Abstract
Objectives:
Past controlled clinical trials centred on virtual reality exposure therapy (VRET) for agoraphobia mostly used multicomponent therapy with success. However, the present paper aimed to evaluate the independent effect of VRET for agoraphobia.
Methods:
A controlled study involving 18 agoraphobic participants assigned to two groups: VRET only and VRET with cognitive therapy. Nine specific virtual environments were developed using an affordable game level editor.
Results:
Questionnaires, behavioural tests and physiological measures indicated a positive effect of VRET. Correlations supported the predictive value of presence towards treatment outcome. The addition of cognitive therapy did not provide significant additional benefit.
Conclusions:
Overall, the isolated effects of VRET did not seem to be significantly less than the effects of VRET combined with cognitive therapy. Future research should explore the use of other components in addition to cognitive therapy and VRET for agoraphobia as well as its possible use in patients’ homes.
Introduction
According to the United Nations (UN, 2010), with more than 6 billion inhabitants on earth, 21 megacities such as Tokyo, the most populous urban agglomeration in the world (36.5 million inhabitants) and more than half of the population on earth living in cities, we are increasingly confronted by urban spaces and infrastructure. This way of life inherently implies places where escape might be difficult or hindered (e.g. crowded places, malls, subways, underground car parks, flats, public toilets, lifts). While a majority of the population deals with these situations with ease, a certain percentage of people suffering from a phobia called agoraphobia perceives these situations as stressful, threatening or panic-provoking, fearing the impossibility of being able to reach an exit in the advent of a panic attack (American Psychiatric Association, 2000). Panic attack is an intense and overwhelming episode of anxiety associated with a cluster of interoceptive cues (e.g. palpitations, tachycardia, sweating, shaking, blurred vision). People with agoraphobia may also fear places where help could be difficult to reach in case of extreme arousal or if a panic attack occurs, such as rural environments, driving, being on a bridge, in a wide open space with homogenous landscape or simply being alone.
Psychosocial treatment has been relatively effective with cognitive behaviour therapy (CBT) being recognised as the standard evidence-based treatment according to guidelines from the National Institute of Health and Clinical Excellence in 2007, which recommended CBT as the treatment of choice for this disorder (NICE, 2007). Despite the existence of efficacious treatments, only a limited number of people with agoraphobia and other anxiety disorders seek professional help (Magee et al., 1996). Among several possible reasons for this failure, it is likely that a key deterrent is the fear of confronting phobogenic stimuli during therapy or the difficulty of systematically building a graduated hierarchy for certain situations (e.g. planes and flight duration, weather conditions, transport schedule, number of people around and their behaviours, train configuration, traffic or other unpredictable events, etc.).
Virtual reality (VR) treatment protocols for phobias were set up to overcome the above issues as well as to exploit a new media for man/machine relationships. During the exposure, reality is replaced by artificially created stimuli inside a controlled computerised world. These procedures aim to facilitate habituation and fear extinction within a virtual environment (VE), just as traditional CBT does within the real world (Rothbaum et al., 2001).
Previous controlled clinical studies have demonstrated a similar efficacy of virtual reality exposure therapy (VRET) to treat participants suffering from agoraphobia when compared to CBT (Botella et al., 2007; Vincelli et al., 2003). However all of these earlier studies have employed multicomponent treatment protocols that combined VRET with other active treatments (e.g. relaxation, cognitive restructuring) (Botella et al., 2004, 2007; Vincelli et al., 2003). Therefore, it is not possible to determine with accuracy whether exposure in a virtual format is a key active component of treatment or whether the positive effects were due to the efficacy of cognitive techniques (Gorini and Riva, 2008). Identifying key active ingredients within a treatment protocol is important for future improvements and for cost efficiency. Consequently, in the current study we aimed to assess the therapeutic effect of VRET presented alone when compared to a dual-component treatment (VRET and cognitive therapy) in a sample of 18 participants diagnosed with agoraphobia.
From the software point of view, it is worth noting that in previous studies, VEs were constructed with exclusive in-house programs or relatively expensive professional software. To reduce costs and improve access, several research teams have successfully employed VEs created from inexpensive game level editors (GLEs) to treat arachnophobia and claustrophobia (Bouchard et al., 2006; Malbos et al., 2008). Given the importance of widely accessible treatment programmes for mental health, in the present study we designed all required VEs for the VRET of agoraphobia within a GLE.
Our objectives were two-fold. First, the study evaluated whether affordable methods such as a GLE could be used by a therapist to construct various VEs that were realistic enough to induce a feeling of presence within sessions and attain therapeutic efficiency. Second, the trial evaluated the isolated effect of VRET when compared to a combined treatment of VRET and cognitive therapy by randomly assigning participants to two different groups. The efficacy of the treatment was assessed with self-report questionnaires as well as behavioural and physiological measures.
