Abstract
Background
Diagnosing acute heart failure in patients presenting with acute dyspnea remains challenging. Current methods, including natriuretic peptide measurement and echocardiography, are time-consuming and not always immediately accessible. A novel biomarker, FILDARIA, may complement natriuretic peptides and enable faster diagnosis.
Methods
In this study (ClinicalTrials.gov: NCT01024049), samples were collected from 235 patients diagnosed via echocardiography: 89 with non-cardiac dyspnea (NCD), 55 with chronic heart failure (CHF), and 91 with acute heart failure (AHF). Levels of BNP and FILDARIA in each patient were measured using both ELISA and lateral flow assay tests.
Results
BNP levels were significantly elevated in AHF and CHF patients compared to NCD patients (905 vs 58 pg/mL, p < .0001; and 447 vs 58 pg/mL, p < .0001, respectively). Similarly, FILDARIA levels were markedly higher in AHF and CHF patients than in NCD patients (1493 vs 223 ng/mL, p < .0001; and 800 vs 223 ng/mL, p < .0001, respectively). The FILDARIA heart failure diagnostic rapid test device accurately identified all 91 AHF patients and correctly excluded 88 of 89 NCD patients, yielding one false positive. Overall diagnostic accuracy was 99.4% (95% CI: 96.9%–99.9%).
Conclusion
FILDARIA biomarker demonstrates strong potential as a rapid
Keywords
Introduction
Heart failure (HF) is a growing global health challenge affecting more than 64 million people. 1 Its prevalence is expected to rise over the next decades due to aging populations, increased survival from myocardial infarction, and rising rates of comorbidities like diabetes, obesity, and hypertension. 2 Projections suggest that by 2030, over eight million people in the U.S. will be affected by heart failure, representing a 46% increase from 2012. 3 The burden in low and middle income countries is also accelerating due to rapid epidemiological transitions.
HF remains one of the most prevalent and complex condition in modern medicine and a leading cause of morbidity and mortality worldwide, characterized by the heart’s inability to pump blood effectively to meet the body’s physiological demands. This condition can manifest in various forms, from chronic stable HF (CHF) to acute exacerbations (Acute heart failure, AHF) that require immediate medical attention, causing frequent and characteristic emergency department (ED) visits and hospitalizations.
Despite therapeutic advances, early AHF diagnosis remains a critical gap. 4 AHF persists as a difficult condition to diagnose, as its symptoms are non-specific, highly variable, and can overlap or mimic with those of other medical conditions, leading to delays in treatments and potentially life-threatening outcomes. Diagnosis traditionally involves a combination of clinical assessment, medical history, physical examination, and multiple diagnostic tests including electrocardiograms, plasma tests for biomarkers such as natriuretic peptides (NPs – BNP/NT-ProBNP), and echocardiography – the gold standard for diagnosing AHF. 5 However, these diagnostic solutions can be time-consuming, especially in emergency settings where time is of the essence, and require equipment and available trained healthcare professionals.
Therefore, a significant unmet need exists for an efficient, specific, standardized reliable, and rapid diagnostic solution. This solution should not only provide accurate identification of AHF but do so with unmatched speed to enable prompt intervention. The urgency for rapid diagnosis is underscored by the fact that during an AHF episode, every moment counts. When the heart enters decompensation, meaning it can no longer maintain adequate blood flow, the heart muscle can suffer additional irreversible damage. 6 The current scenario in many EDs around the world sees patients with suspected HF waiting for hours, sometimes without receiving the critical care they urgently need. This delay can lead to increased hospital stays and higher mortality rates. 7
The use of point-of-care testing (POCT) to promptly support clinical decision-making has demonstrated performance comparable to traditional laboratory-based models in identifying patients at high risk of mortality. With results available within minutes, POCT shows strong potential for early risk stratification in the ED. 8 Furthermore, timely diagnosis and immediate treatment of patients presenting with acute heart failure (AHF) have been reported to reduce disease progression and improve clinical outcomes. 7
Consequently, the development and implementation of a POCT solution for AHF could be transformative. Such a test would ideally be quick to administer and interpret, reducing the diagnostic window from hour(s) to minutes. This would not only allow for quicker initiation of heart-saving treatments thereby preventing further heart damage, saving lives but also potentially decreasing the overall strain on emergency healthcare resources by enhancing triage efficiency and reducing healthcare costs. Fast and accurate diagnostic methods are thus essential to altering the disease trajectory and easing the socioeconomic impact of HF.
The French biomedical R&D company Spartacus-Biomed based in Auterive (Haute-Garonne) which specializes in advanced diagnostic and therapeutic solutions for complex medical conditions has developed a novel rapid in vitro diagnostic test for AHF based on a novel heart failure biomarker called FILDARIA. FILDARIA was discovered through proteomic analysis of dyspnoeic patients samples by capillary electrophoresis coupled to mass spectroscopy, generating one gigabyte of spectral data (i.e. peaks of varied intensity) per patient. After peaks alignment and statistical analysis, differentially represented peaks (the putative biomarkers) between non-cardiac dyspnea and heart failure patients were identified by Orbitrap liquid chromatography-MS and validated by western blot analysis. Spartacus-Biomed subsequently developed a FILDARIA ELISA and a rapid qualitative test (‘FILDARIA heart failure diagnostic rapid test device’) providing results in 10 min from a single drop of blood, aiming to provide effective assistance to healthcare professionals in making a rapid and efficient diagnosis while avoiding the excessive waiting times between the onset of HF decompensation and the initiation of appropriate treatment.
