Abstract
Introduction
Major depressive disorder (MDD) is associated with disturbances in inhibitory control (Langenecker et al., 2007, 2005) such that patients have difficulty disengaging attention due to impairments in response inhibition mechanisms (Jorman, 2004; Joorman and Gotlib, 2008; Jorman et al., 2006; Milliken and Tipper, 1998). Inhibitory control is the ability to deliberately restrain responses to maintain behaviour through thought and action (Fuster, 1997). It allows for dominant, automatic or prepotent responses to be deliberately inhibited to facilitate task-relevant processing (Friedman and Miyake, 2004; MacDonald et al., 2000). However, depression is a heterogeneous disorder (Gold and Chrousos, 2002; Parker, 2000, 2007), which may differentially affect inhibitory control processes. A number of studies have reported that depressed patients display impairments in inhibitory control (George et al., 1997; Langenecker et al., 2007; Wagner et al., 2006), for example depressed patients (n = 20) required greater activation of frontal, limbic and temporal areas during a cognitive Go/No Go inhibitory control task (Langenecker et al., 2007). Similar findings have been reported for executive function tasks, such that depressed patients display greater activity in the frontal cortex to sustain task performance (George et al., 1997; Harvey et al., 2005; Holmes et al., 2005). Increased activation across a range of neural networks may reflect a functional compensation involving greater recruitment of regulatory systems to process information (Langenecker et al., 2007). Moreover, melancholic depression has been associated with perseveration (Gold and Chrousos, 2002) suggesting that patients with melancholia may display impairments in inhibitory processes. The current study aims to investigate the impact of melancholic and non-melancholic depression on inhibitory control mechanisms.
The ability to inhibit irrelevant information has been associated with selective attention, one of the most common cognitive deficits in MDD (Kertzman et al., 2010). Impaired performance on the Stroop interference task (Stroop, 1935), a common measure of inhibitory control and selective attention (Landrø et al., 2001; Moritz et al., 2002), is predicted by psychomotor slowness and vigilance levels in MDD patients (Kertzman et al., 2010), both key features of melancholia (Taylor and Fink, 2006). In addition, patients with melancholia also exhibit cognitive deficits such as perseveration and self-focused recursive thinking (Gold and Chrousos, 2002), i.e. rumination. Rumination has been associated with increased activity of the limbic, medial and dorsolateral prefrontal regions in depression (Cooney et al., 2010) demonstrating some overlap with the areas activated during inhibitory control tasks (George et al., 1997; Langenecker et al., 2007; Wagner et al., 2006). Rumination, vigilance and perseveration have also been associated with anxious arousal (Gold and Chrousos, 2002; Joorman 2006; Taylor and Fink, 2006) and rumination in particular has been associated with reduced inhibition (Cooney et al., 2010). In contrast to melancholia, non-melancholic patients exhibit anxious apprehension and worry (Malhi et al., 2002; Parker, 2000) rather than anxious arousal. As rumination has been associated with reduced inhibition, further investigation of depression subtypes may reveal differences in inhibitory control for MDD patients.
Event-related potential (ERP) studies have examined the temporal processing stream associated with inhibitory control tasks. The N200 and post-stimulus 300 (P300) are major components associated with inhibitory control and evaluative processing (Roche et al., 2005; Schmajuk et al., 2006). The No-Go trial competes and conflicts with the Go trial (Nieuwenhuis et al., 2003) and it is the N200 that plays a key role in conflict processing. The N200 deflection relating to the No-Go trial reflects a cognitive and top-down inhibitory mechanism that suppresses incorrect responses (Kaiser et al., 2003; Kim et al., 2007) evident over frontocentral electrodes at approximately 200–300 ms following stimulus presentation during inhibition (No-Go) trials (Eimer, 1993; Falkenstein et al., 1999; van Boxtel et al., 2001). The P300 also relates to response inhibition in No-Go trials (Bekker et al., 2005; Bruin and Wijers, 2002; Smith et al., 2008) reflecting the termination of response conflict (Dimoska et al., 2006) and inhibition of motor responses (Ruchsow et al., 2008a; Verlerger et al., 2006). The P300 is a positive deflection evident over frontocentral electrodes for No-Go trials around 300–600 ms post-stimulus when participants elicit a behavioural response (Bruin and Wijers, 2002; Falkenstein et al., 1999; Hermann et al., 2003; Pfefferbaum et al., 1985; Strik et al., 1998; Tekok-Kilic et al., 2001).
