Abstract
Objective:
To review the literature to examine the use of clozapine levels to (i) guide therapy and prevent toxicity in clinical care and (ii) determine cause of death in post-mortem examination of patients who were treated with clozapine.
Methods:
MEDLINE was searched in December 2010 using the following keywords: ‘clozapine levels’, ‘clozapine and toxicity’, ‘clozapine and death’, ‘clozapine and mortality’ and ‘post-mortem redistribution’. Data was also collected from the 2010 MIMS Annual.
Results:
The literature reported significant variation in clozapine levels attained with any given dose, and considerable variability in the clinical response achieved at any given clozapine level. The lowest effective clozapine levels ranged from 250 to 550 µg/L, while the recommended upper limit to prevent toxicity varied from 600 to 2000 µg/L. There was minimal correlation between clozapine levels and side effects, with the exception of sedation, hypotension and seizure activity. The risk of seizures increased with plasma clozapine levels greater than 600 µg/L or rapid upward titration. In addition to prescribed dose, there are many factors that influence plasma clozapine levels. After death, the process of post-mortem drug redistribution resulted in 3.00 to 4.89 times increases in clozapine levels in central blood vessels and 1.5 fold increases in peripheral vessels compared to ante-mortem levels.
Conclusions:
The exact range of clozapine levels that corresponds to toxicity remains unclear. However, levels between 350 µg/L and 1000 µg/L achieved with gradual upward titration are more likely to be effective and less likely to cause toxicity. Ongoing clozapine level monitoring is indicated, especially when (i) prescribing higher doses (> 600 mg/day) of clozapine, (ii) there has been a change in a patient’s concomitant pharmacotherapy or cigarette use and (iii) there has been a suboptimal response to treatment. The use of post-mortem clozapine levels to determine clozapine toxicity as a cause of death is unreliable.
Introduction
Clozapine’s superior efficacy compared to other antipsychotics is well established (Bell et al., 1998; Couchman et al., 2010; Green and Schildkraut, 1995; McKean et al., 2008; Meyer, 2001; Miller, 2000; Mitchell, 2001; Oyewumi et al., 2002; Raedler et al., 2008; Sandson et al., 2007; Schulte, 2003; Taylor, 1997; Taylor and Duncan-McConnell, 2000). It rarely causes extrapyramidal side effects (Couchman et al., 2010; Merrill et al., 2005; Miller, 2000; Remington, 2007; Sandson et al., 2007) and is reported to have an anti-suicide effect (Hennen and Baldessarini, 2005; Meltzer, 1998; Meltzer et al., 2003; Merrill et al., 2005; Modestin et al., 2005; Raedler et al., 2008; Tiihonen et al., 2009; Walker et al., 1997). Early use of clozapine has been recommended in those patients with treatment-refractory schizophrenia (Royal Australian and New Zealand College of Psychiatrists, 2005) and prescriptions for clozapine in Australia increased by 78% between 2000 and 2009 (Malalagama et al., 2011).
However, clozapine has a concerning side effect profile. Unlike other antipsychotic medications, a safe therapeutic dosage range for clozapine has not been clearly established (McKean et al., 2008), due to large variations in individual responses (Bell et al., 1998; de Leon and Simpson, 2004; Fabrazzo et al., 2002; Gross et al., 2004; Liu et al., 1996; Llorca et al., 2002; Lowe and Ackman, 2010; Mauri et al., 2004; Spina et al., 2000a; Wong et al., 2006) resulting in a wide range of doses prescribed and corresponding levels accepted during treatment (Davis and Chen, 2004). Although it is possible to measure plasma levels of other antipsychotics (Nazirizadeh et al., 2010; Sparshatt et al., 2011), clozapine is the only antipsychotic where routine monitoring of levels is recommended (Couchman et al., 2010; Flanagan et al., 2005; Perry, 2001; Taylor, 1997).
This literature review was undertaken following the deaths of two patients with treatment-refractory schizophrenia treated with clozapine. A 29-year-old male had received gradually increasing doses of clozapine over 2 years due to ongoing treatment resistance. Plasma clozapine levels in the 3 months prior to his death ranged from 750 to 1200 µg/L. His final clozapine level taken 3 days before he died was 1180 µg/L. His post-mortem clozapine level was 2800 µg/L. He was also found to have a left coronary artery occlusion and hypercholesterolaemia at autopsy. In the second case, a 56-year-old female had been treated with clozapine for 6 years, including a consistent 500 mg daily dose for the 4 years preceding her death. Her clozapine levels varied between 290 and 1370 µg/L over her 6 years of clozapine treatment, although there were no levels greater than 1000 µg/L in the 2 years preceding her death. Her final clozapine level, taken 1 month before she died, was 770 µg/L. Her post-mortem clozapine level was in the range consistent with toxicity. Neither patient had demonstrated clinical signs of clozapine toxicity, but this was determined as the cause of death at autopsy.
