Abstract
Background
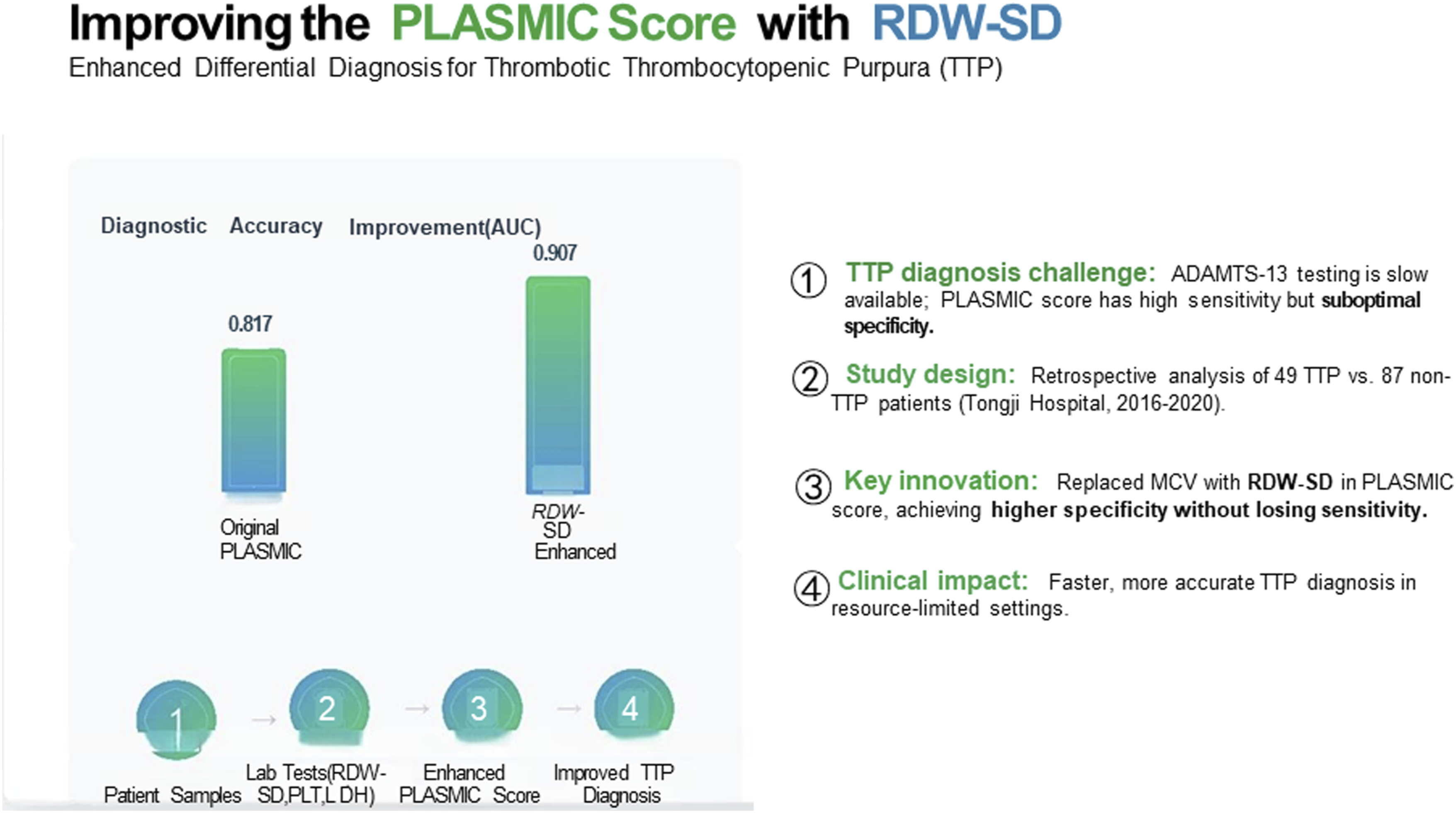
This study aimed to identify laboratory parameters that could optimize the PLASMIC score, thereby improving its diagnostic accuracy for thrombotic thrombocytopenic purpura (TTP).
Methods
We performed a retrospective analysis of 136 patients with suspected TTP who had available ADAMTS-13 activity measurements. Patients were stratified into two groups based on ADAMTS-13 activity: a TTP group (n = 49) and a non-TTP group (n = 87). Routine laboratory parameters—including hemoglobin (HGB), red blood cell distribution width-standard deviation (RDW-SD), mean corpuscular volume (MCV), platelet count (PLT), alanine aminotransferase (ALT), aspartate aminotransferase (AST), direct bilirubin (DBIL), indirect bilirubin (IBIL), lactate dehydrogenase (LDH), creatinine (CR), urea (UREA), international normalized ratio (INR), and D-dimer—were compared between groups. Statistically significant parameters were selected as candidate variables to refine the PLASMIC score. The diagnostic performance of the modified model was evaluated using receiver operating characteristic (ROC) curve analysis.
Results
Six parameters—HGB, PLT, ALT, RDW-SD, IBIL, and LDH—demonstrated significant differences between TTP and non-TTP patients. Multivariate logistic regression identified RDW-SD, PLT, and LDH as independent predictors of TTP. Based on these findings, we revised the PLASMIC score by substituting MCV with RDW-SD. The modified model exhibited a higher area under the curve (AUC) (0.907 vs 0.817) while maintaining sensitivity (95.9%) and improving specificity (70.1% vs 65.9%) compared to the original.
Conclusion
The modified PLASMIC score model may improve diagnostic accuracy for TTP in similar patient populations, but requires external validation in diverse cohorts to confirm its broader utility.