Contradictory findings have been reported across ERP studies that have examined inhibitory control in MDD. For example, Kaiser et al. (2003) employed an auditory inhibitory control task where participants were required to respond to either rare or frequent tones dependent on instruction, and reported that MDD patients (n = 16) display a reduced No-Go-related N200 component, while the P300 component was unaffected. By contrast, Ruchsow et al. (2008b) observed the opposite effect in a visual inhibitory control task in which participants were required to respond to specific letters within congruent and incongruent letter strings (i.e. a cognitive flanker task). This latter study (Ruchsow et al., 2008b) reported that MDD patients (n = 21) display a decreased No-Go-related P300 component, while the N200 component is unaffected relative to controls. An obvious explanation for these incongruent results is task modality. While the cognitive flanker task assesses visuospatial conflict, the Go/No-Go auditory task assesses acoustic conflict. Furthermore, functional MRI (fMRI) evidence indicates that the neural basis of response inhibition is task dependent (Mostofsky et al., 2003; Simmons et al., 2008) such that activation is dependent on whether the task involves visual or auditory discrimination of a stimulus. Our study extends on these findings by examining response inhibition in patients with and without melancholia.
An event-related negativity (ERN) component is observed in response to an error. This occurs even when participants are not explicitly aware they have made an incorrect response and this component may be exacerbated due to negative affect (Hajcak et al., 2004a; Luu et al., 2000). Patients with MDD display increased ERN at 50–100 ms after an incorrect response (Chiu and Deldin, 2007; Falkenstein et al., 1999; Gehring et al., 1993; Holmes and Pizzagalli, 2008; Ruchsow et al., 2004). The ERN is an electrophysiological correlate of response monitoring that occurs in response to signals of negative feedback (Olvet and Hajcak, 2008). This negative feedback signal is increased in depression, consistent with an increased sensitivity to error and negative information (Elliot et al., 1998; Steffens et al., 2001). Furthermore, decreased accuracy is often observed following errors in patients with depression (Holmes and Pizzagalli, 2008; Pizzagalli et al., 2006). As such it will be important to determine whether electrophysiological differences are observed when examining ERPs associated with correct responses only.
We have previously reported increased P200 amplitude, delayed N200/P200 complex and decreased P300 amplitude in patients with clinical depression and participants with depressed mood during a cognitive auditory oddball paradigm (Kemp et al., 2009). These findings were particularly prominent in patients with melancholia (n = 57) relative to patients without melancholia (n = 48) and controls (n = 115) (Kemp et al., 2010) suggesting there may be an effect of depression subtype and severity on temporal processing in MDD during a selective attention task in the auditory domain. The delayed N200/P300 complex was also found to correspond to the slowed behavioural responses to targets in melancholia. These findings were obtained even after removing ERPs associated with incorrect responses. Slowed processing may reflect an overprocessing of stimuli (reflected in an augmentation of the P200 component) which then impairs and delays evaluation of target significance (Kemp et al., 2009). The present study investigated whether these findings could be generalized to inhibitory control.
The primary aim of the current study was to examine the impact of depression on inhibitory control in a large sample of melancholic, non-melancholic and healthy control participants. It was hypothesized that decreased P300 would be observed in melancholia in particular, relative to non-melancholia and controls on a simple visual cognitive Go/No-Go task consistent with findings reported by Ruchsow et al. (2008b). Furthermore we expected errors of commission to be higher in melancholia in comparison to non- melancholia and controls. A secondary aim of the study was to investigate whether other characteristics such as depression severity, anxious arousal, anxious apprehension and psychomotor slowing contribute to behavioural and electrophysiological impairments. We expected that greater levels of anxious arousal would be associated with melancholia and consequent disturbances within the temporal processing stream and behavioural impairments.
Methods
Participants
One-hundred and twenty-eight participants with a primary diagnosis of MDD and 128 healthy control participants were recruited from the community in collaboration with the Brain Resource International Database (Gordon et al., 2005). All depressed participants met the criteria for MDD as determined by trained and supervised research officers using the Mini International Neuropsychological Interview (MINI; Sheehan et al., 1998), a structured psychiatric interview based on the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria. All patient participants were diagnosed with MDD as a primary diagnosis while secondary diagnoses included generalized anxiety disorder (n = 45), panic disorder (n = 29), posttraumatic stress disorder (n = 14) and substance abuse (n = 33). All participants gave written informed consent in accordance with National Health and Medical Research Council (NHMRC) ethical guidelines. Exclusion criteria included a history of brain injury (causing loss of consciousness for 10 min or more), neurological disorder, other serious medical condition or substance abuse or dependence for longer than 1 year. Healthy control participants were excluded if they self-reported a history or presence of psychiatric illness. All participants were medication free for at least five half-lives.
