Abstract
Background and objective
The disaccharide loading test is a method to assess gastric mucosal damage. Since Trelan-G75, which is used for the sugar tolerance test, contains disaccharide maltose, if maltose is detected at a high sensitivity in the sample blood used in the sugar tolerance test, screening for upper gastrointestinal mucosal damage can be made simultaneously with the sugar tolerance test for the diagnosis of diabetes.
Methods
Glucose-6-phosphate is generated by treating maltose with maltose phosphorylase, β-phosphoglucomutase, and glucose-1,6-bisphosphate. Then, change in the absorbance at 405 nm is measured by the enzymatic cycling method using Thio-NADP, β-NADPH, and Glucose-6-phosphate dehydrogenase. After evaluating the optimal condition for this method, it is mounted on an automatic biochemical analyzer, and samples after the sugar tolerance test were assayed.
Results
Regarding the performance of this method, the repeatability was 10–50 μmol/L with a CV of ≤1.1%. Concerning the assay range, a curve passing the origin with a range of linearity up to 120 μmol/L was obtained. No effect of dyes or sugars in the blood was noted. As a result of application to patients with gastric mucosal disorders (those who had a health checkup), significant differences were observed depending on the stage of atrophic gastritis.
Discussion
This method has a high sensitivity and a high precision and can be used for high-speed analysis on an automatic analyzer. It has the potential to be used as a screening test for gastric mucosal damage.
Background
Screening for gastric and esophageal cancers has been made by imaging examinations such as barium X-rays and endoscopy or the measurement of blood pepsinogen. However, both of the former two examinations pose a large burden on examinees, and the latter is effective only for detecting the high-risk group for gastric cancer. 1
In 1993, Meddings et al. 2 reported that, if there is damage in the gastric mucosa, low-molecular-weight disaccharides are transferred to blood after oral intake of sucrose. In healthy individuals, sucrose is absorbed not in the stomach but in the small intestine after being degraded into glucose and fructose, and, therefore, little disaccharide sucrose appears in blood. However, if the gastric mucosa is damaged, sucrose is absorbed without being degraded, so gastric mucosal damage can be detected by measuring the sucrose concentration in blood or urine. Thereafter, this phenomenon has been reported to be useful for the diagnosis of gastric mucosal damage caused by non-steroidal anti-inflammatory drugs or Helicobacter pylori.3–7
Considering that this phenomenon can be utilized in screening for gastric mucosal damage, we developed a highly sensitive method for the measurement of blood sucrose as a 100 g sucrose tolerance test 8 and evaluated it in gastric cancer patients. As a result, gastric cancer could be detected in 100% of the patients with advanced disease and in 90% of those with early disease by the measurement of the blood sucrose concentration 60 minutes after sucrose loading. 9 However, this test has not gained widespread acceptance, because it requires drinking a solution containing 100 g of sucrose.
On the other hand, oral carbohydrate solution (Trelan G75) used in glucose tolerance tests for diabetes diagnosis contains 36% maltose. Therefore, gastric mucosal disorders may be detected by measuring plasma maltose concentration in plasma after a glucose tolerance test.
Reported methods for measuring maltose include mass spectrometry, 10 HPLC methods, 11 infrared spectroscopy, 12 and enzymatic methods. 13 However, mass spectrometry, HPLC, and infrared spectroscopy require special equipment and are complicated to perform. The enzymatic method has the disadvantage of low measurement sensitivity.
We developed a new highly sensitive measurement method using maltose phosphorylase as the starting enzyme, and measured the maltose concentration in plasma after a glucose tolerance test in patients undergoing comprehensive medical checkups. This maltose concentration was compared with the diagnosis results of gastric mucosal disorder determined by gastric X-ray examination or gastroscopy.
Materials and methods
Analysis methods
Maltose phosphorylase (MP, EC 2.4.1.8) and β-phosphoglucomutase (β-PGM, EC 5.4.2.6) were purchased from Kikkoman Corporation (Tokyo, Japan), Thionicotinamide-adenine Dinucleotide Phosphate-oxidized (Thio-NADP) and β-Nicotinamide Adenine Dinucleotide Phosphate Reduced Form (β-NADPH) were purchased from Oriental Yeast Co, Ltd. (Tokyo, Japan), Glucose-6-phosphate dehydrogenase (G-6-PDH, EC 1.1.1.49) was purchased from Asahi Kasei Pharma (Tokyo, Japan), and α-D-Glucose 1,6-bisphosphate tetra (cyclohexylammonium) salt hydrate (G-1,6-diP) was purchased from Sigma-Aldrich Co. LLC (St. Louis, USA). All the other reagents used were those of analytical grade.