In order to better inform clinicians of the role of clozapine level monitoring in relation to treatment, safety and toxicity, we aimed to review the literature to examine the use of clozapine levels to (i) guide therapy and prevent toxicity in clinical care and (ii) determine cause of death in post-mortem examinations of patients who were treated with clozapine.
Methods
The literature search was conducted in December 2010 using the MEDLINE online database. Search terms used were ‘clozapine levels’, ‘clozapine and toxicity’, ‘clozapine and death’, ‘clozapine and mortality’ and ‘post-mortem redistribution’ (PMR). Searches were limited to human and English publications. The 2010 MIMs Annual (Swannick, 2010) was also reviewed for further information pertaining to clozapine levels and toxicity. Articles that discussed the clinical use of clozapine levels or clozapine toxicity and those pertaining to PMR were retained for in-depth review. Publications were excluded if they did not include information on these topics.
Results
The literature search yielded 337 journal articles. A single author (A.S.) read the abstracts of these articles. A total of 204 publications were excluded because they lacked direct relevance to the review topic, resulting in 133 articles retained for in-depth review. The relevant articles included randomised controlled trials, prospective, retrospective and cross-sectional observational studies, literature reviews, letters to editors and case reports.
Clozapine pharmacokinetics
Clozapine is a lipophilic drug with a high volume of distribution (6 L/kg). It is 95–97% bound to plasma proteins and has a mean terminal half-life of 12 hours (range 4–66 hours), reaching peak blood levels in 0.4–4.2 hours. Steady state is attained in 5–7 days. Clozapine is primarily metabolized by the cytochrome P450 enzyme 1A2 (CYP1A2), but P450 enzymes 2D6, 3A4, 2C9 and 2C19 also contribute to its metabolism (Bell et al., 1998; Cormac et al., 2010; Couchman et al., 2010; Flanagan et al., 2005; Frick et al., 2003; Greenwood-Smith et al., 2003; Kerswill and Vicente, 2003; Lowe and Ackman, 2010; Meyer, 2001; Mitchell, 2001; Raedler et al., 2008; Rostami-Hodjegan et al., 2004; Sandson et al., 2007; Spina et al., 2000b; Swannick, 2010; Taylor, 1997). Clozapine’s metabolism demonstrates dose-dependent saturation, resulting in disproportionate increases in clozapine levels with relatively small increases in dose above plasma levels of 600 µg/L in some patients (Couchman et al., 2010).
Clozapine toxicity
Clozapine toxicity can lead to a reduced level of consciousness, delirium, coma, seizures, tachycardia, arrhythmias, hypotension, cardiac arrest, aspiration, and respiratory depression (Bell et al., 1998; Couchman et al., 2010; Swannick, 2010). Serious, uncommon adverse reactions to clozapine that can occur in the absence of toxicity include diabetic ketoacidosis, eosinophilic myocarditis (0.1–1%), cardiomyopathy (0.1%), agranulocytosis (0.7–1%), neutropenia, QTc interval prolongation, overwhelming sepsis, fulminant hepatic failure, venous thromboembolism and neuroleptic malignant syndrome (Ananth et al., 2004; Bell et al., 1998; Dhillon et al., 2011; Hagg et al., 2009; Kilian et al., 1999; Macfarlane et al., 1997; Paciullo, 2008; Swannick, 2010; Wooltorton, 2002).
Clozapine doses and levels in clinical treatment
The 2010 MIMS Annual advises that most patients respond to clozapine doses in the range of 200–450 mg/day (Swannick, 2010). However, one meta-analysis found that doses of clozapine well over 400 mg daily were necessary for optimal treatment of many patients with treatment-refractory schizophrenia (Davis and Chen, 2004). The maximum dose recommended for clozapine is 900 mg/day (Bell et al., 1998; Swannick, 2010).