Methods
Sample
Nineteen participants (12 women, 7 men) meeting DSM-IV criteria for panic disorder with agoraphobia were recruited for the clinical trial. Exclusion criteria included epilepsy, dissociative or non-dissociative chronic psychosis, recent discontinuation of psychotropic drugs and substance dependence.
Diagnoses were established by the first author based on a semi-structured interview, the Anxiety Disorders Interview Schedule for DSM-IV (ADIS-IV) (Di Nardo et al., 1994). One participant dropped out at an early stage due to a severe myopia. Only four participants had previous video game experience. The mean age of the sample was 44.11 years (SD 13.79), ranging from 24 to 72 years. All the participants were Caucasian. Thirteen (72.2%) of the participants were married or had a de facto relationship, five (27.8%) were single, none were divorced. There were 15 (83.3%) who worked or studied full time while three (16.7%) were unemployed. With regard to their level of education, six (33.3%) had a secondary education and 12 (66.7%) had a tertiary education. According to the ADIS semi-structured interview, all the participants were moderately to severely handicapped in their daily life or work due to their agoraphobia. The mean duration of agoraphobia was 18 years (SD 12.53) and the mean age of onset was 26.11 years (SD 7.89). Additionally, 10 participants exhibited at least one co-morbid axis I disorder: major depression (four participants), social phobia (five participants) and specific phobia (nine participants, mostly acrophobia).
When compared to past epidemiological studies or reviews concerning agoraphobia, a relative similarity of the present sample can be found with a predominant number of females suffering from agoraphobia (Somers et al., 2006), a mean age of onset of agoraphobia from mid to late 20s (Antony and Swinson, 2000) and a co-morbidity with depression, social phobia or specific phobia (Starcevic et al., 1992). Concerning marital status, as stated by Marks, at the time of the treatment most agoraphobics are married (Marks, 1987). With respect to education level, an early onset of agoraphobia may interfere with education, making comparison difficult, and lastly, socioeconomic status has weaker associations with this disorder (Noyes and Hoehn-Saric, 1998).
Assessment instruments
In addition to the instruments measuring the psychological, behavioural and physiological impact when using VR, a specific questionnaire related to presence (the presence questionnaire) is usually required. The most common definition of presence that pertains to the immersion in VEs is the one related to the concept of transportation to elsewhere: the feeling of ‘being there’ (Heeter, 1992) that is the pregnant impression of being existent in a real or artificially created place.
In the framework of experimental work where ecologically valid situations are required for studies of behaviour or reactions, presence is of significant importance. Indeed, it is a key requirement that a subject will behave in a VE as they would when confronted with similar cues in everyday reality. Similar characteristics are required for the application of VR to clinical interventions. In this circumstance, the assessment of presence may be especially important.
Side effects of using VR and related apparatus is summarised as cybersickness, a form of motion sickness that induces various symptoms (namely nausea, sweating, dizziness, etc.) when the user is immersed in VEs. Following past recommendations (Robillard et al., 2003), measuring cybersickness through questionnaires such as the Simulation Sickness Questionnaire is a necessary condition because cybersickness may hinder perceived presence and consequently the treatment efficiency.
Presence questionnaire, PQ v3.0 (Witmer et al., 2005). The PQ consists of 32 items rated on a 7-point scale, assessing the participant’s perception of presence. It contains six subscales: involvement, interface quality, adaptation/immersion, consistency with expectation, visual fidelity and auditory fidelity.
Subjective Units of Discomfort (SUD) (Wolpe, 1969). A 100-point scale that measures the perceived level of anxiety at a given time. Scores of 0 represent no fear and 100 represents the most fear the individual has ever felt in their life.
Depression Anxiety Stress Scales (DASS 21) (Lovibond and Lovibond, 1995). A 21-item subjective questionnaire that uses four point scales to measure symptoms of depression, anxiety and stress.
Anxiety Sensitivity Index (ASI) (Peterson and Reiss, 1993). This is a 16-item questionnaire that rates the fear of anxiety-related symptoms (e.g. shortness of breath, tachycardia) using a 5-point scale.
Agoraphobia Cognitions Questionnaire (ACQ) (Chambless et al., 1984). Fourteen 14 items to rate the intensity of thoughts and beliefs common to agoraphobia, featuring two subscales: loss of control and physical concerns.
Mobility Inventory for Agoraphobia (MIA, part 1) (Chambless et al., 1985). This questionnaire uses a 5-point Likert scale to rate the severity of behavioural avoidance of situations dreaded by agoraphobics (each rated sequentially when accompanied or alone). It includes a total of 27 items. An additional 28th item measures the number of panic attacks in the last 7 days (PA/w) and is presented separately.