The aim of this study is to evaluate the diagnostic performance of this novel blood biomarker in AHF patients diagnosed through echocardiography, the ‘gold standard’ diagnostic method 5 by assessing the clinical performance of FILDARIA using both FILDARIA ELISA and FILDARIA rapid test, and conducting a comparative analysis against BNP values, a commonly used biomarker alongside other diagnostic tools to confirm the presence and severity of HF. 9 If proven reliable, this advancement in diagnostic technology would mark a significant step forward in the battle against one of the leading causes of hospitalization, paving the way for better testing and better care for patients worldwide.
Material and methods
Ethical considerations and study population
This study was conducted using blood and plasma samples from the IBLOMAVED cohort. The research protocol was registered in a clinical database (ClinicalTrials.gov: NCT01024049), adhered to the ethical guidelines of the 1975 Declaration of Helsinki, received approval from both the institutional human research committee (COSSEC) and the regional ethics committee (Comité de Protection des Personnes (CPP) # DC2008–452)). Written informed consent was obtained from all participants and/or their legally authorized representatives. This IBLOMAVED cohort included patients at all stages of HF according to the New York Heart Association (NYHA) classification, ranging from Asymptomatic Left Ventricular Dysfunction to Acute Heart Failure. Patients with the following conditions were excluded: end-stage renal disease (stage 5D) or renal transplant (stage 5T), active infectious disease, and known malignancy. All clinical data were recorded via chart review.
AHF patients and non-cardiac dyspnea (NCD) control patients
All selected AHF and NCD patients were admitted at the ED of the University Hospital of Toulouse (France) with acute dyspnea from August 2013 to March 2025. Patients were selected through a simple random sampling method. For HF diagnosis, a cardiac ultrasound device (GE Healthcare – Vivid™) was used to perform bedside echocardiography for each patient. This approach was necessary because natriuretic peptide levels often fall within a diagnostic grey zone, making it difficult to definitively confirm or exclude heart failure based on biomarkers alone.10,11 We obtained two groups of patients: 89 control patients with non-cardiac dyspnea (NCD) and 91 patients diagnosed with AHF. This study focused on AHF patients with Left Ventricular Ejection Fraction (LVEF) < 40%.
Chronic heart failure patients (CHF)
The IBLOMAVED cohort study also enrolled out-patients receiving care at the cardiology department of the University Hospital of Toulouse (France) from August 2013 to March 2025. Our data set consists of 55 stable CHF patients without any acute decompensation for at least 3 months prior to inclusion. All clinical data were collected through detailed chart review, including demographic characteristics, comorbidities, medication history, and relevant laboratory and imaging findings.
Biological sample collection
Blood samples were collected with consent from patients through venesection and drawn into sodium/EDTA tubes to prevent clotting along with the other blood samples routinely collected at the emergency or cardiology departments. Whole blood and plasma samples were tested for AHF and NCD patients using the FILDARIA Heart failure Diagnostic Rapid Test Device. For ELISA quantification test, the blood sample tubes were centrifuged at 2500 g for 10 min at 4°C and the plasma was carefully collected, aliquoted, and stored at −80°C until use. Test results were recorded alongside patient history information and additional investigations for each patient, including BNP values.
Analytical methods
Immune methods
The FILDARIA concentration was measured using the Enzyme-Linked Immunosorbent Assay (ELISA) kit (Spartacus-Biomed Product reference: SB94050608). The assay was performed in accordance with the manufacturer’s protocol. It is a colorimetric sandwich ELISA designed for quantitative measurement in human plasma samples, with a detection range of 58.9 pg/ml to 5000 pg/ml. The intra-assay and inter-assay coefficients of variation were less than 8% and 10%, respectively. Each sample required a volume of 50 µL (diluted plasma), and measurements were obtained at a wavelength of 410 nm using a microplate reader. All samples were analysed in triplicate to ensure reliability and reproducibility. Kit components were stored at 2–8°C or −80°C according to the kit insert. Patients’ plasma samples were stored at −80°C until analysis.
The FILDARIA lateral flow rapid tests (FILDARIA Heart failure Diagnostic Rapid Test Device, Reference: SB94050609) were used as per the manufacturer’s specifications. We used freshly collected samples for optimal test performance and allowed the test device and samples to equilibrate to room temperature (18–30°C) prior to testing. The test cassette was removed from its sealed pouch and immediately used on a level surface. One drop of blood was transferred into the sample well of the cassette with two drops of running buffer (Reference: RBFIL202408A). Results were interpreted after 10 min through visual detection, without reader. A positive result was indicated by the presence of a T line, while a negative result was defined by its absence. The C line, serving as an internal control, was present in all tests, confirming both sufficient sample volume and proper test procedure.
The FILDARIA Heart Failure Diagnostic Rapid Test Device uses an immunochromatographic membrane assay with sensitive monoclonal antibodies directed against the biomarker. The test strip is composed of a sample pad, a conjugate pad, and a reaction membrane. The conjugate pad contains gold nanoparticles covalently conjugated with the first monoclonal FILDARIA antibody. The reaction membrane contains the second FILDARIA monoclonal antibody and the polyclonal antibodies against the mouse immunoglobulin, which are pre-immobilized on the membrane on T and C lines, respectively. When the sample and buffer are added in the sample well of the test cassette, the conjugates dried in the conjugate pad are solubilized and migrate along with the sample. The biomarker, present in the sample forms a complex with the gold conjugate. This complex will be caught by the second monoclonal antibody coated on the ‘T’ line which then is coloured by the bound conjugate, providing the test result. The excess of conjugate will be immobilized by the goat anti-mouse IgG stripped on the C line. Visualization of the latter serves as an internal control, indicating proper test procedure and correct reagent flow across the membrane. The analytical sensitivity of the lateral flow assay (LFA), expressed as the limit of detection (LoD), is established at 500 ng/mL.
