Abstract
Objective
This paper reviews the historical and conceptual background to proposals about prevention of schizophrenia through intervention targeting asymptomatic, high-risk individuals. It also examines the outcomes of a hypothetical model of prevention based on a two-stage risk segmenting approach.
Method
The assumptions and parameters used in the model are derived from actual epidemiological and clinical research. The two risk criteria selected are: (i) genetic risk (having a parent with schizophrenia); and (ii) neurocognitive deficit (abnormal performance on the Continuous Performance Task, CPT). The parameters and risk factors are applied to a hypothetical screening program covering a population of 100 000.
Results
At the end of the second stage of screening the program using the risk criteria to search for preventable cases will have correctly identified only three out of 20 ‘true’ cases and will have incorrectly assigned to treatment two non-cases. The great majority of people at risk who will eventually develop schizophrenia are likely to remain undetected by current screening or preclinical diagnostic programs, while a certain number of people actually not at risk would be falsely identified as high-risk and offered treatment.
Conclusions
Reliably identifying, with intention to treat, asymptomatic people in the community who are presumed to be at high risk of developing schizophrenia is at present epidemiologically non-viable. This caveat should not apply to strategies for early diagnosis and treatment of incipient episodes of schizophrenia where strategies to reduce the duration of untreated psychosis are likely to be both feasible and cost-effective.
According to the Dictionary of Epidemiology [1], the goals of medicine – to promote, preserve or restore health when it is impaired, and to minimise suffering and distress – are best embodied in the word ‘prevention’. Schizophrenia, a disorder which ranks among the top 10 causes of life years lost to disability globally [2], certainly qualifies for close scrutiny with regard to the possibility or prospect of prevention. This paper attempts to place the many issues involved in the context of epidemiological research.
There is little doubt today that schizophrenia is a genetically complex disease, sharing a number of features with many cancers, asthma, hypertension, and diabetes [3]. It is very likely that:
Schizophrenia is transmitted by multiple genes (polygenes) that interact among themselves and with the environment.
The genotype is incompletely penetrant and there exists an unknown number of phenocopies.
Different sets of genes may be involved in the transmission of schizophrenia in different populations and families (locus heterogeneity).
The genes involved in schizophrenia risk are relatively common in the population – some of them probably coding for personality or temperament traits or other basic ingredients of our central nervous system make-up.
Notwithstanding nearly a century of research and significant advances in knowledge over the past two decades, the aetiology of schizophrenia remains enigmatic. Is it possible to even think of preventing a disorder whose causes and mechanisms of occurrence are still outside our understanding? I will counter this sceptic viewpoint by examples demonstrating that prevention of disease is, in principle, feasible and can be successful in the absence of prior complete understanding of its aetiology and pathogenesis. The prototype example is, of course, John Snow's act of removing the handle of the drinking water pump in London's Broad Street in the mid-19th century [4], which effectively interrupted the spread of the cholera epidemic almost 30 years before the bacteriological theory of disease became established and the specific pathogen was discovered.
A more recent example is the practical eradication of a mental disease which, in the early decades of the 20th century, was responsible for the largest number of patients in mental hospitals in Georgia, South Carolina and Illinois in the United States as well as in parts of Europe, particularly southern Italy. The disease was characterised by a periodic or chronic course, typical seasonal exacerbation in spring, depressive and paranoid-hallucinatory symptoms and, in a proportion of cases, transition into dementia. There was a familial clustering of cases that did not fit an exact genetic pattern and gave rise to a suspicion of contagion. This was the factual situation that epidemiologist Joseph Goldberger faced at the start of his investigative journey, which ultimately led to the discovery of the aetiology and the prevention of pellagra [5]. Goldberger systematically applied the epidemiological method, designing a series of case–control and follow-up studies. By refuting the infectious disease hypothesis (hospital workers in contact with cases did not get the disease) and exploring the correlation between incidence and what we would today call ‘lifestyle’ factors, he came to the inevitable conclusion that the disease was the result of a diet that was deficient in animal protein and legumes but containing an excess of maize protein and starch; a nutritional imbalance facilitated by poverty. Goldberger proposed that the substance missing from the diet was a vitamin and that a simple modification of nutrition supported by government subsidies for a balanced diet could prevent the disease. This was soon demonstrated in practice and before long laboratory research identified the aetiological factor as niacin deficiency.