Simulation Sickness Questionnaire (SSQ) (Kennedy et al., 1993). This 16-item instrument on a 4-point scale assesses the manifestation as well as the severity of motion sickness related symptoms exhibited in a simulation or VE.
Behavioural Avoidance Test (BAT). This task comprised a direct, behavioural observation of distress in response to entering a feared situation. A real elevator (2 × 1.8 × 2.4 m) was designated in the psychology department building for the test.
Heart rate (HR) and heart rate variability (HRV). A heart rate monitor was used to measure heart rate in beats per minute for the duration of the BAT. Both HR and HRV (RMSSD and pNN50) were calculated. The HRV indicates the fluctuations of heart rate around an average heart rate and the HR reflects the average heart rate. HR and HRV reflect the autonomic responses involved in emotional arousal, most notably during anxiety where the HR is expected to increase and the HRV to decrease (Bernston and Cacioppo, 2007). HR and HRV represent an alternative objective measurement of anxiety response and presence.
Apparatus
The VR system conformed to the ‘affordable materials’ and portable constraints of the test. It included a ruggedised Virtual Realities HMD 42 Pro Head-Mounted Display (HMD, 800 × 600 non-stereoscopic OLED screen with 42° field of view) coupled with an embedded three degrees of freedom head tracker (angular resolution: 0.02°, latency 4 ms). The steering wheel exploited for the driving VE was a Logitech Momo with vibration and force feedback capabilities. The VEs were generated and ran on an ordinary graphics-orientated notebook with an Intel I7 processor (four cores), 4 Go DDR2 RAM, an ATI M7740 graphic card with 1 Go RAM and a 1440 × 900 resolution screen. Required software was Microsoft Windows XP (32 or 64 bits edition), Microsoft DirectX 9.0 or higher and the equipment’s drivers. The heart rate monitor was a Polar S810i.
Software and virtual environments
The main software exploited to create and run the VEs was Sandbox 2, an inexpensive (US$30–) and commercially available GLE developed by Crytek GmbH. Prior to its full use for the trial, this GLE was tested and compared to seven other commercially available GLEs by considering several distinct criteria and requirements previously reported (Malbos et al., 2011).
To construct the VEs, the experimenter (a PhD student in psychology) exploited the aforementioned GLE to build nine specific context-graded VEs. These nine VEs offered distinct anxiety-inducing places for agoraphobics. In order to respect the principle of graded exposure, the hierarchy in terms of anxiogenic cues present in the VEs was established, taking into account previous works (American Psychiatric Association, 2000; Barlow, 2007; Beck and Emery, 1985; Marks, 1987; North et al., 1996; Vincelli et al., 2003): being alone, crowded places, homogeneous wide open space, restricted exit possibilities, difficulty of reaching help, degree of confinement. As evoked by Marks (1987), other secondary cues were integrated such as the degree of illumination and light hue. Moreover, to enhance the graded context during the VRET, the experimenter could trigger the onset of an internal cue (simulated bodily symptoms such as heartbeat sounds/breathing sounds at different speeds, and visual disturbances) as well as specific events within the VE (e.g., number of virtual passengers or crowds, increased queue length, turbulences, technical difficulties, etc.). These options allowed increases in the intensity of induced fear to modulate the degree of exposure at various times. The VEs also provided the participant with direct realistic interactions (e.g. doors, responding virtual humans, grabbing objects, physical or mechanical reactions to the user’s presence, etc.). These nine VEs comprised a valley for training purposes, a town square with a building featuring lifts of different size, a supermarket in a mall with the possibility of standing in a queue, bridges in the Australian outback, a subway station with a working train, an underground car park, a cinema with a viewable movie, an airplane with all flight procedures and a highway with tunnel/traffic jam where the participants can drive a car.
Procedure
Prior to its set-up, the experimental protocol was reviewed and approved by the Macquarie University Human Research Ethics Committee.
Following the intake assessment and the diagnostic interview, the participants were randomly assigned to two therapeutic groups: one group receiving VRET only (VRO) and one group receiving VRET and cognitive therapy (VRC). The allocation to each group was done using a randomisation table generated by a computerised sequence generator. The protocol included 10 weekly sessions lasting 90 minutes for both groups. Both groups received eight sessions of VRET and two initial sessions. For the VRC group, in order to keep the present clinical trial comparable to previous successful works involving VRET and cognitive therapy (Botella et al., 2004, 2007), the protocol included only two sessions of cognitive therapy, which took place during the two initial sessions. For the VRO group the initial sessions do not offer cognitive therapy but instead comprised ‘neutral’ conversations pertaining to leisure, family, history and activities. For the VRC group, cognitive therapy consisted of psychoeducation, anxiety acceptance, cognitive restructuring, positive self-statements and relapse prevention, as outlined by several authors (Barlow, 2007; Beck and Emery, 1985; Craske, 1999). Moreover, throughout the eight VRET sessions for the VRC group, participants were regularly encouraged to review and apply methods acquired during the two cognitive therapy periods. The VR exposure followed standard recommendations for the delivery of therapeutic exposure and desensitisation (Barlow, 2007; Marks, 1987; Vincelli et al., 2000).
