Abstract
Background
Point of care (POC) tests may improve accessibility and reduce costs of blood tests including in prostate cancer. The Man Van project was a pilot designed to address health inequalities that affect prostate cancer with novel community-based targeting of high-risk groups on a mobile clinical unit.
Methods
The i-CHROMA-II™ POC machine is a quantitative assay for the measurement of total prostate specific antigen (PSA) from capillary blood using fluorescence immunoassay technology. Laboratory based Serum PSA testing was compared with capillary blood POC testing using the i-CHROMA-II™ to determine its accuracy and impact on clinical decision making on the Man Van.
Results
28 men participated. The median age was 53 years (range 45–74). One POC test result was invalid. Nine POCT samples gave a result of <0.5 μg/L and were not included in the analysis. Of the remaining results (N = 18) the median PSA was 1.97 μg/L (range 0.54-31.22 μg/L). Using Lin’s Concordance Correlation Coefficient of Absolute Agreement gave a value of 0.392 (N = 17). A Bland-Altman plot showed a mean difference of 0.377 μg/L.
Conclusions
We report the first testing of PSA using the i-Chroma-II™ machine, and the first real-world mobile testing using any POC PSA test. Our study did not show correlation between the laboratory and i-Chroma-II™, although it did replicate the positive bias seen in previous studies. Further testing and refinement of POC tests may help to achieve the goal to developing reliable POC PSA tests.
Impact statement
This study presents the first evaluation of prostate-specific antigen (PSA) testing using the i-CHROMA-II™ point-of-care (POC) device on a mobile clinic – the Man Van. Among 28 participants, POC results showed 92.6% clinical concordance with standard laboratory testing, though lacked strong analytical correlation and demonstrated a positive bias. Despite these limitations, the study highlights the feasibility and promise of mobile POC testing to improve access and address health disparities in prostate cancer care. Further refinement of POC technology is needed to ensure accuracy and support its broader adoption in community-based health initiatives.
Introduction
Prostate cancer is the most common male cancer in the UK with 55,000 cases and 12,000 deaths per year. 1 Although PSA testing is widely used in the detection of prostate cancer, its role in routine screening remains controversial. Critics argue that PSA testing can lead to over-diagnosis and overtreatment due to false positives, contributing to unnecessary medical interventions and patient anxiety. These concerns have sparked debate over whether PSA screening should be used universally or reserved for high-risk populations, balancing the benefits of early detection against potential harms.
The Man Van was a pilot project developed by the Royal Marsden Hospital and Institute of Cancer Research as a novel approach to address health inequalities in prostate cancer screening. Socioeconomic and systemic barriers, including limited access to healthcare, geographical constraints, and lower health literacy, disproportionately affect high-risk groups such as Black men and those from lower-income backgrounds. 2 To mitigate these disparities, the Man Van provided community-based targeting of these groups through mobile clinical units. Men were invited to participate in a Man Van check, with counselling offered for a prostate-specific antigen (PSA) test as well as the blood test itself. An integral part of the project was its long-term sustainability with the costs of the project being balanced to support this.
Given the scale of the issue of prostate cancer, with trends suggesting this burden will only increase, 2 minimising costs and making tests more accessible are key targets for future endeavours. Point of care (POC) tests offer the advantage of providing faster results compared to traditional laboratory-based tests. Use of POC tests particularly accelerated during the COVID-19 pandemic and is particularly relevant for times of health crises, 3 demonstrating their relevance in times of health crises, and their potential has also been recognized in community-based settings. Advantages to the use of POC tests have also been noted in community-based locations, 3 including streamlining services4,5 with potentially fewer appointments 3 and improved cost-effectiveness.6–10 However, while these benefits are promising, POC tests are often disadvantaged by lower sensitivity compared with hospital-based laboratory testing, 9 raising concerns about the reliability of results, particularly in high-stakes diagnoses like prostate cancer. Furthermore patient perspectives of near-instantaneous results have not fully been explored. 3 Addressing these limitations is essential if POC testing is to be used as a reliable alternative for community-based prostate cancer screening.
