Abstract
Background
Adopting the WHO protocol for glucose analysis is arguably impractical in the routine clinical setting. Deviations may develop due to a lack of understanding regarding the impact of glycolysis on the accuracy of results.
Aim
We sought to assess the stability of glucose in two different blood collection tubes (BCT), BD Vacutainer® FX ‘Fl-Ox’ and Greiner Vacuette® FC-Mix ‘FC-Mix’ stored at room temperature (RT:18–22°C) and 4°C over 8.5 days.
Method
Each participant provided venous whole blood collected into 51 BCTs; ‘Fl-Ox’ (n = 26) and ‘FC-Mix’ (n = 25). One Fl-Ox sample from each participant was handled according to the WHO recommended method. The remaining BCTs were stored at 4°C/RT prior to analyses at designated study timepoints. Glucose was measured using the hexokinase assay on the Cobas® 8000 platform.
Results
Participants (n = 8, Male = 2) were aged 24–56 years. Plasma glucose measured in FI-Ox BCTs according to the WHO sample-handling method had a median concentration of 5.73 mmol/L (Range: 5.39–10.37 mmol/L). Glucose decreased by greater than minimal difference (>0.26 mmol/L) in blood collected into Fl-Ox and stored @4°C/RT within 24 h of phlebotomy. FC-Mix BCT maintained glucose <0.26 mmol/L @4°C over a period of 8.5 days and up to 4 days @RT when compared to the WHO recommended method.
Conclusion
Glucose in FC-Mix BCT stored @4°C demonstrated the best agreement with results determined using the WHO specifications. When FC-Mix tubes were stored @RT, glucose was stable for 4 days. These findings suggest that the FC-Mix BCT effectively inhibits glycolysis and should be introduced into routine clinical practice.
Introduction
Diabetes mellitus (DM) is a metabolic disorder characterized by elevated blood glucose concentration (hyperglycaemia). Diagnostic tests for DM in the non-pregnant adult include glycated haemoglobin (HbA1C) and/or venous plasma glucose thresholds in samples collected in an individual at random or fasting (at least 8h) and at 2h post ingestion of a standard 75-g anhydrous glucose load.
Gestational diabetes mellitus (GDM), defined as hyperglycaemia with onset or first recognition during pregnancy, 1 is associated with short- and long-term adverse outcomes for both mother and baby. 2 Identification of women with GDM is important as randomized controlled trials have determined that treatment ameliorates adverse perinatal outcomes.3–5 In Ireland, diagnosis is made using a one-step oral glucose tolerance test (OGTT) with venous plasma glucose measurements made in a woman fasted overnight and at 1h and 2h post ingestion of a 75-g anhydrous glucose load, and adjudicated based on the seminal Hyperglycaemia and Adverse Pregnancy Outcome (HAPO) Study published in 2008. 6 The HAPO study aimed to clarify the risks of adverse outcomes which would be associated with degrees of maternal glucose intolerance considered less severe than those with overt diabetes during pregnancy. 7 A key finding of the HAPO study was that the associations of various adverse outcomes with OGTT results were continuous, with no clear inflection points identified.
Post the publication of the HAPO study, a task force of the International Association of Diabetes in Pregnancy Study Groups (IADPSG) convened to develop a consensus regarding appropriate diagnostic criteria based on the HAPO cohort data and recommended that the diagnosis of GDM be made when any of the following OGTT thresholds are met or exceeded: Fasting 5.1 mmol/L, 1h 10.0 mmol/L and 2h 8.5 mmol/L. 7 These diagnostic criteria were endorsed by the World health Organization (WHO) in 2013, and importantly glycated haemoglobin (HbA1c) is not recommended as a diagnostic test for GDM. 8
Acknowledging that the diagnosis of GDM depends exclusively on the measurement of glucose, and may be made on relatively small changes in glucose concentration, the IADPSG report recommends that an enzymatic method with high accuracy and precision be used to measure glucose, and that proper sample collection and processing be employed to minimize preanalytical glycolysis. This is to ensure a standardized approach to GDM diagnosis that mirrors the stringent sample-handling and processing conditions of the HAPO study methodology; immediate immersion of OGTT specimens (sodium fluoride (NaF) blood collection tube (BCTs)) in crushed ice/ice-slurry with plasma separated within 30 mins to 1h of the completion of the OGTT test. 9
Implementing recommended preanalytical standards to ensure the quality of glucose results that minimize the potential for misdiagnosis and mismanagement of patients cannot be overstated. 10 Glucose in whole blood ex-vivo continues to be metabolized (glycolysis) by blood cells to generate energy lowering its concentration. 11 The estimated rate of loss of glucose due to glycolysis is 5–7% per hour 12 which can lead to a missed diagnosis of diabetes which is particularly important during pregnancy. This preanalytical loss of glucose at 1h is greater than the desirable limit of total analytical error for glucose based on biological variation (6.9%).13,14 Hence, prevention of glycolysis is critical to ensure the accuracy of results of glucose measurement.11,15 To minimize glycolysis ex-vivo, the World Health Organization (WHO) recommend that plasma is separated immediately from venous whole blood, or alternatively, that the sample is collected in a blood collection tube (BCT) with a glycolytic inhibitor and placed in an ice-water slurry and plasma separated within 30 minutes.12,16 The purpose of the ice-water slurry is to inhibit cellular metabolism by cooling. However, recent studies have highlighted that although rapid cooling reduced the rate of glycolysis, it did not inhibit it completely. Prompt centrifugation of the BCT reduces the impact of cellular metabolism on plasma glucose concentration by the physical separation of blood cells from plasma.