Prior to immersion, SUD and HR/HRV were measured at rest across 5 minutes in a standing position (baseline recording). The eight sessions of VRET comprised exposure to nine different context-graded VEs related to common agoraphobic fears (e.g. airplane, subway, driving, etc.). To explore the VEs elaborated within a selected GLE, participants donned a head-mounted display with a motion tracker. The participants were exposed to each VE over a period of 50–60 minutes. Additionally and as suggested by Botella et al. (2007), participants from both groups were invited not to practice voluntary self-exposure between sessions until the clinical trial was completed. In this way the method examined the effects of pure virtual exposure without confounding with additional in vivo exposure.
Questionnaires, behaviour tests and physiological measures were registered before and after the treatment procedure. The PQ was completed after each VR exposure session. At the end of the 3-month follow-up period, the same battery of tests was repeated.
Results
Demographic values, pre-tests and presence between groups
The two groups did not differ significantly in terms of sex ratios: χ2 (1, n = 18) = 0.23, p < 0.63, age: t (16) = −1.65, p < 0.12, η2 = 0.14, education: χ2 (1, n = 18) = 0.0, p < 1, marital status: χ2 (1, n = 18) = 2.25, p < 0.13 or duration of agoraphobia: (t = −1.39), p < 0.18, η2 = 0.10. In a similar vein, no difference on perceived presence throughout the VR exposure was detected between the two groups (t = −0.22), p < 0.83, η2 = 0.00. They also did not differ significantly on measures of psychopathology: DASS (t = −0.349), p < 0.73, η2 = 0.00, ASI (t = −0.69), p < 0.50, η2 = 0.02, ACQ (t = −0.66), p < 0.52, η2 = 0.02 and MIA (t = −0.71), p < 0.67, η2 = 0.03.
Presence effects