PSA is a 30−34 kDa kallikrein-related peptidase which is secreted by both normal prostate cells and prostate cancer cells. 11 Disruption of the cell architecture due to prostate cancer causes increased release of PSA into the blood stream 11 with higher-levels of PSA being progressively more suggestive of underlying prostate cancer. Its use as a screening tool for prostate cancer is controversial screening studies having mixed outcomes12–14 although there is mounting evidence supporting this with the number needed to screen now lower than breast cancer. 15 This is on a background of evolution in the diagnostic pathway for prostate cancer with pre-biopsy MRI and modern biopsy techniques helping to reduce the risk of harms and increase the diagnosis of clinically significant disease.16–18
Despite the controversy of PSA use as a diagnostic or screening tool its use is widely recommended for surveillance following prostate cancer treatment. 11 This is set to increase with the European Union Council recently recommending PSA-based prostate cancer screening 19 and countries developing screening programmes.20,21
Currently the gold-standard method for PSA testing is performed using automated immunoassays within laboratory settings, requiring specialised staff and equipment. 11 For a mobile clinic and other resource limited settings, a POC PSA test could be beneficial and cost-effective by removing the need for logistics for lab-transportation and reducing the need of follow-up appointments. 22 Feasibility of mobile screening for prostate has been shown with a review showing six previous studies showing its potential. 23 More recently the lead author published pilot data from the Man Van project showing higher rates of prostate cancer detection compared with previous screening studies. 24
NHS recommendations support the use of point-of-care testing to reduce the need of clinic attendances 25 but despite a multitude of POC PSA tests being developed 22 none have yet displaced laboratory PSA analysis in the NHS, most often due to a lack of accuracy/precision. 11
The i-CHROMA™ (Boditech Med Inc., Korea) POC machine is a quantitative lateral flow assay for the measurement of total PSA from either whole or capillary blood using fluorescence immunoassay technology. 11 When more antigen is contained in the sample this leads to more complexes forming and a stronger fluorescence signal26,27 which is detected once the cartridge is inserted into the i-CHROMA™ machine. When sealed cartridges are stable for 20 months if stored at 4–30°C. 26
Bolodeoku et al. first published a real-world evaluation of the i-CHROMA™ test with volunteers attending a screening event based at a hospital. 22 55 men underwent testing comparing POC capillary blood samples with venous blood samples sent to a laboratory with good correlation shown (r2 = 0.9084) and 90% of data falling between 2 standard deviations with good clinical correlation. 22 Further internal and external quality testing assessments were carried out showing a positive bias with the i-Chroma™ of more than 1.0 ng/ml in over 50% of the methods tested. 27 A further study of the i-Chroma™ using serum blood showed similar correlation (r 2 = 0.9664) with a positive correlation. 28
The system was updated with release of the i-CHROMA-II™ machine with: “improved user interface, display, an advanced optical system, in-built printer, and better connectivity”. 29 Till date no mobile testing of either machine has been published and no data has been published on the performance of PSA testing with the i-CHROMA-II™ machine.
Objectives
(1) To determine whether there is a significant difference in clinical decision outcomes between laboratory and POC PSA testing with the i-CHROMA-II™ (2) To evaluate concordance between laboratory and real-world POC PSA tests with the i-CHROMA-II™.
Materials and methods
To evaluate the real-world mobile use of the i-CHROMA-II™ PSA tests we trialled the machine on our Man Van mobile health clinic. As part of the Man Van health check eligible men were offered and counselled for PSA testing based on NICE guidance 30 : Black men over 45 years old and other ethnicities over 50 years old. Participants were recruited based on their eligibility under NICE guidance, with an emphasis on high-risk groups. Additional efforts were made to recruit a diverse sample across different socioeconomic backgrounds and locations within the region, ensuring representation from underserved communities.
Patients with known prostate cancer were excluded. Formal PSA blood tests were performed on the Man Van with blood bottles couriered to the Royal Marsden laboratory which utilises the Ortho Clinical Diagnostics VITROS Immunodiagnostic Total PSA method.
Paired whole blood capillary samples were taken alongside serum blood samples. As per the manufacturer’s instructions 75 μL of capillary blood was pipetted into a buffer solution, mixed then added to a cartridge for 15 min (incubation period at room temperature) before being scanned by the i-CHROMA-II™ machine. 26 The buffer solution contains anti-PSA antibodies which bind to the antigen (PSA) forming antigen-antibody complexes. 26 When the buffer is applied to a test strip (contained within the cartridge) the solution migrates onto a nitrocellulose matrix, merging with another immobilised antibody on the test strip. 26 The system has a range of 0.5–100 μg/L. 26 The i-CHROMA-II™ was selected due to its portability, ease of use in field settings, and prior studies demonstrating acceptable correlation with laboratory results in controlled environments. However, its known limitations, including lower sensitivity at the lower end of PSA detection, were considered when evaluating its performance in this pilot.