Other examples of successes of prevention include:
The prevention of mental retardation in children affected by phenylketonuria, a genetic disorder in which the brain is severely damaged by the accumulation of phenylalanine due to an enzyme deficiency. A diet free of phenylalanine during childhood and adolescence ensures normal brain development [6].
The reduction of the incidence of trisomy 21 (Down syndrome) by genetic counselling and prenatal diagnosis targeting prospective mothers aged 35 and over [6].
The dramatic reduction in mortality due to ischaemic heart disease in areas such as northern Karelia (Finland) where the interaction between genetic liability and a high cholesterol diet had contributed to an epidemic of myocardial infarction among males [7].
What are the common features of these examples of successful prevention and the lessons that can be drawn from them? In the instance of pellagra, reduction of the incidence of psychosis by eradication of the underlying disease was achieved: (i) before the exact nature of the nutritional deficiency was elucidated; (ii) without the intervention of psychiatrists; and (iii) by applying a mix of biological (nutritional), social and economic measures. In all examples cited, prevention was achieved either by interrupting the contact between a presumed pathogen and the host or by reducing the impact of a major risk factor. What is common to these examples of success is, first, that a potent risk factor with a high relative risk or odds ratio was identified. We do not know exactly what the cholera odds ratio had been for those using water delivered by the Broad Street pump, but it is likely to have been of the order of 60 or 70. So the simple act of interrupting the supply of contagion had a very high chance of success. Second, the risk factor identified or surmised in those cases had a very high positive predictive value. Third, not only was the risk factor shown to be a key mechanism in the transmission of the disease, but a simple and feasible intervention was available. All this is to show that epidemiology, especially when fortified by laboratory research, can give clear guidance to public health strategies of prevention. The question is, does the epidemiology of schizophrenia provide us with similarly clear clues that could eventually lead to the development of similarly effective preventive strategies?
The important idea of primary prevention in schizophrenia is at present predicated on the ‘… possibility of identifying potential patients long before onset using vulnerability markers and of making more feasible primary prevention efforts’ [8]. This is not a recent proposition. Its origins go back to E. Bleuler who set the stage for the search of pre- or subclinical forms of schizophrenia by asserting that ‘under all circumstances, latent schizophrenias are fairly common, so that the “disease” schizophrenia must be a much broader concept than the overt psychosis of the same name’ [9]. Later, this was echoed by Gross et al. who claimed that ‘early diagnosis and early treatment of these prepsychotic basic stages, preceding very often the psychosis by many years, is very important. The aim is prevention of psychosis by therapeutic intervention as early as possible, i.e. in the basic stages, long before the onset of the first psychotic manifestation’ [10].
‘Long before the onset’ implies being able to reliably identify people who are asymptomatic but very likely to eventually develop the disease and to intervene in some way early on, in order to prevent its onset. In public health, this is usually the objective of screening programs. There is a fairly well-developed body of knowledge concerning the design of screening programs, as well as a set of criteria helping to determine whether a screening program is likely to do more good than harm [11]. In a brief summary, these criteria boil down to several key questions: (i) Does the burden of disease or disability warrant screening? (ii) Is there an efficacious preventive intervention? (iii) Is there a good screening test? (iv) Is the program likely to reach those who could benefit from it? (v) Can the health-care system cope with the screening program? (vi) Will the screen-positive individuals comply with the proposed intervention?
To answer these questions in the instance of a hypothetical primary prevention of schizophrenia, it would be helpful to review briefly the basic epidemiological evidence about the disorder. The prevalence of schizophrenia, according to a large number of studies conducted at different times and in different populations is, on average, 5 cases per 1000 population at risk, in the age range 15–54 years (Table 1). In the Australian National Mental Health Survey (the study of low-prevalence disorders), the combined mean point prevalence rate for Brisbane, Canberra, Melbourne and Perth was 3.6 per 1000 (range 3–7 per 1000). Adding to this the untreated morbidity (which was estimated at 0.3 per 1000), the mean prevalence becomes 3.9 per 1000, which is well within the range of the majority of studies [12].