Statistical analysis
Normal distribution of values was evaluated for all variables using the d'Agostino–Pearson test for Normal distribution. Continuous data were presented as mean +/− standard deviation, while categorical variable were presented as number (percentage). Comparisons among the groups of patients (NCD vs AHF; NCD vs CHF; CHF vs AHF) were done using the Student’s test. Spearman correlation coefficient was assessed to detect potential associations of FILDARIA and BNP levels with other parameters. A two-tailed P-value <.05 was used to determine significant associations. All statistical analyses were performed with MedCalc® Statistical Software version 23.1.3 (MedCalc Software Ltd, Ostend, Belgium).
Results
Demographic and clinical data
Compared to NCD patients, AHF patients were significantly older and had higher heart rates, smoking, hypertension, C reactive protein, ALT, and diabetes rates but lower LVEF, mean blood pressure, obesity, creatinine clearance, Na+, and dyslipidemia. AHF patients also had a greater history of cardiac diseases and were more frequently prescribed cardiac medications, except a similar prescription of corticoids, statins, and ARAII.
Compared to NCD patients, CHF patients were significantly older and had higher heart rates, and C reactive protein concentrations while maintaining lower LVEF, mean blood pressure, creatinine clearance, Na+, and dyslipidemia rates. CHF patients also had a greater history of cardiac diseases except for hereditary HCM and were more frequently prescribed cardiac medications, except a similar prescription of corticoids, statins and ARAII.
Compared to CHF patients, AHF patients were significantly older and had higher diabetes rate with lower creatinine clearance and LVEF. AHF patients also had a similar history of cardiac diseases and prescribed cardiac medications, with the exception of a higher prescription of diuretics.
These findings suggest an association of the HF severity with increased renal dysfunction and altered hemodynamic parameters. Inflammatory markers such as C reactive protein rate were significantly elevated in AHF and CHF patients, indicating a potential link between systemic inflammation and heart failure progression.
ALT was more elevated in AHF patients vs NCD, also indicative of liver inflammation or cell damage. Finally, blood sodium was slightly reduced in AHF and CHF patients vs NCD, indicative of frequent mild hyponatremia in HF patients. 12
FILDARIA plasma levels in AHF, CHF and NCD patients
The box plot (Figure 1(a)) illustrates the distribution of FILDARIA levels across three patient groups: non-cardiac dyspnoeic patients (NCD), chronic heart failure patients (CHF), and acute heart failure patients (AHF). Statistically significant differences were observed between all groups (p < .0001). FILDARIA levels were significantly increased in the plasma of AHF and CHF patients compared to NCD patients (1493 vs 223 ng/ml, p < .0001, i.e., an average of 7-fold increase) and (800 vs 223 ng/ml, p < .0001, i.e., an average of 3-fold increase), respectively (Table 1). Comparison of (a) FILDARIA plasma levels (ng/ml) and (b) B-type natriuretic peptide (BNP) plasma levels (pg/ml) in non-cardiac dyspnea (NCD), chronic heart failure (CHF), and acute heart failure (AHF) patients assessed by ELISA. IBLOMAVED cohort clinical and demographic parameters. Abbreviations: BMI: Body Mass Index, ACE Inhibitor: Angiotensin-Converting Enzyme Inhibitor, ARA2: Angiotensin II Receptor Blockers (ARBs), BNP: B-Type Natriuretic Peptide, ALT: Alanine Aminotransferase, LVEF: Left Ventricular Ejection Fraction.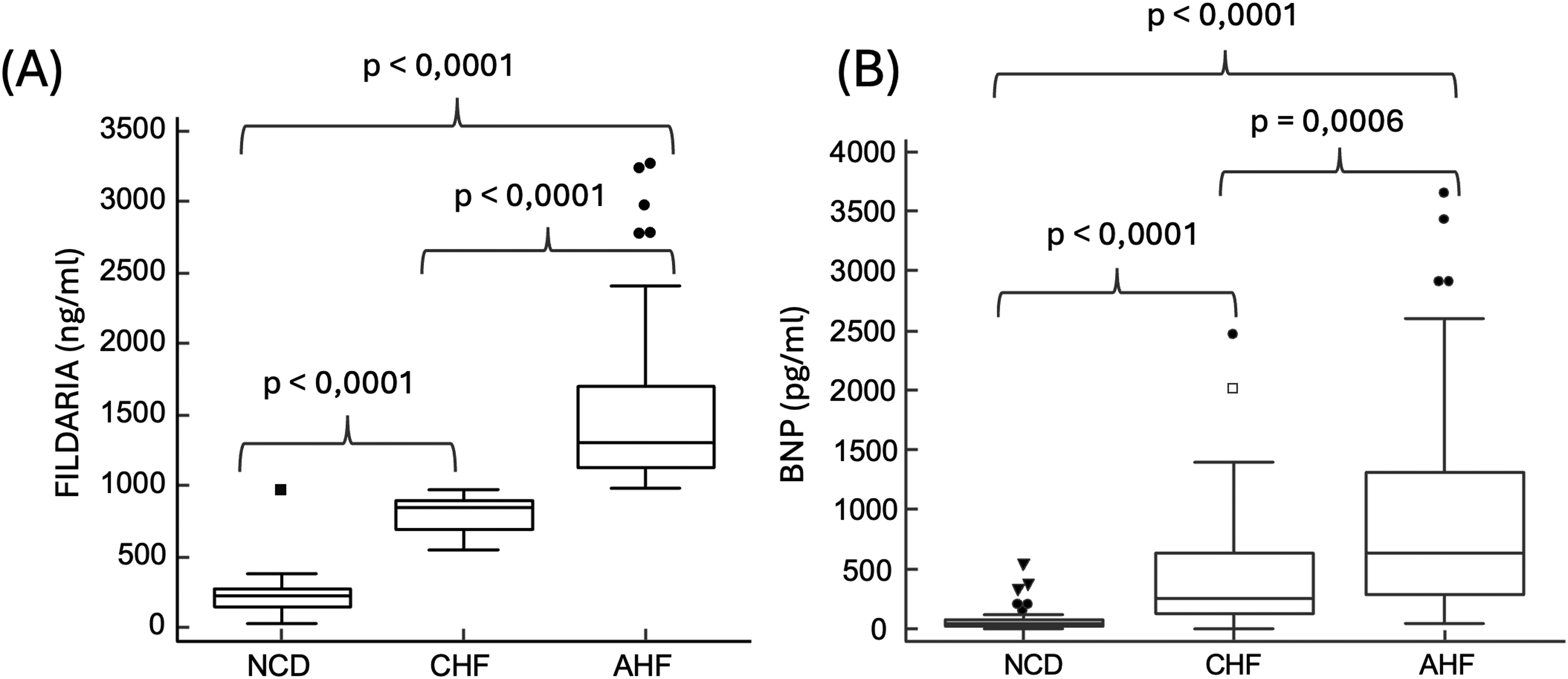
BNP plasma levels in AHF, CHF, and NCD patients
Another box plot (Figure 1(b)) depicts the BNP levels in the three patient groups (NCD, CHF, and AHF). As expected, BNP levels were significantly elevated in AHF patients and CHF patients compared to NCD patients (905 vs 58 pg/ml, p < .0001, and 447 vs 58 pg/ml, p < .0001, respectively (Table 1). The median BNP value was lowest in NCD patients, followed by CHF patients, with AHF patients exhibiting the highest BNP levels. Outliers were also observed, particularly in the NCD group.