The BCT commonly used in clinical laboratories for glucose measurement contains sodium fluoride potassium oxalate (Fl-Ox). Sodium fluoride is an enolase or ‘late’ inhibitor of glycolysis and potassium oxalate inhibits coagulation. This downstream mechanism of action leads to complete inhibition of glycolysis only after 1h to 2h post blood collection. Such BCT should not be relied on to prevent glycolysis. Instead, BCT with a rapidly effective inhibitor of glycolysis should be used. Recent evidence has shown that BCTs containing a citrate buffer (sodium-fluoride-ethylenediaminetetraacetic-acid-citrate (NaF-EDTA-citrate)), hereafter termed as ‘FC-Mix’ are better inhibitors of glycolysis than BCTs containing sodium-fluoride–potassium-oxalate alone.12,17 Citrate primarily inhibits the enzyme hexokinase and is therefore an ‘early’ glycolytic pathway inhibitor. Citrate acidifies venous blood (pH 5.3–5.9) which in turn leads to the inhibition of the glycolytic pathway at this early stage (Figure 1). The inhibition of glycolysis earlier in the pathway has been shown to maintain glucose concentrations in blood specimens much more effectively, therefore leading to more accurate glucose measurements.17–19 The tube manufacturers state that the additives in the FC-Mix/Fl-Ox BCTs can stabilize the sample for up to 48 hours if processed according to their instructions.
20
Blood collection tube (BCT) additives and their inhibitory effect on enzymes of the glycolytic pathway.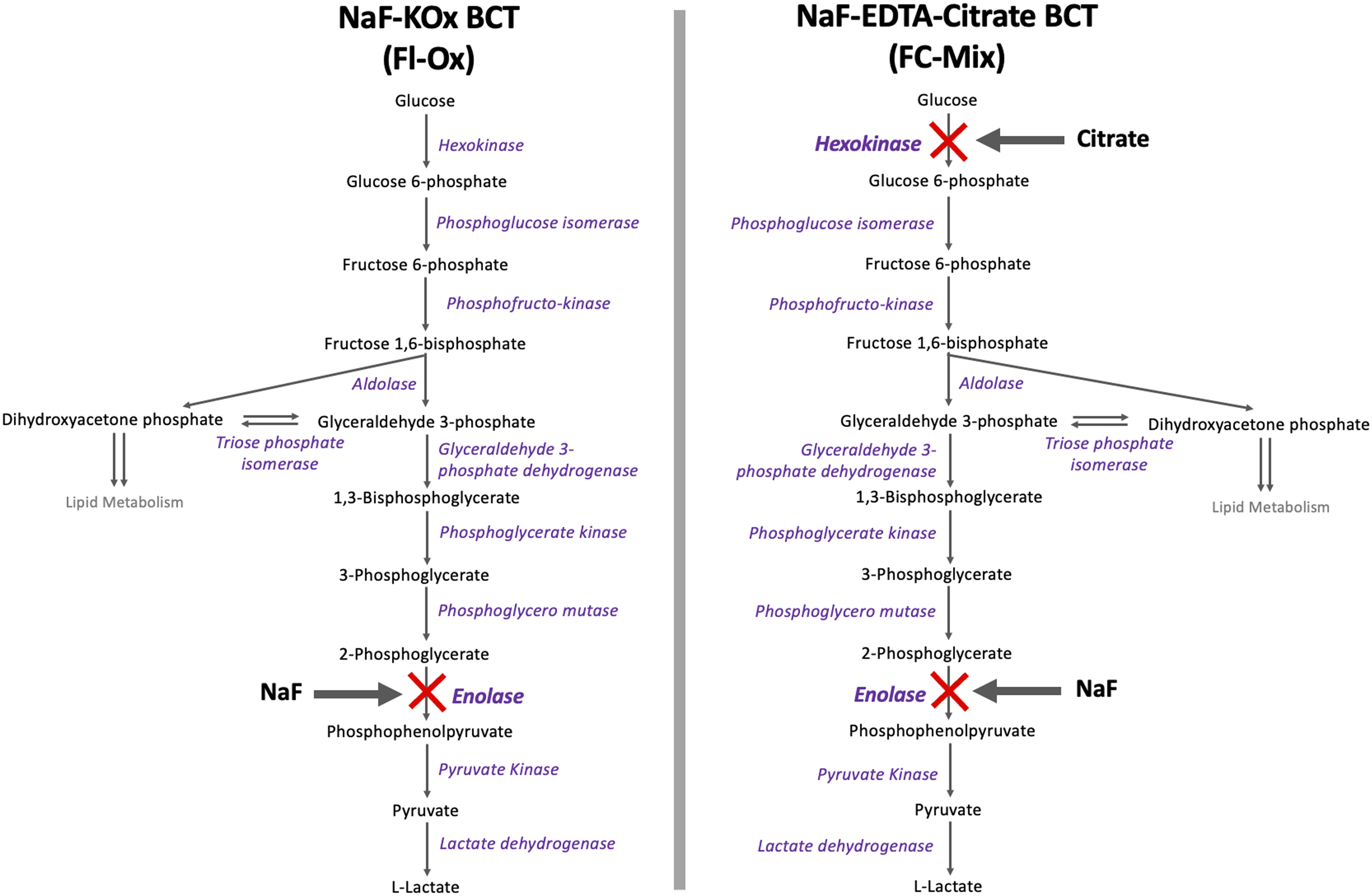
The quality of specimens for glucose analysis is also impacted by grossly haemolysed samples because red blood cells contain lower glucose concentrations than plasma. This issue is further compounded by extended specimen transit times that lead to a delay in centrifugation and separation of plasma from cells. ‘Spurious haemolysis’, happens when erythrocytes in whole blood specimens lose their ability to maintain osmotic homeostasis resulting in cell lysis. This is due to the metabolic consumption of ATP reserves, which are finite ex-vivo. Moreover, prolonged contact of erythrocytes with anti-glycolytic agents causes the release of intracellular fluid, free haemoglobin (Hb) and the enzyme catalase. Catalase can further lower glucose concentration. 21 Together this affects the reliability of glucose analysis in specimens that are grossly haemolysed 17