Compared to mean scores from a previous study using the presence questionnaire score (PQ = 104.39, SD = 18.99) (Robillard et al., 2002), as well as our previous trial involving the significant effectiveness of VRET with claustrophobia (PQ = 113.03, SD = 9.34) (Malbos et al., 2008), it can be seen that the mean PQ score in the current study (144.05, SD = 15.78) indicated excellent immersion in the VEs.
Effects of treatment on symptoms
Scores of questionnaires, physiological and behaviour measures are detailed in Tables 1 and 2. A two-way mixed analysis of variance (ANOVA) was used to compare the two groups over time (pre vs. post). Significant main effects of time were demonstrated on the DASS, the ASI, the ACQ, the MIA, and the number of panic attacks per week. Examination of group by time interactions indicated no significant interactions for any variable: DASS, ACQ, MIA, number of panic attacks per week. Mean scores, ANOVA and effect sizes are reported in Table 1.
Means, standard deviations of the dependent variables, results of ANOVA and effect sizes between pre-and post-test period (Time) and for time × group comparison (Interaction).
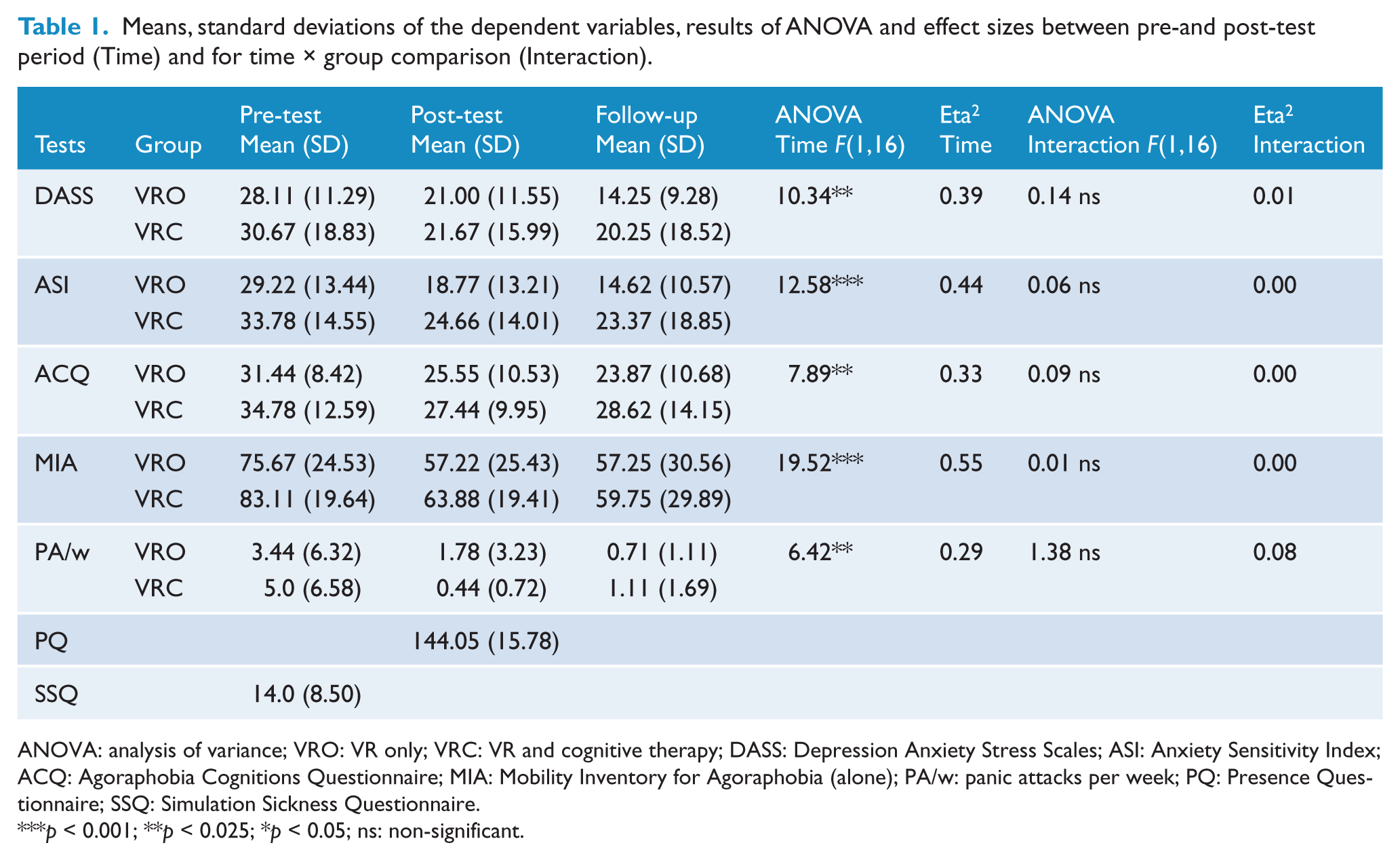
ANOVA: analysis of variance; VRO: VR only; VRC: VR and cognitive therapy; DASS: Depression Anxiety Stress Scales; ASI: Anxiety Sensitivity Index; ACQ: Agoraphobia Cognitions Questionnaire; MIA: Mobility Inventory for Agoraphobia (alone); PA/w: panic attacks per week; PQ: Presence Questionnaire; SSQ: Simulation Sickness Questionnaire.
p < 0.001;
p < 0.025;
p < 0.05;
ns: non-significant.
Means, standard deviations of the dependent variables related to the BAT tests, results ANOVA and effect size between pre- and post-test period (Time) and for time × group comparison (Interaction).
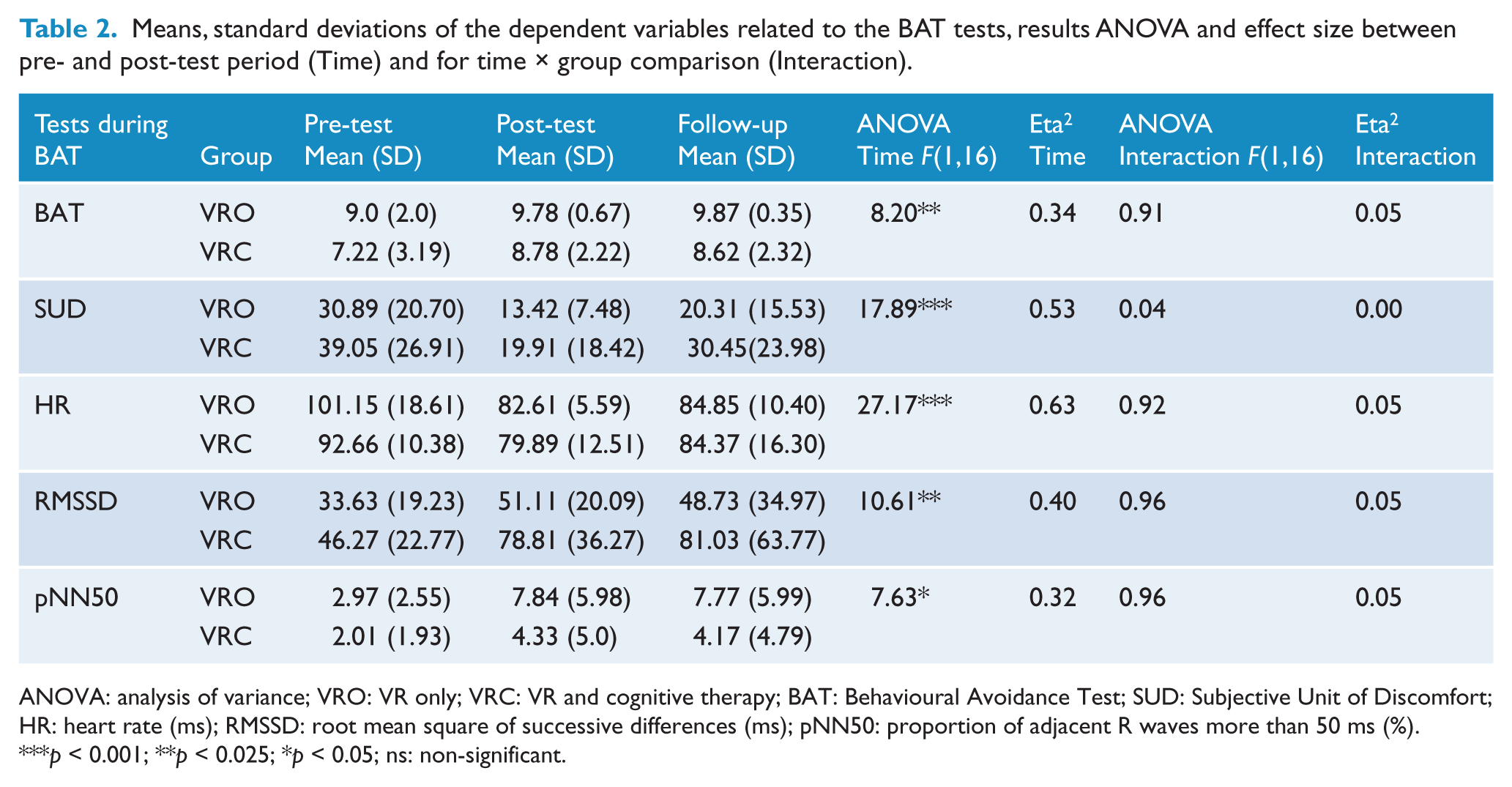
