corp., Labopharm, Inc., Lorex Pharmaceuticals, Lundbeck, Inc., MedAvante, Inc., Merck & Co., Inc., Methylation Sciences, Neuronetics, Inc., Novartis AG, Nutrition 21, Organon Pharmaceuticals, PamLab LLC, Pfizer, Inc., PharmaStar, Pharmavite® LLC, Precision Human Biolaboratory, Prexa Pharmaceuticals, Inc., PsychoGenics, Psylin Neurosciences, Inc., Rexahn Pharmaceuticals, Inc., Ridge Diagnostics, Inc., Roche, RCT Logic LLC, Sanofi-Aventis US LLC, Sepracor, Inc., Schering-Plough Corporation, Solvay Pharmaceuticals, Inc., Somaxon Pharmaceuticals, Inc., Somerset Pharmaceuticals, Inc., Synthelabo, Takeda Pharmaceutical Company Limited, Tetragenex Pharmaceuticals, Inc., TransForm Pharmaceuticals, Inc., Transcept Pharmaceuticals, Inc., Vanda Pharmaceuticals, Inc., and Wyeth-Ayerst Laboratories.
MF has been involved in speaking/publishing for the following: Adamed, Co., Advanced Meeting Partners, American Psychiatric Association, American Society of Clinical Psychopharmacology, AstraZeneca, Belvoir Media Group, Boehringer Ingelheim GmbH, Bristol-Myers Squibb, Cephalon, Inc., Eli Lilly and Company, Forest Pharmaceuticals, Inc., GlaxoSmithKline, Imedex LLC, MGH Psychiatry Academy/Primedia, MGH Psychiatry Academy/Reed Elsevier, Novartis AG, Organon Pharmaceuticals, Pfizer, Inc., PharmaStar, United BioSource, Corp., and Wyeth-Ayerst Laboratories.
MF has equity holdings in Compellis.
MF holds a patent for the Sequential Parallel Comparison Design (SPCD) and a patent application for a combination of azapirones and bupropion in MDD. Dr Fava has copyright royalties for the MGH CPFQ, SFI, ATRQ, DESS, and SAFER.
TD’s research has been funded by NIMH, NARSAD, TSA, and OCF. He has received honoraria, consultation fees and/or royalties from the MGH Psychiatry Academy, BrainCells, Inc., Systems Research and Applications Corporation, Boston University, the Catalan Agency for Health Technology Assessment and Research, the National Association of Social Workers Massachusetts, Massachusetts Medical Society and Oxford University Press. He has also participated in research funded by NIH, NIA, Janssen Pharmaceuticals, The Forest Research Institute, Shire Development, Inc., Medtronic, Cyberonics, and Northstar.