The ‘WHO 2006 sample handling protocol’ is markedly different to the routine specimen handling procedure and laboratory workflow at our institution. Here, OGTT specimens are collected into ‘Fl-Ox’ BCTs, retained together at ambient temperature with one request form until test completion. On completion, samples together with the request form are transported to the laboratory specimen reception where they are labelled according to time of collection, with patient demographics and test request inputted into the laboratory information system. Samples are then sent to the core laboratory for centrifugation and analysis. This practice of batching OGTT samples prior to analysis means that the effect of glycolysis is greatest on the fasting sample as it has the longest delay to analysis. Moreover, samples received prior to a weekend are potentially stored at room temperature (@RT) for up to 2 days post phlebotomy and prior to centrifugation.
In the case of samples (random, fasting and OGTT) for glucose analysis that originate from General Practice, these regularly arrive to the laboratory more than 2 days post blood draw, in fact glucose measurement can be delayed by as much as 5 days if collected prior to bank holidays. There is an unmet clinical need for BCTs that are highly effective in inhibiting glycolysis, allowing accurate measurement and interpretation of glucose levels, and minimizing the risk of post sampling degradation of glucose. 12
This study sought to assess glucose stability and haemolysis in two BCTs, the Becton Dickinson® (BD) fluoride-oxalate (Fl-Ox) and the Greiner® citrate-fluoride-oxalate (FC-Mix) stored at room temperature (RT) and 4°C over a period of 8.5 days, compared to the WHO sample-handling protocol. 16
Methods
Study design
Recruitment was by word of mouth. All participants were fully informed of the nature of the study by initial meeting and detailed Participant Information Leaflet. Following informed written consent, participants ≥18 years and non-pregnant were invited to partake. Each participant was required to provide ∼127mL of venous blood (51 BCTs). Phlebotomy was performed with a 50mL syringe used to collect and transfer blood into BCT’s rapidly. A total of 8 individuals with and without diabetes (self-declared) were recruited to this study, Figure 2 illustrates the study design. Recruitment schematic.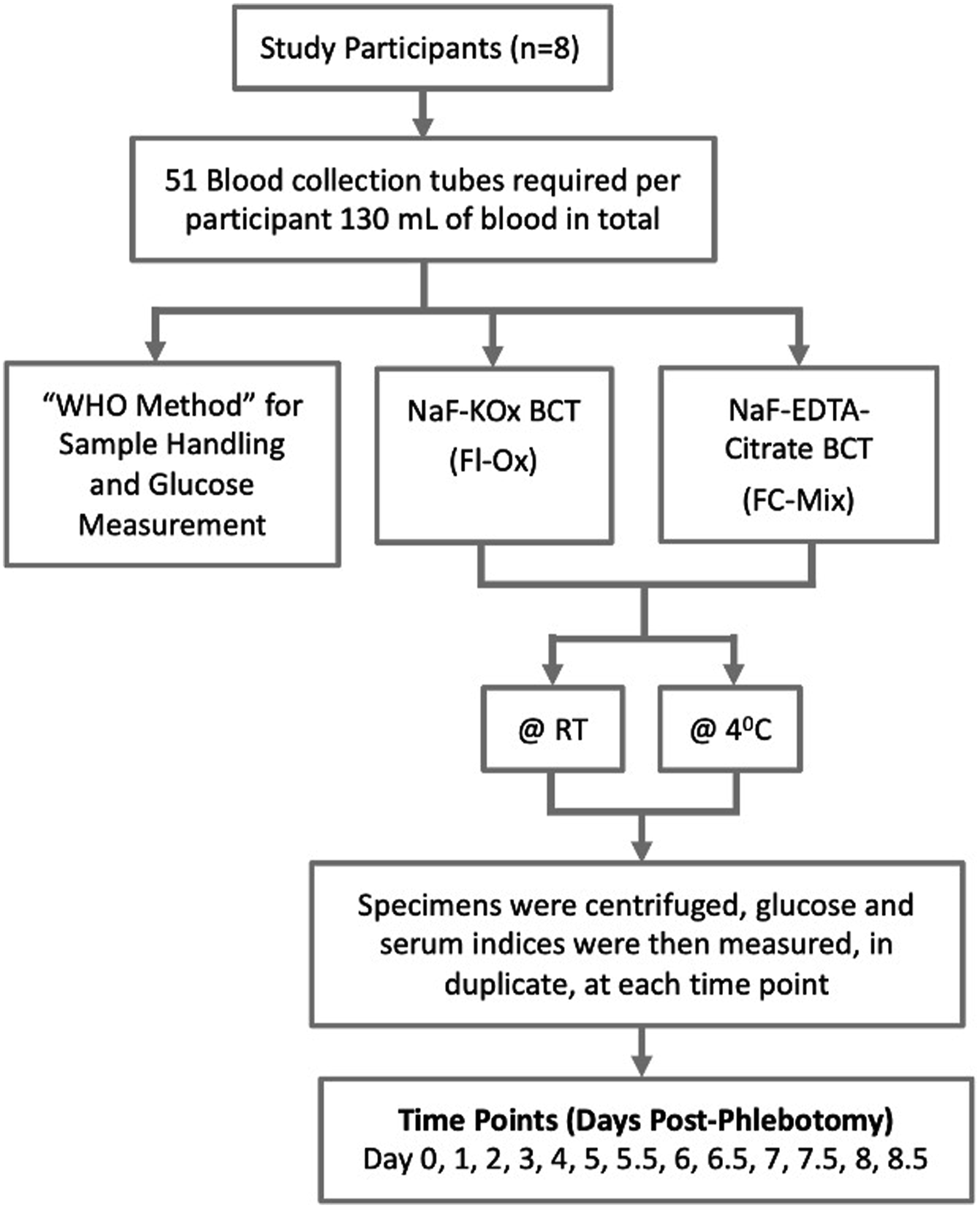
Sample collection and handling
Venous blood specimens were collected into two BCT types; the BD Vacutainer® FX ‘Fl-Ox’ (Catalogue No: 368920, capacity: 2mL) and the Greiner Vacuette® FC-Mix ‘FC-Mix’ (Catalogue No: 454514, capacity: 3mL). Each participant provided whole blood material for 51 BCTs; ‘Fl-Ox’ (n = 26) and ‘FC-Mix’ (n = 25).