Selected prevalence studies of schizophrenia
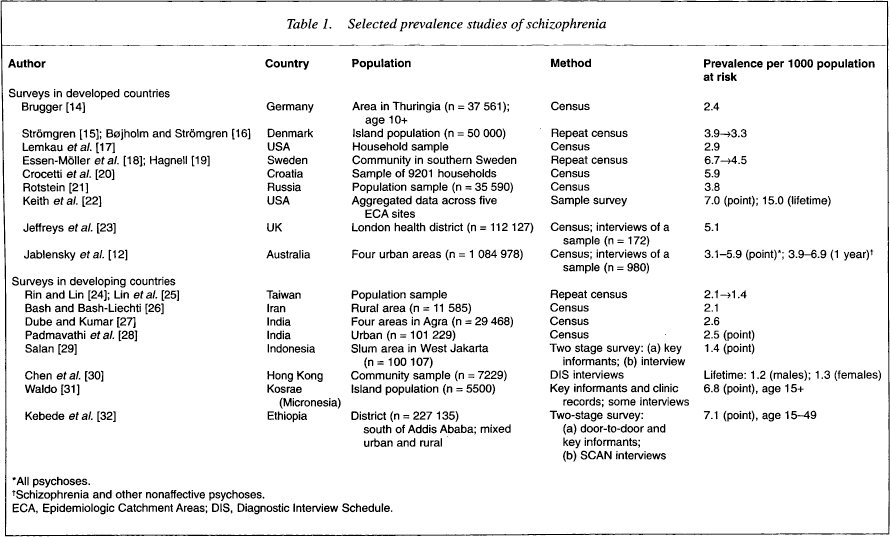
All psychoses.
Schizophrenia and other nonaffective psychoses.
ECA, Epidemiologic Catchment Areas; DIS, Diagnostic Interview Schedule.
The incidence of a complex disorder such as schizophrenia is likely to be of a greater interest for potential preventive programs than its prevalence. Incidence tells us something about the so-called ‘force of morbidity’ in the population and provides us with a window on the events proximate to onset, which may be aetiologically relevant. However, as schizophrenia has a low population incidence, stable estimates with a narrow confidence interval can only be obtained if we have a sufficiently large population denominator. For example, it would be extremely difficult to obtain reliable estimates of incidence in populations smaller than, say, 250 000. Thus, one needs to study very large population cohorts or, in a smaller population, to mount continuous, prospective case finding over a number of years to get a sufficient number of cases. The ideal design would, of course, be to follow up an entire birth cohort. However, this is likely to be both costly and laborious.
In the World Health Organization (WHO) 10-Country Study on the incidence of schizophrenia, identical instruments and methods were applied simultaneously in 13 catchment areas with a view to intercepting at the point of first contact every new case suggestive of psychosis [13]. The rates estimated on the basis of a 2-year prospective case finding are shown in Table 2. The incidence of schizophrenia is approximately less than one-tenth of the prevalence; that is, between 0.2 and 0.5 new cases per 1000 population per year. This gives a lifetime risk, or disease expectancy, of 0.8–1.0% in the general population. Although the mean age at onset is earlier in males than in females, in terms of lifetime risk there is no significant gender difference. As far as we know from previous research, including the WHO multicountry studies, the incidence rates are similar across different cultures and we know of no population that is free of the disorder. However, the striking aspect of the results of the WHO study is that the variation of incidence across the populations is constrained within a rather narrow band. This is quite unlike most of the multifactorial diseases (e.g. ischaemic heart disease) where the incidence rates differ between populations by a factor of 10 or greater. A possible interpretation of this striking dissimilarity between schizophrenia and other diseases is provided by the multifactorial threshold model, which assumes that the polygenic liability to develop disease is a quantitative trait with a population distribution close to the normal one [33]. According to the model: (i) clinical disease occurs when a certain threshold of vulnerability is crossed at the ‘severe’ end of the distribution; and (ii) the threshold itself can shift towards greater or lesser vulnerability under the influence of major environmental or genetic factors. Significant threshold shifts seem to occur in, say, heart disease as a result of environmental and behavioural factors such as diet and smoking, but not in schizophrenia where no single major environmental factor has been detected as influencing its incidence. The corollary would be that vulnerability to schizophrenia can be described as a continuous trait that is similarly distributed in most populations and exhibits comparable, relatively stable thresholds of manifestation of clinical disorder.