Complementary statistical approaches were utilised to assess concordance between POC and laboratory PSA values, providing a more comprehensive evaluation. Non-parametric regression modelling with Passing-Bablok regression was used to analyse correlation, Lin’s Concordance Correlation Coefficient to quantify agreement, and Bland-Altman to assess bias. Binary outcomes were based on clinical thresholds for further investigation. Missing data was handled through sensitivity analyses, and outliers were examined to determine their influence on overall model fit.
To account for potential confounders, such as age, ethnicity, and health status, these variables were collected and included in sensitivity analyses to assess their influence on PSA values and concordance between test methods.
Results
In July of 2023, 28 men had a POC PSA blood test alongside a formal laboratory PSA test on the Man Van mobile clinical unit. During this period the mobile unit was located at Chelsea Football Club, Stamford Bridge Stadium, in West London. The median age was 53 years (range 45–74). One POC test result was invalid, and 27 tests were successfully processed. The median laboratory PSA value was 1.08 μg/L (range 0.19–3.89 μg/L). Nine POCT samples gave a result of <0.5 μg/L and therefore could not be included in the analysis. Of the remaining (N = 18) values the median PSA was 1.97 μg/L (range 0.54–31.22 μg/L). Given the small sample size (N = 28), and the exclusion of nine samples with POC results <0.5 μg/L, the statistical power of the analysis is limited. These factors should be considered when interpreting the reliability and generalisability of the findings.
Correlation
A Passing-Bablok regression was plotted, however, the data was skewed by one erroneous result (i-Chroma-II™ PSA: 31.22 μg/L, laboratory PSA: 1.35 μg/L). This gave a R value of −0.0244 (N = 18). The erroneous result was removed, and data replotted (Figure 1) giving an R value of 0.4307 (N = 17). Sensitivity analyses were conducted to assess how the exclusion of this data point influenced the overall findings. Passing-Bablok regression plot of i-CHROMA (POC test) versus Laboratory test PSA results. Encircled in purple and green are those results that were clinically disconcordant.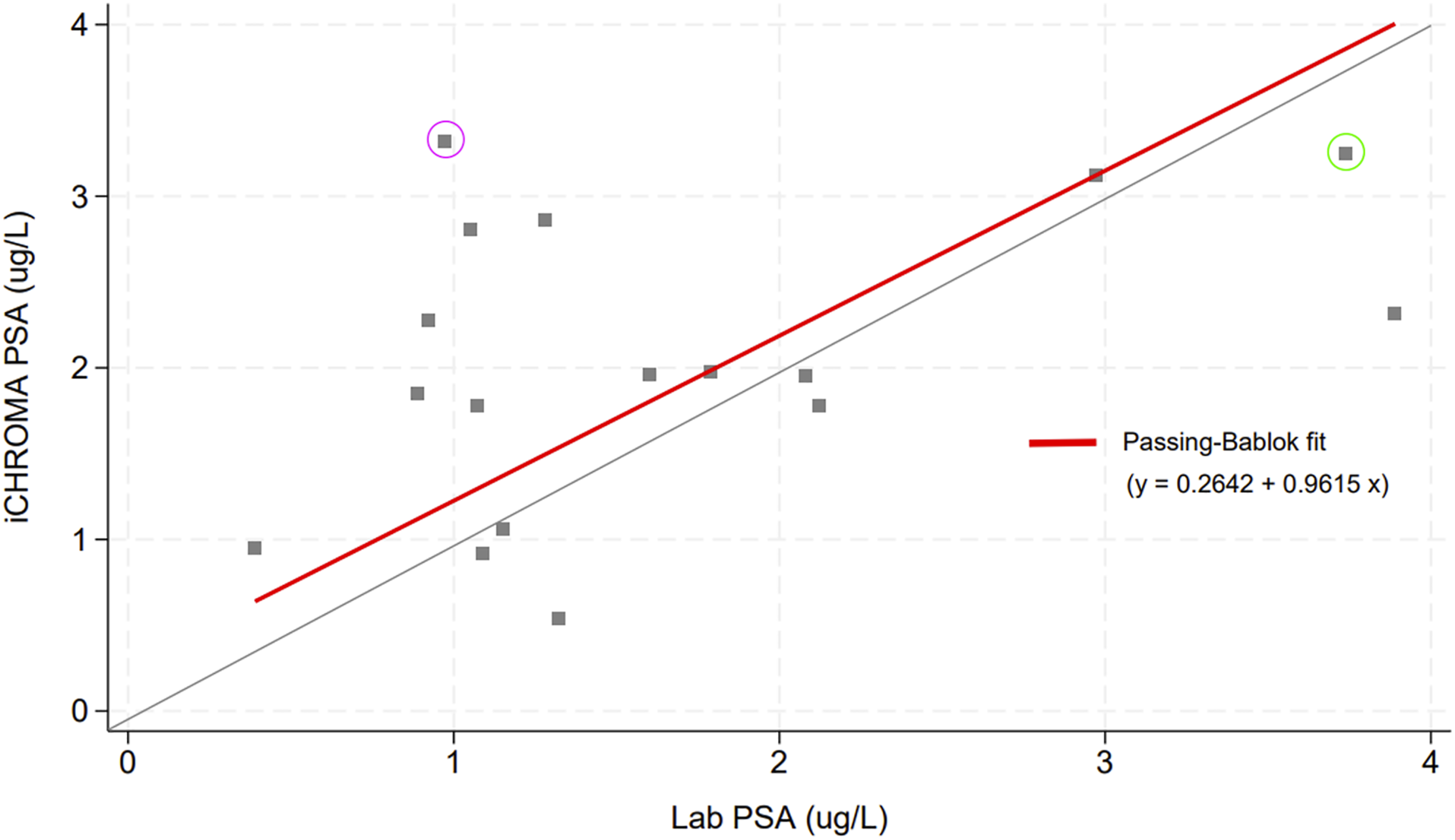