The assay principle is depicted in Figure 1. Maltose is phosphorylated by the action of MP and eventually forms Thio-NADPH via enzymatic cyclic reaction using Thio-NADP, β-NADPH, and G-6-PDH. The change in absorbance at 405 nm as a result of the reaction is measured. The principle of enzymatic assay for maltose.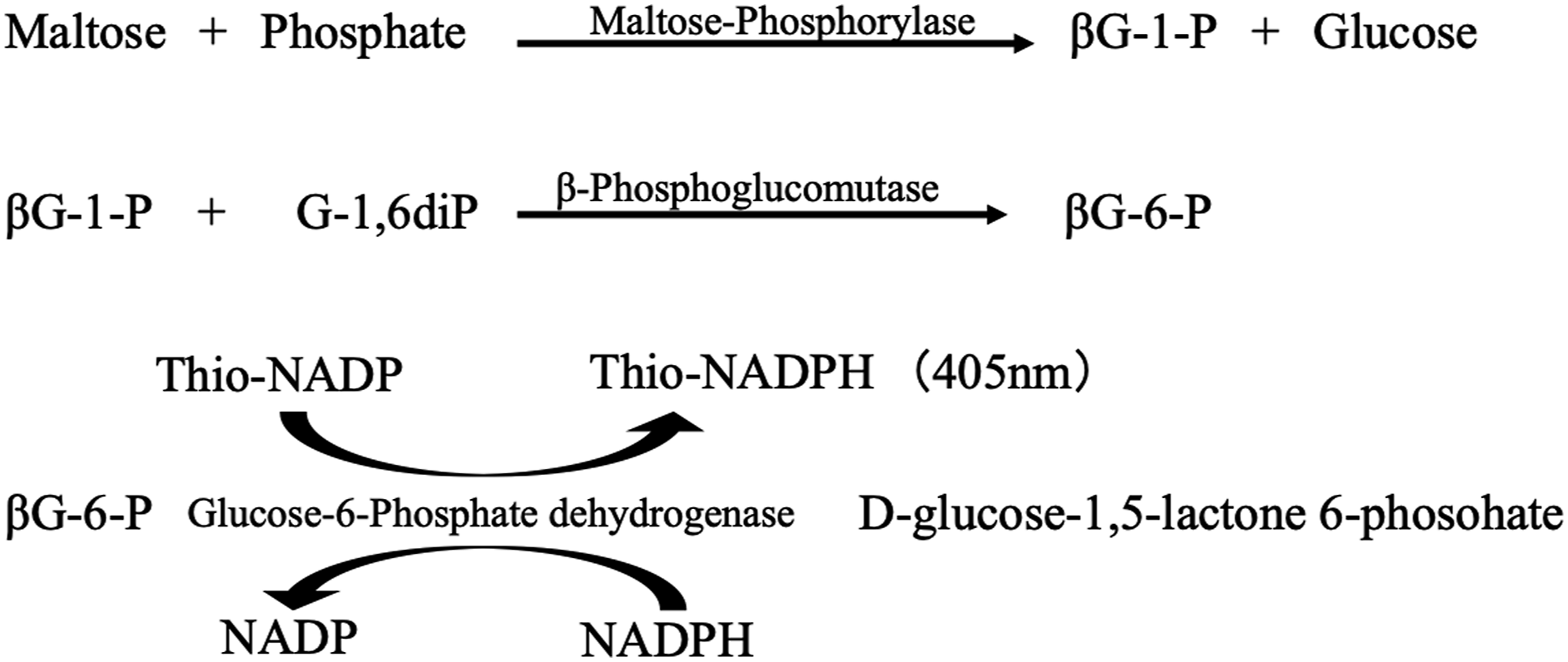
A model 7180 Hitachi automatic analyzer (Hitachi High-Tech Corporation, Tokyo, Japan) was used for the maltose assay. The concentration of each reagent was determined after evaluation of the optimal condition for each reagent. Changes in absorbance at a main wavelength (405 nm) and a complementary wavelength (600 nm) were measured automatically using 5 μL of plasma, 120 μL of the first reagent, and 60 μL of the second reagent by the endpoint assay.