Procedure
Depression severity was assessed using the Hamilton Rating Scale for Depression (HRSD; Hamilton, 1960) and psychomotor disturbance was measured by the CORE Assessment of Psychomotor Change (CORE; Hickie, 1996). Patients were diagnosed as melancholic (MEL; N = 60) or non-melancholic (NMEL; N = 54) on the basis of the MINI. The self-report, Depression Anxiety and Stress Scale (DASS; Lovibond and Lovibond, 1995) was completed by participants at completion of the clinical interview. Participants then completed a cognitive Go/No-Go task while electroencephalogram (EEG) data was collected. Participants were fitted with a Quik-Cap (Neuroscan, Compumedics Limited, Abbotsford, Victoria, Australia) for EEG recording. Task instructions were pre-recorded and delivered in a standardized computer format using visual and audio instructions (via headphones).
Go/no-go task
In this task, subjects were required to respond during ‘Go’ trials and then to withhold this pre-potent response during ‘No-Go’ trials; inhibition of responding during ‘No-Go’ trials was employed as a measure of executive inhibitory control (Aron, 2007; Rubia et al., 2001). The inability to withhold a response, known as the commission error (CE), reflects a deficit in inhibitory control. Go stimuli (‘PRESS’, presented in green-coloured type in the centre of a black screen) and No-Go stimuli (‘PRESS’ in red-coloured type in the centre of a black screen) were presented to participants on a computer screen. The word ‘PRESS’ was presented 6 times in a row, with a total of 28 sequences. Twenty-one trials were randomly presented in green and seven in red. Thus ‘Go’ stimuli were presented 75% of the time (total Go stimuli = 126), creating a tendency to respond, and the ‘No-Go’ stimuli 25% of the time to gain an accurate measure of response inhibition (total ‘No-Go’ stimuli = 42). The stimuli were presented on screen for 500 ms with an interstimulus interval (ISI) of 1 s. The total task duration was 5 min. Participants received standardized instructions to press a response box as quickly as possible when the Go stimulus appeared and to withhold responding to No-Go stimuli. Participants were instructed to press the response box with their left and right thumbs simultaneously. Speed and accuracy were equally stressed in task instructions. Behavioural measures included omission errors (OEs: failure to respond when required), CEs (inability to withhold a response when required), response time (RT) and variability of response time (RTsd).
Measurement
The EEG data was recorded from 26 standard electrode sites positioned according to the 10/20 international system. Electrode sites included Fp1, Fp2, F7, F3, Fz, F4, F8, FC3, FCz, FC4, T3, C3, Cz, C4, T4, CP3, CPz, CP4, T5, P3, Pz, P4, T6, O1, Oz and O2. Data was recorded relative to the average of the A1 and A2 (mastoid) electrode sites and an electrode on the forehead was used as a ground. Horizontal eye movements were recorded with electrodes placed 1.5 cm lateral to the outer canthus of each eye, and vertical eye movements were recorded with electrodes placed 3 mm above the middle of the left eyebrow and 1.5 cm below the middle of the left bottom eyelid. Electrode impedance was generally maintained below < 5kOhm. A continuous acquisition system was employed with a sampling rate of 500 Hz and data were corrected offline for electro-oculogram (EOG) artefact analysis (Gratton et al., 1983).
ERP data focusing on each No-Go stimulus trial were extracted from the EEG recordings. Each single-trial waveform was filtered at 25 Hz with a Tukey or cosine taper to 35 Hz, above which no signal was passed. Conventional ERP averages were then formed for each correct response to the No-Go stimuli, thus controlling for incorrect responses. Error rates have been found to confound ERP amplitude depending on the number of errors made (Amodio et al., 2008; Gehring et al., 1993; Hajcak et al., 2003, 2004b; Yeung, 2004). ERPs were scored using an automated algorithm and validated by experienced scorers. Variables were screened for outliers and cases with data > 3.5 standard deviations from the mean of all cases (regardless of group) were removed from the data set. Eight per cent of the data set was removed following this procedure.
Statistical analyses
All statistical analyses were performed using SPSS, version 17 (SPSS Inc., Chicago, Illinois, USA). Significant effects were set at p < 0.05, while findings were labelled as trends if p < 0.1 and p > 0.05. The impact of depression subtype on inhibitory control was investigated using repeated measures ANOVAs for amplitude and latency of each ERP component (P200, N200 and P300) that occurred to the No-Go stimuli. As a data reduction technique, the N100 component was not examined in this study. This component is associated with early attentional orienting (Doallo et al., 2005) rather than response inhibition, and is not considered to be associated with the top-down processing mechanism associated with inhibitory processes (Bokura et al., 2001). A group (MEL, NMEL, CTRL) by electrode site (midline: Fz, Cz, Pz; frontal: F3, F4) design was utilized. In order to establish a baseline and provide greater clarity on the influence of MDD on inhibitory control, we initially examined the differences between MDD (including both melancholic and non-melancholic patients) and control participants. No ERP differences were observed. Main and interaction effects of group were analysed using post-hoc, pairwise, between-group comparisons and the Tukey correction for multiple comparisons. ANOVAs were also conducted to examine group differences (MEL vs NMEL vs CTRL) on behavioural data including total number of CEs, total number of OEs, RT and RTsd. The relationships between clinical and behavioural variables were also investigated.