Keywords
Introduction
Thrombotic thrombocytopenic purpura (TTP), formally classified as ADAMTS-13 deficiency-mediated thrombotic microangiopathy (TMA), represents a rare but life-threatening hematologic emergency with an estimated incidence of 10–15 cases per million population annually. 1 The pathophysiology centers on severe deficiency of ADAMTS-13, a metalloprotease that cleaves ultra-large von Willebrand factor (vWF) multimers released from activated endothelial cells. 2 This enzymatic deficiency leads to accumulation of pathologically large vWF multimers, which promote excessive platelet adhesion and aggregation under high shear stress conditions, ultimately resulting in widespread microvascular thrombosis and end-organ ischemia. 2 Current standard-of-care involves emergent plasma exchange (PEX) to replenish functional ADAMTS-13 and remove inhibitory autoantibodies, combined with immunosuppressive therapy.3,4 In complicated cases, rituximab and caplacizumab are increasingly incorporated into initial treatment strategies.4–6
Prior to the advent of PEX therapy, acquired TTP carried a dismal prognosis with mortality rates exceeding 90%. 7 The landmark 1991 randomized controlled trial demonstrated that prompt PEX initiation could dramatically improve outcomes, achieving a 78% survival rate. 8 Contemporary studies have identified several prognostic factors associated with adverse outcomes, including delayed diagnosis, platelet transfusion, elevated cardiac troponin, central nervous system involvement, increased lactate dehydrogenase (LDH) levels, age, and ethnicity. 3 These findings underscore the critical importance of early recognition and intervention. However, the diagnosis remains challenging due to the nonspecific nature of TTP’s clinical features, which overlaps considerably with other thrombotic microangiopathies. In this context, quantitative ADAMTS-13 activity testing has emerged as the diagnostic gold standard, enabling reliable differentiation of TTP from alternative etiologies of microangiopathic hemolysis. 9
Despite its diagnostic importance, ADAMTS-13 activity testing remains limited by variable availability and potential delays in turnaround time, particularly when samples require referral to specialized laboratories. To facilitate rapid clinical decision-making regarding PEX initiation while awaiting confirmatory testing, Bendapudi et al. developed the PLASMIC score, a validated clinical prediction tool for TTP. 10 This scoring system originally designed as a screening tool for TTP incorporates seven key parameters: platelet count <30 × 109/L, evidence of intravascular hemolysis (elevated indirect bilirubin, decreased haptoglobin, or schistocytes), absence of active cancer or organ transplantation, normal MCV, INR <1.5, and creatinine <2 mg/dL. Scores of 0–4 indicate low risk (TTP unlikely), 5–6 suggest intermediate risk (requiring ADAMTS-13 testing), and 6–7 points denote high risk (strongly suggesting TTP and necessitating immediate plasma exchange). This stratification helps clinicians prioritize urgent management of this hematologic emergency while avoiding unnecessary interventions in low-risk cases. However, despite high sensitivity, the specificity of PLASMIC score remains suboptimal. Thus further optimization is warranted to improve the accuracy and guide timely therapeutic decisions. Recent studies have explored modifications to enhance the score’s performance. Tang et al. reported that incorporating an elevated LDH-to-aspartate aminotransferase (AST) ratio improved predictive accuracy in pregnant women, while MCV could be safely omitted without compromising diagnostic power. 11 Lee et al. proposed adjusting MCV and INR thresholds to ≥90 fL and >1.1, respectively, though external validation is pending. 12 Additionally, Reddy et al. found that including neurological symptoms as a scoring component further increased diagnostic yield. 13 These findings suggest that tailored adjustments to the PLASMIC score may optimize its clinical utility across different patient populations.
This study aimed to identify readily available laboratory parameters that could improve TTP diagnosis and potentially enhance the PLASMIC score’s predictive performance. We conducted a retrospective analysis of patients presenting with TMA symptoms who had available ADAMTS-13 activity results at Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology between January 2016 and August 2020.