Due to the wide dose range and the variable clinical response to different doses, plasma levels are important in guiding clozapine treatment. The lower limit of clozapine levels thought to be effective in most patients ranged from 200–550 µg/L, with most reports favouring levels of at least 350–420 µg/L (Bell et al., 1998; Buckley et al., 2001; Cormac et al., 2010; Couchman et al., 2010; de Leon and Simpson, 2004; Flanagan et al., 2005; Freeman and Oyewumi, 1997; Kronig et al., 1995; Liu et al., 1996; Llorca et al., 2002; Miller et al., 1994; Paz et al., 2008; Potkin et al., 1994; Raedler et al., 2008; Rostami-Hodjegan et al., 2004; Schulte, 2003; Spina et al., 1998; VanderZwaag et al., 1996; Wong et al., 2006; Xiang et al., 2006). One study showed clozapine levels lower than 200 µg/L or a reduction greater than 60% in baseline level where response was achieved predicted relapse (Xiang et al., 2006). One study demonstrated equal efficacy between clozapine levels of 200–300 µg/L and 350–450 µg/L (VanderZwaag et al., 1996). One review recommended an adequate trial of clozapine should include at least 8 weeks on a plasma trough level above 350–400 µg/L (Schulte, 2003). Another review concluded that in the absence of clinical improvement at these levels, clozapine should be further increased until intolerable side effects occurred or a maximum dose of 900 mg/day was reached (Bell et al., 1998).
One challenge for clinicians prescribing clozapine is the significant variation in plasma levels attained with any given dose, both between individuals (8–45 fold difference) and within each individual (18–53% variance). This is thought to be caused by wide variations in metabolic enzyme activity that are influenced by genetic factors, patient characteristics (gender and weight), concomitant medications, cigarette use and other environmental factors (Bell et al., 1998; Bennett and Keck, 1996; Buckley et al., 2001; Chang et al., 1997; Chong et al., 1997; Couchman et al., 2010; de Leon and Simpson, 2004; Fabrazzo et al., 2002; Greenwood-Smith et al., 2003; Meyer, 2001; Mitchell, 2001; Oyewumi et al., 1995; Ozdemir et al., 2001; Potkin et al., 1994; Raedler et al., 2008; Tang et al., 2007). The timing of clozapine trough levels also contributes to this variability. Two studies demonstrated up to 60% variation in CYP1A2 activity between individuals (Ozdemir et al., 2001; Raedler et al., 2008). Couchman et al. (2010) reported wide variation of clozapine levels in more than 25,000 patients measured through a therapeutic drug monitoring service. Although 1.2% of blood samples had clozapine levels greater than 1000 µg/L at prescribed doses of less than 150 mg/day, 23.3% of samples had clozapine levels less than 350 µg/L at doses equal to or greater than 850 mg/day. For a proportion of patients, the recommended maximum dosage of 900 mg/day may not be adequate to achieve recommended therapeutic plasma levels.
Despite the variability between clozapine doses and plasma levels, most studies did find a significant correlation between clozapine levels and clinical response (Bell et al., 1998; Greenwood-Smith et al., 2003; Kronig et al., 1995; Miller et al., 1994; Piscitelli et al., 1994; Potkin et al., 1994; Spina et al., 2000b; Taylor, 1997) with only one report finding no significant relationship (van Troostwijk et al., 2003). This suggests that clozapine levels are more clinically reliable than recommended dosage ranges when determining safe and effective clozapine doses for individual patients.
Factors affecting clozapine levels
Cigarette smoking and concomitant medications are the most dynamic and therefore the most clinically important factors affecting plasma clozapine levels. Clozapine levels are significantly reduced by polycyclic aromatic hydrocarbons contained in cigarette smoke via induction of CYP1A2 (Cormac et al., 2010; Couchman et al., 2010; Greenwood-Smith et al., 2003; Lowe and Ackman, 2010; McKean et al., 2008; Meyer, 2001; Ozdemir et al., 2001; Palego et al., 2002; Sagud et al., 2009; Sandson et al., 2007; Swannick, 2010; Taylor, 1997). Daily smoking of between seven and twelve cigarettes is sufficient for maximum induction of clozapine metabolism (Lowe and Ackman, 2010). The literature does not provide evidence that smoking cannabis without tobacco induces CYP1A2 (Lowe and Ackman, 2010; Zullino et al., 2002). Studies reported 20–70% increases in clozapine levels following smoking cessation, suggesting a theoretical risk of clozapine toxicity in these circumstances. Nicotine replacement therapy does not change clozapine levels (Cormac et al., 2010; Couchman et al., 2010; Sandson et al., 2007). Clinicians prescribing clozapine need to consider patient smoking behaviours and should measure plasma clozapine levels when there is a change in smoking patterns. These changes are most likely to occur with a change in treatment environments between community and inpatient care.