World Health Organization 10-Country Study: annual incidence rates per 1000 population at risk, age 15–54, for ICD-9 (‘broad’) and a ‘restrictive’ case definition of schizophrenia [13]
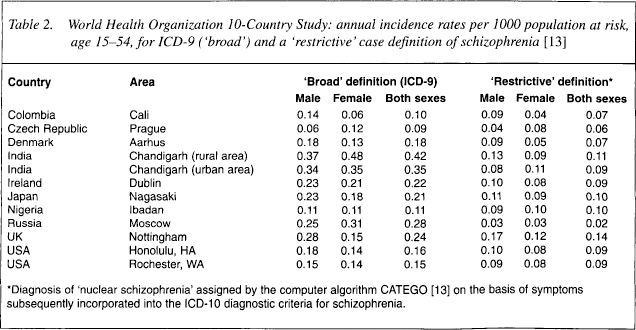
Diagnosis of ‘nuclear schizophrenia’ assigned by the computer algorithm CATEGO [13] on the basis of symptoms subsequently incorporated into the ICD-10 diagnostic criteria for schizophrenia.
But what about subclinical states and the spectrum of transitions between such states and clinical disorder? Although we have some clinically meaningful and biologically plausible theoretical models, our epidemiological knowledge on these issues is very limited. Paul Meehl has been the first to postulate a fundamental trait he termed schizotaxia [34], which is the product of a specific genetic defect expressed at the neural cellular level but not giving rise to any clinical symptoms. Under certain conditions, for example, under the influence of other background genes or as a result of defective social learning in infancy and childhood, schizotaxia may convert to schizotypal disorder. This latter term was originally used by the psychoanalyst Sandor Rado to describe a particular constellation of anhedonia, ambivalence, ‘interpersonal aversiveness’ and mild ‘cognitive slippage’ – features he observed in some of his patients [35]. This was not an entirely novel concept. As already pointed out, Bleuler believed that there are latent schizophrenias that never become manifest [9]. This elusive entity had been described by many clinicians under a bewildering variety of names such as ‘soft schizophrenia’, ‘ambulatory schizophrenia’, ‘microprocess schizophrenia’ or ‘defaulted shizophrenia’, etc. However, it was not until the Danish adoptive study [36], which demonstrated that latent schizophrenia was prevalent in the biological relatives of those adoptees who eventually developed schizophrenia, that the concept gained acceptance among researchers. Renamed as schizotypal personality or disorder, latent schizophrenia was included in DSM-III-R/-IV and ICD-10.
Let us now examine the evidence concerning the risk factors. A number of risk factors for schizophrenia have been tentatively identified (Table 3) among which genetic vulnerability is by far the most prominent one. The risk of schizophrenia for a first-degree biological relative of a person with schizophrenia ranges between 5% and 12%; that is, a significant increase over the general population risk of about 1% [37]. Thus, the child of a parent with schizophrenia runs a 10–12% statistical risk of developing schizophrenia in later life. Everything else being equal, individuals born of a complicated pregnancy, with birthweight less than 2500 g, and signs of neurodevelopmental impairment manifest in brain structural abnormalities (increased ventricle: brain ratio) or functional deficits (attention dysfunction), are at an increased risk of schizophrenia in adult life. The combination of genetic predisposition and neurodevelopmental impairment is likely to particularly increase the risk and, as such, should attract attention as a potential biological marker of the disease. It is tempting to consider that, by using a combination of risk factors, we might be able to identify those individuals who are likely to experience the transition from schizotaxia to schizotypy and then to overt schizophrenia.