As seen for both biomarkers, the mean levels between CHF and AHF were doubled (Figure 1), but BNP displayed a higher variability of concentration than FILDARIA in each tested group. Both box plots indicate that FILDARIA followed a similar trend to that of BNP in differentiating between non-cardiac dyspnea, chronic heart failure, and acute heart failure in our cohort of patients diagnosed by echocardiography. However, BNP levels (pg/mL) showed overlapping values between chronic heart failure (CHF) and acute heart failure (AHF) groups, with some CHF patients having BNP levels similar to AHF patients (Figure 1(b)). This overlap makes it more challenging to distinguish CHF from AHF based solely on BNP, which aligns with previous studies showing BNP’s diagnostic uncertainty in intermediate ranges (∼100-500 pg/mL). 10 FILDARIA in Figure 1(A) shows a more distinct separation between CHF and AHF groups, with less overlap compared to BNP, making it a potentially more reliable tool for distinguishing AHF from CHF and from non-cardiac dyspnea, therefore potentially reducing misclassification and improving diagnostic accuracy.
Correlation between BNP plasma levels and LVEF
Panel (Figure 2(a)) shows the relationship between BNP plasma levels (pg/mL) and LVEF (%). A significant negative correlation was observed (r = −0.54, p < .0001), indicating that higher BNP levels are associated with lower LVEF which is consistent with the established role of BNP as a biomarker reflecting ventricular dysfunction. Correlation between (a) BNP plasma levels, (b) FILDARIA plasma levels, and Left Ventricular Ejection Fraction (LVEF) for all patients groups (AHF, CHF, and NCD patients). (c) Correlation between BNP plasma levels and FILDARIA plasma levels for all patients groups (AHF, CHF, and NCD patients).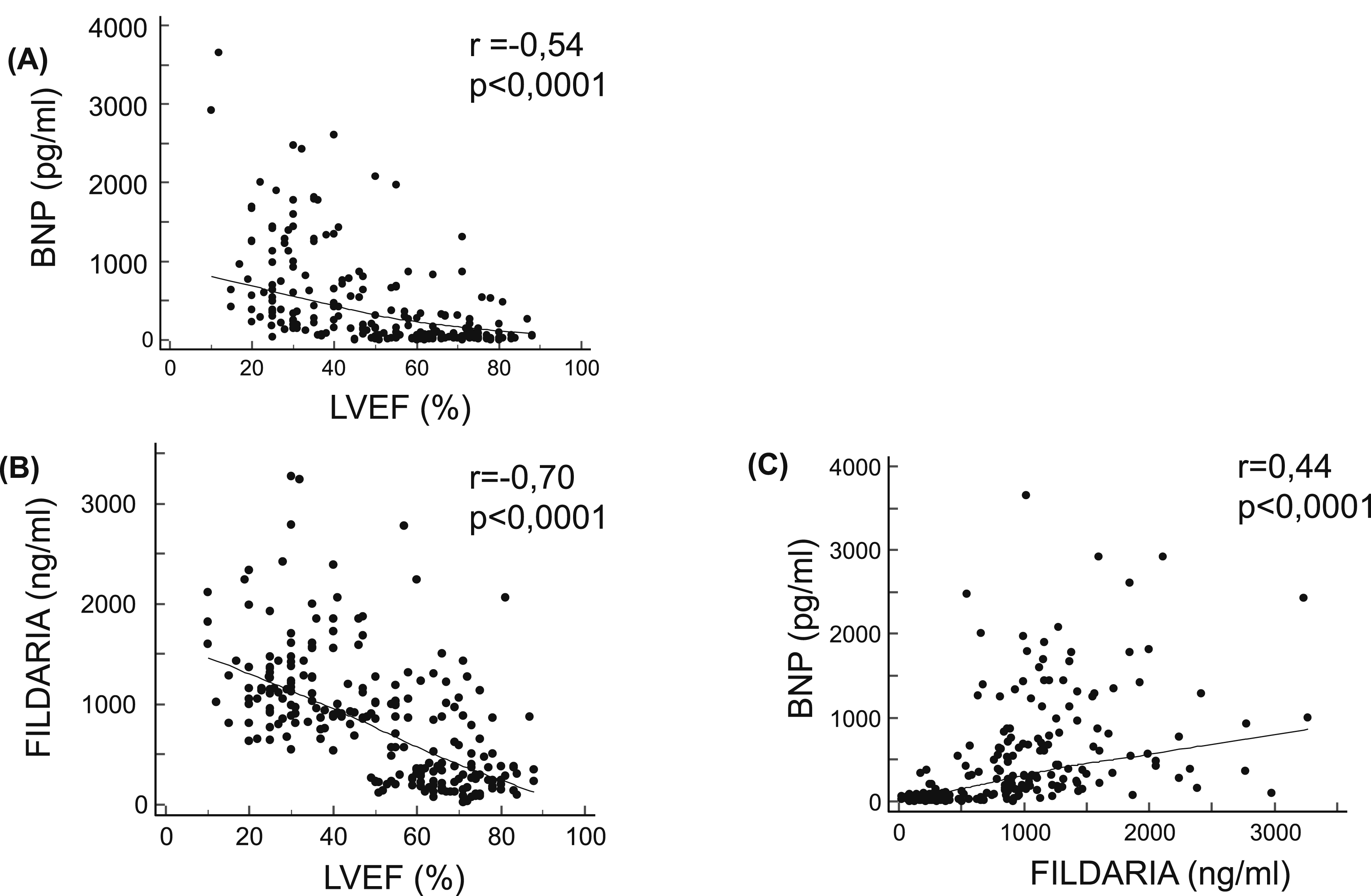
Correlation between FILDARIA plasma levels and LVEF
Panel (Figure 2(b)) depicts the correlation between FILDARIA (ng/mL) and LVEF (%). In comparison with BNP, a similarly strong negative correlation was identified (r = −0.70, p < .0001), suggesting that increasing levels of FILDARIA are also linked to left ventricular systolic dysfunction. Interestingly, the correlation coefficient for FILDARIA is slightly stronger than that of BNP, implying that it may provide comparable or superior value in assessing cardiac dysfunction.
Correlation between FILDARIA and BNP plasma levels