Changes in clozapine levels may occur with the commencement or cessation of other medications (Greenwood-Smith et al., 2003; Swannick, 2010). Importantly some psychotropic medications change plasma clozapine levels. For example, the selective serotonin reuptake inhibitors (SSRIs) and haloperidol are CYP2D6 inhibitors that can reduce clozapine metabolism, thereby increasing plasma clozapine levels. Increases in clozapine levels that arise from the concomitant prescribing of psychotropic medications could potentially increase the risk of clozapine toxicity. Changes in clozapine levels are also possible in patients taking medications that are highly plasma protein bound such as warfarin (Greenwood-Smith et al., 2003; Swannick, 2010). Medications that have the potential to alter clozapine levels are shown in Table 1.
Influence of medications on clozapine levels.
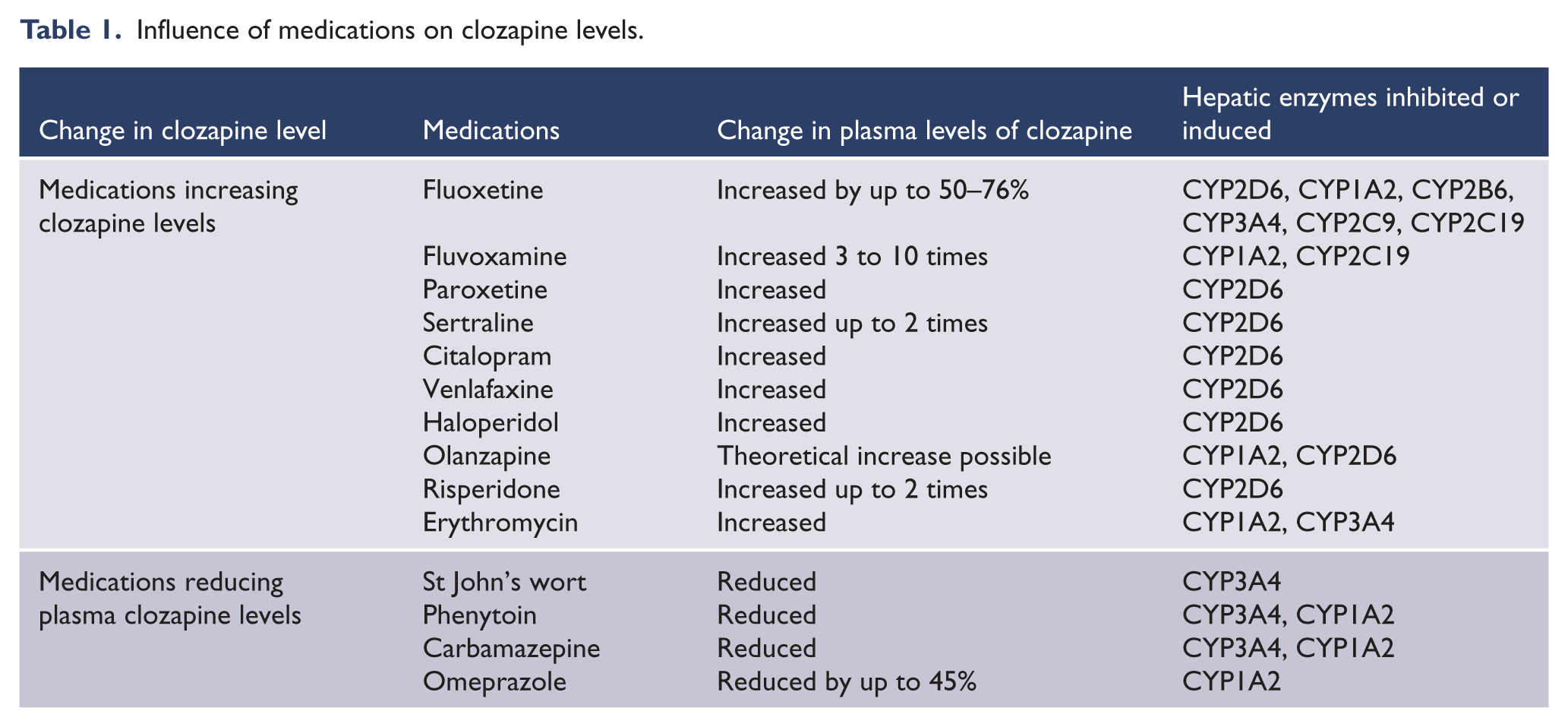
There are a large number of other possible causes for changes in clozapine metabolism. Clozapine levels can be increased by caffeine consumption, due to competition for metabolism by CYP1A2 (Couchman et al., 2010; Greenwood-Smith et al., 2003; Haring et al., 1990; Raaska et al., 2004; Swannick, 2010; Taylor, 1997; van Troostwijk et al., 2003). Two literature reviews reported that chargrilled meat induced CYP1A2 (Greenwood-Smith et al., 2003; Taylor, 1997), resulting in a reduction in plasma clozapine levels. Stress, hypoalbuminaemia, and renal and hepatic failure have the potential to increase clozapine levels (Bell et al., 1998). Clozapine levels are relatively higher in people of Asian heritage compared to those of Caucasian background, probably related to lower CYP1A2 activity in people of Asian descent (Chang et al., 1997; Chong et al., 1997; Greenwood-Smith et al., 2003; Matsuda et al., 1996; Nielsen et al., 2010; Wong et al., 2006). Clozapine levels are also generally higher in females compared to males, with rates of increase varying from 17.0% to 24.9% (Aichhorn et al., 2006; Couchman et al., 2010; Flanagan et al., 2005; Greenwood-Smith et al., 2003; Haring et al., 1990; Lane et al., 1999; Palego et al., 2002; Tang et al., 2007).