Results
Participant characteristics
Patients with melancholia displayed increased scores on all clinical measures than those without melancholia and controls respectively (see Table 1). HRSD [F(2, 225) = 981.066, p < 0.001], DASS-D [F(2, 225) = 430.453, p < 0.001], DASS-A [F(2, 225) = 95.686, p < 0.001], DASS-S [F(2, 225) = 223.795, p < 0.001] and the CORE total score [F(2, 225) = 85.394, p < 0.001]. Increased levels of depression severity, psychomotor disturbance and anxious arousal are consistent with current conceptualizations of melancholia (Parker, 2007, 2000; Hickie, 1996). There were no significant differences for group on gender [χ2 (2, N = 228) = 0.418, p = 0.812] or age [F(2, 225) = 0.890, p = 0.412].
Participant characteristics
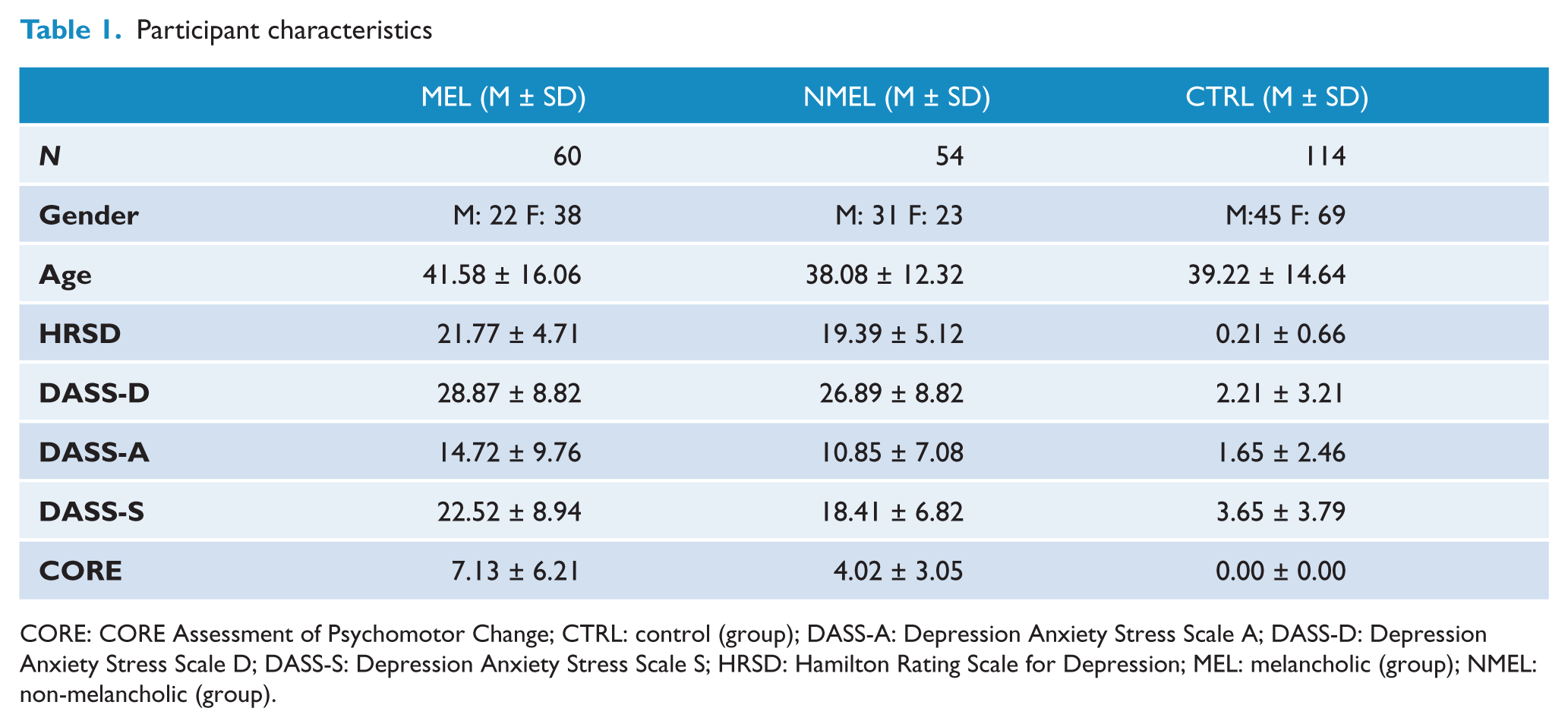
CORE: CORE Assessment of Psychomotor Change; CTRL: control (group); DASS-A: Depression Anxiety Stress Scale A; DASS-D: Depression Anxiety Stress Scale D; DASS-S: Depression Anxiety Stress Scale S; HRSD: Hamilton Rating Scale for Depression; MEL: melancholic (group); NMEL: non-melancholic (group).