Using Lin’s Concordance Correlation Coefficient of Absolute Agreement gave a value of −0.006 (N = 18), whilst the removal of the erroneous result gave a correlation coefficient of 0.392 (N = 17). Both coefficients suggested no strong concordance between the laboratory and POC test methods. This level of concordance raises concerns about the reliability of the POC test in real-world settings.
To further assess the agreement between the two methods, a Bland–Altman plot analysis was performed (Figure 2) which showed a mean difference of 0.377 μg/L (95% CI: −0.14, 0.89; limits of agreement: −1.58, 2.33). The wide limits of agreement suggest considerable variability between the POC and laboratory test results. In clinical practice, this level of variability could lead to misinterpretation of PSA levels, particularly for borderline cases where small fluctuations in PSA levels are crucial for clinical decision-making in both diagnostic and post treatment settings. Bland-Altman plot of i-CHROMA (POC test) versus Laboratory PSA results.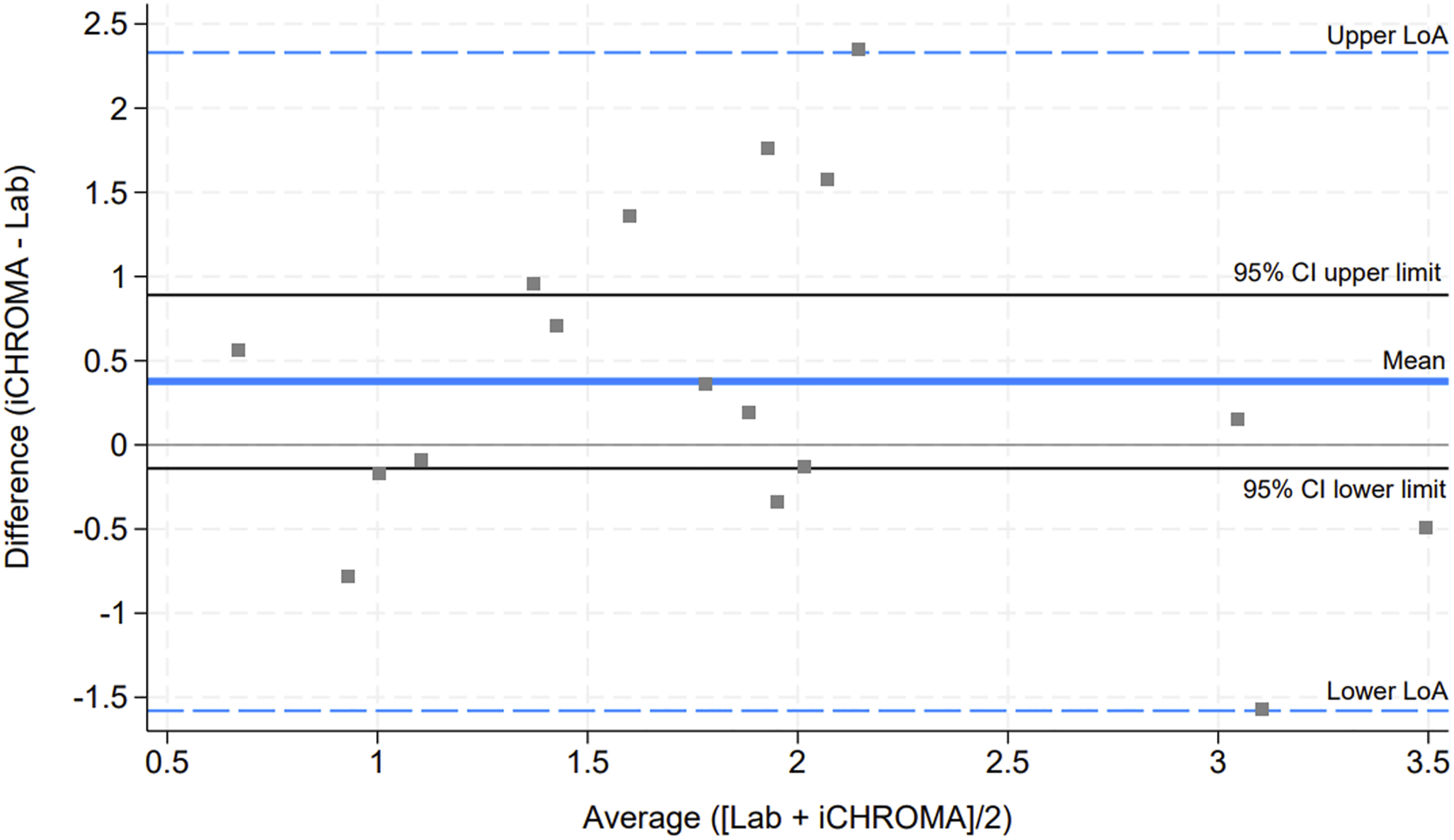
Sensitivity analysis
As part of sensitivity analyses, the nine patients who had been excluded from the above analysis (with a POC test PSA value of <0.5 μg/L) were assigned a PSA value to assess the concordance and agreement results, under the assumption that their POC test PSA values were known.
Different methods were performed as part of the sensitivity analyses, each followed by the calculation of Lin’s Concordance Correlation Coefficient and the Bland–Altman mean and 95% limits of agreement; these was calculated across a total of 26 patients with both a POC test and laboratory PSA result available under the sensitivity analyses.
Sensitivity analyses were performed by assigning PSA values below 0.5 μg/L based on various assumptions, such as random assignment or assigning a value of 0.5 μg/L. These methods were chosen to account for the missing data points while minimizing bias. However, the assumptions made in the sensitivity analyses may introduce additional variability into the results. (1) The first sensitivity analysis method involved the 9 POC test PSA values being randomly assigned a value from a uniform distribution between 0 and 0.5 (using seed number 1). (2) The second sensitivity analysis method involved the 9 POC test PSA values being assigned a value of 0 μg/L. (3) The third sensitivity analysis method involved the 9 POC test PSA values being assigned a value of 0.5 μg/L.