One venous blood sample from each of the 8 participants collected into Fl-Ox BCT was immediately placed in an ice slurry and analysed within 30mins of phlebotomy (WHO recommended sample-handling method). The remaining 8x 25 Fl-Ox and 8x 25 FC-Mix BCTs collected were stored at 4°C or RT prior to analyses at the designated study timepoint. Glucose was subsequently measured using the hexokinase assay on the Cobas® 8000 platform at baseline (and every 24h for 5 days followed by every 12h for a further 3.5 days (Figure 2)). The baseline (day 0) measurement of glucose was made in both BCTs types at 2.5h post blood draw. This time interval reflects the average ‘real world’ turnaround time for glucose analyses received from non-acute wards and clinics at our institution. To minimize analytical variability, all samples were analysed on the same analyser with identical reagent lots. Three levels of Internal Quality Control (IQC) materials were run pre- and post-analysis and evaluated in accordance to the best laboratory practice. 22 The mean (SD) concentration of glucose in each of the 3 levels of IQC material employed was, 2.27 (0.04) mmol/L, 6.78 (0.08) mmol/L and 15.11 (0.16) mmol/L, respectively.
Plasma glucose measurement
Following assay calibration and confirmation that IQC conditions were met, BCTs were centrifuged at 3000 RPM (Hitachi Rotina 420 centrifuge) for 10 minutes to separate plasma from cells. Glucose was analysed in duplicate using the hexokinase assay (Roche Cobas® Glu HK Gen.3) ISO 15189: 2012 accredited method on the Roche Cobas® 8000 modular analyser series (Roche Diagnostics Limited, West Sussex, UK). The assay is an enzymatic, UV spectrophotometric method which measures an analyte’s concentration at two wavelengths (700/340nm). It employs two enzymes: hexokinase and glucose-6-phosphate dehydrogenase. Hexokinase catalyzes the phosphorylation of glucose to glucose-6-phosphate via ATP. Glucose-6-phosphate dehydrogenase (in the presence of NADP) then oxidizes glucose-6-phosphate to gluconate-6-phosphate to produce NADPH. The amount of NADPH formed is then measured spectrophotometrically. The rate at which it is produced is directly proportional to the concentration of glucose present in the specimen. 23 In addition, serum indices (Haemolysis (H), Lipaemia (L) and Icterus (I)) that monitor an instrument’s response to detect and flag preanalytical interference affecting analysis were performed on all samples.
Statistical analysis
Statistical analyses were performed using Analyze-it® (Version 6.15) and MedCalc® Statistical Software (Version 20.112).