Methods and materials
Study design
Patients underwent ADAMTS-13 activity testing due to clinical suspicion of TTP with available complete laboratory data, including complete blood count (CBC), ALT, AST, direct bilirubin (DBIL), indirect bilirubin (IBIL), LDH, CR, blood urea nitrogen (UREA), INR, and D-dimer, were enrolled in the study through a retrospective review of medical records from January 2016 to August 2020 at Tongji Hospital in Wuhan. Patients lacking evidence of intravascular hemolysis were excluded. The detailed patient flow diagram was shown in Figure 1. ADAMTS-13 activity was measured in untreated patient at the moment of conducting the PLASMIC score using a fluorescence resonance energy transfer (FRETS)-based assay with the VWF86 substrate, employing a commercial ACTIFLUOR ADAMTS-13 Activity Kit (Sekisui Diagnostics, Stamford, CT, USA) on a Mithras LB 940 Multimode Microplate Reader (Berthold Technologies, Bad Wildbad, Germany). The Bethesda assay was used to detect ADAMTS-13 inhibitors when activity levels were below 30%. ADAMTS-13 gene sequencing was performed if activity was less than 20% and no inhibitors were detected. Patients were diagnosed with TTP if ADAMTS-13 activity was below 10%, or between 10% and 20% with presence of inhibitors or pathogenic mutations. Finally, the study population was divided into two groups: the TTP group (n = 49) and the non-TTP group (n = 87). Flowchart of patient screening and enrollment.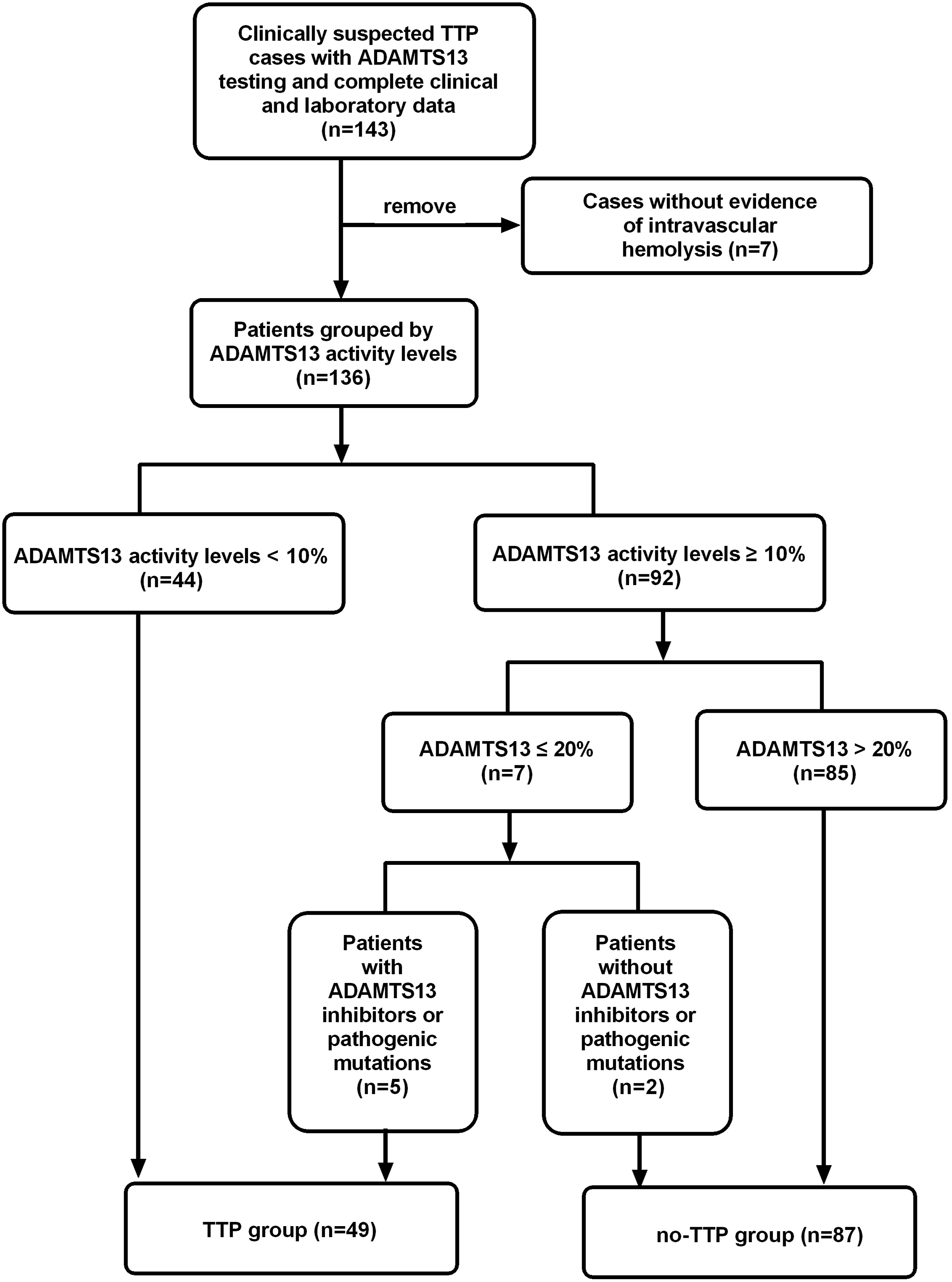
For each patient, relevant clinical and laboratory data were collected, including age, sex, diagnosis, presenting symptoms, and the earliest available laboratory results within 3 days prior to ADAMTS-13 testing. The laboratory parameters recorded were hemoglobin (HGB), red cell distribution width (RDW-SD), MCV, PLT, alanine aminotransferase (ALT), AST, DBIL, IBIL, LDH, CR, UREA, INR, and D-dimer. Laboratory tests were performed using the XN-9000 Sysmex analyzer (Sysmex Co., Kobe, Japan), Roche Cobas 8000 system (Roche Diagnostics, Mannheim, Germany), and STA-R coagulation analyzer (Diagnostic Stago, France). The RDW-SD measurement was performed using the XN-9000 hematology analyzer employing semiconductor laser light scattering technology combined with nucleic acid fluorescent staining in the WNR channel. This methodology allows for precise determination of red cell distribution width by directly measuring the standard deviation of erythrocyte volume distribution. According to manufacturer specifications, the analytical precision of this measurement is maintained at <2.0% coefficient of variation in whole blood mode. Our laboratory implements comprehensive management and control measures covering instrument status, reagent quality, internal quality control conditions, and environmental temperature to ensure the accuracy of the results.
This study was approved by the Ethics Committee of Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology. Informed consent was waived due to the retrospective nature of the study and the anonymization of all patient data, which did not involve individual privacy.
Statistical analyses
Continuous variables with non-normal distribution were summarized as medians (interquartile ranges) and compared using the Mann–Whitney U test. Categorical variables were presented as frequencies or percentages and analyzed using the chi-square test or Fisher’s exact test, as appropriate. Statistical analyses were conducted using SPSS 25.0 (SPSS, Chicago, IL, USA) and GraphPad Prism 6 (GraphPad Software, San Diego, CA, USA), with statistical significance defined as a two-sided P-value <.05. Parameters showing significant differences between the TTP and non-TTP groups were selected as candidates. Spearman correlation analysis was performed to assess correlations among these parameters; those highly correlated with others were excluded to avoid multicollinearity. The remaining parameters were further evaluated using multivariate logistic regression analysis to identify independent predictors. Receiver operating characteristic (ROC) curve analysis was conducted to determine the optimal cut-off values, area under the curve (AUC), sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) for each parameter and diagnostic model.