There are conflicting opinions as to whether age impacts on clozapine levels, with some studies supporting increased levels with increasing age (Couchman et al., 2010; Haring et al., 1990; Lane et al., 1999; Meyer, 2001) and others finding no relationship (Mauri et al., 2004; Ozdemir et al., 2001; Palego et al., 2002; Tang et al., 2007). Two studies reported a significant negative correlation between clozapine levels and weight (Couchman et al., 2010; Haring et al., 1990), but three found no relationship (Lane et al., 1999; Ozdemir et al., 2001; Palego et al., 2002). There were case reports where infection or inflammation was linked to an increase in clozapine levels in the absence of dose changes. It was postulated that inflammatory cytokines reduced CYP1A2 activity (Couchman et al., 2010; de Leon and Diaz, 2003; Haack et al., 2003). Alternative explanations of reduced smoking or reduced ability to absorb the hydrocarbons from the smoke during respiratory infection have been suggested (Couchman et al., 2010).
Accuracy of laboratory measurement of clozapine has been identified as a potential factor affecting reported plasma clozapine levels. Although two studies reported that laboratory measurements of plasma clozapine were reliable (Bell et al., 2001; Freudenreich et al., 1996), two other studies found significant discrepancies in clozapine levels between different laboratories using equal, calibrated samples (Greenwood-Smith et al., 2003; Oo et al., 2006), suggesting measurement error. No differences were detected in clozapine levels associated with the different brands of clozapine (Couchman et al., 2010).
Clinicians prescribing clozapine need to consider both static and dynamic factors that can affect clozapine levels. Monitoring of plasma clozapine levels, particularly when there are changes in a patient’s pharmacotherapy or cigarette use, is recommended to ensure safe prescribing of clozapine.
Clozapine levels and adverse events
Most studies found minimal correlation between clozapine levels and side effects, with the exception of sedation, hypotension and seizure activity (Bell et al., 1998; de Leon and Simpson, 2004; Greenwood-Smith et al., 2003; Jaskiw et al., 2008; Liu et al., 1996; Mauri et al., 2004; Spina et al., 2000a; Taylor, 1997; Wong et al., 2006; Yusufi et al., 2007). Higher clozapine levels (> 600 µg/L) or doses (> 600 mg/day) and faster rate of upward titration are associated with increased risk of seizure activity (Bell et al., 1998; Cooper, 1996; Couchman et al., 2010; Devinsky et al., 1991; Greenwood-Smith et al., 2003; McKean et al., 2008; Miller, 2000; Welch et al., 1994). Higher clozapine levels may also be associated with metabolic side effects, hypersalivation, constipation, tachycardia, QTc prolongation, myocarditis and cardiomyopathy (Couchman et al., 2010; Simon et al., 2009).