Behavioural data
Groups differed significantly on CEs [F(2, 225) = 3.78, p = 0.024] and standard deviation of reaction time [F(2, 225) = 3.05 p = 0.049]. Means and standard deviations per group on cognitive measures are listed in Table 2. Post-hoc tests revealed that patients with melancholia display a greater number of CEs compared to non-melancholic patients (CE: MD = 0.823, p = 0.042) and controls (MD = 0.697, p = 0.043) respectively (see Figure 1) and an increase in reaction time variability (i.e. increased standard deviation) in comparison to controls (MD = 17.020, p = 0.051). No differences were observed between non-melancholic patients and controls on these measures (see Figure 2). No significant differences were found between groups on OEs or reaction time.
Behavioural findings
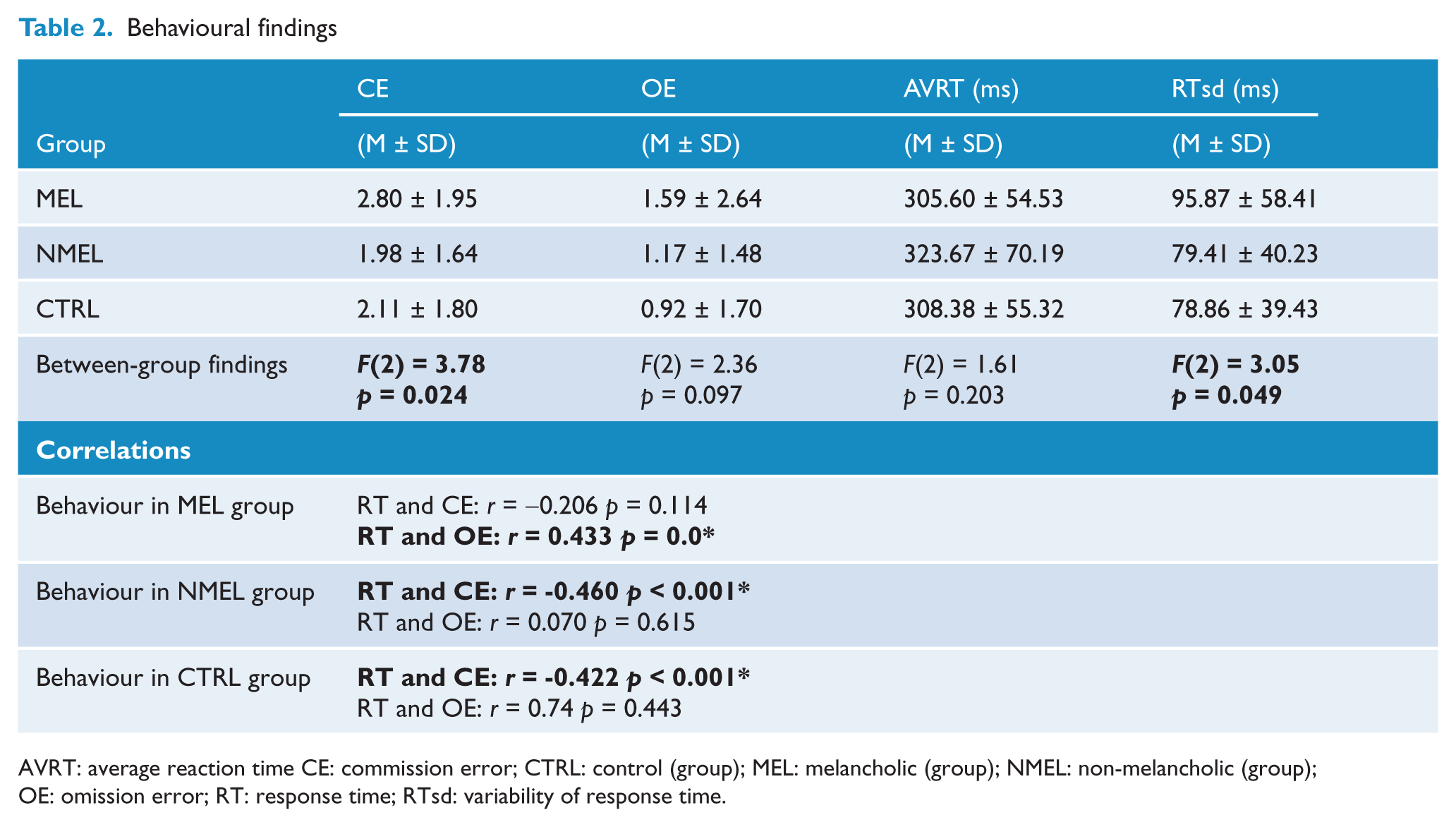
AVRT: average reaction time CE: commission error; CTRL: control (group); MEL: melancholic (group); NMEL: non-melancholic (group); OE: omission error; RT: response time; RTsd: variability of response time.