Optimal condition for each reagent
Since this study was performed using an automatic biochemical analyzer, all measurements were conducted at 37°C. The spectrophotometer used was H7012 (Hitachi High-Tech Corporation, Tokyo, Japan).
Amount of MP
Using a 200 μmol/L maltose aqueous solution as a sample, MP activity was confirmed by changing the MP concentrations from 2.8 to 17.1 U/mL, and the strongest reaction was observed at 8.5 U/mL.
Amount of β-PGM
Using a 200 μmol/L maltose aqueous solution as a sample, β-PGM was confirmed by changing the β-PGM concentrations from 3.6 to 5.4 U/mL, and the strongest reaction was observed at 3.6 U/mL.
Amount of G-1,6-diP
Using a 200 μmol/L maltose aqueous solution as a sample, G-1,6-diP was confirmed by changing the G-1,6-diP concentrations from 0.046 to 0.069 mmol/L, and the strongest reaction was observed at 0.046 mmol/L.
Amount of G-6-PDH
Using a 200 μmol/L G-6-P aqueous solution as a sample, G-6-PDH activity was confirmed by changing the G-6-PDH concentrations from 50∼200 U/mL, and the strongest reaction was observed at 50 U/mL
Amount of Thio-NADP
Using a 200 μmol/L G-6-P aqueous solution as a sample, Thio-NADP was confirmed by changing the Thio-NADP concentrations from 0.23∼0.46 mmol/L, and the strongest reaction was observed at 0.23 mmol/L
Amount of β-NADPH
Using a 200 μmol/L G-6-P aqueous solution as a sample, β-NADPH was confirmed by changing the β-NADPH concentrations from 1.3∼5.3 mmol/L, and the strongest reaction was observed at 1.3 mmol/L
Amount of TritonX-100
Using a 200 μmol/L maltose aqueous solution as a sample, TritonX-100 was confirmed by changing the TritonX-100 concentrations from 0.10∼1.00% (v/v), and the strongest reaction was observed at 0.25%.
The final concentration
The first reagent was set at pH7.0 in consideration of the stability of various enzymes and Thio-NADP. The first reagents were included: β-PGM (3.6 U/mL), G-6-PDH (50 U/mL), G-1,6-diP (0.046 mmol/L), Thio-NADP (0.23 mmol/L), Phosphate Buffer (50 mmol/L), MgCl2 (3.1 mmol/L), KCl (31 mmol/L), TritonX-100 0.25%. The second reagent was set at pH8.0 in consideration of the stability of MP and β-NADPH. The second reagents were included: MP (8.5 U/mL), β-NADPH (1.3 mmol/L), Tris-HCl Buffer (50 mmol/L), MgCl2 (3.1 mmol/L), KCl (31 mmol/L), TritonX-100 (0.25%). The pH during the reaction was adjusted to pH7.5.
Samples
The samples were obtained from patients who underwent a health checkup at International University of Health and Welfare (IUHW) Narita Hospital (plasma that remained after the sugar tolerance test for diabetes screening performed simultaneously with barium X-rays or gastroscopy). Two individuals in whom no abnormality was detected by gastroscopy, 3 who were diagnosed to have fundic gland polyp, and 8 with atrophic gastritis (closed type in 4, open type in 4) were involved as the subjects. The samples used for condition studies and performance evaluation of this reagent were prepared by pooling plasma collected from healthy volunteers and adding maltose. This study was carried out with approval by the Institutional Review Board of IUHW (23-Im-001).
Results
Repeatability
Three plasma samples containing maltose at 10, 30, and 50 μmol/L were assayed 20 times under repeatability conditions. As a result, CV was 1.1, 0.7, and 0.2% at the respective concentrations. The inter assay CV was determined by measuring plasma sample on 14 different days. As a result, CV was 3.2, 2.1, and 1.9%, respectively.