The clozapine level associated with toxicity remains unclear, but most articles suggested levels greater than 600–1000 µg/L were more likely to cause adverse effects, particularly seizure activity (Bell et al., 1998; Buckley et al., 2001; Cooper, 1996; Cormac et al., 2010; Couchman et al., 2010; Flanagan et al., 2003, 2005; Freeman and Oyewumi, 1997; Greenwood-Smith et al., 2003; Jaskiw et al., 2008). One article reported that plasma clozapine levels associated with acute toxicity are usually above 2000 µg/L (Flanagan et al., 2005). However, there have been reports of patients with clozapine levels over 3000 µg/L with no evidence of associated toxicity (Flanagan et al., 2003). It is important to note that toxicity is more likely in novice users who have not yet developed tolerance to clozapine, or in non-adherent patients inconsistently exposed to their prescribed dose. In these circumstances, 200–400 mg could be life-threatening (Couchman et al., 2010; Flanagan et al., 2005; Swannick, 2010). For this reason, slow upward titration as recommended by protocols is important when initiating clozapine therapy. Conversely, covert accumulation of clozapine can occur in patients who have developed tolerance to clozapine over a prolonged period, as they may not manifest signs of toxicity at higher levels (Couchman et al., 2010). Therefore, risk of clozapine toxicity is dependent on dosage of medication, accumulated dosage, clozapine levels and tolerance to the medication. We suggest ongoing monitoring of clozapine levels in patients who are prescribed higher doses (> 600 mg/day) to help in assessing the risk of toxicity.
Use of clozapine levels post-mortem
Clozapine toxicity is a potential cause of death in patients treated with this medication. We provide an overview of what is known about the use of clozapine levels post- mortem, specifically considering their application for establishing clozapine toxicity as a cause of death.
Clozapine post-mortem redistribution
After death, most drugs undergo a process called post-mortem redistribution (PMR). Drugs diffuse between tissues and blood vessels, thereby altering their concentrations at varying sites (Butzbach, 2010; Cook et al., 2000; de Leon and Simpson, 2004; Drummer and Gerostamoulos, 2002; Ferner, 2008; Flanagan et al., 2003; Hilberg et al., 1993; Rodda and Drummer, 2006). In the post-mortem period, drugs can be released from the gastrointestinal tract, liver, lungs, myocardium and striated muscle into central blood vessels and to a lesser extent peripheral vessels (Cook et al., 2000; Flanagan et al., 2003; Hilberg et al., 1993; Pelissier-Alicot et al., 2003; Pounder and Jones, 1990; Yarema and Becker, 2005). PMR is more pronounced with lipophilic drugs with large volumes of distribution exceeding 3 L/kg, such as clozapine, as they have higher ante-mortem tissue concentrations (Cook et al., 2000; Drummer and Gerostamoulos, 2002; Ferner, 2008; Flanagan et al., 2003, 2005; Jaskiw et al., 2008; Kerswill and Vicente, 2003; Pelissier-Alicot et al., 2003; Rodda and Drummer, 2006; Yarema and Becker, 2005).
Table 2 shows the known factors that influence post-mortem drug concentrations. Clinical treatment prior to death, storage of the body post-mortem, time to sampling and the method and location of collection may increase or decrease the PMR of drugs and therefore alter post-mortem drug concentrations (Cook et al., 2000; Flanagan et al., 2003; Hargrove and McCutcheon, 2008; Kerswill and Vicente, 2003; Pelissier-Alicot et al., 2003; Pounder and Jones, 1990; Yusufi et al., 2007).
Factors that influence post-mortem drug concentrations.