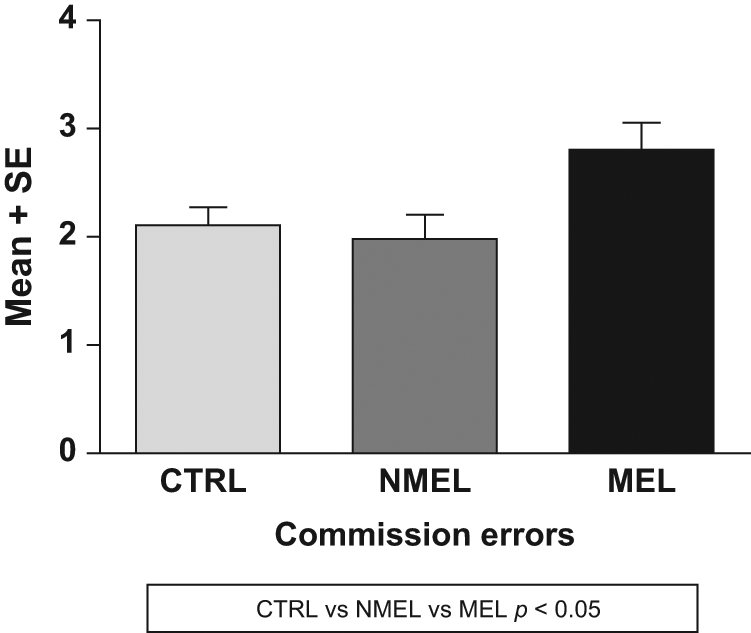
Means and standard errors of commission errors per group. CTRL: control (group); MEL: melancholic (group); NMEL: non-melancholic (group).
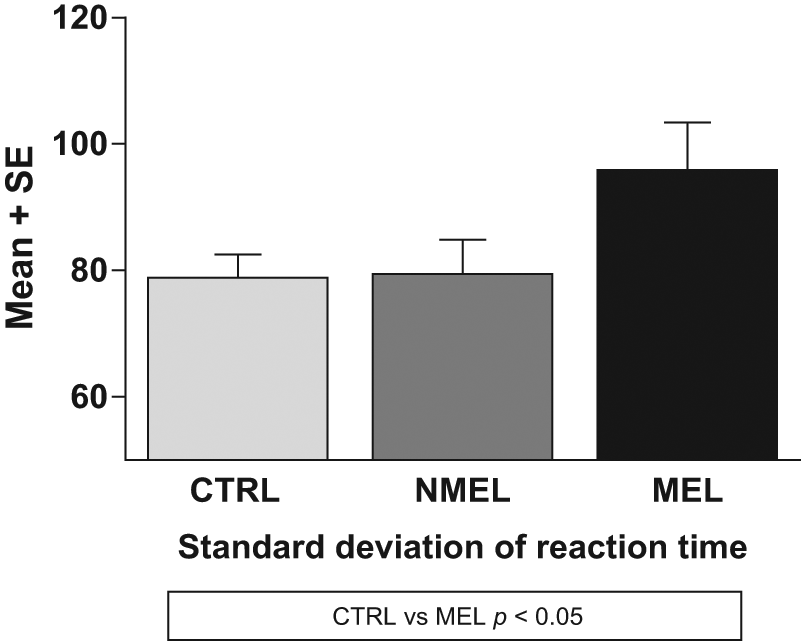
Means and standard errors of the standard deviation of reaction time per group. CTRL: control (group); MEL: melancholic (group); NMEL: non-melancholic (group).