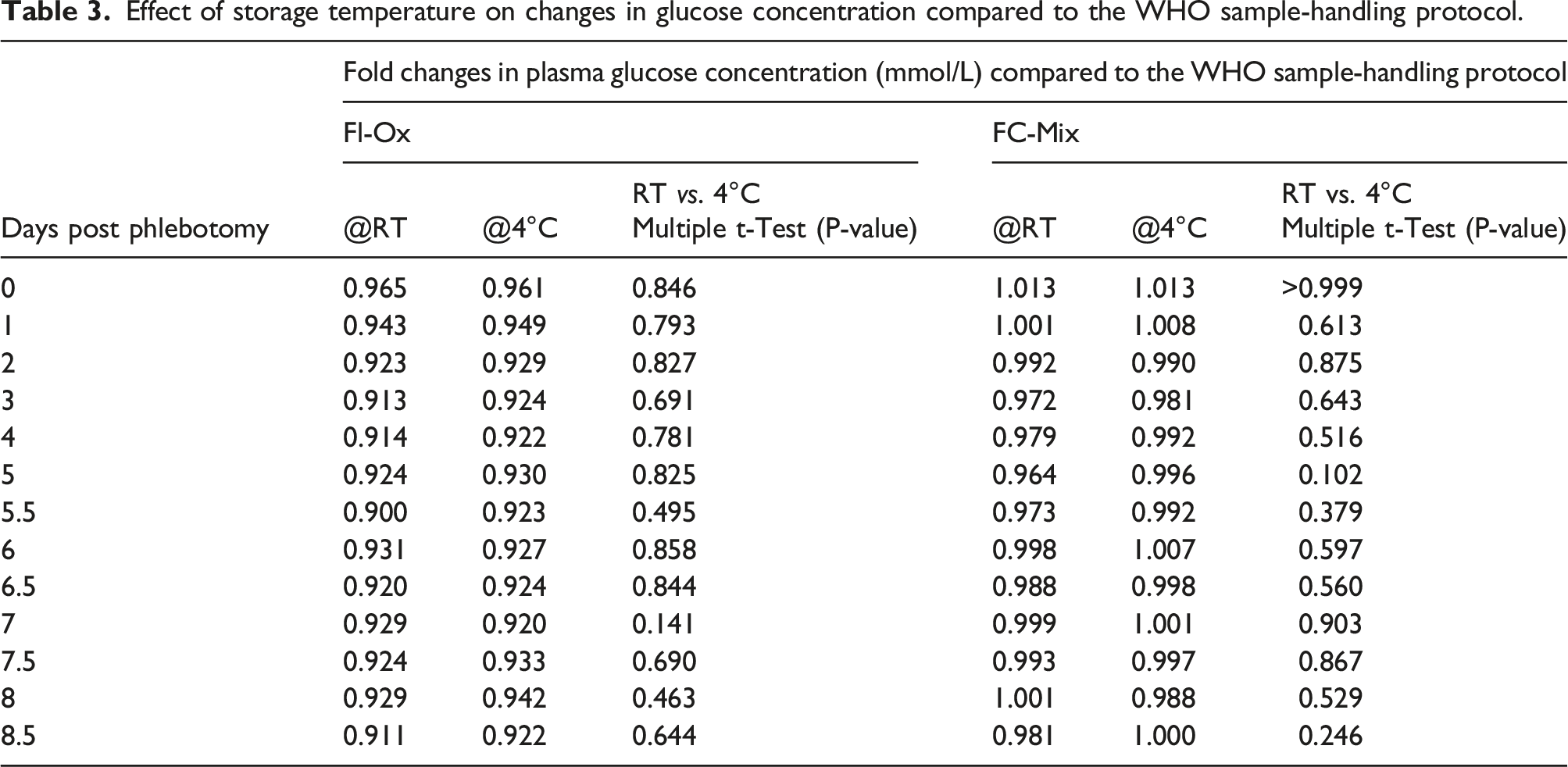
To compare changes in glucose concentration in two different BCTs at different time points, fold changes in glucose concentration were calculated by dividing each value with the glucose concentration determined by WHO sample-handling method. The statistical differences in the median glucose values from the two BCTs at different time points were analysed using multiple comparisons t-test corrected by Bonferroni–Dunn method. A p-value of <0.05 was deemed statistically significant.
In addition, to convert analytical imprecision into the unit of the measurand (mmol/L), the minimal difference (MD) was calculated.24,25 The MD is the smallest difference between two consecutive measurements that is statistically significant. The formula is
The standard deviation (SD) of IQC materials level 1, 2 and 3 for the Roche Cobas® Glu HK Gen.3 assay was 0.04, 0.08 and 0.16 mmol/L, respectively, giving an average SD of 0.09 mmol/L. The resultant MD value was calculated as 0.26 mmol/L. The MD value of 0.26 mmol/L was then used to determine which specimens yielded glucose concentrations that differed significantly from the baseline result achieved using the WHO sample-handling protocol.