Comparison of Methods of Assigning POC Test PSA Results.

Clinical evaluation
Using current NICE thresholds for PSA referral only one patient had a referral based on the laboratory reading, this patient would not have been referred using the POC test. This participant underwent an MRI scan of the prostate which was normal and therefore did not have a biopsy or diagnosis of prostate cancer (highlighted in green within Figure 1). Similarly there was one positive result on the POC test that should not have been referred (highlighted in purple within Figure 1). 25 out of 27 results were clinically concordant, giving a clinical correlation of 92.6%.
Discussion
Although the sample set is limited, we have shown the first data reporting the reliability of POC capillary blood PSA testing, for the first time in a mobile clinic. While this study represents the first reported data on the use of POC capillary PSA testing in a mobile clinic, the lack of strong concordance with laboratory results raises concerns about the clinical utility of this technology. These findings suggest that, despite the promise of increased accessibility, significant improvements in the accuracy of POC tests are necessary before they can replace laboratory testing, particularly in critical decision-making settings.
Although previous studies22,27,28 have shown good correlation between the i-Chroma™ this was not replicated with our findings in the mobile clinic. Our study did replicate the positive bias found in previous studies with the i-Chroma™.22,27,28 Interestingly of the three studies published on the i-Chroma™ only one contains data on capillary blood. 28 This reinforces the need to have real-world testing of POC tests prior to community based roll-out. In terms of clinical decision making the results of both tests correlated well, but the lack of actual result correlation makes uptake challenging especially when looking at trends in PSA changes which is often important in decision making. The lack of sensitivity of the i-Chroma-II™ below 0.5 ug/L is also problematic for using the machine in post-treatment disease settings.