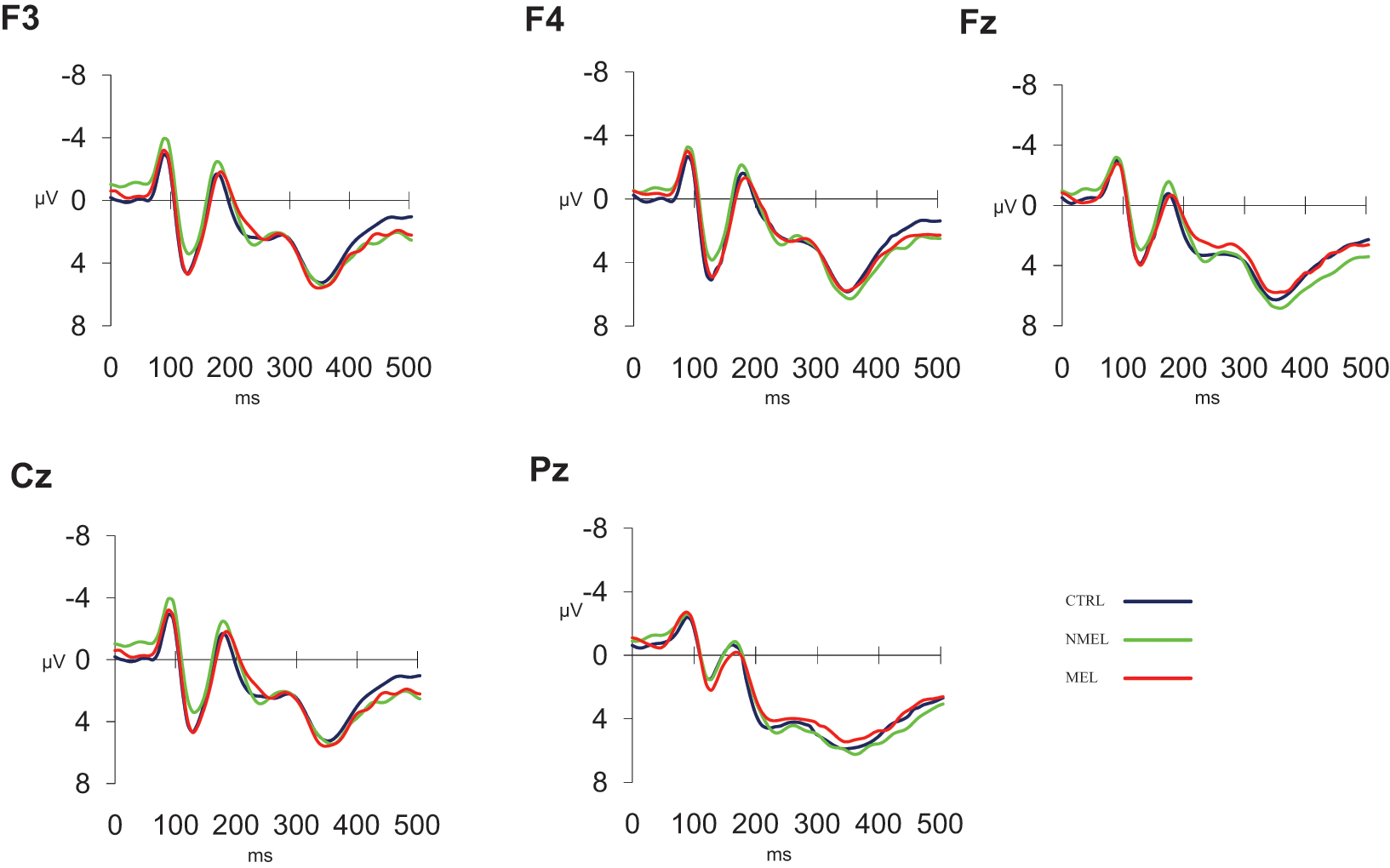
ERP waveforms in response to No-Go trials for frontal (F3, F4) and midline (Fz, Cz, Pz) components.
CEs were positively correlated with psychomotor disturbance (r = 0.40, p < 0.01) and depression severity (as measured by the HRSD; r = 0.34, p = 0.01) for melancholic patients only.
ERP data
No significant differences were found between groups (MEL vs NMEL vs CTRL) at frontal or midline locations for P200, N200 or P300 components.
Discussion
The current study examined the impact of depression heterogeneity on inhibitory control. Patients with melancholia made a greater number of CEs relative to patients with non-melancholia and control participants. Furthermore, CEs were correlated with depression severity and psychomotor disturbance for melancholic patients only. While anxious arousal was greater in patients with melancholia relative to those without melancholia and controls, anxious arousal did not correlate with any behavioural variable for either subtype. Behavioural results indicate that patients with melancholia generate more incorrect responses and these responses are, in part, associated with depression severity and psychomotor disturbance. After controlling for differences in CEs, no ERP abnormalities were observed between depression subtypes and healthy controls. These findings demonstrate that conflict processing and the termination of response conflict is similar in depressed patients and controls. As such when patients with depression generate correct responses, there is no apparent electrophysiological impairment in the brains of melancholic or non-melancholic patients with depression. Furthermore, unlike our prior studies on selective attention (Kemp et al., 2009, 2010), cognitive inhibition does not appear to be associated with an overprocessing of stimuli in patients with depression.
Decreased accuracy is often observed following errors during behavioural tasks in patients with depression (Holmes and Pizzagalli, 2008; Pizzagalli et al., 2006). In the current study, we observed an increased variability in reaction time in patients with melancholia, which may be due to patients making adjustments to increase their accuracy. Interestingly, CEs are correlated with increased psychomotor disturbance, suggesting that any attempt to improve accuracy – as indicated by variability in reaction time – will be unsuccessful. By contrast, when correct responses are made, no electrophysiological differences are observed suggesting that the divergence between behavioural and ERP findings may relate to error processing for the Go/No-Go task.