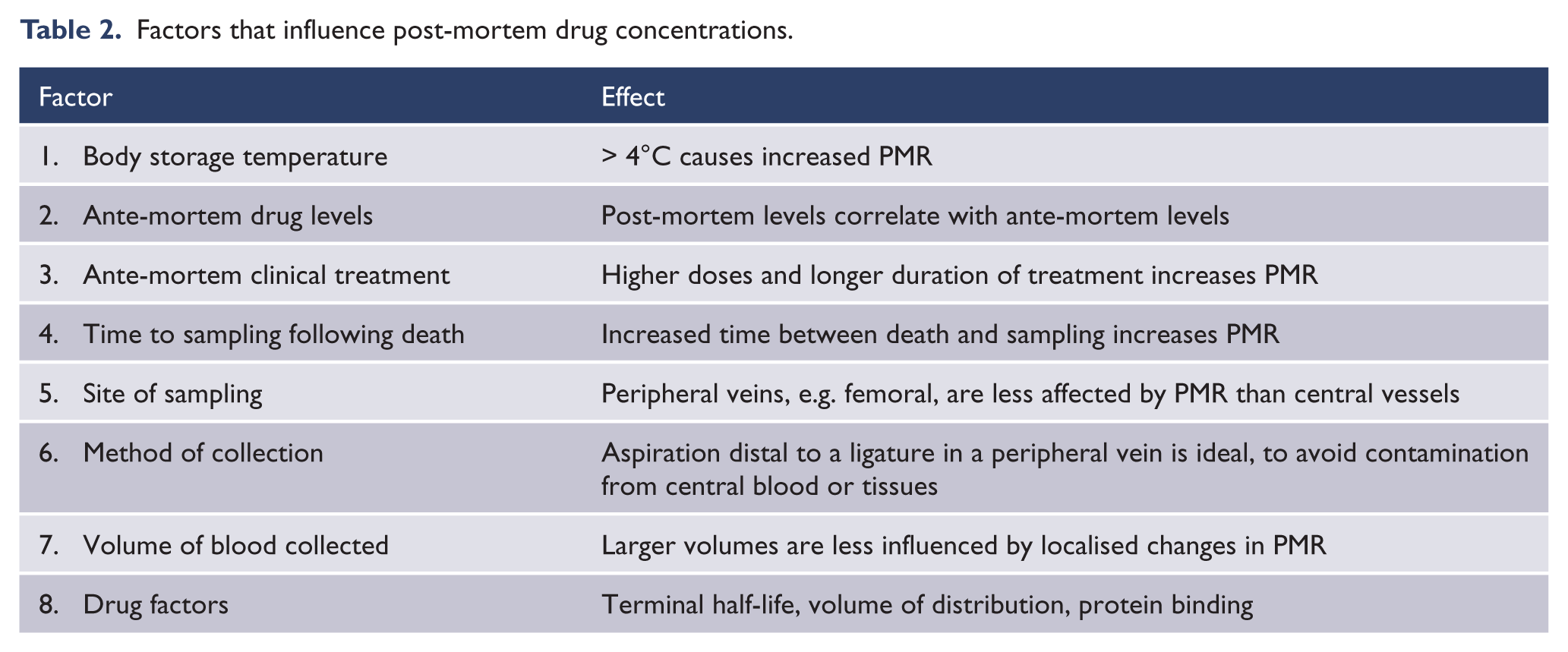
Unfortunately there are a paucity of studies comparing ante-mortem and post-mortem plasma clozapine levels, due to lack of available ante-mortem samples close to the time of death and inadequate documentation of post-mortem sampling techniques (Cook et al., 2000; Flanagan et al., 2003, 2005). Existing case reports found a 3.00 to 4.89 fold elevation of clozapine levels (and 3.70 fold increase in norclozapine levels) in central blood vessels in cases where recent ante-mortem levels were available for comparison (de Leon and Simpson, 2004; Flanagan et al., 2005; Jaskiw et al., 2008). Another study found clozapine levels in peripheral blood vessels post-mortem were up to 1.5 times higher than the corresponding ante-mortem levels (Flanagan et al., 2003). Post-mortem clozapine levels in myocardium and striated muscle have been recorded up to 2–3 times higher than ante-mortem blood levels (Flanagan et al., 2003). There are multiple case reports of significant elevation of post-mortem concentrations of other antipsychotics (chlorpromazine, thioridazine and risperidone) and the tricyclic antidepressants. As these medications have similar lipophilic properties to clozapine, these case reports represent indirect evidence of the PMR that is also likely to occur with clozapine (Cook et al., 2000; Drummer and Gerostamoulos, 2002; Ferner, 2008; Hilberg et al., 1993, 1999; Pounder and Jones, 1990; Rodda and Drummer, 2006).
The literature suggests that post-mortem clozapine level increases due to PMR would be higher in patients who had been on long-term or high-dose clozapine therapy, due to an increased ante-mortem tissue burden of clozapine in these patients (Couchman et al., 2010; Flanagan et al., 2003, 2005). Two articles recommended quantifying the clozapine to norclozapine ratio (normally around 1.32) in post-mortem blood, as this ratio is increased between 4.4 and 7.6 times in cases of recent clozapine overdose (Flanagan et al., 2003, 2005).