The limited sensitivity of the i-Chroma-II™ machine below 0.5 ug/L poses significant challenges, particularly in post-treatment surveillance, where small PSA fluctuations are critical for detecting recurrence. This technological shortcoming underscores the need for further refinement of POC tests before they can be relied upon in such high-stakes clinical settings.
POC tests for PSA will continue to be developed and the basis for doing so remains with increased accessibility and potentially reduced costs and resources. 11 For the time being, however, the issues regarding the lack of reliability, and poor sensitivity and specificity remain, 11 particularly as we have shown in real-world environments. Ultimately this will hinder their uptake as the greatest benefit from their use will be in remote/rural environments and further testing is required in these types of settings to ensure that the tests are robust enough to maintain reliability.
Future efforts should focus on increasing the sensitivity of POC tests, particularly at low PSA levels, and on validating these tests across diverse clinical settings. Additionally, large-scale studies are necessary to confirm the generalisability of POC testing in real-world environments, including rural and underserved populations.
There are several limitations to this study, including: the small sample size and single geographic location limit the generalisability of the study’s findings as well as the usage of the newer i-Chroma-II™ machine with no previous studies published using this to test PSA. Furthermore, the exclusion of PSA values below 0.5 ug/L, due to the limitations of the machine restricts the interpretation of the results. However the sensitivity analysis showed incorporating these data may result in higher concordance, the lack of concordance at higher values may be problematic as referral thresholds for possible prostate cancer generally begin at 2.5 ug/L. 30
Future studies should aim for larger, more diverse samples and explore alternative POC devices that can accurately detect PSA at lower levels.
To conclude, although POC PSA testing has significant promise for improving accessibility and reducing the costs of prostate cancer screening, its current limitations in reliability and sensitivity mean it cannot yet replace formal laboratory testing imminently. However, if these challenges are addressed, POC testing could play a critical role in reducing health disparities by expanding access to prostate cancer screening in underserved populations and remote settings.
Footnotes
Acknowledgements
The authors thank JB Consulting (MDP) Limited for kindly donating all reagents and use of the i-CHROMA-II™ reader for this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Phase 2 of the Man Van study was funded by a grant from NHS England. No additional funding was used for this sub-study.
Ethical approval
The Man Van project has ethical approval from Wales Research Ethics Committee (REC) 6. Study title: The Man Van Project: An evaluation of the effectiveness of a mobile outreach screening clinic model for earlier detection of prostate cancer and other male cancers. REC reference: 22/WA/0113. Protocol number: CCR5573. IRAS project ID: 304320.
Guarantor
MM and NDJ accept full responsibility for the integrity of the work as a whole, including the accuracy of the data and analysis.
Contributorship
The specific author contributions are shown below following CRediT (Contribution Roles Taxonomy):- MM: Conceptualisation, Data Curation, Formal Analysis, Funding acquisition, Investigation, Resources, Methodology, Project Administration, Visualisation, Validation, Writing – Original Draft Preparation, Writing – Review & Editing- HB: Methodology, Validation, Formal Analysis, Investigation, Resources, Data Curation, Writing – Review & Editing- FM: Methodology, Validation, Formal Analysis, Investigation, Resources, Data Curation, Writing – Review & Editing- AT: Methodology, Validation, Formal Analysis, Resources, Data Curation, Writing – Review & Editing- FM: Conceptualisation, Data Curation, Investigation, Writing – Review & Editing- EW: Conceptualisation, Data Curation, Investigation, Writing – Review & Editing- FC: Conceptualisation, Data Curation, Investigation, Resources, Project Administration, Writing – Review & Editing- RS: Conceptualisation, Data Curation, Investigation, Resources, Project Administration, Writing – Review & Editing- NK: Conceptualisation, Data Curation, Investigation, Methodology, Resources, Project Administration, Supervision, Writing – Review & Editing- PK: Conceptualisation, Project Administration, Supervision, Writing – Review & Editing- DC: Conceptualisation, Project Administration, Supervision, Writing – Review & Editing- NDJ: Conceptualisation, Funding acquisition, Methodology, Resources, Project Administration, Supervision, Writing – Review & Editing.