Participants
The study cohort included 136 patients (93 female, 43 male) who underwent ADAMTS-13 activity testing for suspected TTP. Based on diagnostic criteria, 49 patients (36.0%) were classified into the TTP group, including 44 patients with ADAMTS-13 activity <10% and 5 patients with mactivity 10–20% plus detectable inhibitors, among which 47 cases were acquired TTP (inhibitor-positive) and 2 were congenital TTP confirmed by genetic testing. The remaining 87 patients formed the non-TTP group, consisting of 19 cases (21.8%) of HELLP syndrome, 13 (14.9%) infectious diseases, 8 (9.2%) systemic lupus erythematosus, 7 (8.0%) hemolytic uremic syndrome, 7 (8.0%) brain injury, 5 (5.7%) hemophagocytic syndrome, 5 (5.7%) renal dysfunction, 4 (4.6%) immune thrombocytopenia, and 4 (4.6%) pregnancy-associated thrombocytopenia, along with 15 cases of other conditions including 2 each of autoimmune hemolytic anemia, Sjögren’s syndrome, leukemia and vasculitis, and single cases of drug-induced liver injury, myocarditis, Evans syndrome, megaloblastic anemia, adult-onset Still’s disease, DIC, and PNH. The TTP group had a median age of 45 years, with no significant differences in age or sex distribution compared to the non-TTP group.
Results
Results of laboratory parameters in TTP and non-TTP groups
Demographic characteristics and laboratory test results in TTP and non-TTP groups.
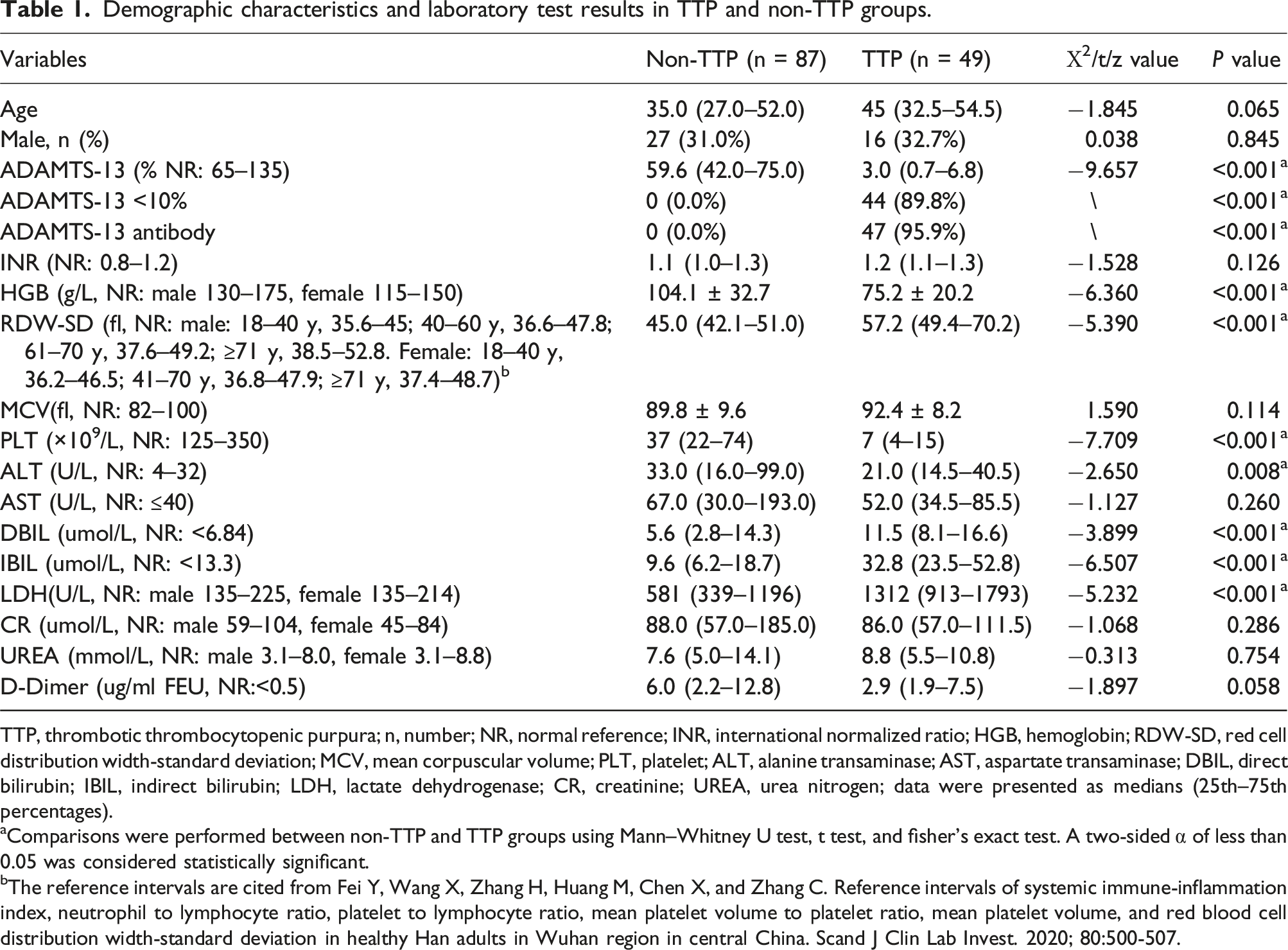
TTP, thrombotic thrombocytopenic purpura; n, number; NR, normal reference; INR, international normalized ratio; HGB, hemoglobin; RDW-SD, red cell distribution width-standard deviation; MCV, mean corpuscular volume; PLT, platelet; ALT, alanine transaminase; AST, aspartate transaminase; DBIL, direct bilirubin; IBIL, indirect bilirubin; LDH, lactate dehydrogenase; CR, creatinine; UREA, urea nitrogen; data were presented as medians (25th–75th percentages).
aComparisons were performed between non-TTP and TTP groups using Mann–Whitney U test, t test, and fisher’s exact test. A two-sided α of less than 0.05 was considered statistically significant.
bThe reference intervals are cited from Fei Y, Wang X, Zhang H, Huang M, Chen X, and Zhang C. Reference intervals of systemic immune-inflammation index, neutrophil to lymphocyte ratio, platelet to lymphocyte ratio, mean platelet volume to platelet ratio, mean platelet volume, and red blood cell distribution width-standard deviation in healthy Han adults in Wuhan region in central China. Scand J Clin Lab Invest. 2020; 80:500-507.