ANOVA: analysis of variance; VRO: VR only; VRC: VR and cognitive therapy; BAT: Behavioural Avoidance Test; SUD: Subjective Unit of Discomfort; HR: heart rate (ms); RMSSD: root mean square of successive differences (ms); pNN50: proportion of adjacent R waves more than 50 ms (%).
p < 0.001;
p < 0.025;
p < 0.05;
ns: non-significant.
Effects of treatment on response to BAT
A two-way mixed ANOVA was used to compare the two groups on the physiological and self-report indices taken during the behavioural tests (see Table 2). There was a significant time effect (pre vs. post) for SUD, as well as for HR/HRV: RMSSD and pNN50 indicating significant reductions in arousal. There was a similar significant improvement over time on the BAT score, suggesting reduced avoidance of the lift. Examination of group by time interactions indicated no significant interactions for any variable: BAT, HR and HRV: RMSSD, pNN50. Mean scores, ANOVA and effect sizes can be found in Table 2.

Screenshots of two VEs built with the Sandbox 2 for the present clinical trial. Note the high-resolution textures, the lighting effects, the high range rendering shadows, the avatar attitude and the contemporary 3D objects including a drivable vehicle (supermarket and highway).

Screenshots of two other phobogenic VEs elaborated with Sandbox 2 for the present study (subway station and an airplane cabin).
In order to ascertain that the absence of a significant group by time interaction, difference of dependent variables between the two groups was not due to the small size of the sample, the number of subjects needed to reach statistical difference was calculated. Computation was carried out given the effect size of each result, a type 1 error of 0.05 and a satisfactory power of 0.8. Results are outlined in Table 3.
Required number of participants to reach statistical difference for group by time interaction for each test.