Panel (Figure 2(c)) presents the correlation between FILDARIA (ng/mL) and BNP (pg/mL) plasma levels. A significant positive correlation was found (r = 0.44, p < .0001), indicating that higher levels of FILDARIA correspond to higher BNP concentrations. This suggests that FILDARIA could serve as a supportive tool alongside BNP, offering further information on the underlying cardiac status. However, the moderate strength of the correlation indicates that the two biomarkers may capture partially distinct physiological processes.
Taken together, these findings suggest that FILDARIA exhibits a strong inverse correlation with LVEF, comparable to that of BNP. Furthermore, its significant association with BNP levels supports its potential role as a new biomarker for heart failure diagnosis and severity assessment.
BNP and FILDARIA correlation analysis with diastolic Function
Positive correlations were observed with E/A (BNP: r = 0. 4797, FILDARIA: r = 0.3045) and E/Ea (BNP: r = 0.2716, FILDARIA: r = 0.3745) indicating that higher biomarker levels are associated with reduced diastolic function.
Positive correlations were also recorded with Left Atrial (LA) size (BNP: r = 0. 5417, FILDARIA: r = 0.4403) suggesting that higher biomarker levels are associated with LA size, an important marker for the chronicity of elevated left ventricular filling pressures and a powerful predictor of adverse cardiovascular outcomes, including stroke, development of atrial fibrillation, congestive heart failure, and death.13,14
BNP and FILDARIA correlation analysis with ventricular size and function
Positive correlations were documented with LVEDD (BNP: r = 0.4538, FILDARIA: r = 0.3108), LVESD (BNP: r = 0.5801, FILDARIA: r = 0.3491), and LVEDV (BNP: r = 0.3944, FILDARIA: r = 0.4803) implying that higher biomarker levels are associated with enlarged left ventricular dimensions, a key feature of ventricular dysfunction in heart failure.
Right ventricular function and pulmonary pressure markers
Both biomarkers exhibited significant negative correlations with TAPSE (BNP: r = −0.3784, FILDARIA: r = −0.5257) and tricuspid annulus area (BNP: r = −0.423, FILDARIA: r = −0.5063). This suggests that higher biomarker levels are associated with impaired right ventricular function, which is common in advanced heart failure with pulmonary hypertension. PAPSE (a marker of pulmonary arterial systolic pressure) correlates with both biomarkers, indicating their relevance in evaluating pulmonary congestion.
Lack of correlation with septal thickness (SIV) and LVESV
Septal thickness (SIV) showed non-significant correlations with BNP (r = 0.0707, P = .3932) nor FILDARIA (r = 0.1022, p = .2134). LVESV did not show significative correlation, particularly with FILDARIA (r = 0.03201, p = .7187) or with BNP (r = 0.1535, p = .0836), suggesting that the two biomarkers’ levels may not be associated with end-systolic volume.