Risk factors and antecedents of schizophrenia *
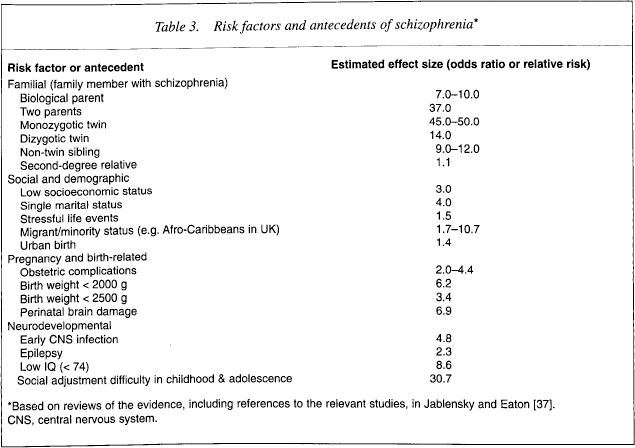
Based on reviews of the evidence, including references to the relevant studies, in Jablensky and Eaton [37]. CNS, central nervous system.
To estimate the probabilities that we would be able to detect those at high risk and apply some sort of preventive intervention, we need some parametric data. In the example presented, ‘real life’ data are used, derived from the WHO 10-Country Study and the Western Australian family study on schizophrenia, in which 121 families comprising 502 individuals have been extensively investigated with a battery of neurocognitive tasks tapping critical functional deficits, such as sustained attention, verbal memory, lexical retrieval and executive control. In particular, I propose that we use the following parameters: (i) lifetime risk of schizophrenia = 0.5%; (ii) risk if one parent is affected = 11%; (iii) incidence rate at age 15–19 years = 1.8 per 10 000; (iv) natural history of the first episode of psychosis = 30% remission rate; (v) efficacy of intervention (response rate to an ‘atypical’ antipsychotic) = 65%; (vi) sensitivity of the screening test = 80%; and (vii) specificity of the screening test = 60%.
Let us now assume that we are dealing with a total population of 100 000; that is, the size of a major town. In this population we expect to find a total of 500 individuals (300 males and 200 females) affected by schizophrenia. We expect that about 100 out of the 500 persons affected (80 females and 20 males) would be parents having at least one child. The estimated number of children aged 15–19 years who have a parent with schizophrenia would be about 10. We postulate further that the incidence of schizophrenia in the age group 15–19 years is two new cases per 10 000 population per year. Having a parent with schizophrenia would result in a 10-fold increase of the risk.
If we now apply the strategy of segmenting the risk [38], which requires that we first apply the least expensive or intrusive measurement that has a relationship to risk and then apply a second criterion that would further concentrate the risk population, we can expect to end up with a group that is highly likely to eventually ‘convert’ into psychosis and therefore constitute a prime target for prevention. The two risk criteria that we might wish to apply in this example are: (i) having a parent diagnosed with schizophrenia; and (ii) performing abnormally on the Continuous Performance Task, Identical Pairs (CPT-IP), which discriminates significantly between patients with schizophrenia, ‘at risk’ biological relatives without any clinical symptoms and normal controls. The mean scores (dL or signal detection score) on the CPT-IP for groups of patients with schizophrenia, first-degree relatives of such patients and normal controls, which are given in Table 4, have been calculated from data collected in the Western Australian Family Study of Schizophrenia. It should be noted that, although the group means separate the three groups quite well, a proportion (15%) of patients with schizophrenia have ‘normal’ scores and a proportion (5–10%) of the asymptomatic normal controls have scores in the abnormal range, characterising the majority of patients with schizophrenia.