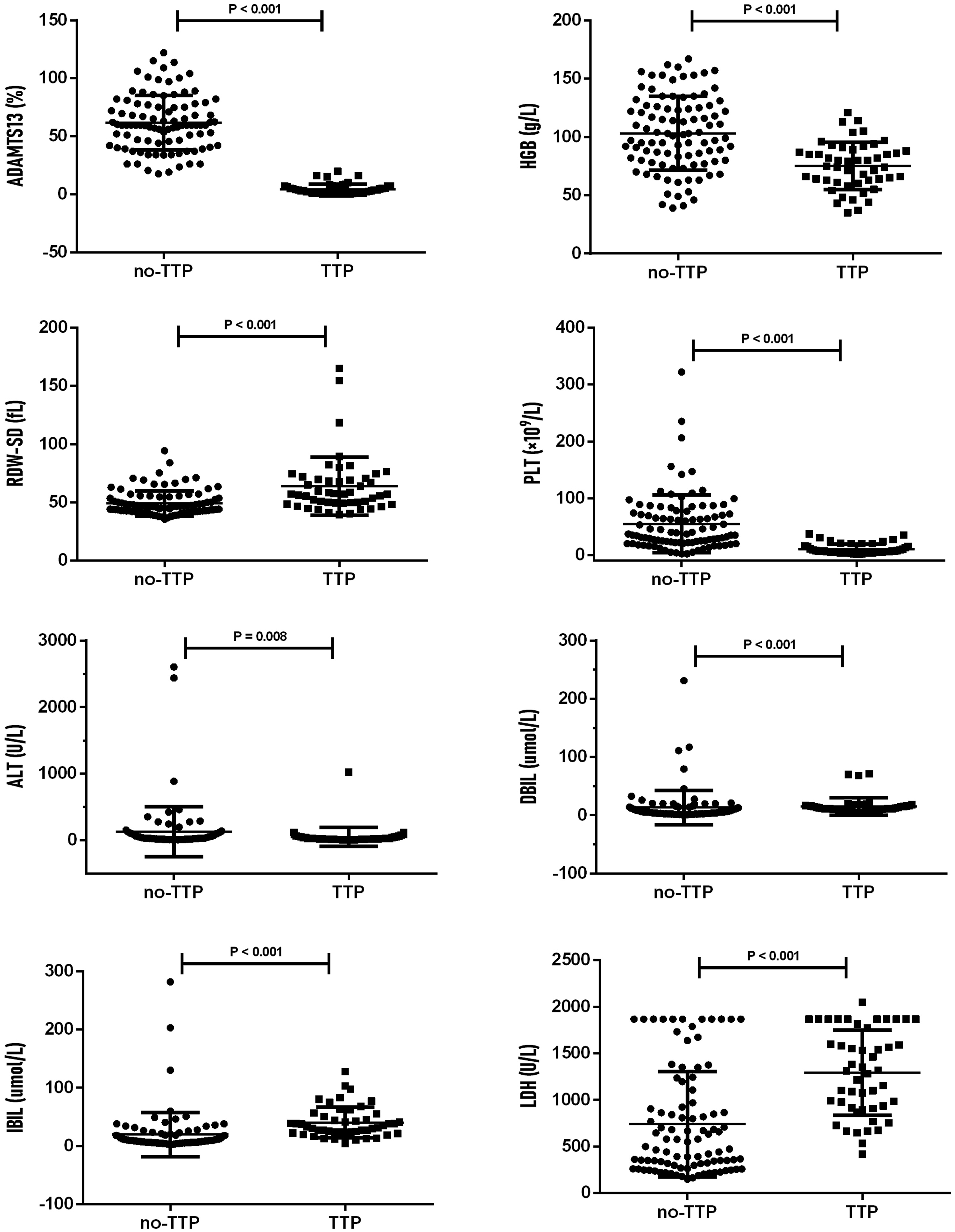
Distribution of ADAMTS-13, HGB, RDW-SD, PLT, ALT, IBIL, and LDH levels in TTP and non-TTP groups.