Liver tissue, hair or nail analysis may provide further information on the nature and magnitude of longer-term clozapine exposure, but there are few facilities able to test these samples and there is limited guidance available on interpretation of the results of such testing (Flanagan et al., 2003, 2005; Paz et al., 2008). Biopsy of heart muscle is also essential to look for eosinophilic myocarditis as a possible cause of death in clozapine-treated patients (Flanagan et al., 2003, 2005).
The literature was consistent in advising against extrapolations of ante-mortem medication levels from post-mortem levels, due to the unpredictable nature of PMR and the limited understanding of its quantitative impact (Butzbach, 2010; Cook et al., 2000; de Leon and Simpson, 2004; Drummer and Gerostamoulos, 2002; Ferner, 2008; Flanagan et al., 2003, 2005; Haring et al., 1990; Hilberg et al., 1999; Jaskiw et al., 2008; Kerswill and Vicente, 2003; Pelissier-Alicot et al., 2003; Pounder and Jones, 1990; Rodda and Drummer, 2006; Shepherd et al., 1992; Yarema and Becker, 2005). The importance of using the ante-mortem medical history in conjunction with post-mortem findings when determining cause of death was emphasised (Ferner, 2008).
Discussion
This review highlights the many challenges facing clinicians prescribing clozapine in the clinical care of their patients. Recommended clozapine dose ranges will not be appropriate for many patients. Although there are limitations for using plasma clozapine levels to inform efficacy, risk of adverse events and future risk of toxicity, there is emerging consensus that they are of value in prescribing clozapine safely and effectively. In spite of the challenges in clozapine monitoring, it remains our most effective antipsychotic medication for treatment-refractory schizophrenia (Bell et al., 1998; Couchman et al., 2010; Green and Schildkraut, 1995; McKean et al., 2008; Meyer, 2001; Miller, 2000; Mitchell, 2001; Oyewumi et al., 2002; Raedler et al., 2008; Sandson et al., 2007; Schulte, 2003, Taylor, 1997; Taylor and Duncan-McConnell, 2000). It has been associated with a significant reduction in morbidity and mortality despite its potential for causing serious adverse effects. Clinicians must be vigilant in detecting factors that can change clozapine levels and patients receiving clozapine should be monitored regularly for side effects.
Post-mortem clozapine levels may be significantly and unpredictably increased by PMR, which is influenced by a large number of factors (Flanagan et al., 2003; Hargrove and McCutcheon, 2008; Kerswill and Vicente, 2003; Pelissier-Alicot et al., 2003; Pounder and Jones, 1990; Yarema and Becker, 2005). The sole use of post-mortem clozapine levels for determining ante-mortem clozapine toxicity is unreliable. The majority of the ante-mortem and post-mortem clozapine level comparative data in this review was derived from case studies. It is unlikely that studies using better methodology will be readily available given the inherent difficulty in assuring ante-mortem clozapine levels are done in close proximity to an unpredictable event such as death.
When considering the results of this literature review, combined with the absence of clinical evidence of ante-mortem clozapine toxicity in the two patients who died whilst receiving clozapine, it seems likely that the elevated post-mortem clozapine levels obtained at autopsy were caused by PMR rather than ante-mortem clozapine toxicity. An understanding by psychiatrists, pathologists and coroners that PMR increases clozapine levels after death will reduce the risk of clozapine toxicity being misattributed as a cause of death in similar cases.
This paper uses the methodology of a narrative review with the accompanying risk of biased decisions on article relevance and interpretation of data within the literature. In spite of this limitation, this review provides an overview that may assist clinicians in safely prescribing clozapine and interpreting plasma clozapine levels in their patients. It may also assist in providing information for future determinations of cause of death in clozapine-treated patients.
Conclusions
Clozapine level monitoring is indicated, especially when (i) prescribing higher doses (> 600 mg/day) of clozapine, (ii) where there has been a change in a patient’s concomitant pharmacotherapy or cigarette use and (iii) where there has been a sub-optimal response to treatment.
The prediction of ante-mortem clozapine levels based on post-mortem findings is unreliable. In concluding cause of death in clozapine-treated patients, evaluation of the ante-mortem clinical presentation, especially considering signs of toxicity and use of ante-mortem clozapine levels, is more reliable than post-mortem clozapine levels in determining the role of clozapine toxicity as a contributing factor.
Footnotes
Acknowledgements
The authors would like to thank Mr Michael Duhig for his contribution to managing the references for the paper.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