Linearity
By serially diluting plasma containing maltose with physiological saline, a curve passing the origin with a linear range up to 120 μmol/L was obtained (Figure 2). Linearity.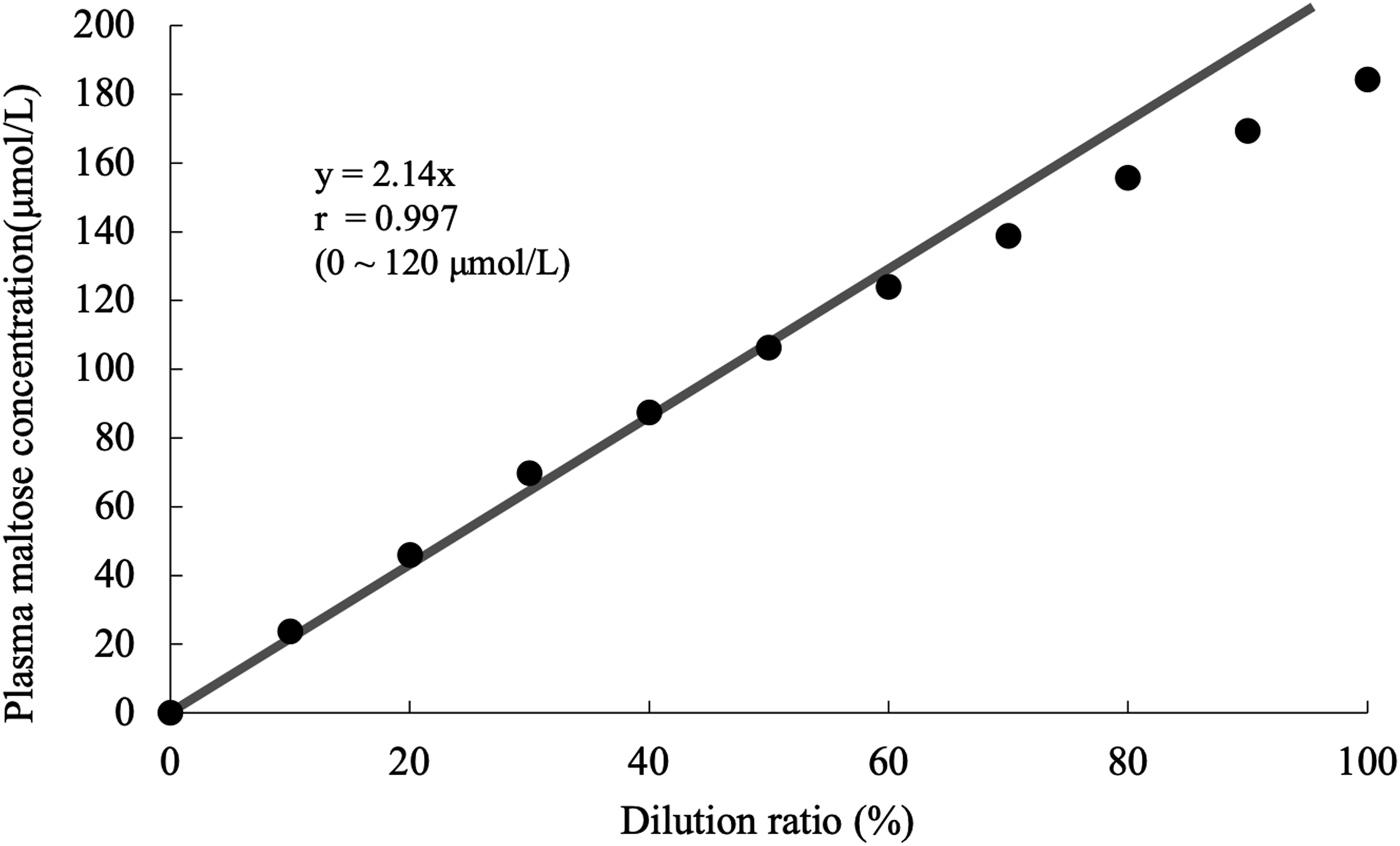
Analytical recoveries
The maltose-added plasma samples were prepared by adding one volume of maltose solution (100–500 Amol/l) to nine plasma containing 38 Amol/l maltose. Analytical recoveries were 102–103%.