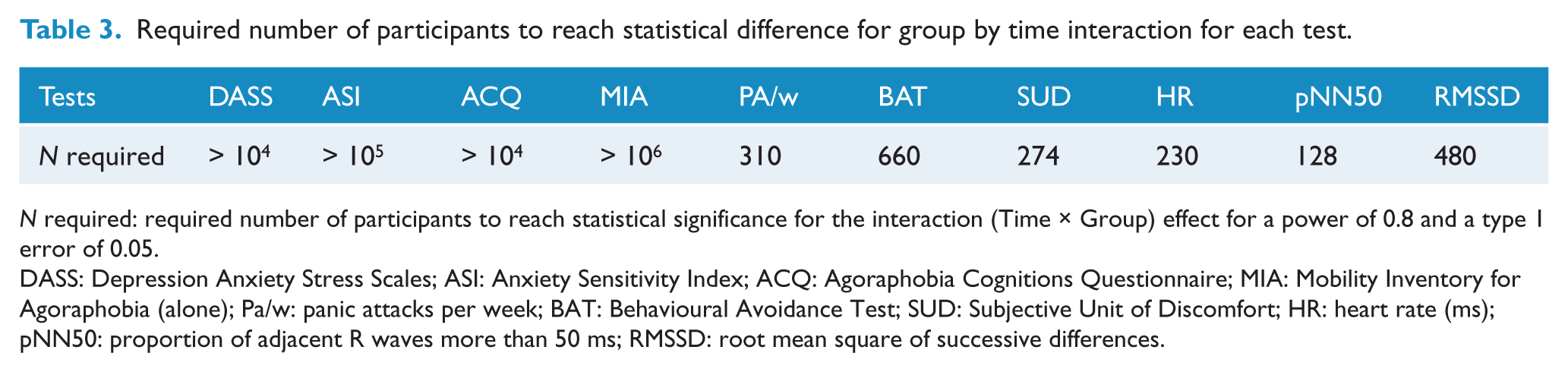
N required: required number of participants to reach statistical significance for the interaction (Time × Group) effect for a power of 0.8 and a type 1 error of 0.05.
DASS: Depression Anxiety Stress Scales; ASI: Anxiety Sensitivity Index; ACQ: Agoraphobia Cognitions Questionnaire; MIA: Mobility Inventory for Agoraphobia (alone); Pa/w: panic attacks per week; BAT: Behavioural Avoidance Test; SUD: Subjective Unit of Discomfort; HR: heart rate (ms); pNN50: proportion of adjacent R waves more than 50 ms; RMSSD: root mean square of successive differences.
Correlations with presence
We assumed that the first VR exposure was very likely to produce anxiety related to exposure to virtual stimuli due to the absence of habituation or fear extinction at this early stage in the treatment. Consequently, correlations were searched between the presence (PQ) and the subjective and physiological measures during the first session. For this session, small and non-significant positive correlations were demonstrated between PQ and SUD (r = 0.22), p < 0.37, PQ and HR (r = 0.11), p < 0.68, PQ and pNN50 (r = 0.24), p < 0.33, but not between PQ and RMSSD (r = −0.09), p < 0.74.
To investigate any relationship between the perceived presence and the treatment efficacy, difference scores were calculated between the pre-test and the post-test for all dependent variables and correlations were calculated between these scores, the mean PQ and the BPT across all sessions. Small to medium significant or insignificant positive correlations were shown between PQ and pre-post difference for DASS (r = 0.36), p < 0.15, ASI (r = 0.23), p < 0.36, ACQ (r = 0.15), p < 0.56, MIA (r = 0.10), p < 0.68, PA/w (r = 0.33), p < 0.17, BAT (r = 0.49), p < 0.04, HRbat (r = 0.25), p < 0.31, pNN50bat (r = 0.49), p < 0.04. However a minimal correlation was found between PQ and SUDbat (r = 0.07), p < 0.76 and a negative correlation was shown between PQ and RMSSDbat (r = −0.16), p < 0.53.
Follow-up (3 months)
Sixteen participants (89%) returned data 3 months after the end of treatment. Two-way ANOVA compared the groups between post-treatment and follow-up. No significant main effects of time were found on any measures: DASS F(1,14) = 2.62, p < 0.12, η2 = 0.16, ASI F(1,14) = 2.98, p < 0.10, η2 = 0.17, ACQ F(1,14) = 0.17, p < 0.69, η2 = 0.01, MIA F(1,14) = 0.70, p < 0.41, η2 = 0.05, number of panic attacks per week F(1,14) = 0.90, p < 0.36, η2 = 0.06, BAT F(1,14) = 1.0, p < 0.33, η2 = 0.07, SUD F(1,14) = 3.20, p < 0.09, η2 = 0.18, HR F(1,14) = 1.36, p < 0.26, η2 = 0.09 and HRV: RMSSD F(1,14) = 0.02, p < 0.88, η2 = 0.00, pNN50 F(1,14) = 0.81, p < 0.38, η2 = 0.05. Likewise, no significant group by time interactions were detected on any measures: DASS F(1,14) = 0.67, p < 0.43, η2 = 0.05, ASI F(1,14) = 1.35, p < 0.26, η2 = 0.08, ACQ F(1,14) = 1.04, p < 0.32, η2 = 0.07, MIA F(1,14) = 0.01, p < 0.75, η2 = 0.00, number of panic attacks per week F(1,14) = 2.14, p < 0.16, η2 = 0.13, BAT F(1,14) = 1.0, SUD F(1,14) = 0.09, p < 0.77, η2 = 0.00, HR F(1,14) = 0.12, p < 0.73, η2 = 0.00 and HRV: RMSSD F(1,14) = 0.18, p < 0.68, η2 = 0.01, pNN50 F(1,14) = 1.89, p < 0.19, η2 = 0.12.