Multivariate logistic regression and ROC curve analysis
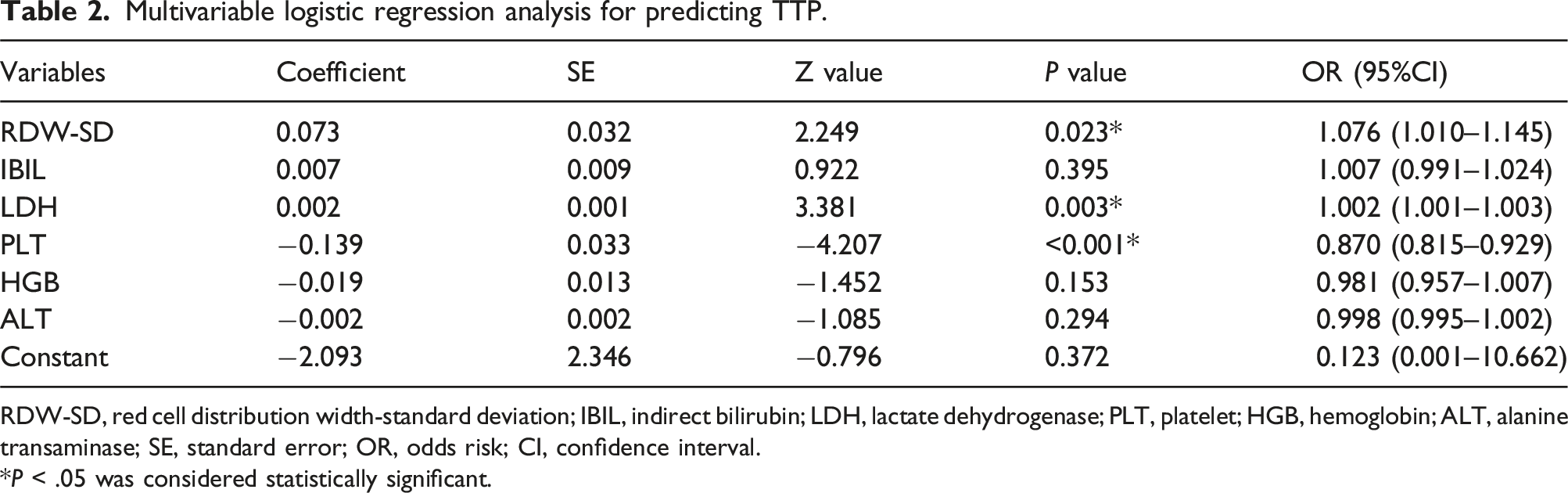
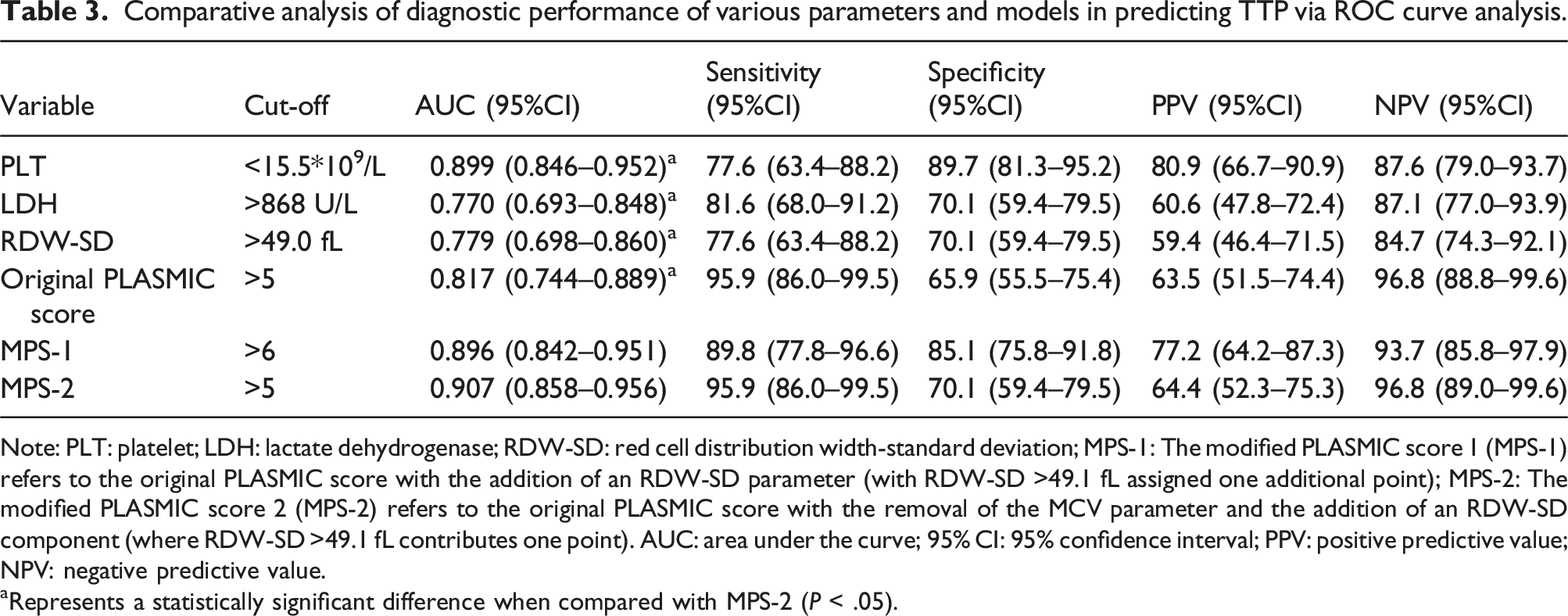
Spearman correlation analysis revealed a strong positive correlation between DBIL and IBIL (r = 0.711, P < .001), necessitating the exclusion of DBIL to prevent multicollinearity in subsequent process. The remaining six parameters (HGB, PLT, ALT, RDW-SD, IBIL, and LDH) were entered into multivariate logistic regression analysis, which identified three independent predictors of TTP: elevated RDW-SD (adjusted OR 1.076, 95% CI 1.010–1.145; P = .023), decreased PLT (adjusted OR 0.870, 95% CI 0.815–0.929; P < .001), and increased LDH (adjusted OR 1.002, 95% CI 1.001–1.003; P = .001). This model demonstrated excellent discrimination (C-statistic 0.964, 95% CI 0.937–0.991) and explained 77.6% of variance (Nagelkerke’s R2 = 0.776). Given that PLT and hemolysis (reflected by LDH) are already components of the original PLASMIC score, we optimized the score by replacing MCV with RDW-SD, which showed superior diagnostic performance (AUC 0.907, 95% CI 0.858–0.956) compared to alternative modifications. At the optimal cut-off (>5 points), the revised score achieved 95.9% sensitivity (95% CI 86.0%–99.5%), 70.1% specificity (95% CI 59.4%–79.5%), 64.4% positive predictive value (95% CI 52.3%–75.3%), and 96.8% negative predictive value (95% CI 89.0%–99.6%). For details, see Figure 3, Tables 2 and 3. Receiver operating characteristic curve analysis of various parameters and models for predicting TTP. PLT: platelet; LDH: lactate dehydrogenase; RDW-SD: red cell distribution width-standard deviation; MPS-1: the modified PLASMIC score 1 (MPS-1) refers to the original PLASMIC score with the addition of an RDW-SD parameter (with RDW-SD >49.0 fL assigned one additional point); MPS-2: The modified PLASMIC score 2 (MPS-2) refers to the original PLASMIC score with the removal of the MCV parameter and the addition of an RDW-SD component (where RDW-SD >49.0 fL contributes one point). Multivariable logistic regression analysis for predicting TTP. RDW-SD, red cell distribution width-standard deviation; IBIL, indirect bilirubin; LDH, lactate dehydrogenase; PLT, platelet; HGB, hemoglobin; ALT, alanine transaminase; SE, standard error; OR, odds risk; CI, confidence interval. *P < .05 was considered statistically significant. Comparative analysis of