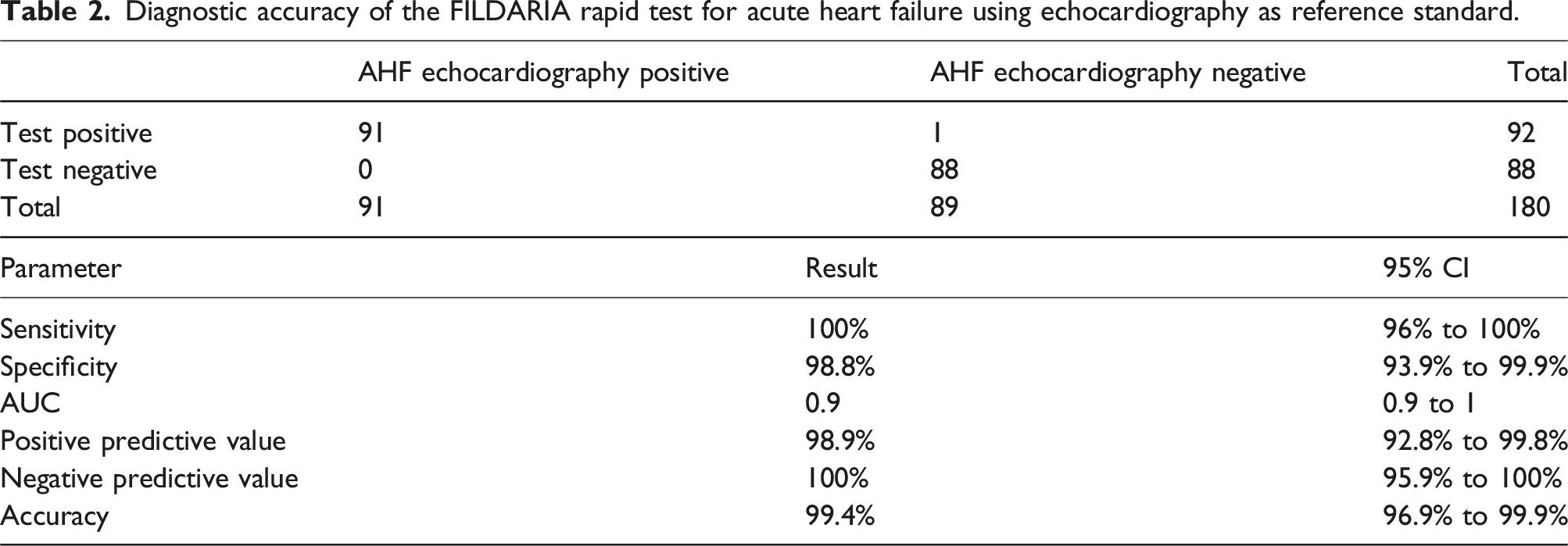
Diagnostic performance of the FILDARIA rapid test
Diagnostic accuracy of the FILDARIA rapid test for acute heart failure using echocardiography as reference standard.

FILDARIA rapid test analysis of AHF (A) and NCD (N) patients whole blood samples. Patients 1 to 15 from each group are displayed.
Discussion
Comparative analysis of FILDARIA and BNP in acute heart failure
The results presented in Figure 1 revealed a strong correlation between FILDARIA and BNP in differentiating between NCD, CHF, and AHF. Both markers exhibit a stepwise increase from NCD to CHF and both peak in AHF, suggesting their role in heart failure diagnostics. However, several key points merit further discussion:
Diagnostic challenges of BNP in acute heart failure and the emerging role of FILDARIA
BNP is a well-established biomarker and is widely used for AHF diagnosis in patients presenting with acute dyspnea. 15 However, the findings presented in Figure 1 also highlight a limitation of BNP: the presence of an overlap between CHF and AHF values. Another well-known limitation of natriuretic peptides is a diagnostic grey zone affecting 25–30% of patients tested at ED, BNP values (∼100–500 pg/mL) lacking specificity and leading to diagnostic uncertainty.10,11,16 Furthermore, NPs are highly influenced by other factors such as atrial fibrillation 17 or are significantly impacted by age, BMI, or renal function, affecting diagnostic thresholds. 18 Differentiation between AHF and non-AHF conditions can therefore be uncertain, making it difficult to confirm or exclude HF in patients with elevated NP levels. This has important clinical consequences, particularly in emergency settings where timely and accurate diagnosis is crucial for guiding treatment decisions. As shown in Figure 1, FILDARIA appears to have clearer cutoffs, with less overlap between the different groups of patients. This means that clinicians could use FILDARIA to more confidently distinguish acute heart failure, potentially reducing unnecessary hospitalizations or delays in appropriate treatment. Patients with borderline BNP levels or uncertain AHF diagnosis but elevated FILDARIA levels could be prioritized for treatment, while those with low levels could avoid unnecessary interventions. While FILDARIA shows promise as a standalone diagnostic tool, combining it with BNP could therefore further improve accuracy. This multi-marker approach could allow for more precise cutoffs for ruling in or out AHF, and for personalized treatment strategies based on biomarker profiles.
Diagnostic performance and potential advantages of FILDARIA in clinical practice
Since FILDARIA concentration levels in plasma or blood are measured in nanograms per millilitre (ng/mL), whereas BNP/NT-proBNP levels are in picograms per millilitre (pg/mL), the significantly higher concentration of FILDARIA enabled the development of a reliable and efficient rapid diagnostic qualitative test (FILDARIA Heart Failure Diagnostic Rapid Test Device, Spartacus-Biomed, Auterive, France). Unlike BNP/NT-proBNP assays, which often require sensitive laboratory techniques and specialized equipment to detect very low biomarker concentrations that remain in the picogram range even in very severe situations (BNP: 50 to 10000 pg/ml
19
and NT-proBNP up to 100000 pg/ml
20
), the FILDARIA rapid test can be performed without the need for a dedicated reader or signal enhancement methods requiring complex and expensive laboratory instrumentation. Notably, the FILDARIA rapid test performs with both plasma and blood, and therefore also eliminates the need for centrifugation to obtain plasma. Moreover, due to the important difference in concentrations between NCD and AHF patients, NCD patients exhibit no detectable signal, whereas AHF patients display a strong signal that is easily visible to the naked eye. These advantages make it highly suitable for point-of-care testing, particularly in emergency settings where quick diagnosis is critical. By eliminating the need for advanced medical equipment, the test could be easily implemented in a variety of clinical environments, including EDs, ambulances, and resource-limited healthcare facilities. Furthermore, the FILDARIA rapid diagnostic test led us to discriminate AHF patients from NCD patients in our cohort within 10 min using a single drop of blood and provided a high sensitivity (100 %) and specificity (98,876%) demonstrating excellent diagnostic performance. FILDARIA rapid testing would not necessarily be able to reliably determine between CHF and AHF patients, as it is not intended for this purpose and does not provide quantitative measurements. Patients with established CHF are generally aware of their diagnosis, are usually already under medical supervision and would not typically require LFA testing. This limitation aligns with its intended use as a qualitative, point-of-care tool to aid in the diagnosis of AHF, particularly in patients with no prior history of HF who often present with non-specific or ambiguous symptoms (undiagnosed or ‘de novo’ cases of AHF). It provides
Pathophysiological insights and biomarker origin and kinetics
Correlation analysis between echocardiographic parameters and biomarkers (BNP and FILDARIA); all patients groups are included in the correlation analysis.