Continuous Performance Task, Identical Pairs version (CPT-IP): results from the Western Australian Study of Schizophrenia

The results of the first phase of the screening procedure are illustrated in Table 5. As the majority of people (over 80%) who eventually develop schizophrenia do not have a parent affected with schizophrenia, our ‘genetic risk’ criterion will, at best, identify correctly four out of the 20 individuals who will eventually develop schizophrenia (low sensitivity). Although the specificity of this criterion is very high (> 99%), the positive predictive value of the ‘parent with schizophrenia’ criterion remains unacceptably low (40%). This also implies that we would have to treat six persons who would never develop schizophrenia and miss; that is, fail to treat, 16 persons who would develop the disease. Overall, we would misclassify and erroneously treat (or not treat) on average 5.5 individuals per each correctly identified and treated case.
Outcome of exposure to a risk factor in a hypothetical population of 100 000
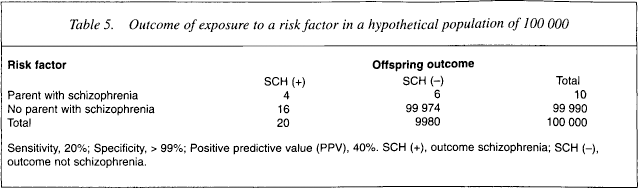
In the hope of improving this unsatisfactory rate of success, we may now decide to apply a neurocognitive test (CPT-IP) to those 10 individuals who are at high genetic risk by having a parent with schizophrenia. The likely outcome of this second screening phase is shown in Table 6. The end result is that within the group of 10 individuals at high genetic risk, we would still have to treat two non-cases and miss one ‘true’ case; that is, misclassify on average one case per each individual correctly identified. However, this latter result could be quite misleading if we omit from consideration the initial population of 100 000 with which we started. In the context of this total population, we will have, at the end of the second stage of our search for preventable cases, correctly identified only three out of 20 such cases (i.e. missed 17 cases) and incorrectly assigned to treatment two non-cases. Thus, the strategy of segmenting the risk in this instance may help reduce the number and proportion of false-positive case identification but will have no effect on the large number of cases that would remain undetected due to the low sensitivity of our screening tools.
Outcome of sequential application of a second screening test (CPT-IP)

The evaluation of the end result of this kind of screening and intervening preventively is further complicated by the fact that early episodes of psychosis have a strong tendency to recover spontaneously. Thus, at a 2-year follow-up we can expect that up to three-quarters of the treated cases will be symptom-free, while one quarter will still exhibit symptoms. Among those who will be symptom-free, it would not be possible to distinguish remissions attributable to the intervention from remissions occurring irrespective of intervention.
In conclusion, proposals to apply on a population scale a strategy of prevention of schizophrenia based on identifying individuals at high risk for the disorder in the preclinical stage and offering them a pharmacological intervention (plus any psychosocial measures) need to address three serious issues related to the nature of the disorder and the current state of our knowledge:
The likelihood that the great majority of people at risk who will eventually develop schizophrenia would remain undetected by current screening or preclinical diagnostic programs.
The likelihood that a certain number of people actually not at risk would be falsely identified as high-risk and offered treatment, with concomitant arousal of anxiety and possible adverse effects of labelling,
The likelihood that the effectiveness of the intervention would not be evaluated properly because of the tendency of the early symptoms and signs of psychosis to remit spontaneously and irrespective of treatment.
Considering the logistics and cost of setting up a population-based program for preclinical identification of people at high risk for schizophrenia, and the complexity of ensuring adherence to any proposed intervention, monitoring progress and evaluating outcome, it would appear that the cost-effectiveness of any such program would be extremely low. However, it is of great importance to stress that this conclusion applies strictly and exclusively to the prospect of identifying and treating asymptomatic people in the community who are presumed to be at high risk of developing schizophrenia. It does not apply to strategies for early intervention and treatment of incipient, first episodes of schizophrenia. There is a strong justification for such strategies in the light of evidence – albeit still limited – that irreversible neural network alteration does occur in the course of the first psychotic breakdown and that the duration of untreated psychosis (DUP) is significantly related to the risk of such neurological damage [39]. Reduction of DUP is therefore a worthwhile goal and strategies to achieve it may be far more cost-effective.