Results
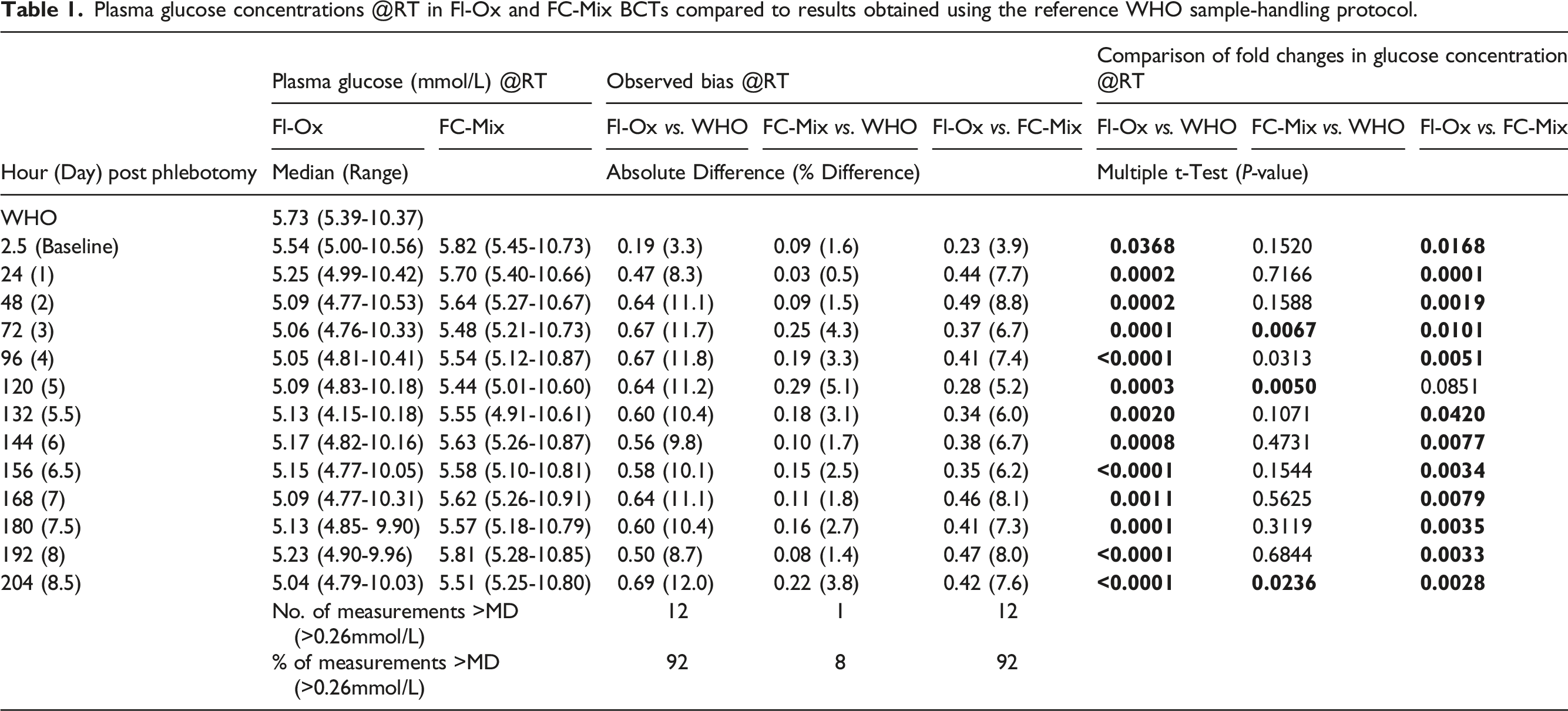
Participants (n = 8) ranged in age between 24 and 56 years and 2 were male. Volunteer plasma glucose concentrations measured in FI-Ox BCTs collected according to the WHO sample-handling method had a median concentration of 5.73 mmol/L (Range: 5.39–10.37 mmol/L).
Plasma glucose concentrations @RT in Fl-Ox and FC-Mix BCTs compared to results obtained using the reference WHO sample-handling protocol.
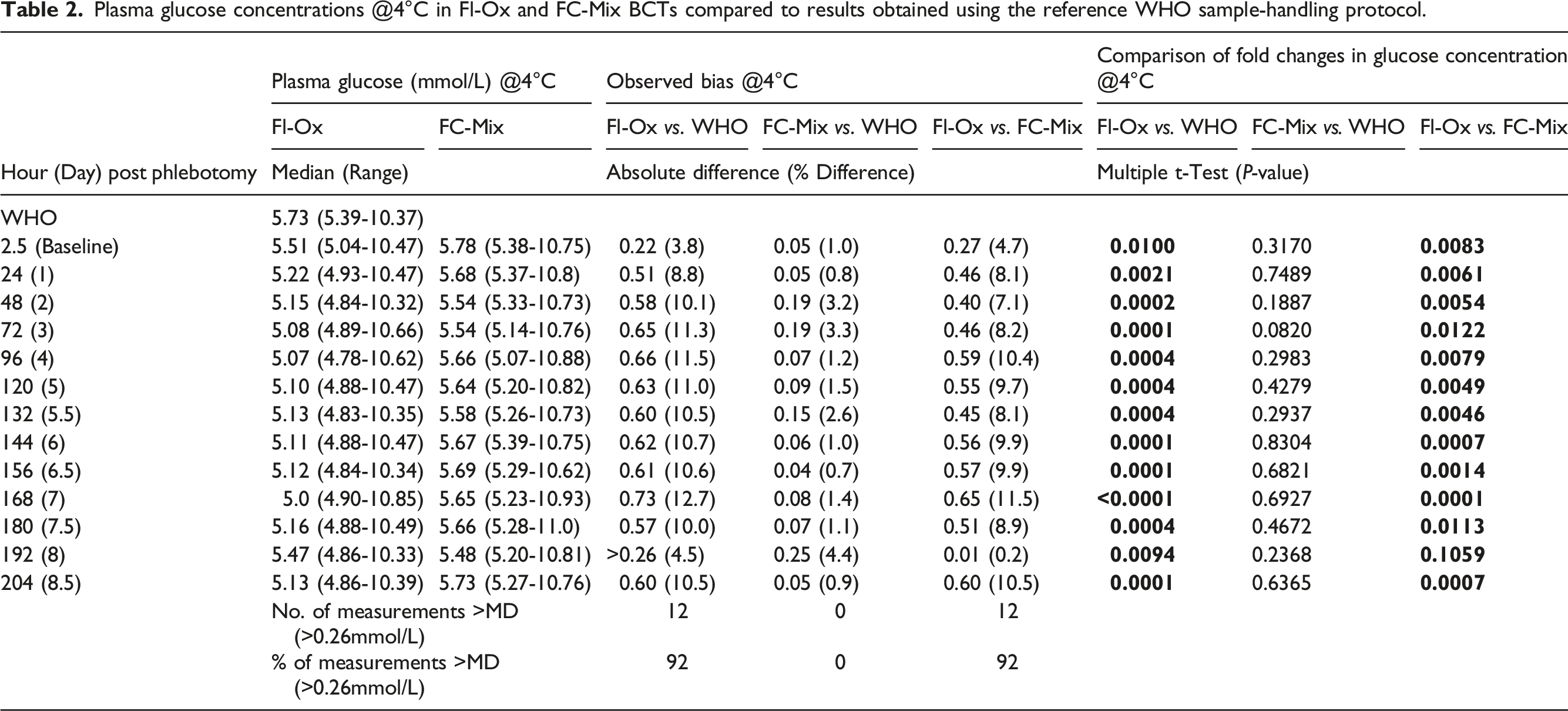
Plasma glucose concentrations @4°C in Fl-Ox and FC-Mix BCTs compared to results obtained using the reference WHO sample-handling protocol.