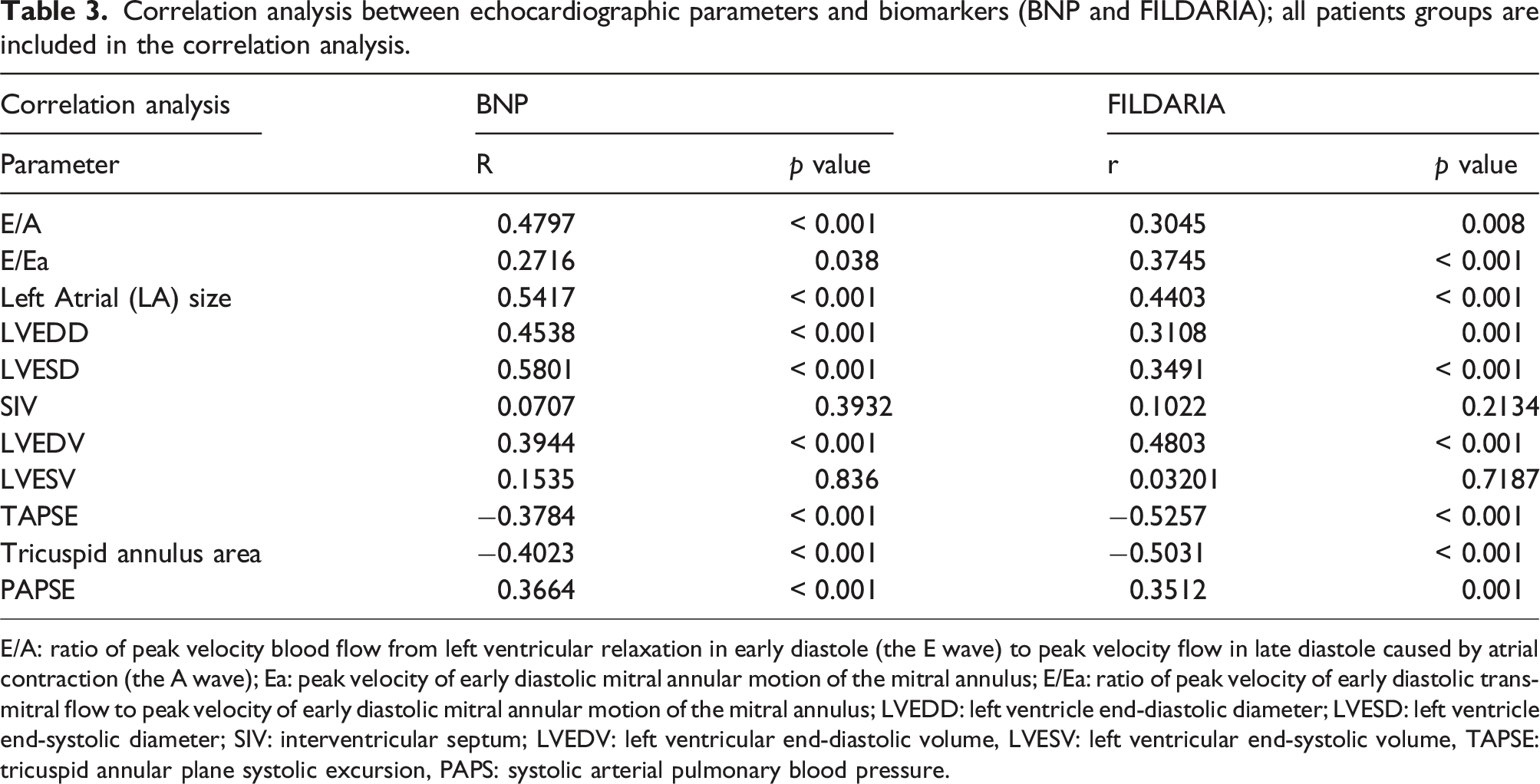
E/A: ratio of peak velocity blood flow from left ventricular relaxation in early diastole (the E wave) to peak velocity flow in late diastole caused by atrial contraction (the A wave); Ea: peak velocity of early diastolic mitral annular motion of the mitral annulus; E/Ea: ratio of peak velocity of early diastolic trans-mitral flow to peak velocity of early diastolic mitral annular motion of the mitral annulus; LVEDD: left ventricle end-diastolic diameter; LVESD: left ventricle end-systolic diameter; SIV: interventricular septum; LVEDV: left ventricular end-diastolic volume, LVESV: left ventricular end-systolic volume, TAPSE: tricuspid annular plane systolic excursion, PAPS: systolic arterial pulmonary blood pressure.