Effects of interfering substances
Interference by dyes that may appear in the body was examined. Various substances were added at 1/10 to 30 μmol/L of maltose-containing plasma. The final concentrations of the added substances were: ascorbic acid (10 mmol/L), bilirubin-conjugate (20.1 mg/dL), bilirubin-free (19.7 mg/dL), chyle (1480 FTU), and hemoglobin (0.48 g/L). The effects of the substances at the concentrations were within ±5%
The selectivity study for the detection of maltose
In order to confirm the selectivity in maltose measurements, addition experiments were conducted with the following sugars. Seven types of sugars were added: glucose (50 mmol/L), xylose (10 mmol/L), fructose (10 mmol/L), lactose (10 mmol/L), sucrose (10 mmol/L), rhamnose (10 mmol/L), and mannose (10 mmol/L). Various substances were added at 1/10 to 30 μmol/L of maltose-containing plasma. The effects of the substances at the concentrations were within ±5%
Maltose concentration in patients with gastric mucosal damage
Changes in the maltose concentration in the plasma samples of patients who underwent the sugar tolerance test in the health checkup are shown in Figures 3(a) and (b). The maltose concentration in the plasma before the sugar tolerance test was 7.6–56.4 μmol/L, and the changes 1 and 2 hours after sugar loading compared with the baseline (0 μmol/L) were examined (Figure 3(a)). The maltose concentration was higher in patients with fundic gland polyp or severe atrophic gastritis (open type) than in those with no abnormality or those with mild atrophic gastritis (closed type) (Figure 3(b); P < .01, Welch’s t test). (a) The time course of the appearance of maltose in plasma after a glucose tolerance test in people diagnosed with nothing in particular on endoscopy and patients with gastric disease.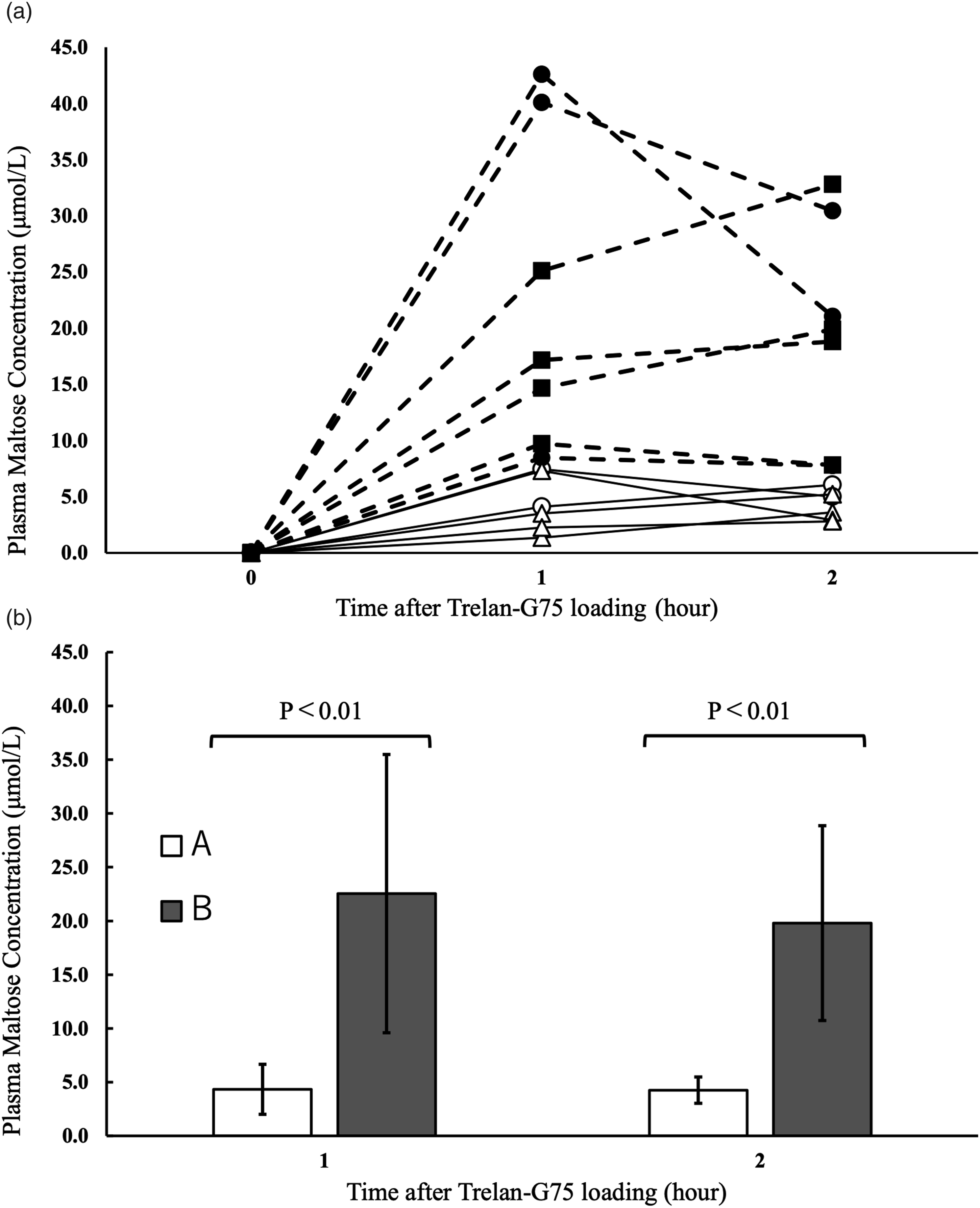
Discussion
We focused on the fact that the Trelan G solution used in the glucose tolerance test contained maltose and assumed that gastric mucosal damage could be detected by measuring the maltose concentration in plasma from a glucose tolerance test for diabetes. To measure maltose concentration inexpensively and quickly for this purpose, analysis using an automatic biochemical analyzer is required. The enzymatic assay of maltose was reported by Shirokane et al. 13 in 2000, but it is designed to measure maltose in water, and its sensitivity is insufficient for maltose assay in plasma. Enzyme cycling methods were used to increase detection sensitivity14–16 when quantifying low-concentration substances. To further increase sensitivity, Thio-NADP (ε = 11,900; 398 nm), a highly sensitive chromogenic substrate, was used. As a result, the test showed a sufficient detection sensitivity and little effect of coexisting interfering substances including sugars.