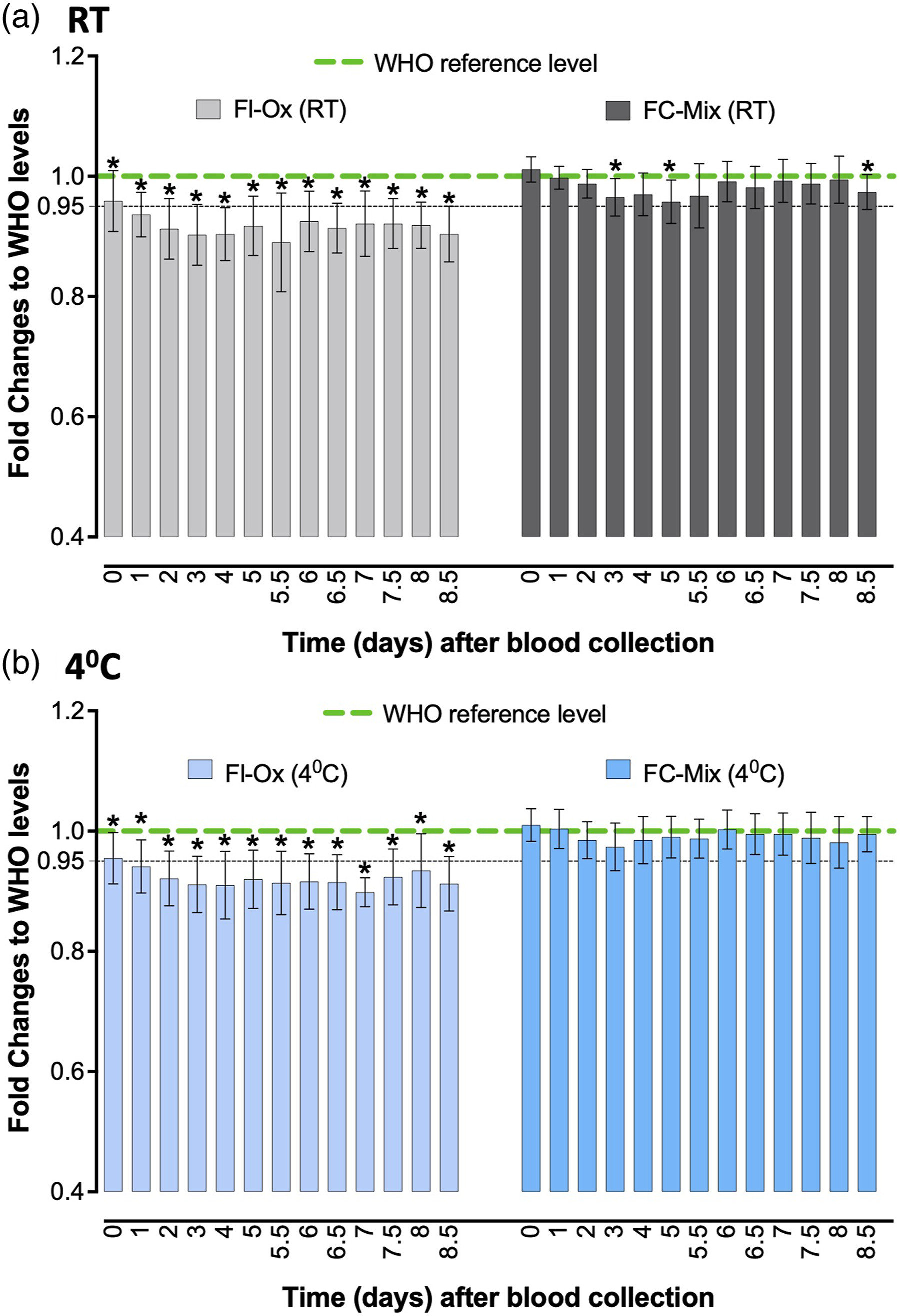
Fold changes in glucose concentration by Fl-Ox or FC-Mix BCTs compared to the WHO reference levels. Data represented as median (minimum to maximum) fold change of glucose concentrations; *p < .05 is statistically significant.