Discussion
The present clinical trial demonstrated the overall therapeutic effectiveness and presence-eliciting effect of VEs constructed using an affordable GLE. These outcomes were reached even though agoraphobia is usually considered to be a more complex disorder than specific phobias (Botella et al., 2007) as it involves fear and avoidance of a wide range of situations. Concerning correlations between presence and self-reported anxiety, physiological parameters and behavioural tests, these were calculated from the first session’s measures and were positive but not significant. It is therefore difficult to infer a possible relationship between these variables with the present sample but it pointed towards similar findings regarding the relation between presence and anxiety (Price and Page, 2007; Regenbrecht et al., 1998; Robillard et al., 2003; Schuemeie et al., 2000).
In contrast to previous clinical trials with agoraphobia (Botella et al., 2007; Vincelli et al., 2003), which involved a multicomponent therapeutic set-up involving VR exposure as one of its components, in the current study, the VRO group benefitted from VRET only. The overall results of all self-report, behavioural and physiological measures for the VRET only group and the VRET combined with cognitive therapy group, corroborated the positive outcome of VRET for agoraphobia: mood was improved, anxiety and avoidance behaviour lessened, the fear towards agoraphobic situations as well as agoraphobic cognitions reduced. Because there were no significant group by time interactions, the two groups did not seem to differ in their therapeutic effects over time. Therefore the isolated effects of VRET did not seem to be significantly less than the effects of VRET combined with brief cognitive therapy.
The especially small interaction effect sizes indicated that any possible differences are unlikely to have clinically significant implications. These outcomes replicate similar findings from studies using in vivo exposure where the addition of cognitive therapy to traditional exposure in vivo has often failed to demonstrate significant benefits (Bouchard et al., 1996; Burke et al., 1997).
The positive impact of the VR exposure treatment was maintained across the 3-month follow-up. There was no significant difference between the two groups, indicating that the addition of cognitive therapy did not provide significant longer-term improvements.
Several key limitations need to be kept in mind when interpreting clinical results. First, the participants were well educated with two-thirds having benefitted from tertiary education and a majority studying or working full-time. Such proportions might not be representative of the agoraphobic population in the Australian community. Second, the size of the sample was especially small and limits conclusions regarding group differences. As described earlier the very small effect sizes provide some confidence in the clinical value of the results because many hundreds of participants would have been required to show significant differences between groups. Nevertheless, replication with a larger sample is warranted. Third, the lack of a waiting list group and an in vivo exposure group limits the conclusions that can be drawn about the absolute efficacy of the treatments. However, past controlled clinical trials have already demonstrated a similar efficacy of VRET for agoraphobia when compared to CBT with an in vivo exposure component (Botella et al., 2007; Vincelli et al., 2003). Additionally, spontaneous remission of agoraphobia is exceptional (Wittchen and Essau, 1991), hence it is unlikely that such large changes could have been due to the simple effects of time.
The degree to which participants felt immersed in the VEs (presence) appeared to be a positive, moderate predictor of response to the therapy based on behavioural and physiological measures. This result supports the hypothesis that the more a participant felt immersed in the VE, the more efficacious the treatment. Clinically, this would suggest that the more that presence can be maximised, the greater the efficacy of the intervention (Wiederhold and Wiederhold, 2005). Finally, in the context of the recruitment process, three persons suffering from agoraphobia could not access the treatment protocol due to the severity of this disorder, which prevented them from leaving the boundaries of their home. In this present study, the VR equipment and the graphics-orientated notebook exploited during the clinical trial were light-weighted, thus suggesting the possible use of VRET outside of a clinical structure. Later studies could be dedicated to the effect of ambulatory VR treatment of agoraphobics in the patient’s home. Alternatively, internet-based CBT, or freely downloadable VR content (such as Neuro VR 2.0), might represent an intermediate step to prompt the patient to step out of their home and then gain access to more immersive solutions in specialised structures.
Future research should replicate this experiment on a larger scale, explore the effect of adding more cognitive therapy sessions or the use of other components in addition to cognitive therapy and VRET (e.g. relaxation, mental imagery, biofeedback, mindfulness).
Overall, the aim of VR intervention remains the same as that of traditional treatment: to give the patients the « cognitive click » (Beck and Emery, 1985) that allows them to ‘reconceptualise’ the real world, once they step out of the cyberspace.
Footnotes
Acknowledgements
The authors would like to thank Yao Ying, Andy Maesen (Sigma), Jonathan Gaston, Eric Fassbender, Debbie Richards, Iwan Kartiko, Vickie Williams, John Porte, Meredith Taylor, Psyfalc, Dan Dombrowski, Carl Kenner and Racer, as well as the reviewers, for their help and advice.
Funding
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Declaration of interest
No competing financial interests exist. No conflicts of interests exist towards any of the companies mentioned in this article.