diagnostic performance of various parameters and models in predicting TTP via ROC curve analysis. Note: PLT: platelet; LDH: lactate dehydrogenase; RDW-SD: red cell distribution width-standard deviation; MPS-1: The modified PLASMIC score 1 (MPS-1) refers to the original PLASMIC score with the addition of an RDW-SD parameter (with RDW-SD >49.1 fL assigned one additional point); MPS-2: The modified PLASMIC score 2 (MPS-2) refers to the original PLASMIC score with the removal of the MCV parameter and the addition of an RDW-SD component (where RDW-SD >49.1 fL contributes one point). AUC: area under the curve; 95% CI: 95% confidence interval; PPV: positive predictive value; NPV: negative predictive value. aRepresents a statistically significant difference when compared with MPS-2 (P < .05).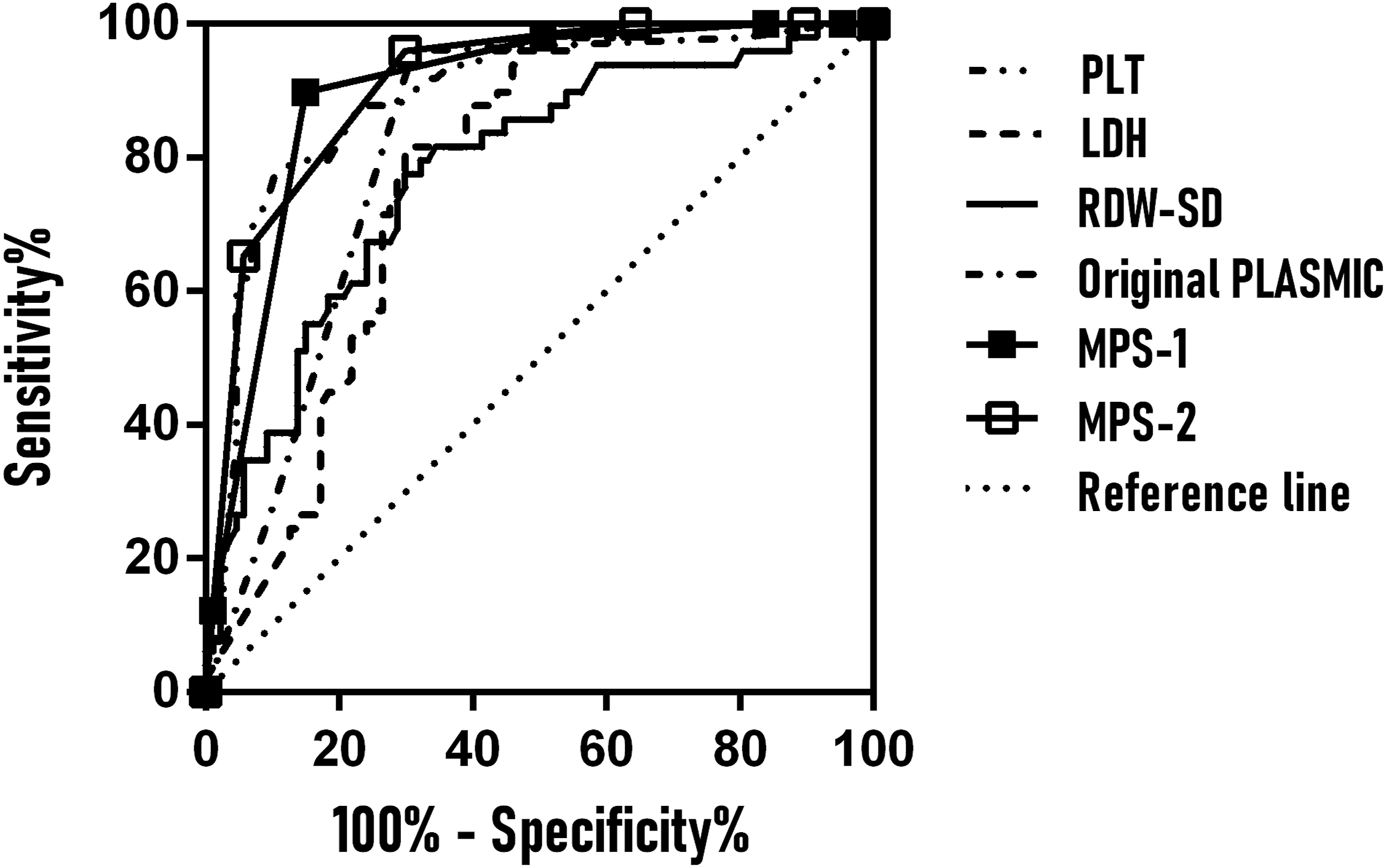
Discussion
This study’s laboratory findings align closely with the established pathophysiology of TTP. The observed significant reductions in HGB, PLT, and ALT, coupled with elevated RDW-SD, bilirubin fractions (DBIL and IBIL), and LDH in the TTP group, directly reflect the consequences of severe ADAMTS-13 deficiency. The characteristic accumulation of ultra-large vWF multimers promotes widespread platelet aggregation and microvascular thrombosis, leading to the hallmark triad of profound thrombocytopenia (typically <30 × 109/L), 9 microangiopathic hemolytic anemia (evidenced by reduced HGB and elevated RDW-SD), and consequent hemolysis markers (increased DBIL, IBIL and LDH). The absence of significant differences in coagulation parameters (INR, D-dimer), MCV, AST, CR, and UREA between groups corroborates previous research findings,10,11,14–16 reflecting TTP’s distinct pathogenesis where microthrombosis results primarily from vWF-mediated platelet aggregation rather than coagulation cascade activation. 4 These biochemical patterns provide important diagnostic clues, as the lack of prominent liver enzyme elevation helps differentiate TTP from HELLP syndrome, 16 while preserved renal function in most cases contrasts with hemolytic uremic syndrome (HUS), 17 though severe TTP can occasionally present with acute kidney injury, 18 emphasizing that renal involvement doesn’t exclude the diagnosis. The pathophysiological consistency of these laboratory findings supports their utility in distinguishing TTP from other thrombotic microangiopathies.