In contrast, when FC-Mix tubes were stored @RT, glucose results on day 3, 4, 5 and 8.5 post blood draw were significantly lower than those measured according to the WHO protocol with the MD exceeded on day 5 post phlebotomy. Plasma glucose results obtained when FC-Mix tubes were stored @4°C were comparable and not significantly different to those achieved using the WHO sample-handling criteria (Table 2). Moreover, at this temperature, the MD was not exceeded at any of the study time points.
Effect of storage temperature on changes in glucose concentration compared to the WHO sample-handling protocol.
Haemolytic index (HI), measurements that are indicative of the degree of haemolysis were performed in duplicate on all study samples. The rate of haemolysis in BCTs containing Fl-Ox was higher than that of FC-Mix BCTs (Supplemental Figure 1). The highest HI results were evident in Fl-Ox BCTs and the lowest in the FC-Mix stored @RT. However, in total, only 4 HI measurements breached the rejection limit (HI >1000 S.I. units). These plasma glucose results were not reported and were excluded from statistical analyses. This HI breach occurred in one participant’s samples at days 5.5, 6, 6.5 and 8.
Discussion
This study examined the measurement of glucose levels in blood collected into Fl-Ox and FC-Mix BCTs according to local practice and compared the results observed to those achieved using the gold standard WHO sample-handling protocol. A sharp decline in median glucose concentration was observed within 24h of phlebotomy in whole blood collected into Fl-Ox BCT and stored @ either 4°C or RT. The pattern of initial decline and then relative stability in glucose concentrations in Fl-Ox BCTs aligns to the mechanism of action of sodium fluoride, a ‘late’ stage glycolytic inhibitor. In contrast, plasma glucose results in FC-Mix tubes stored @4°C were comparable to those achieved using the WHO protocol and at each study time point all results were within the MD. Of note, glucose results in FC-Mix tubes stored @RT were not comparable to those of the WHO protocol after day 4 post phlebotomy. Haemolysis in Fl-Ox BCTs was higher than that of FC-Mix BCTs an observation that agrees with the findings of del Pino et al. 26 Despite this finding, in the Fl-Ox samples assessed, a breach of the HI limit prohibiting the reporting of glucose results was relatively infrequent (<1%). Together these findings confirm that BCTs with enolase inhibitors alone (Fl-Ox) should not be relied upon to prevent glycolysis. 12
Adopting the WHO protocol for glucose analysis is challenging in the routine clinical setting. Deviations may develop due to a lack of understanding regarding the impact of glycolysis on the quality and accuracy of results, the manner and point of action of different anti-glycolytic agents, resource and logistical limitations. Stapleton et al, demonstrated that time and temperature affect glycolysis with the potential to underdiagnose diabetes. These authors estimated that if the fasting glucose result was amended to reflect the quantity of glucose loss due to glycolysis when the WHO recommended approach is not followed, the diagnosis of GDM in their population would more than double. 27 It is important to recognize that most of the glucose loss in Fl-Ox fasting samples on ice happens within 30 mins of blood draw. 28 This may be due to delayed cooling of Fl-Ox samples impeding the immediate arrest of glycolysis even when samples are rapidly analysed. 19
To prevent glycolysis and to ensure a solution that is practicable in routine clinical settings, a rapid inhibitor of glycolysis is required. Previous studies support citrate as a superior glucose sample stabilizer to fluoride.19,29,30 The standardized introduction of FC-Mix tubes would reduce and eliminate variability in diagnostic sensitivity of testing for GDM and facilitate epidemiology studies in the field. Moreover, such tubes would also have value in the diagnosis of DM, impaired fasting glycaemia and glucose intolerance, where use of glycated haemoglobin is unreliable. Potential limitations of the current study are its small sample size and the concentration range of glucose assessed.
Conclusion
Glucose in FC-Mix BCT stored @4°C demonstrated the best agreement to results determined using the WHO specifications. When FC-Mix tubes were stored @RT glucose was stable for 4 days. These findings suggest that the FC-Mix BCT effectively inhibits glycolysis and should be introduced into routine clinical practice to ensure the correct classification of people undergoing testing for DM and GDM.
Supplemental material
Supplemental Material - Maintaining glucose integrity ex-vivo: Impact of pre-analytical specimen handling
Supplemental Material for maintaining glucose integrity ex-vivo: Impact of preanalytical specimen handling by Md Nahidul Islam, Claire Lyons, Tomas P Griffin, Siobhan Hamon, Fidelma P Dunne and Paula M O’Shea in Annals of Clinical Biochemistry.
Footnotes
Acknowledgements
We wish to express our gratitude to all the participants, medical, nursing and scientific staff of Galway University Hospitals Ireland, who made this research possible. We gratefully acknowledge the commitment and dedication of everyone involved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was granted by the Clinical Research Ethics Committee, Galway University Hospitals (Ref: C.A. 1865).
Guarantor
PMO’S.
Contributorship
PMO’S was responsible for study concept and design. PMO’S, MNI, CL, TPG and FPD were responsible for patient recruitment. PMO’S, CL and SH were responsible for the analytical measurements and quality of analysis. PMO’S and CL were responsible for data collation. PMO’S, MNI, TPG and CL were responsible for statistical analysis. MNI drafted the initial manuscript. All authors (MNI, CL, TPG, SH, FPD and PMO’S) made substantial contributions to the interpretation of data, critically reviewed the manuscript for important intellectual content and approved the final version of the manuscript. PMOS is responsible for the integrity of the work as a whole.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