The application of this test to samples for the sugar tolerance test in health checkup, no association was observed between the maltose concentration before the test and the disease. However, examination of the association between the changes 1 and 2 h after sugar loading of maltose concentration and the grade of gastric mucosal damage showed that the increase in the maltose concentration from the baseline was greater in patients with fundic gland polyp, and severe atrophic gastritis (open type) compared with no abnormality or mild atrophic gastritis (closed type). The staging of atrophic gastritis was reported by Kimura et al. 17 It is classified into the closed type, in which the border of atrophy does not extend beyond the cardia, and the open type, in which the border of atrophy extends beyond the cardia to the greater curvature. It has been reported that patients with open type atrophic gastritis have a higher incidence of gastric cancer and undifferentiated cancer. 18
Therefore, the assay of maltose concentration in plasma after the sugar tolerance test for diabetes screening was suggested to be effective also as a screening test for gastric mucosal damage. For the future, it is necessary to evaluate whether the test is usable as a screening test of the upper gastrointestinal tract by applying it to normal individuals and esophageal and gastric cancer patients.
Conclusion
We developed a highly sensitive method for the assay of maltose concentration in plasma aimed to be used as a screening test for upper gastrointestinal disorders. This method is applicable to high-sensitivity and high-speed assays on automatic analyzers and is considered to have a potential to be used as a screening test for upper gastrointestinal disorders.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a project of the Kanto Branch Subcommittee of the Japanese Society of Clinical Chemistry.
Ethical approval
Ethics Committee of International University of Health and Welfare (approval number, 23-Im-001).
Guarantor
MS.
Contributorship
TK, MS, SO, EH designed the research; TK, KoK, and KiK performed the research; TK, ToS, TaS, were involved in analysis and interpretation of data; TK, MS were involved in gaining ethical approval; and TK and MS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