This study identified PLT, LDH, and RDW-SD as the most discriminative laboratory parameters for differentiating TTP from other conditions. While statistical analysis revealed that a PLT threshold of <15.5 × 109/L provided optimal diagnostic accuracy (maximizing the Youden index), we maintained the original PLASMIC score cut-off to preserve its critical sensitivity for clinical decision-making, as modifying this threshold could compromise its ability to identify true TTP cases. The marked elevation of LDH, resulting from mechanical hemolysis through platelet-rich microthrombi,2,3 serves as both a diagnostic marker and important prognostic indicator, with persistently elevated levels post-plasma exchange correlating with poorer outcomes.11,14,16 However, since the existing “evidence of hemolysis” criterion in the PLASMIC score already incorporates this parameter, we determined that additional modifications based on LDH would not substantially improve the score’s performance.
RDW-SD, which quantifies erythrocyte size heterogeneity, has emerged as a prognostic biomarker in various critical conditions including including traumatic brain injury, stroke, myocardial infarction, and pediatric sepsis.19–23 While its role in TTP remains understudied, our findings support Nagajothi et al.'s preliminary observation of elevated RDW-SD in TTP, 24 demonstrating significantly higher values in TTP patients compared to non-TTP cases (P < .001). This phenomenon likely reflects increased erythrocyte size variation secondary to schistocyte formation, even when MCV remains unchanged—a finding that explains why MCV shows limited diagnostic utility for TTP discrimination in multiple studies.11,14 The relative insensitivity of MCV may stem from the typically low proportion of schistocytes in peripheral blood smears, which affects size distribution more than average volume. In optimizing the PLASMIC score, we evaluated two approaches: (1) adding RDW-SD as an eighth parameter (1 point), or (2) replacing MCV with RDW-SD. The substitution strategy proved superior, significantly enhancing the score’s specificity (70.1% vs 65.9%) while maintaining its high sensitivity (95.9%), thereby improving overall diagnostic accuracy (AUC 0.907 vs 0.817) without increasing complexity.
This study has several important limitations that warrant consideration. First, as a single-center investigation conducted in an ethnically homogeneous population, the generalizability of our findings to more diverse patient groups remains uncertain. The model’s performance in populations with varying genetic backgrounds, environmental exposures, or healthcare systems requires validation through multicenter studies incorporating ethnically diverse cohorts. Second, the retrospective design and requirement for ADAMTS-13 testing as an inclusion criterion may have introduced selection bias, as not all clinically suspected TTP cases were consecutively enrolled. Third, while RDW-SD has demonstrated clinically significant variations across demographic groups (particularly by age and gender) in prior studies,25,26 our sample size limitations prevented meaningful subgroup analyses to establish age- and sex-specific cut-off values—an important consideration for future research. Additionally, all laboratory measurements were performed on a single analytical platform (Sysmex XN-9000), potentially limiting the transferability of our established cut-off values to other instrumentation systems due to known inter-platform variability. These limitations highlight the need for larger-scale, prospective multicenter studies to validate and refine the model across diverse populations and laboratory settings.
Conclusion
This study is the first to incorporate the routine laboratory parameter RDW-SD into the PLASMIC scoring model by replacing MCV. Compared to the original model, the revised version demonstrates equivalent sensitivity but enhanced specificity. This may improve diagnostic accuracy for TTP in similar patient populations, but requires external validation in diverse cohorts to confirm its broader utility.
Footnotes
Acknowledgements
We thank the clinical staff at Tongji Hospital for their assistance in data collection. We are grateful to Professor Liming Chen for laboratory support.
Author contributions
CZ, LX, ZX, and YF have contributed to the intellectual content of this paper and drafted the manuscript. XL and WS have contributed to collected and analyzed the experiment data. All the authors have read and accepted the final version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethics Committee of Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology. Informed consent was waived due to the retrospective nature of the study and the anonymization of all patient data, which did not involve individual privacy.
Contributorship
CZ, LX, ZX and YF have contributed to the intellectual content of this paper and drafted the manuscript. XL and WS have contributed to collected and analyzed the experiment data.
Guarantor
CZ.