The ERN is a potential endophenotype for depression, anxiety and substance use disorders (Olvet and Hajcak, 2008). This negative feedback signal is enhanced in depression where patients exhibit increased sensitivity to error and negative information (Elliot et al., 1998; Steffens et al., 2001). The ERN complex found in response to errors and incorrect responses may be exacerbated due to negative affect (Hacjak et al., 2004b; Luu et al., 2000), a core component of depression and anxiety. One explanation for the behavioural impairments observed in melancholia is overactivity of the cholinergic system which may be associated with increased levels of arousal (Dagyté et al., 2010). Overactivity in this system may lead to a reduction in signal detection threshold, an overprocessing of stimuli and hyperattentive impairments which may contribute to the negative biases observed in depression (Kemp et al., 2009, 2010 ). Here we observed melancholic patients to have greater levels of anxious arousal relative to non-melancholic patients and controls, consistent with other reports (Gold and Chrousos, 2002). Cholinergic dysfunction may account for mood and cognitive deterioration in depression and reduced hippocampal neurogenesis (Dagyté et al., 2010). Prior studies have highlighted that delays in P300 component latency are associated with cognitive deterioration or melancholic symptoms (Bruder et al., 1991; Schlegel et al., 1991; Vandoolaeghe et al., 1998) suggesting a relationship between melancholia, hyperarousal and behavioural impairment. We have suggested previously that overprocessing of stimuli may account for the slower reaction times observed in melancholic patients relative to controls during an auditory oddball task (see Kemp et al., 2010).
The current study did not replicate our prior behavioural and electrophysiological findings of differences between patients with melancholia and controls when errors were explicitly controlled for (see Kemp et al., 2010). This may be due to differences in task modality and requirements, as the auditory oddball task is a measure of selective attention and the Go/No-Go task a measure of inhibitory control. The findings pertaining to the auditory oddball task are consistent with the hypothesis that overprocessing of stimuli and hyperattentive impairments may contribute to the negative biases observed in depression, which is relevant for selective attention. Regarding inhibitory control, Ruchsow et al. (2008b) reported decreases in the No-Go P300 for depressed patients in comparison to controls. This study involved more complex stimuli than those involved in the current study. The paradigm used by Ruchsow et al. (2008b) required participants to look for a target letter among competing stimuli whereas in the current study the target word was the same for each stimulus presentation only varying by colour to indicate whether to respond or withhold. Therefore, the level of attention required to complete the Go/No-Go task used in the current study may not be sufficient for the expected hyperattentive impairments in stimulus processing to be observed after controlling for errors.
Research suggests that visual stimuli have a greater capacity to capture attention than auditory stimuli (Spence et al., 2001; Turatto et al., 2002), supporting theories on visual dominance (Spence, 2009). It is possible, therefore, that auditory attentional mechanisms may actually be poorer than visual attention mechanisms in depression. According to Miller and Cohen (2001) the prefrontal cortex exerts control over input (sensory) and output (response) neurons and in turn the prefrontal cortex mediates reciprocal connections with the sensory and motor cortices and the limbic system. The results from our oddball study (Kemp et al., 2010) suggest that melancholic patients have reduced auditory attentional capacity, while findings from the current study suggest that these same patients display similar visual attentional capacity to controls, suggesting that depressed patients are better at utilizing visual information than auditory information. This may be due to visual dominance, with attention mechanisms being prioritized for visual information over and above auditory information. As such the capacity to attend to visual information may remain equivalent to control participants, whereas the capacity to attend to auditory stimuli may be reduced in depressed participants. This finding has significant implications for understanding how attentional mechanisms differ in depression.
In conclusion, our study reveals that electrophysiological processing associated with a simple inhibitory task does not differ between depressed patients and healthy controls when examining ERPs associated with correct behavioural responses. Melancholic patients exhibited a greater numbers of CEs relative to non-melancholic patients and controls respectively and also exhibited a greater variability in reaction time in comparison to controls. Behavioural responses suggest that depression subtypes are differentially affected by inhibitory control processes with the melancholic subtype exhibiting the greatest number of errors. Key clinical indicators of melancholic depression, including depression severity and psychomotor disturbance, appear to relate to some of the observable behavioural impairments during cognitive inhibition. Strengths of the study include a relatively large sample size, a medication-free sample and controlling for behavioural differences between groups when inspecting ERPs.
Footnotes
Acknowledgements
Funding
CRQ was supported by an Australian Postgraduate Award and AHK by a National Health and Medical Research Council Career Development Award Fellowship (571101). This research was supported by a National Health and Medical Research Council (NHMRC) project grant (464863) and an Australian Research Council Discovery Grant (DP0987332)
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