Prognostic and potential therapeutic implications of FILDARIA
BNP is not only a diagnostic marker but also carries prognostic significance, with higher levels correlating with increased mortality and readmission rates. 18 In our study, we observed a statistically significant elevated positive correlation of FILDARIA and BNP levels (Figure 2). Using FILDARIA ELISA quantification, we observed that circulating FILDARIA levels are low in NCD patients, around 220 ng/mL, and are seven times higher in AHF patients, averaging 1500 ng/mL (Table 1). In CHF patients, the average circulating FILDARIA level is lower, around 750 ng/mL, which is three times the level observed in NCD patients. Since CHF patients receive treatment, this could explain their reduced circulating FILDARIA levels compared to AHF patients, and therefore FILDARIA could also be indicative of the disease severity and possible outcome. Treated AHF patients commonly become CHF patients and remain at risk of cardiac decompensation and rehospitalization. They are typically monitored by their cardiologist in outpatient settings or within cardiology departments, with regular follow-up visits to assess cardiac function. However, decompensation may still occur between scheduled consultations, highlighting the need for better monitoring strategies, with studies reporting a 40 to 50 % survival rate 5 years after AHF diagnosis. 23 A major contributor to this high mortality and frequent hospital readmissions is inadequate follow-up, a pattern previously observed in diabetes before effective self-monitoring and management strategies were introduced. An ideal approach would be a Home Monitoring Study of CHF Patients empowered with user-friendly FILDARIA rapid diagnostic tests designed for home use. CHF patients would periodically test themselves at home in combination with a connected reader for remote monitoring. This system would enable detection of early physiological signs of decompensation, allowing for timely medical intervention and anticipations, reducing the risk of hospitalizations or potentially life-threatening outcomes. Just as diabetic patients manage their condition through home glucose monitoring, this innovation could help heart failure patients maintain a more stable condition and a better quality of life. We could also ideally envision a FILDARIA threshold value at which the connected reader sends an alert directly to the healthcare professionals and the Spartacus-Biomed team stated that they are already working on a prototype for such a connected reader. FILDARIA could then serve as a valuable prognostic tool, a new CHF monitoring solution, and may guide therapy and risk stratification more effectively than NPs alone.
Conclusion
This study aimed to evaluate the diagnostic performance of FILDARIA and compare it to the established BNP in patients presenting with acute dyspnea suspected of AHF. Our findings demonstrate that FILDARIA exhibits a promising pattern similar to that of BNP in distinguishing between NCD, CHF, and AHF while overcoming some of BNP’s limitations. Demonstrating arguably higher sensitivity and specificity (100% and 98.8% respectively), the FILDARIA rapid test could emerge as a valuable diagnostic and potentially prognostic tool for AHF. In addition, BNP/NT-proBNP measurements require laboratory instruments that are generally located in central laboratories because of their cost and the need for specialized staff for their use which partially explain why de novo HF diagnosis takes time. Unfortunately, it has been proposed that ‘time is prognosis’ in HF, 7 observation driven by the ‘time is muscle’ concept. 24 The time to establish an accurate diagnosis, mandatory to start an effective treatment, was shown to be a prognostic factor in AHF patients. In this context, the use of this new biomarker rapid test that can be performed at the patient’s bedside on a blood sample in just 10 min without the need for additional equipment could be transformative. FILDARIA’s stability, cost-effectiveness, and ease of measurement would certainly determine its adoption. Its high performance, driven by a highly sensitive lateral flow assay, is consistent with the well-established capabilities of this technology, which has matured over the past 60 years and demonstrated sensitivities of up to 100% in specific evaluation studies. 25 Although this single-centre study had a limited sample size, the findings support FILDARIA’s clinical utility while confirming that the test is operational and delivers reliable performance. A strength of this study is its focus on a well-defined population of AHF patients with a left ventricular ejection fraction (LVEF) below 40%, confirmed through echocardiographic screening. This targeted approach likely contributed to the high overall diagnostic performance observed. To enhance the generalizability of these findings, future research should include more diverse patient populations, such as those with preserved (HFpEF, LVEF ≥50%) or mildly reduced LVEF (HFmrEF, LVEF 40–49%). This study also demonstrates a high degree of specificity and promising stability of FILDARIA values across diverse patient profiles. Despite the presence of patients with prior cardiac diseases and age variations in each group, our boxplot (Figure 1) shows clearly distinct cutoff values with minimal to no overlap between the different groups. While we did not specifically assess the influence of multiple confounding factors as extensively studied with natriuretic peptides (NPs), the consistency observed in our results supports the robustness of FILDARIA in varied clinical contexts. Further research may also expand on its prognostic value, potential use in treatment monitoring, and integration into multi-marker strategies or AI-assisted diagnostic tools for heart failure. The incorporation of FILDARIA into emergency diagnostic protocols could enhance the accuracy and speed of AHF diagnosis, particularly in patients where BNP interpretation is limited, improving triage efficiency, reducing unnecessary hospital admissions, and healthcare costs. In conclusion, this study supports FILDARIA’s diagnostic value as a potentially more precise and clinically useful alternative to BNP, paving the way for more nuanced and effective approaches to AHF diagnosis, potentially allowing better testing and better care for patients worldwide.
Limits of the study
We conducted a study in which only 235 patients were enrolled. This limited recruitment was primarily due to strict inclusion criteria, the exclusion of patients participating in other clinical protocols, the need for patient consent, the requirement for complete clinical data, and the availability of healthcare professionals to perform study-related procedures. To enhance the generalizability and robustness of our findings, future collaborations with other centres are welcome and encouraged.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study received approval from both the institutional human research committee (COSSEC) and the regional ethics committee (Comité de Protection des Personnes (CPP) # DC2008–452).
Guarantor
PR.
Contributorship
KR and PR conceived the study and drafted the initial manuscript. PR and MG contributed to protocol development, obtained ethical approval, managed patient recruitment, and performed data analysis. FK conducted the literature review and contributed to data analysis and manuscript preparation. All authors reviewed, revised, and approved the final version of the manuscript.
